Introduction
Lung cancer is one of the leading causes of
cancer-associated mortality worldwide, resulting in >1 million
deaths annually (1). Non-small cell
lung carcinoma (NSCLC) accounts for ~85% of lung cancer cases
(2). As an antitumor drug, cisplatin
(DDP) is commonly used for non-small cell lung cancer as a
first-line treatment (3); however,
this treatment is associated with serious adverse side effects.
Hence, the identification of novel target drugs or an effective
combination of existing drugs is required for the improved
treatment of lung cancer.
The WNT signaling pathway serves a key role in the
regulation of cell proliferation, differentiation, apoptosis and
migration. Abnormal expression and activation of WNT signaling
pathway components have been observed to induce tumorigenesis
(4,5).
Tankyrase (TNKS) is an important regulator of canonical
WNT/β-catenin signaling (6). TNKS was
initially discovered to bind with telomerase repeat binding
sequences and belongs to the family of proteins responsible for
poly ADP-ribosylation (PARylation), which includes TNKS1 and TNKS2
(6). Busch et al (7) demonstrated that TNKS-knockdown reduced
the proliferation of murine and human lung cancer cell lines, and
decreased tumor formation in mouse models. TNKS inhibition may
therefore represent a novel therapeutic strategy for cancer
treatment. XAV939 is a small molecule that selectively inhibits
TNKS family members, including TNKS1 and TNKS2. XAV939 can inhibit
the transcription regulated by β-catenin, which is a key
transcription factor in the WNT signaling pathway. Kulak et
al (8) revealed that XAV939
inhibited the activation of β-catenin; after binding to TNKS,
XAV939 was able to stimulate β-catenin phosphorylation and abolish
the function of axin in the ‘destruction complex’. This action
resulted in the inhibition of the WNT pathway and stabilization of
axin by PARylation (9,10). Targeting components of the
WNT-TNKS-β-catenin pathway, together with epidermal growth factor
receptor (EGFR) inhibition, may improve clinical outcomes in
patients with NSCLC (11). Through
its effect on TNKS activity, XAV939 also inhibited WNT signaling in
cancer of the breast, colon and other tissues (12). However, XAV939-mediated regulation of
the WNT signaling pathway in lung cancer has not been clearly
elucidated. Therefore, the aim of the present study was to
investigate whether different concentrations of XAV939 influenced
the proliferation and migration of the lung adenocarcinoma A549
cell line, via with the WNT signaling pathway. The present study
demonstrated that TNKS (TNKS1 and TNKS2) and β-catenin expression
were increased in lung adenocarcinoma and that this increase
positively correlated with expression of the other. It is hoped
that the present study provides a novel approach and experimental
evidence for the basis for the future clinical treatment of lung
cancer.
Materials and methods
Patient samples
The present study consisted of 72 patients
(including 37 men and 35 women; 34–83 years old, with a mean age of
61.24±3.56) with single-subtype alveolar-like lung adenocarcinoma
(the lung adenocarcinoma group) and 67 patients (including 34 men
and 33 women; 34–78 years old, with a mean age of 60.44±4.23) with
normal lung tissue adjacent to carcinoma specimens (the
adjacent-to-carcinoma group) who underwent lung adenocarcinoma
puncture and bronchoscopy between January 2011 and June 2015.
Formalin-fixed and paraffin-embedded patient samples were obtained
from the Department of Pathology at First Teaching Hospital of
Tianjin University of Traditional Chinese Medicine (Tianjin,
China). Sections from all cases were reviewed and confirmed to be
single-subtype alveolar-like lung adenocarcinoma by two senior
pathologists affiliated with the First Teaching Hospital of Tianjin
University of Traditional Chinese Medicine.
All patients were diagnosed with lung cancer and had
no indication of surgery, nor had any undergone chemotherapy or
radiotherapy. Retrospective clinicopathological data, including
age, gender and degree of cancer cell differentiation, were also
obtained from the patients. A total of 21 pairs of single subtype
alveolar-like lung adenocarcinoma samples and matched adjacent
normal lung tissues (including 11 men and 10 women; 33–78 years
old, with a mean age of 60.23±3.44) were selected following lung
puncture and bronchoscopy between January 2014 and June 2015, and
frozen in liquid nitrogen at −80°C, within 20 min of collection.
The use of the tissue samples for this study was approved by the
First Teaching Hospital of Tianjin University of Traditional
Chinese Medicine Medical Ethics Committee.
Reagents
The primary antibodies used were mouse
anti-tankyrase (cat no. ab13587; Abcam, Cambridge, UK), mouse
anti-β-catenin (cat no. ZS-7963; OriGene Technologies, Inc.,
Beijing, China) and mouse anti-β-actin (cat no. MAB8929; OriGene
Technologies, Inc.), mouse anti-Myc proto-oncogene protein (c-Myc)
(cat no. sc-40; Santa Cruz Biotechnology, Inc., Dallas, TX,
USA).
Immunohistochemistry (IHC)
Formalin-fixed and paraffin-embedded sections were
deparaffinized by sequential washing with xylene, and graded
ethanol (100, 95, 80 and 70%), and subsequently heated with sodium
citrate buffer 0.01M, pH 6.0 in a pressure cooker to ~95°C for 2
min to perform antigen retrieval. The sections were incubated with
3% H2O2 in methanol for 15 min at 25°C,
blocked using goat isolated serum for 30 min at 25°C, and then
incubated with mouse anti-TNKS monoclonal antibody (1:200
dilution), mouse anti-β-catenin monoclonal antibody (1:200
dilution), and mouse anti-cMyc monoclonal antibody (1:100
dilution), overnight, at 4°C. The slides were then incubated with
horseradish peroxidase-conjugated goat anti-rabbit/mouse secondary
antibodies (cat no. PV9000; OriGene Technologies, Inc.) at 37°C for
30 min. Samples known to express TNKS, β-catenin, and c-Myc, among
others, served as positive control, whereas PBS was used instead of
the primary antibody as negative control. Positive samples were
obtained from the Department of Pathology, First Teaching Hospital
of Tianjin University of Traditional Chinese Medicine. The
intensity of staining was evaluated using the following criteria:
1, negative; 2, positive (brown). The extent of staining was scored
as 0, 0% of cells stained; 1, 1–25% stained; 2, 26–50% stained; 3,
51–100% stained. The final score was calculated by multiplying the
intensity score by the extent score. The staining results were
divided into two categories, according to the final score: <3,
negative, ≥3, positive. If 10% of cancer cells were positive for
nuclear staining, they were defined as positive.
Cell culture
The human A549, Calu-3 and SK-LU-1 cells were
provided by Dr Shiwu Zhang (Tianjin Union Medicine Center, Nankai
University Affiliated Hospital, Tianjin, China). Cells were
maintained in RPMI-1640 medium (Hyclone; GE Healthcare, Chicago,
IL, USA), supplemented with 10% (v/v) fetal bovine serum (Hyclone)
and cultured in a humidified incubator with 5% CO2 at
37°C.
MTT cell viability assay
A549 cells in the logarithmic growth phase were
seeded in 96-well plates, at a density of 2×104
cells/well, and subsequently treated with various XAV939 (Selleck
Chemicals, Houston, TX, USA) concentrations (0.1, 0.5, 1, 5, 10
µmol/l) for 24, 48, 72 and 96 h. MTT was added to the cells 4 h
prior to harvesting. Cells from each group were incubated for 4 h
with 10 µl MTT, which was subsequently replaced by 150 µl DMSO. The
96-well plates were then placed on a shaking table for 10 min. Cell
viability was determined by colorimetry, and measured the
absorbance using a microplate reader (Thermo Fisher Scientific,
Inc., Waltham, MA, USA) at a wavelength of 490 nm (A490 value).
