Introduction
The results of several prospective randomized trials
have shown the benefits of laparoscopic surgery for localized
colorectal cancer in terms of postoperative outcomes, including
reduction of pain, earlier intestinal peristaltic recovery,
shortening of postoperative stay, cosmetic advantages, and
equivalent effectiveness on long-term cancer outcomes (1–4). However,
locally advanced colon cancers sometimes invade or adhere to
neighboring organs or structures, and en bloc multivisceral
resection is necessary. According to the European Association of
Endoscopic Surgery (EAES) consensus, potentially curative resection
of colon cancer suspected of invading the abdominal wall or
adjacent structures should be undertaken by open surgery (5). In the Society of American
Gastrointestinal and Endoscopic Surgeons (SAGES) guidelines, as
open approach is suggested if a laparoscopic en bloc resection
cannot be performed adequately for locally advanced adherent colon
tumors (6). Because of increased the
experience with laparoscopic approach for locally advanced colon
cancer, the opportunities for adopting laparoscopic surgery for
advanced colon cancer invading or adhering to neighboring organs or
structures are considered to be increasing. Several studies have
demonstrated that laparoscopic multivisceral resection for advanced
colorectal cancer was safe and feasible in selected patients
(7–9).
The first single-port surgery (SPS) procedure was
described for right colectomy in 2008 (10). Compared with conventional multi-port
surgery, the benefits of SPS include better cosmetic outcomes, less
postoperative pain (11–13), faster postoperative recovery (14), and earlier discharge from the hospital
(13,14). SPS for colorectal cancer was shown to
be safe, feasible (11,12,15), and
able to provide perioperative and short-term oncological outcomes
equal to those of multi-port surgery (16,17).
However, cases of locally advanced colon cancer invading or
adhering to neighboring organs were excluded in these studies
(11–17), and there is no study of SPS for
locally advanced left colon cancer invading or adhering to
neighboring organs. Therefore, in this study, we aimed to evaluate
the safety and feasibility of single-port laparoscopic
multivisceral resection (S-MVR) for locally advanced left colon
cancer invading or adhering to neighboring organs, based on a
retrospective comparison with multi-port laparoscopic multivisceral
resection (M-MVR).
Patients and methods
Patient profiles
Multivisceral resection was defined as en bloc
resection of any organ or structure to which the primary tumor was
adherent. The preoperative diagnosis of cancer was established with
a barium enema, a colonoscopy, and computed tomography for
evaluation of local disease and distant metastases. Locally
advanced colon cancer was diagnosed with preoperative computed
tomography when the tumor and surrounding structures were unclear.
In cases with unclear boundaries, we judged whether the tumor
invaded adjacent organs or structures during the intraoperative
period. Patients who had a diagnosis of Stage IV disease who
underwent macroscopic complete resection of the primary tumor were
included in this study, but those who underwent palliative
resection of the primary tumor were excluded.
In our department, the first SPS for benign disease
was carried out in May 2009. Before January 2010, SPS was performed
only for benign disease and early colon cancer only, but the
indication was expanded later to include advanced colorectal
cancer. SPS for locally advanced colon cancer invading or adhering
to neighboring organs has been performed since January 2011, and
since then, it has been considered the primary treatment for colon
cancer in those eligible for conventional multi-port surgery.
Between January 2008 and December 2014, a total of
38 patients underwent multivisceral resection for locally advanced
left colon cancer invading or adhering to neighboring organs at our
department. Among these patients, 9 patients underwent open surgery
because of an emergency operation (n=3), a tumor associated abscess
(n=2). In addition, we performed open surgery when urinary
diversion and total cystectomy were needed (n=4). In total, 29
consecutive patients who underwent S-MVR or M-MVR for locally
advanced left colon cancer with adjacent organs or structures in
this study. Patients received written information describing the
differences between S-MVR and M-MVR. All of the patients agreed to
receive S-MVR and provided written informed consent. The study was
approved by the Ethics Committee of Osaka Police Hospital.