Colony formation assay
For the colony formation assays, A549 cells were
plated at a density of 200 cells/well in 6-well tissue culture
plates (Corning Incorporated, Corning, NY, USA) in triplicate for
24 h prior to drug (XAV939; Selleck Chemicals) or control
treatments. After 24 h, A549 cells were treated with doses of 0.1,
1, 10 µmol/l XAV939. After 48 h of culture in treatment, the
colonies were fixed with 4% paraformaldehyde for 15 min at 25°C,
stained with 0.5% crystal violet solution for 20 min at 25°C, and
four fields of view were counted. The level of colony formation was
observed using an inverted light microscope.
Wound healing assay
A549 cells were seeded into 6-well plates at a
density of 1×105 cells/well. After 24 h, a sterilized
200 µl pipette tip was used to generate a wound across the cells.
Cells migrating into the wounded area were observed at different
time points (0 and 24 h) under an inverted light microscope at a
magnification of ×200. A total of 9 fields of view were randomly
analyzed in each well.
Reverse
transcription-semi-quantitative polymerase chain reaction
(RT-PCR)
A549 cells were treated with various concentrations
of XAV939 (0.1, 1 and 10 µmol/l) for 24 h. Total RNA was extracted
from A549 cells using TRIzol (Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. cDNA was generated using
gene-specific random hexamers (Sangon Biotech Co., Ltd, Shanghai,
China) (Table I) and RT kit (Tiangen
Biotech Co., Ltd., Beijing, China). The PCR reaction conditions
were as follows: 95°C for 5 min followed by 40 cycles of 95°C for
30 sec, 60°C for 30 sec, 72°C for 30 sec and a final extension step
of 72°C for 5 min. Samples were then left at 4°C until further use.
Agarose gel (1.5%) was used to run the PCR product which scanned
pictures using a gel imaging system (Fluor Corporation, Irving, TX,
USA). All data were normalized to endogenous β-actin, which used as
an endogenous control. Image Pro Plus 6.0 software (Media
Cybernetics, Inc., Rockville, MD, USA) was applied to analyze the
band intensity.
 | Table I.Polymerase chain reaction primers
used in this study. |
Table I.
Polymerase chain reaction primers
used in this study.
| Gene | Sequence,
5′-3′ | Product size,
bp |
|---|
| β-catenin |
| 104 |
|
Forward |
ATCATTCTGGCCAGTGCTGG |
|
|
Reverse |
GACAGCACCTTCAGCACTCT |
|
| c-Myc |
| 110 |
|
Forward |
GAGTCAGGGTCATCCCCATCA |
|
|
Reverse |
CCAAGACGTTGTGTGTCCGC |
|
| β-actin |
| 115 |
|
Forward |
TCTGTGTGGATTGGTGGCTCT |
|
|
Reverse |
AGAAGCATTTGCGGTGCAC |
|
Immunofluorescence
A549 cells were cultured in 24-well plates for 24 h.
Once they had adhered to glass slides at 80% density, cells were
divided into four groups: The control group (treated with NaCl),
and the 0.1, 1 and 10 µmol/l XAV939 treatment groups. After 48 h of
treatment, cells were fixed with 4% paraformaldehyde for 15 min at
25°C. Cells were incubated with 0.5% Triton X-100 (PBS compound)
for 20 min at 25°C and blocked using goat isolated serum (cat no.
ZLI-9022; OriGene Technologies, Inc.), for 30 min at 25°C. Then,
cells were incubated with a monoclonal antibody against β-catenin
(1:100), at 4°C overnight. The following day, a FITC-conjugated
goat anti mouse IgG (1:100; cat no. ZF0312; OriGene Technologies,
Inc.) secondary antibody was added, and cells were incubated in the
dark for 60 min at 37°C. DAPI was used to counterstain the nuclei
for 5 min at 25°C. Positive and negative control groups were also
prepared and images were captured using a fluorescence microscope
(at a magnification of ×200).
Western blot analysis
A total of 21 pairs of lung adenocarcinoma samples
and adjacent normal lung tissues were lysed in SDS lysis buffer.
(Beyotime Institute of Biotechnology, Shanghai, China). The protein
concentration was measured using a BCA Protein Assay kit (Beyotime
Institute of Biotechnology). A total of 40 µg protein lysates were
separated by SDS-PAGE (10%) and transferred by electrophoresis to
polyvinylidene difluoride (PVDF) membranes. The membranes were
blocked with 5% non-fat milk for 2 h at room temperature and
incubated overnight at 4°C with primary antibodies. The target
proteins were detected by western blot analysis with the following
primary antibodies: Mouse anti-human TNKS monoclonal antibody
(1:400), β-actin antibody (1:2,000) overnight at 4°C. After washing
with PBS-Tween-20 (PBST), membranes were incubated with goat
anti-mouse horseradish peroxidase-conjugated polyclonal secondary
antibodies (cat no. ZDR5307, 1:1,000; OriGene Technologies, Inc.)
for 2 h at 25°C. Proteins were detected using an enhanced
chemiluminescence kit (Tanon Science and Technology Co., Ltd.,
Shanghai, China). All quantitative data were normalized to β-actin,
which used as an endogenous control. Image Pro Plus 6.0 software
(Media Cybernetics, Inc., Rockville, MD, USA) was applied to
analyze the band intensity.
A549, Calu-3 and SK-LU-1 cells were lysed with
ice-cold RIPA buffer (Beyotime Institute of Biotechnology,
Shanghai, China), and protein concentrations were determined by BCA
method. Equal amounts of 40 µg protein lysates were separated by
SDS-PAGE (10%) and then transferred to PVDF membranes. Membranes
were incubated overnight at 4°C with the following primary
antibodies: Anti-TNKS (1:400), and β-actin (1:2,000). After washing
with PBST, the corresponding concentrations of goat anti-mouse
IgG-FITC secondary antibody (cat no. ZDR5307; 1:1,000; OriGene
Technologies, Inc.) was joined for 2 h at 25°C. Proteins were
detected using an enhanced chemiluminescence kit (Tanon Science and
Technology Co., Ltd). All quantitative data were normalized to
β-actin, which was used as an endogenous control. Image Pro Plus
6.0 software was applied to analyze the band intensity.
A549 cells were treated with DDP (0.002 g/l;
approval no. H21020212; Jinzhou Jiutai Pharmaceutical Co., Ltd.,
Jinzhou, China), 1 µmol/l XAV939, 0.002 g/l DDP and 1 µmol/l XAV939
for 24 h. Then the cells lysed with ice-cold RIPA buffer (Beyotime
Institute of Biotechnology), and protein concentrations were
determined by BCA method. Equal amounts of 40 µg protein lysates
were separated by SDS-PAGE (10%) and then transferred to PVDF
membranes. Membranes were incubated overnight at 4°C with the
following primary antibodies: Anti-TNKS (1:400), anti-β-catenin
(1:500), anti-c-Myc (1:500) and β-actin (1:2,000). After washing
with PBST, the corresponding concentrations of goat anti-mouse
IgG-FITC secondary antibody (cat no. ZDR5307; 1:1,000; OriGene
Technologies, Inc.) was joined for 2 h at 25°C. Proteins were
detected using an enhanced chemiluminescence kit (Tanon Science and
Technology Co., Ltd). All quantitative data were normalized to
β-actin, which was used as an endogenous control. Image Pro Plus
6.0 software was applied to analyze the band intensity.
Statistical analysis
Data are presented as the mean ± standard deviation
and all statistical analyses were performed using SPSS 17.0
software (SPSS, Inc., Chicago, IL, USA). Data counts in all cases
were expressed as a percentage; a χ2 test was performed
to compare between groups. The correlation between TNKS, β-catenin,
and c-Myc expression were evaluated using the Spearman correlation
coefficient. Comparative Student's unpaired t-test was used to
assess the statistical significance between two groups. One-way
analysis of variance was performed to statistically compare two or
more groups. The post-hoc test used was Student-Newman-Keuls (SNK).
P<0.05 was considered to indicate a statistically significant
difference.