Data collection
For each patient, the following variables were
collected from our prospectively maintained database; age, sex,
body mass index (BMI), American Society of Anesthesiology Score
(ASA score), previous abdominal surgery, tumor location, tumor
size, operative procedure, stoma construction, operative time,
blood loss, conversion rate, morbidity, mortality, length of
hospital stay, and pathological findings. Postoperative
complications were classified according to the Dindo et al
Classification (18). Operative
mortality was defined as death on the same admission or within 30
days of surgery. All patients were followed for at least 30 days
after surgery.
Surgical procedure
SPS was performed, as described previously (19). A flexible laparoscope 10 mm in
diameter and only standard straight laparoscopic instruments were
used. Every procedure was performed with an Ultracision harmonic
scalpel regardless of whether it was single or multi-port surgery.
High ligation of feeding vessels and en bloc resection of the
primary tumor with a safe margin around the adjacent organs or
structures were performed. For left colectomy, an extracorporeal
functional end-to-end anastomosis was then created. For
sigmoidectomy and high anterior resection, the sigmoid colon or
upper rectum was divided laparoscopically with staplers. An
end-to-end anastomosis was created with the trans-anal insertion of
a circular stapler.
Statistical analysis
Continuous data are expressed as median (range),
unless specifically noted. Preoperative factors, operative factors,
and pathological outcomes were analyzed retrospectively.
Statistical analyses were performed with the use of JMP 11.0
software (SAS Institute Inc., Cary, NC, USA). All group data are
reported as median values. Differences in age, BMI, tumor size,
operative time, blood loss, length of hospital stay, and number of
harvested lymph nodes were analyzed with Mann-Whitney's U test.
Other factors were analyzed with Fischer's exact probability test.
Disease-free survival and overall survival rate were determined
using Kaplan-Meier analysis with a log-rank test. All analyses were
performed on an intention-to-treat basis: patients who had S-MVR
converted to M-MVR were included in the S-MVR group. Patients who
had M-MVR converted to open surgery were included in the M-MVR
group. P<0.05 was considered to indicate a statistically
significant difference.
Results
Table I lists the
profiles of the patients analyzed by group. There were no
significant differences between the S-MVR and M-MVR groups in terms
of age, gender, BMI, ASA score, previous abdominal surgery, tumor
location and tumor size. None of the patients received neoadjuvant
chemotherapy or chemoradiotherapy in this study.
 | Table I.Patient profiles. |
Table I.
Patient profiles.
| Characteristic | S-MVR (n=14) | M-MVR (n=15) | P-value |
|---|
| Median age, years
(range) | 71 (36–89) | 69 (53–91) | 0.878 |
| Sex, n |
|
|
|
|
Male/female | 6/8 | 10/5 | 0.272 |
| Median BMI,
kg/m2 (range) | 21.5
(14.1–32.9) | 19.4
(12.5–30.5) | 0.176 |
| ASA score, n |
|
| 0.814 |
| 1 or
2/≥3 | 12/2 | 12/3 |
|
| Previous abdominal
surgery, n | 4 | 3 | 0.682 |
| Tumor location,
n |
|
| 0.330 |
|
Descending/sigmoid | 3/11 | 1/14 |
|
| Median tumor size, mm
(range) | 60
(25–110) | 70
(40–110) | 0.333 |
Table II summarizes
the operative factors. The conversion rate in the S-MVR group was
14.3% (2/14 patients). None of the patients who underwent S-MVR was
converted to open surgery. The reason for conversion to multi-port
surgery was strong inflammatory adhesion to the small intestine in
one patients and bladder invasion in one patient. In the M-MVR
group, 5 patients were converted to open surgery because of tumor
invasion of the small intestine in three, invasion of the right
ureter and the abdominal wall in one, and invasion of the uterus in
one. There were no significant differences between groups in terms
of operative procedure, stoma construction rate (14.3% in the S-MVR
group vs. 26.7% in the M-MVR group, P=0.651), operative time (222
min in the S-MVR group vs. 255 min in the M-MVR group, P=0.163),
blood loss (60 ml in the S-MVR group vs. 220 ml in the M-MVR group,
P=0.295), and the median number of harvested lymph nodes (30 in the
S-MVR group vs. 25 in the M-MVR group, P=0.328).
 | Table II.Comparison of operative factors. |
Table II.
Comparison of operative factors.
| Factors | S-MVR (n=14) | M-MVR (n=15) | P-value |
|---|
| Operative procedure,
n |
|
| 0.591 |
| Left
hemicolectomy | 3 | 1 |
|
|
Sigmoidectomy | 8 | 9 |
|
| High
anterior resection | 3 | 5 |
|
| Stoma construction,
n | 2 | 4 | 0.651 |
| Median operative
time, min (range) | 222
(129–323) | 255
(174–395) | 0.163 |
| Median blood loss, ml
(range) | 60 (5–600) | 220 (5–2670) | 0.295 |
| Median number of
harvested lymph nodes, (range) | 30 (17–58) | 25 (10–72) | 0.328 |
| Conversion to MPS,
n | 2 | – | – |
| Conversion to open
surgery, n | 0 | 5 | – |
Table III lists the
adjacent organs or structures resected en bloc. The most commonly
affected organs were the small intestine (28.6% in the S-MVR group
and 46.7% in the M-MVR group). The number of resected organs or
structures was similar between the two groups (P=1.000).
 | Table III.Organs or structures resected at
multivisceral resection. |
Table III.
Organs or structures resected at
multivisceral resection.
| Organ or
structure | S-MVR (n=14) | M-MVR (n=15) | P-value |
|---|
| Small intestine | 4 | 7 | 0.450 |
| Abdominal wall | 3 | 2 | 0.651 |
| Urinary bladder | 3 | 2 | 0.651 |
| Ovary | 3 | 1 | 0.330 |
| Appendix | 1 | 1 | 0.776 |
| Seminal
vesicle | 1 | 0 | 0.483 |
| Uterus | 1 | 2 | 0.527 |
| Ureter | 0 | 1 | 0.517 |
| Total | 17 | 18 |
|
| Number of resected
organs or structure |
|
|
|
|
1/2/ | 11/3 | 12/3 | 0.709 |
Table IV summarizes
the postoperative complications in each group. The overall
complication rate was 28.6% in the S-MVR group. There were no
significant differences between groups in terms of postoperative
complication. Length of hospital stay was significantly shorter
with S-MVR than with M-MVR (11 vs. 18 days, P=0.048). Reoperation
was performed in one patient in the M-MVR group because of
anastomotic leakage. Perioperative death was not recorded in both
groups.
 | Table IV.Postoperative complications. |
Table IV.
Postoperative complications.
| Complication | S-MVR (n=14) | M-MVR (n=15) | P-value |
|---|
| Bleeding | 1 | 0 | 0.483 |
| Anastomotic
leakage | 0 | 1 | 0517 |
| Wound
infection | 2 | 3 | 0.814 |
| Abdominal
abscess | 2 | 1 | 0.598 |
| Pneumonia | 0 | 1 | 0.517 |
| Urinary tract
infection | 1 | 2 | 0.527 |
| Bowel
obstruction | 0 | 2 | 0.483 |
| Colitis | 1 | 0 | 0.483 |
| Reoperation | 0 | 1 | 0.517 |
| Perioperative
death | 0 | 0 | – |
| Overall
complications | 4 | 7 | 0.450 |
| Length of hospital
stay, days (range) | 11 (7–97) | 18 (8–90) | 0.048 |
Table V summarizes the
pathological findings for each group. The R0 resection rate of the
primary tumor was similar between groups (100% in the S-MVR group
and 93.3% in the M-MVR group). The rate of pathological T4b disease
was 35.7% in the S-MVR group and 33.3% in the M-MVR group
(P=1.000). There were no significant differences between groups in
terms of the grade of differentiation, pathological lymph node
metastasis, and TNM classification.
 | Table V.Pathological outcomes. |
Table V.