Results
Expression of TNKS, β-catenin, and
c-Myc in lung adenocarcinoma and adjacent carcinoma tissues
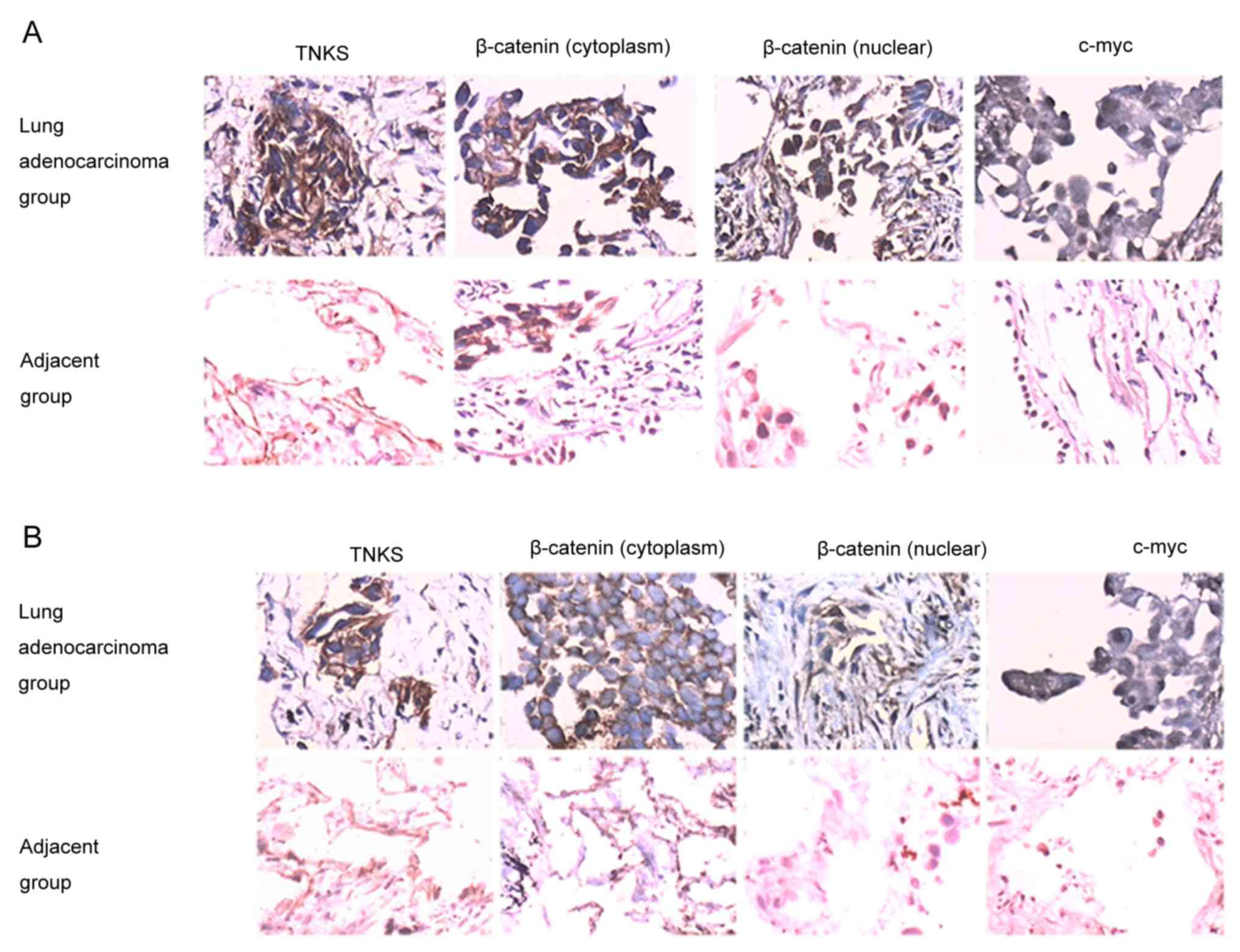
TNKS-positive immunostaining was identified by brown
staining in the cytoplasm of tumor cells (Fig. 1A and B). β-catenin was mainly found in
the cytoplasm and nucleus of the lung adenocarcinoma group, whereas
it was expressed only in the cytoplasm of the paired adjacent
carcinoma group (Fig. 1A and B).
c-Myc was predominantly observed in the nucleus of cells belonging
to the lung adenocarcinoma group (Fig. 1A
and B). The positive expression of TNKS, c-Myc and β-catenin
was significantly increased in the lung adenocarcinoma group
compared with the paired adjacent group (P<0.05; Fig. 1 and Table
II). The correlation between TNKS expression and other
WNT-associated proteins was further investigated by comparing
changes in TNKS expression levels to those of β-catenin and c-Myc,
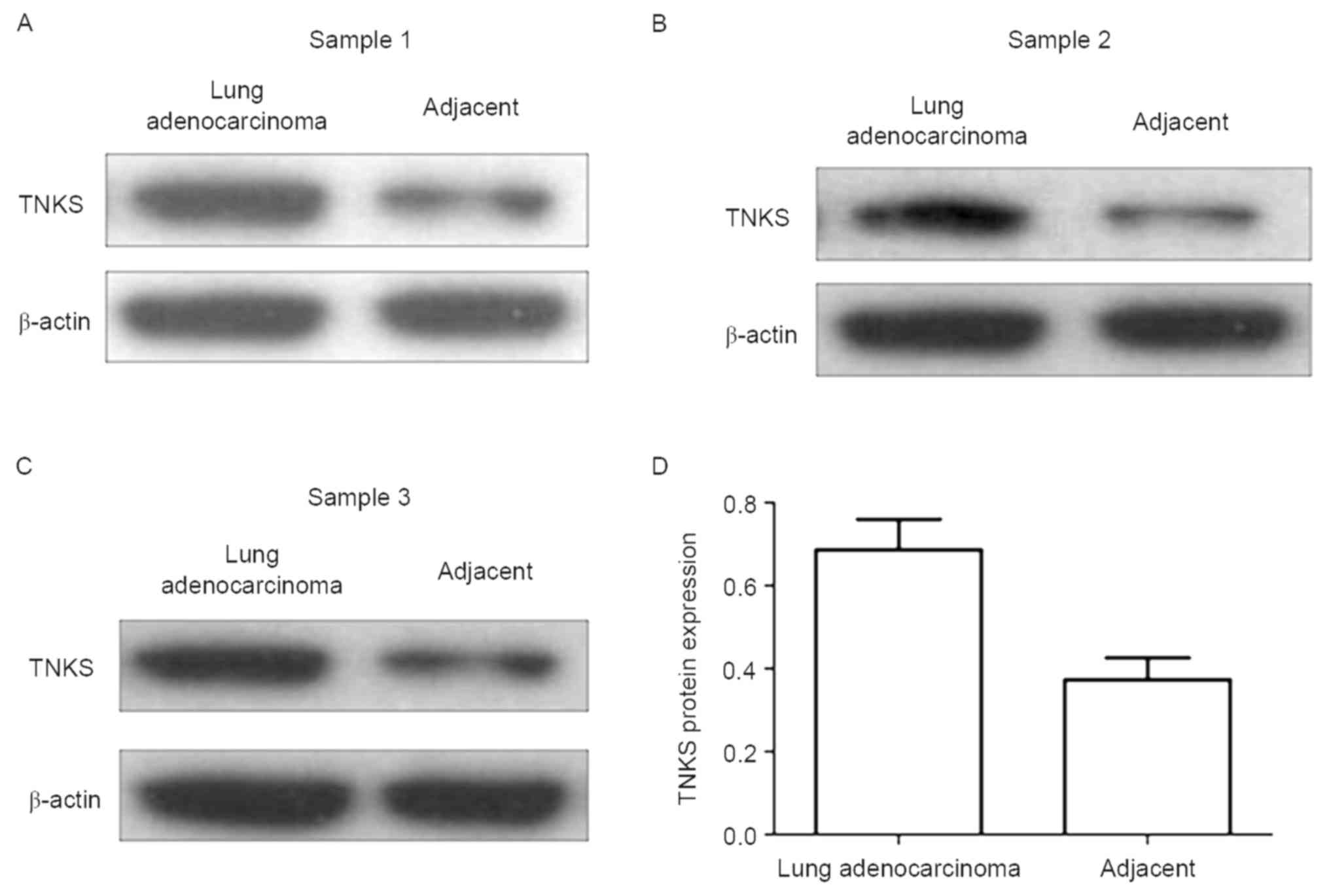
revealing a significant positive value (Table III). Western blot analysis was
conducted to detect relative TNKS expression levels in 21 lung
adenocarcinoma and paired adjacent carcinoma tissues.
Representative blots of three paired samples demonstrate that TNKS
expression is significantly higher in the lung adenocarcinoma group
than in the paired adjacent carcinoma group (P<0.05; Fig. 2).
 | Table II.Expression of TNKS, β-catenin and
c-Myc in lung adenocarcinoma and adjacent group. |
Table II.
Expression of TNKS, β-catenin and
c-Myc in lung adenocarcinoma and adjacent group.
|
|
|
| β-catenin
expression |
|
|---|
|
|
|
|
|
|
|---|
| Group | Total patients | TNKS, n (%) | Cytoplasm | Nuclear | Positive c-Myc
expression |
|---|
| Adenocarcinoma, n
(%) | 72 | 65 (90.28) | 64 (88.89) | 51 (70.83) | 48 (66.67) |
| Adjacent, n
(%) | 67 | 18 (26.86) | 20 (29.85) | 8 (11.94) | 9 (13.43) |
| χ2 |
| 58.01b | 50.588b | 49.273b | 40.66a |
 | Table III.Correlation between the expression of
β-catenin, TNKS, and c-Myc proteins in 72 cases of lung
adenocarcinoma. |
Table III.
Correlation between the expression of
β-catenin, TNKS, and c-Myc proteins in 72 cases of lung
adenocarcinoma.
|
| TNKS expression,
n |
| c-Myc expression,
n |
|
|---|
|
|
|
|
|
|
|---|
| Group | Positive | Negative | R-value | Positive | Negative | R-value |
|---|
| β-catenin
(cytoplasm) |
|
|
|
|
|
|
|
Negative expression | 5 | 3 | 0.682a | 1 | 7 | 0.814a |
|
Positive expression | 60 | 4 |
| 47 | 17 |
|
| β-catenin
(nucleus) |
|
|
|
|
|
|
|
Negative expression | 16 | 5 | 0.619a | 10 | 11 | 0.495a |
|
Positive expression | 49 | 2 |
| 38 | 13 |
|
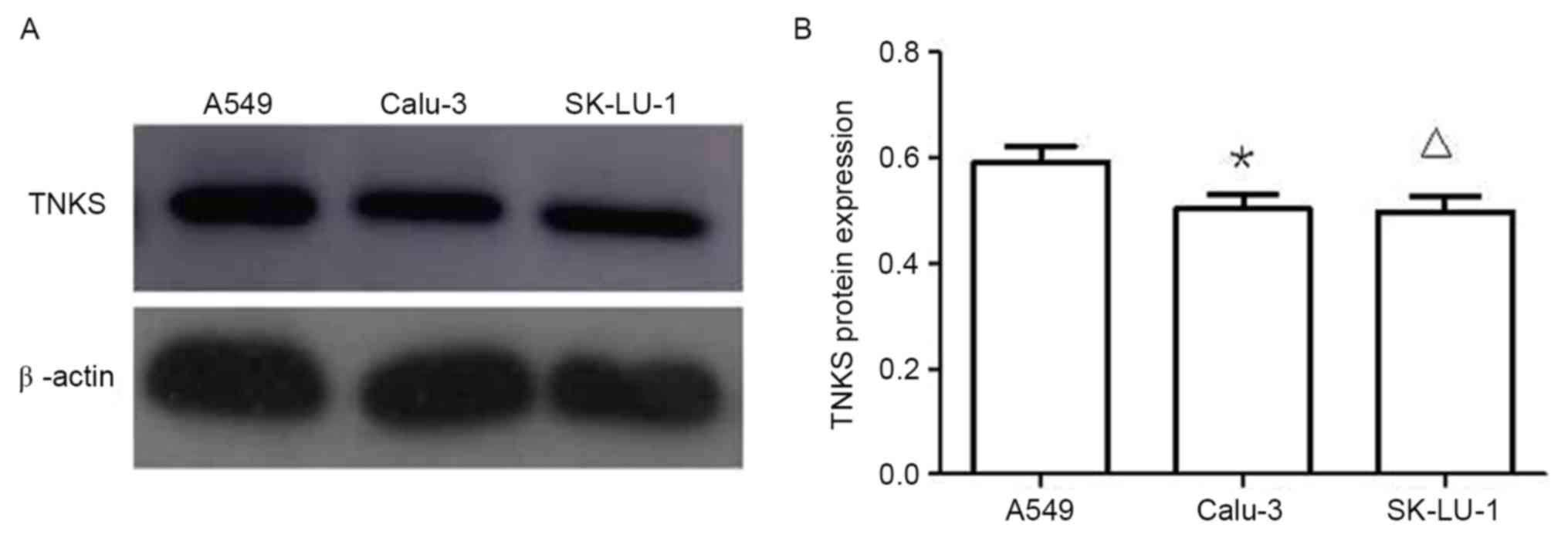
Protein expression of TNKS in three
lung adenocarcinoma cell lines
Western blotting results (Fig. 3) demonstrated that TNKS protein
expression levels in A549 cells were significantly higher than
those in Calu-3 and SK-LU-1 cells (P<0.05; Fig. 3). Owing to the higher TNKS protein
expression levels observed in A549 cells compared with Calu-3 and
SK-LU-1 cells, A549 cells were selected for subsequent
experiments.
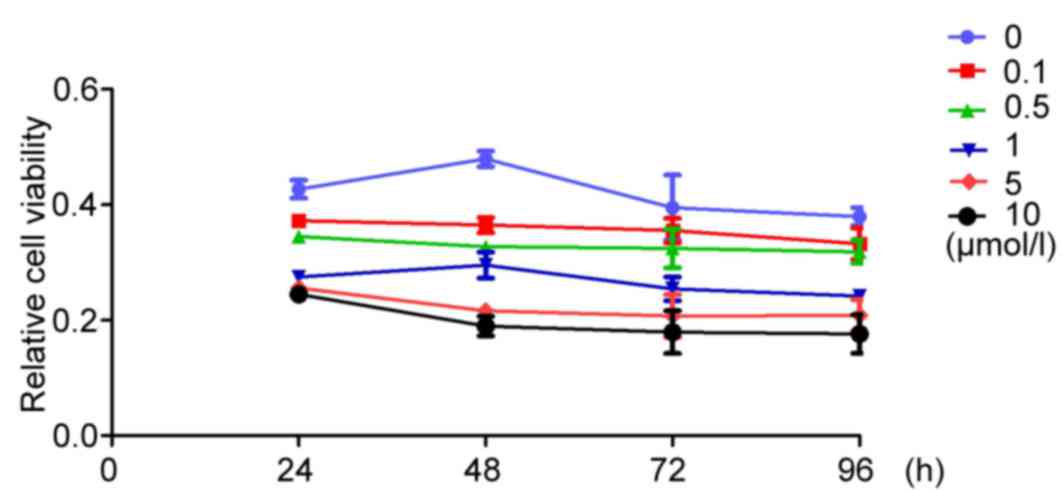
XAV939 inhibits the proliferation of
A549 cells in vitro
To analyze the inhibitory effects of XAV939 at
different concentrations (0.1, 0.5, 1, 5 and 10 µmol/l) on A549
cell proliferation, an MTT assay was performed at specific time
points (24, 48, 72 and 96 h). At all experimental time points,
XAV939 treatment was able to significantly inhibit A549 cell
proliferation compared with the control group
(F24h=30.382, F48h=52.463,
F72h=56.635, F96h=59.274; P<0.05), with
the exception of the 5-and 10-µmol/l groups at the 24 h time point
(P=0.147).
XAV939 inhibits the proliferation of
A549
Fig. 4 presents the
values of inhibition of A490 proliferation at different XAV939
concentrations (0.1, 0.5, 1, 5 and 10 µmol/l) and different times
(24, 48, 72 and 96 h). Between groups treated with the same
concentration of XAV939, the differences were statistically
significant at the different time points tested (P<0.05), except
the 0.1 µmol/l XAV939 treatment group.
The SNK test revealed that the differences were
statistically significant at the different time points (24 vs. 48
h, 24 vs. 72 h, 24 vs. 96 h, 48 vs. 72 h and 72 vs. 96 h) at the
same concentration of XAV939.
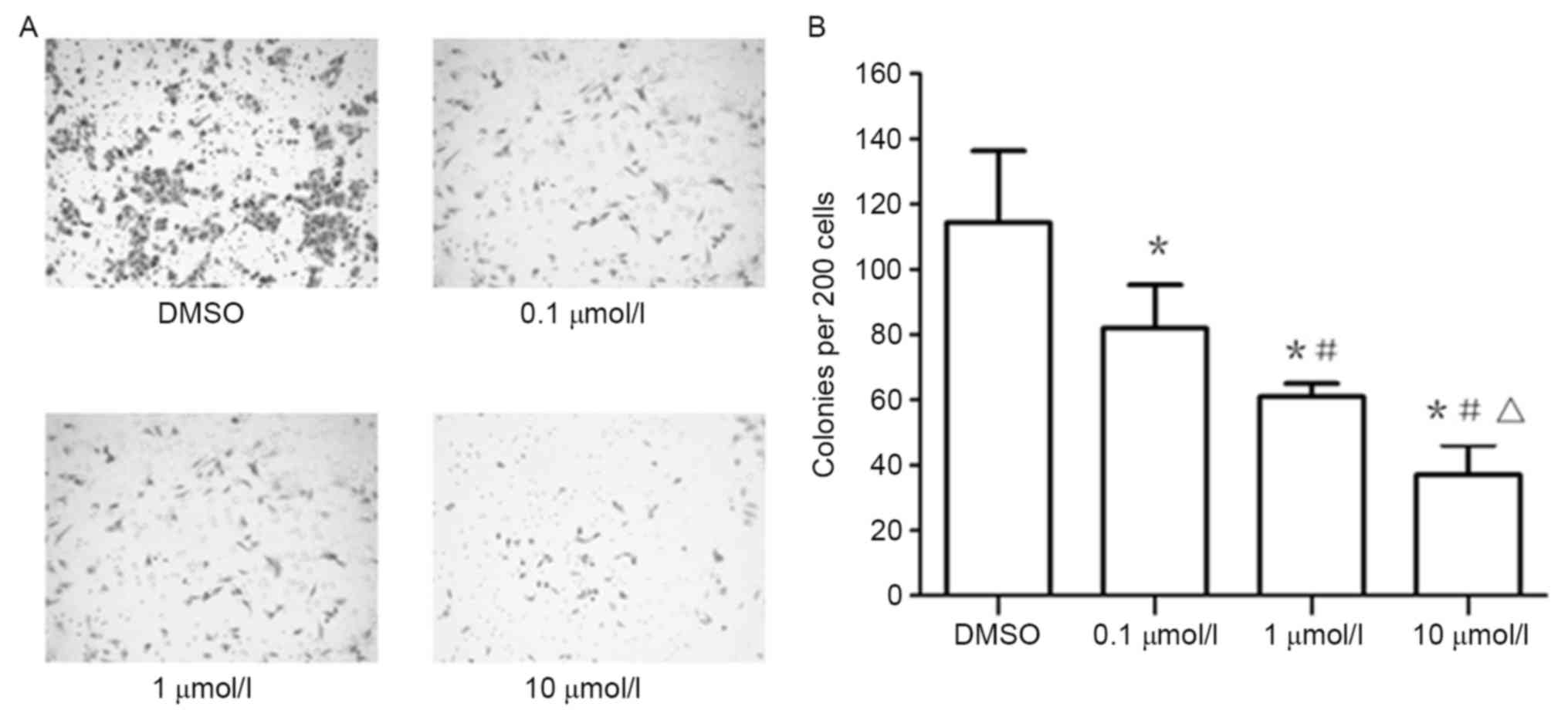
XAV939 inhibits the clonogenicity of
A549 cells
Treatment of the A549 cell line, plated under colony
forming conditions, with 0.1, 1 and 10 µmol/l of XAV939,
significantly inhibited colony formation ability compared with
cells under control treatment, as shown with colony number
quantification (Fig. 5).
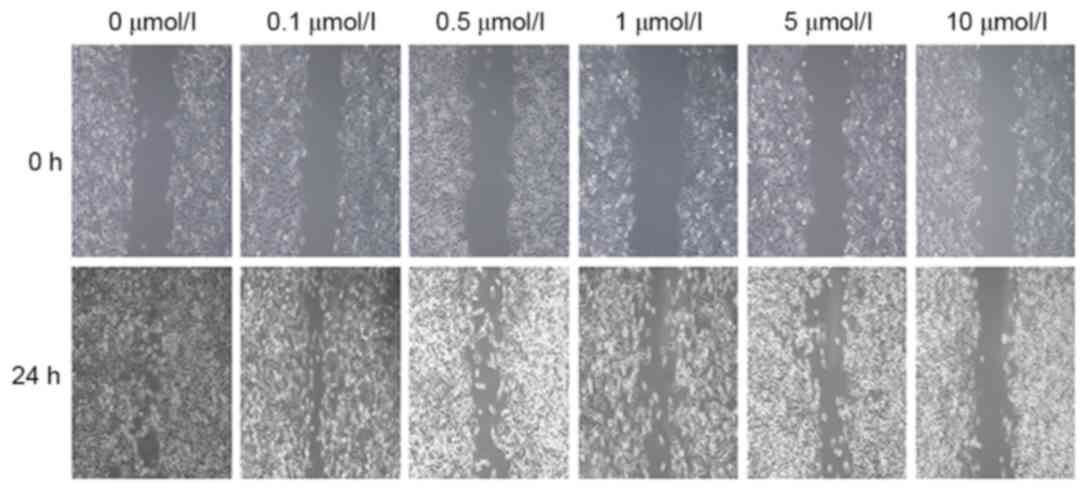
XAV939 inhibits the migration of A549
cells in vitro
To assess whether XAV939 treatment inhibited A549
cell migration in vitro, the results of the wound-healing
assay (Fig. 6) demonstrated that the
wound width in groups treated with 0.1, 0.5, 1, 5 and 10 µmol/l
XAV939 was 165.8±12.3, 176.6±11.9, 267.4±13.5, 328.7±18.1 and
445.4±21.6 µm, respectively.
These gaps were all significantly wider than that in
the control group (106.4±10.5 µm, P<0.5), demonstrating that the
cell migratory ability was decreased. When comparisons within
different treatment groups were performed, the differences were
statistically significant (F=786.294; P<0.05) other than for the
0.1 and 0.5 µmol/l groups (P>0.05). These results revealed that
XAV939 was able to inhibit the migration of A549 cells in a
concentration-dependent manner.
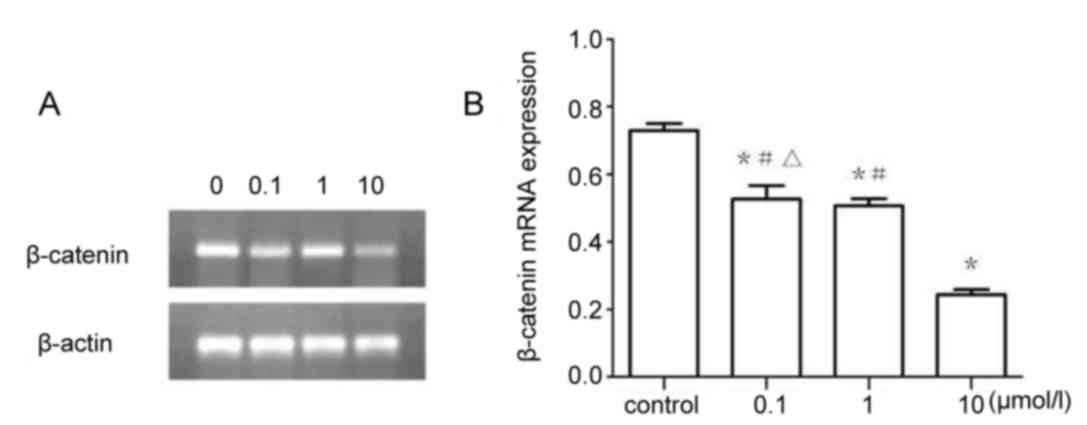
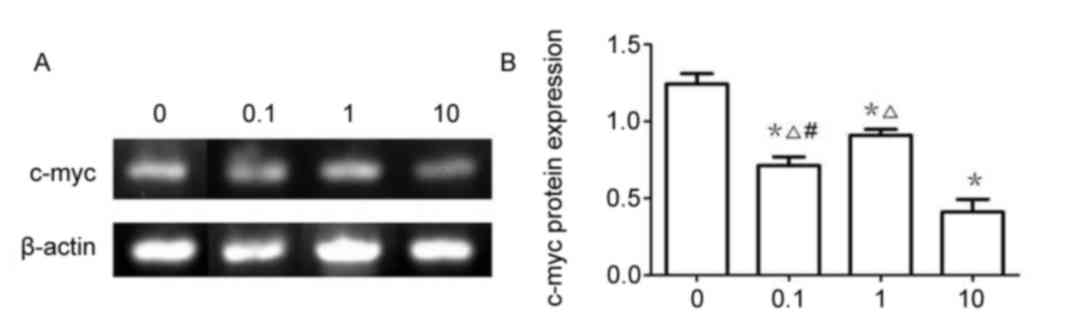
β-catenin mRNA expression
The results of RT-sqPCR demonstrated that β-catenin
(Fig. 7) and c-Myc (Fig. 8) mRNA expression were significantly
downregulated in 0.1, 1 and 10 µmol/l XAV939 treatment groups
compared with the control group (P<0.05). The expression of
c-Myc was demonstrated to be significantly different between
treatment groups, 0.1, 1 and the 10 µmol/l group (P<0.05).
However, the differences in β-catenin expression were not
statistically significant between the 0.1 and the 1 µmol/l groups
(P>0.05).
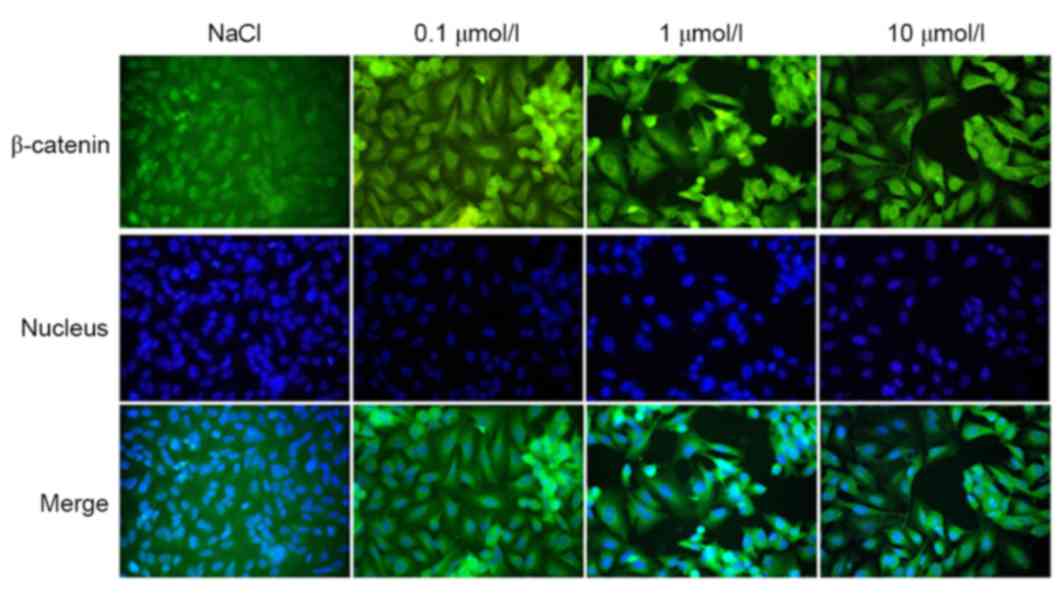
Immunofluorescence
The localization of β-catenin expression, as
observed by immunofluorescence, revealed that the protein had a
tendency to translocate to the cytoplasm/membrane from the
cytoplasm/nucleus (Fig. 9); this
translocation was directly proportional to the concentration of
XAV939 used.
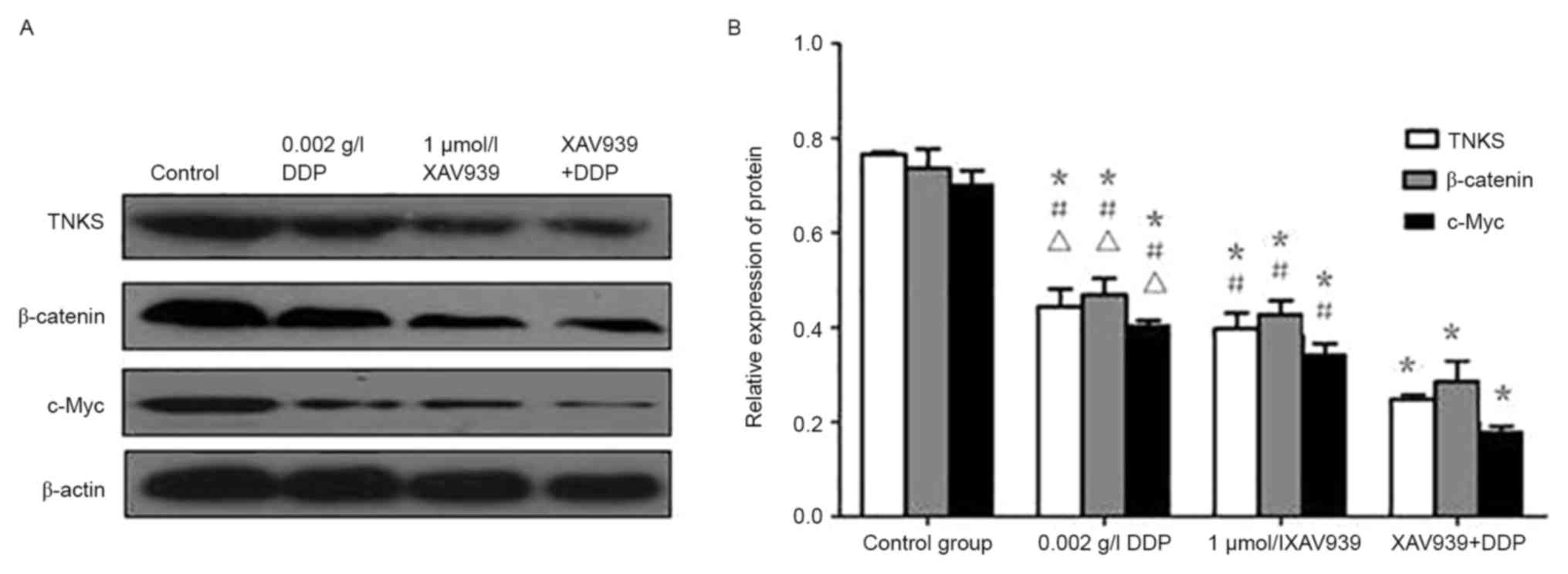
Western blotting
Analysis by western blotting (Fig. 10) demonstrated that the level of
β-catenin, TNKS, and c-Myc protein expression in A549 cells treated
with XAV939 or DDP was significantly reduced compared with the
control group (P<0.05). However, the combined effect of the two
drugs further attenuated the inhibitory effect of each drug when
used alone (P<0.05).
Discussion
The majority of patients with lung cancer frequently
develop tumor metastasis and relapse shortly following surgery,
radiotherapy, or chemotherapy (13).
The 5-year relative survival rate is only 17% (13). WNT/β-catenin signaling is part of a
functional network present in the first metazoans and is implicated
in a broad range of biological functions, which are fundamental in
stem cells, embryonic development and adult organs (14). Deregulation of components of the
WNT/β-catenin signaling pathway has been implicated in a wide
spectrum of diseases, including several types of cancer (14). In lung adenocarcinoma, the frequently
upregulated WNT/β-catenin signaling pathway represents a promising
target for molecular therapies. Currently, multiple key factors
belonging to the WNT/β-catenin signaling cascade represent
potential therapeutic targets (15).
Previous research has confirmed that multiple physiological and
pathological processes that are activated by WNT signals are
regulated by TNKS and its corresponding inhibitors (8,16,17).
The data from the present study revealed that TNKS
and β-catenin expression was increased in lung adenocarcinoma
tissue, and were also positively correlated. However, β-catenin is
a target of the WNT signaling pathway, and we hypothesize that
enhanced TNKS expression levels are associated with the abnormal
activation of the WNT signaling pathway. XAV939 is a small molecule
that selectively inhibits TNKS, and also inhibits the transcription
of β-catenin. Using XAV939 to inhibit the WNT pathway is a
therapeutic strategy used for the treatment of several types of
cancer (18–20). In the breast cancer MDA-MB-231 cell
line, inhibition of TNKS1 was revealed to weaken cell migration,
induced by WNT3a (11). Tian et
al (21) demonstrated that TNKS1
inhibition may, in part, block the WNT/β-catenin signaling cascade
and reduce the expression of anti-apoptotic proteins. In addition,
it was further demonstrated that TNKS1 inhibition decreased in
vitro colony formation. De la Roche et al (22) demonstrated that TNKS inhibitors also
inhibited WNT-induced transcription, and reduced β-catenin
transcriptional activity in colorectal cancer cells. Busch et
al (7) revealed that knockdown of
TNKS1 and/or TNKS2 with small interfering RNAs or short hairpin
RNAs reduced lung cancer cell proliferation and repressed tumor
formation in murine xenograft and syngeneic lung cancer models. The
present study provides initial evidence to support the notion that
TNKS enzymes, in association with WNT/β-catenin signaling
components, are feasible targets for lung adenocarcinoma
treatment.
The data from the present study indicated that high
TNKS expression was more frequently detected in the lung
adenocarcinoma group (90.28%) than in the adjacent to carcinoma
group (26.86%). In addition, TNKS expression level was demonstrated
to be significantly higher in tumor cells in the lung
adenocarcinoma group compared with the control group, indicating
that TNKS in lung adenocarcinoma is highly expressed. Aberrant WNT
signaling caused by mutations in the β-catenin gene, a key
regulator of the canonical WNT-signaling pathway, is frequently
detected in lung cancer (23,24). However, the mechanisms underlying the
aberrant activation of WNT/β-catenin signaling in lung cancer have
not yet been fully characterized.
In the present study, lung adenocarcinoma cancer
cells were observed to consistently express high levels of nuclear
and cytoplasmic β-catenin protein (70.83 and 88.89%, respectively).
By contrast, β-catenin was predominantly expressed in the
cytoplasm, with a small proportion in the nucleus in the
adjacent-to-carcinoma group (29.85 and 11.94%, respectively). The
correlation between TNKS and WNT/β-catenin signal was confirmed by
comparing the level of β-catenin (nuclear) expression. (TNKS vs.
β-catenin, r=0.619, P<0.05; c-Myc vs. β-catenin, r=0.495,
P<0.05). β-catenin is an oncoprotein that is normally localized
in the cytoplasm (18). The results
of the present study revealed that oncogenic WNT signals promote
the cytosolic accumulation of β-catenin and nuclear translocation,
leading to the activation of downstream targets, including the
oncogene c-Myc, which is consistent with a previous report by Zhang
et al (25). TNKS inhibition
attenuates WNT/β-catenin signaling by promoting dynamic assemblies
of functional active destruction complexes into a TNKS-containing
scaffold, even in the presence of an adenomatous polyposis coli
(APC) truncation (26). Wang et
al (9) demonstrated that XAV939
significantly inhibited the activation of WNT/β-catenin signaling
and attenuated bleomycin-induced lung fibrosis in mice, thus
improving the survival of mice with lung injury.
Using cell viability and colony formation assays, it
was revealed that XAV939 inhibited the proliferation and colony
formation ability of A549 cells in vitro. The results of the
wound-healing assay revealed the inhibitory effect of treatment
with XAV939 on the migration of A549 cells in a dose-dependent
manner. Taken together, these results demonstrated that XAV939
inhibited the proliferative, colony formation and migratory
capacity of A549 cells.
β-catenin is a key molecule in the WNT pathway; its
stable accumulation in the cytoplasm and translocation into the
nucleus are critical events in this pathway. In normal cells,
β-catenin is mainly located in the membrane. Once β-catenin
accumulates in the cytoplasm, it is be translocated into the
nucleus and binds with T-cell factor/lymphoid enhancer factor to
induce transcription factors responsible for the transcription of
target genes involved in the activation and regulation of cell
proliferation and differentiation (27). Thus, the abnormal activation of the
WNT pathway induces tumorigenesis (28). The RT-sqPCR data demonstrated that,
compared with the control group, β-catenin and c-Myc expression in
XAV939 treatment groups (0.1, 1 and 10 µmol/l) clearly decreased.
Additionally, following treatment with 10 µmol/l XAV939 for 24 h,
the expression of β-catenin mRNA was significantly lower than that
of other treatment groups. Immunofluorescence analysis demonstrated
that β-catenin had a tendency to translocate from the
cytoplasm/nucleus to the cytoplasm/membrane, depending on the
dose-dependent groups (0, 0.1, 1 and 10 µmol/l). Therefore, it was
concluded that XAV939 might induce β-catenin degradation and
inhibit its accumulation in the cytoplasm, leading to a weakening
or inhibition of the abnormal activation of the WNT signaling
pathway. This may potentially be a mechanism through which XAV939
inhibits A549 cell proliferation and migration.
DDP is used as a first-line drug for the treatment
of advanced non-small cell lung cancer. However, the drug is
usually prescribed at a low dosage owing to its toxicity. In the
present study, the expression level of β-catenin, TNKS and c-Myc
proteins was detected by western blotting. A549 cells treated with
XAV939 or DDP exhibited decreased the expression of TNKS protein
compared with untreated control cells. c-Myc, a downstream target
of β-catenin, was also downregulated. In addition, the effect of
combined XAV939 and DDP treatment decreased the expression of TNKS,
β-catenin and c-Myc protein to a greater degree than when either
drug was used alone. This result highlighted the potential utility
of combination therapy in increasing the attenuation of the
WNT/β-catenin signaling pathway. The results of the present study
indicate that XAV939 could be considered as a novel therapeutic
agent for the treatment of lung cancer. XAV939 may also improve the
curative effect of DDP and reduce adverse reactions.
In summary, the present study revealed that TNKS and
β-catenin expression levels were increased in lung adenocarcinoma
tissue and were positively correlated with each other. Thus, it can
be concluded that the enhanced expression level of TNKS is
associated with the abnormal activation of the WNT signaling
pathway. The WNT/β-catenin pathway is considered a particularly
difficult target for molecular interventions owing to its numerous
non-evident enzyme targets, which may disrupt necessary and
beneficial biological processes (29). To the best of our knowledge, this is
the first study to reveal the effect of the TNKS inhibitor XAV939
on the WNT/β-catenin signaling pathway in lung adenocarcinoma; TNKS
inhibitors should thereby reduce β-catenin levels and activity. As
such, TNKS enzymes represent promising candidate targets for
anticancer molecular therapies (30).
The present study provides experimental evidence for the basis of
novel therapeutic strategies for the clinical treatment of lung
cancer. Future studies are required to delineate further this
mechanism and its role in suppressing lung tumor progression in
vivo.
Acknowledgements
The authors would like to thank Dr Shiwu Zhang
(Tianjin People's Hospital, Tianjin, China) for assistance with the
experiments and valuable discussion.
Funding
The present study was supported by National Natural
Science Foundation (grant no. 81774054).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CL contributed substantially to the conception and
design of the study, and acquisition of data. XZ performed the
immunohistochemical experiments and was a major contributor in
writing the manuscript. YH performed the western blot analysis,
reverse transcription-polymerase chain reaction examination and
analysis and interpretation of data. YL was responsible for
pathological diagnosis and image analysis. FL contributed to cell
culture and data collection. JZ produced the paraffin sections and
performed some cellular functional tests. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The use of the tissue samples for this study was
approved by the First Teaching Hospital of Tianjin University of
Traditional Chinese Medicine Medical Ethics Committee (Tianjin,
China). Ethical lot number: TYLL2018 [K] character005.
Consent for publication
Identifying information, including names, initials,
date of birth or hospital numbers, images or statements were not
included in the present study.
Competing interests
The authors have declared that there are no
competing interests.
References
|
1
|
Zahir ST and Mirtalebi M: Survival of
patients with lung cancer, Yazd, Iran. Asian Pac J Cancer Prev.
13:4387–4391. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cancer Genome Atlas Research Networ:
Comprehensive molecular profiling of lung adenocarcinoma. Nature.
511:543–550. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liu ZL, Jin BJ, Cheng CG, Zhang FX, Wang
SW, Wang Y and Wu B: Apatinib resensitizes cisplatin-resistant
non-small cell lung carcinoma A549 cell through reversing multidrug
resistance and suppressing ERK signaling pathway. Eur Rev Med
Pharmacol Sci. 21:5370–5377. 2017.PubMed/NCBI
|
|
4
|
Holland JD, Klaus A, Garratt AN and
Birchmeier W: Wnt signaling in stem and cancer stem cells. Curr
Opin Cell Biol. 25:855–863. 2013. View Article : Google Scholar
|
|
5
|
Anastas JN and Moon RT: WNT signalling
pathways as therapeutic targets in cancer. Nat Rev Cancer.
13:11–26. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
McGonigle S, Chen Z, Wu J, Chang P,
Kolber-Simonds D, Ackermann K, Twine NC, Shie JL, Miu JT, Huang KC,
et al: E7449: A dual inhibitor of PARP1/2 and tankyrase1/2 inhibits
growth of DNA repair deficient tumors and antagonizes Wnt
signaling. Oncotarget. 6:41307–41323. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Busch AM, Johnson KC, Stan RV, Sanglikar
A, Ahmed Y, Dmitrovsky E and Freemantle SJ: Evidence for tankyrases
as antineoplastic targets in lung cancer. Bmc Cancer. 13:2112013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kulak O, Chen H, Holohan B, Wu X, He H,
Borek D, Otwinowski Z, Yamaguchi K, Garofalo LA, Ma Z, et al:
Disruption of Wnt/β-catenin signaling and telomeric shortening are
inextricable consequences of tankyrase inhibition in human cells.
Mol Cell Biol. 35:2425–2435. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang C, Zhu H, Sun Z, Xiang Z, Ge Y, Ni C,
Luo Z, Qian W and Han X: Inhibition of Wnt/β-catenin signaling
promotes epithelial differentiation of mesenchymal stem cells and
repairs bleomycin-induced lung injury. Am J Physiol Cell Physiol.
307:C234–C244. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kamal A, Riyaz S, Srivastava AK and Rahim
A: Tankyrase inhibitors as therapeutic targets for cancer. Curr Top
Med Chem. 14:1967–1976. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Casás-Selves M, Kim J, Zhang Z, Helfrich
BA, Gao D, Porter CC, Scarborough HA, Bunn PA Jr, Chan DC, Tan AC
and DeGregori J: Tankyrase and the canonical Wnt pathway protect
lung cancer cells from EGFR inhibition. Cancer Res. 72:4154–4164.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Arqués O, Chicote I, Puig I, Tenbaum SP,
Argilés G, Dienstmann R, Fernández N, Caratù G, Matito J,
Silberschmidt D, et al: Tankyrase inhibition blocks Wnt/β-catenin
pathway and reverts resistance to PI3K and AKT inhibitors in the
treatment of colorectal cancer. Clin Cancer Res. 22:644–656. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Voronkov A and Krauss S: Wnt/beta-catenin
signaling and small molecule inhibitors. Curr Pharm Des.
19:634–664. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Polakis P: Drugging Wnt signalling in
cancer. EMBO J. 31:2737–2746. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Huang SM, Mishina YM, Liu S, Cheung A,
Stegmeier F, Michaud GA, Charlat O, Wiellette E, Zhang Y, Wiessner
S, et al: Tankyrase inhibition stabilizes axin and antagonizes WNT
signaling. Nature. 461:614–620. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu X, Luo F, Li J, Zhong X and Liu K:
Tankyrase 1 inhibitior XAV939 increases chemosensitivity in colon
cancer cell lines via inhibition of the Wnt signaling pathway. Int
J Oncol. 48:1333–1340. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Clevers H and Nusse R: Wnt/β-catenin
signaling and disease. Cell. 149:1192–1205. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lecarpentier Y, Claes V, Duthoit G and
Hébert JL: Circadian rhythms, Wnt/beta-catenin pathway and PPAR
alpha/gamma profiles in diseases with primary or secondary cardiac
dysfunction. Front Physiol. 5:4292014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dregalla RC, Zhou J, Idate RR, Battaglia
CL, Liber HL and Bailey SM: Regulatory roles of tankyrase 1 at
telomeres and in DNA repair: Suppression of T-SCE and stabilization
of DNA-PKcs. Aging (Albany NY). 2:691–708. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tian XH, Hou WJ, Yan F, Fan J, Tong H, Bai
SL, Chen Q, Xu H and Li Y: XAV939, a tankyrase 1 inhibitior,
promotes cell apoptosis in neuroblastoma cell lines by inhibiting
Wnt/β-catenin signaling pathway. J Exp Clin Cancer Res. 32:1002013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
de la Roche M, Ibrahim AE, Mieszczanek J
and Bienz M: LEF1 and B9L shield β-catenin from inactivation by
Axin, desensitizing colorectal cancer cells to tankyrase
inhibitors. Cancer Res. 74:1495–1505. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen X, Meng J, Yue W, Yu J, Yang J, Yao Z
and Zhang L: Fibulin-3 suppresses Wnt/β-catenin signaling and lung
cancer invasion. Carcinogenesis. 35:1707–1716. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lim JH, Park JW and Chun YS: Human arrest
defective 1 acetylates and activates beta-catenin, promoting lung
cancer cell proliferation. Cancer Res. 66:10677–10682. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang X, Lou Y, Zheng X, Wang H, Sun J,
Dong Q and Han B: Wnt blockers inhibit the proliferation of lung
cancer stem cells. Drug Des Devel Ther. 9:2399–2407.
2015.PubMed/NCBI
|
|
26
|
Thorvaldsen TE, Pedersen NM, Wenzel EM,
Schultz SW, Brech A, Liestøl K, Waaler J, Krauss S and Stenmark H:
Structure, dynamics and functionality of tankyrase
inhibitor-induced degradasomes. Mol Cancer Res. 13:1487–1501. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Krishnamurthy N and Kurzrock R: Targeting
the Wnt/beta-catenin pathway in cancer: Update on effectors and
inhibitors. Cancer Treat Rev. 62:50–60. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang X, Lou Y, Zheng X, Wang H, Sun J,
Dong Q and Han B: Wnt blockers inhibit the proliferation of lung
cancer stem cells. Drug Des Devel Ther. 9:2399–2407.
2015.PubMed/NCBI
|
|
29
|
Kahn M: Can we safely target the WNT
pathway? Nat Rev Drug Discov. 13:513–532. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Riffell JL, Lord CJ and Ashworth A:
Tankyrase-targeted therapeutics: Expanding opportunities in the
PARP family. Nat Rev Drug Discov. 11:923–936. 2012. View Article : Google Scholar : PubMed/NCBI
|