Pathological outcomes.
| Outcome | S-MVR (n=14) | M-MVR (n=15) | P-value |
|---|
| Residual tumor
status of the primary tumor |
|
| 0.517 |
|
R0/R1 | 14/0 | 13/1 |
|
| Grade of
differentiation, n |
|
| 0.814 |
| Well or
moderate/poor or mucinous | 12/2 | 11/3 |
|
| Depth of tumor
invasion, n |
|
| 0.992 |
| T3 | 2 | 1 |
|
|
T4a | 7 | 9 |
|
|
T4b | 5 | 5 |
|
| Lymph node
metastasis, n |
|
| 0.449 |
| N0 | 5 | 9 |
|
| N1 | 4 | 3 |
|
| N2 | 5 | 3 |
|
| TNM classification,
n |
|
| 0.461 |
| II | 4 | 6 |
|
|
III | 7 | 4 |
|
| IV | 3 | 5 |
|
Table VI lists the
detail of cancer recurrence in both groups. 4 patients in the S-MVR
group and 3 patients in the M-MVR group had distant metastases. 4
patients had a diagnosis of pathological T4b. 5 of 7 patients had
received postoperative adjuvant chemotherapy. None of 7 patients
had received a secondary intervention. There was no significant
difference between groups in the 3-year overall survival rate
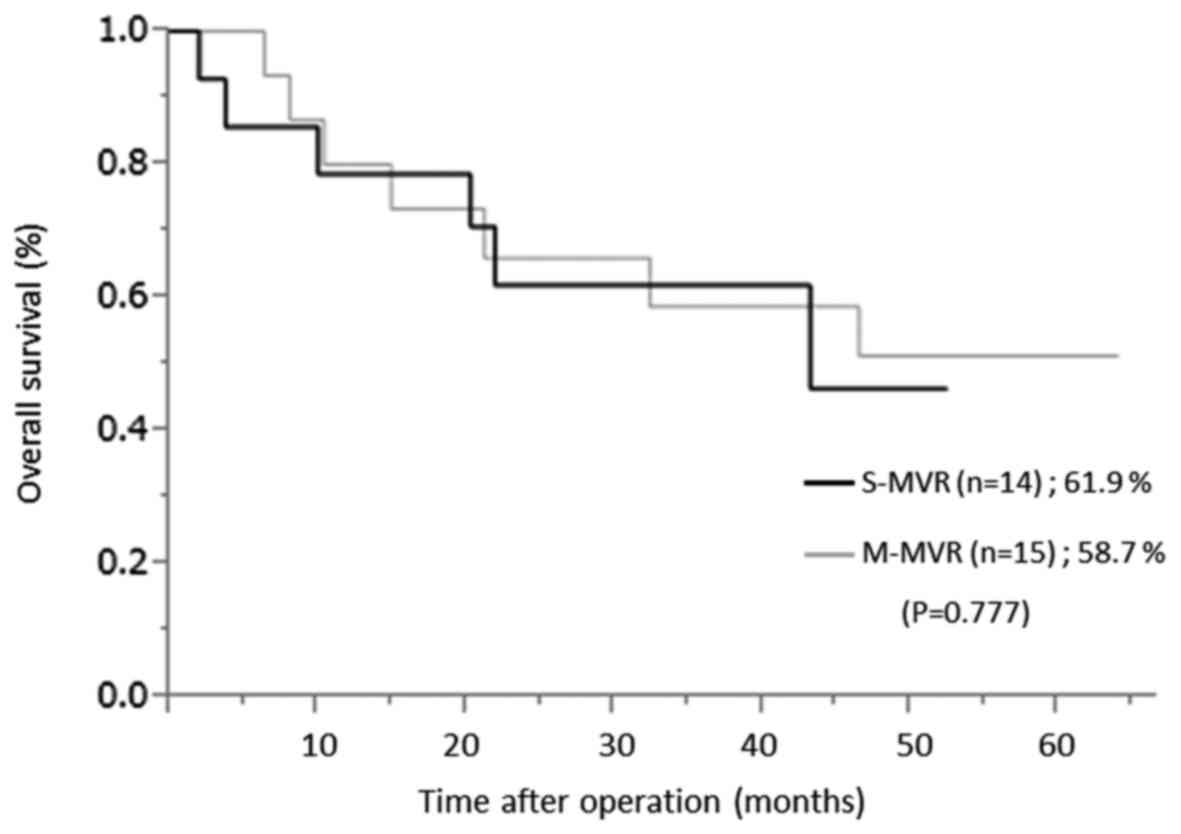
(61.9% in the S-MVR group vs. 58.7% in the M-MVR group, P=0.777)
over a median follow-up period of 30.7 (2.0–63.9) months (Fig. 1). In pathologic Stage II or III
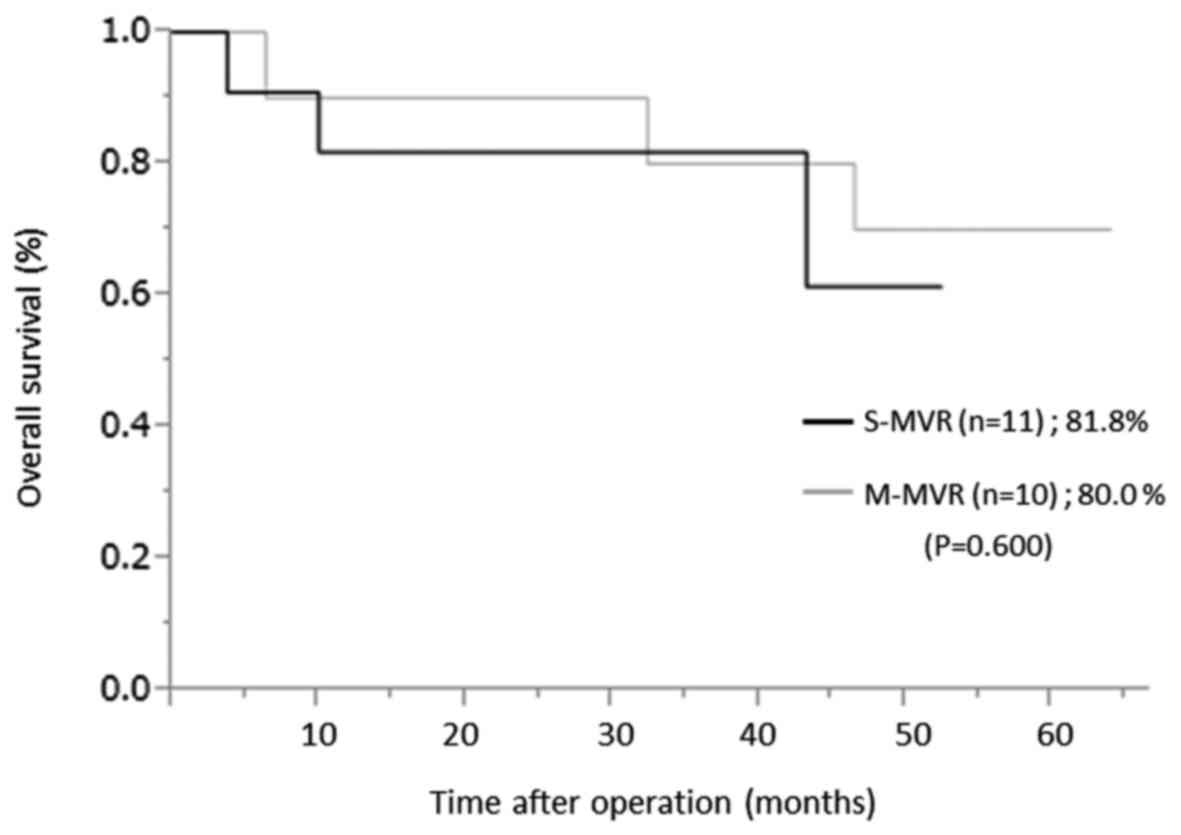
patients, the 3-year overall survival rate was 81.8% in the S-MVR
group and 80.0% in the M-MVR group (P=0.600) (Fig. 2). The 3 year disease-free survival
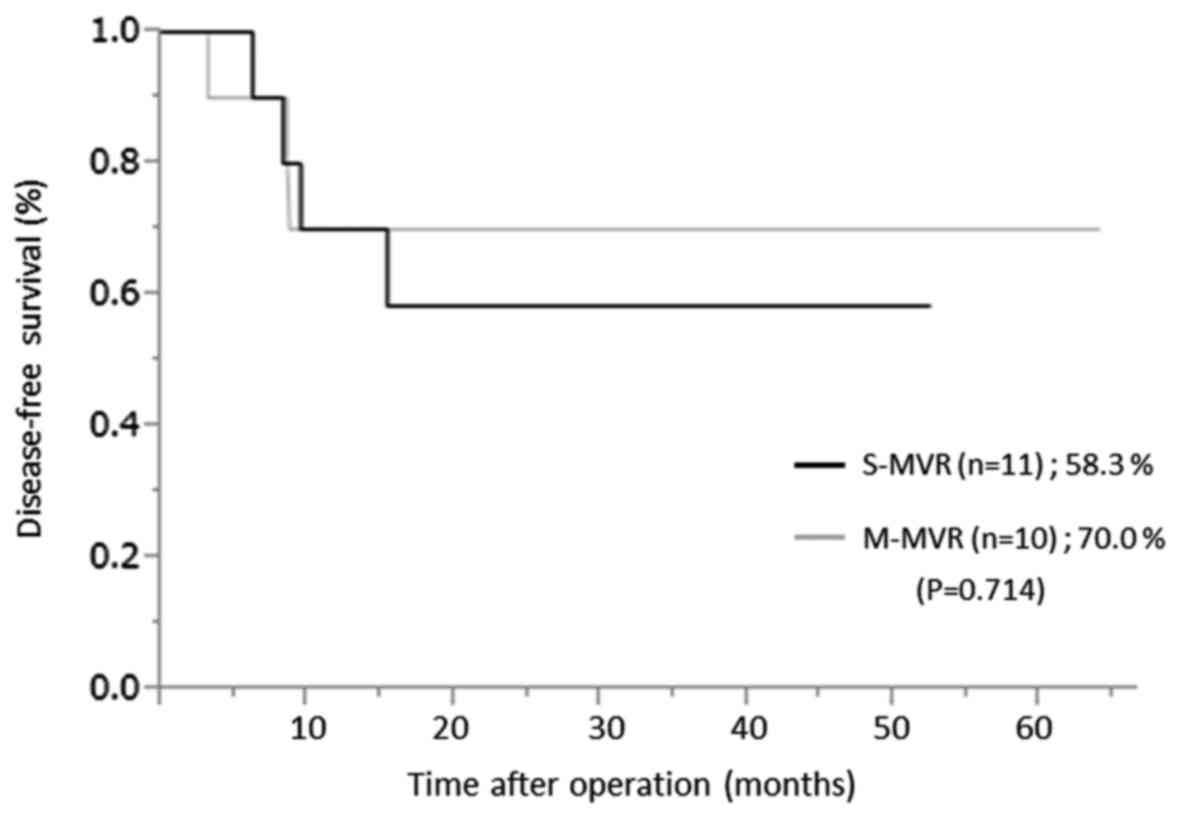
rate was 58.3% and in the S-MVR group and 70.0% in the M-MVR group
(P=0.714) over a median follow-up period of 34.0 (2.0–63.9) months
(Fig. 3).
 | Table VI.Cancer recurrence. |
Table VI.
Cancer recurrence.
| Sex | Age | Operation type | Tumor depth | Stage | Timing of
recurrence | Recurrence
pattern | Prognosis |
|---|
| M | 43 | S-MVR | T4b | III | 6 m | Liver,
peritoneum | 3 y 6 m
deceased |
| M | 51 | S-MVR | T4b | III | 6 m | Liver, lung,
peritoneum, para-aortic lymph node | 7 m deceased |
| F | 85 | S-MVR | T4a | III | 6 m | Lung | 2 y 10 m alive |
| F | 76 | S-MVR | T4a | III | 1 y 4 m | Lung, | 3 y 2 m alive |
| F | 83 | M-MVR | T4b | III | 7 m | Peritoneum,
para-aortic lymph node | 3 y 6 m
deceased |
| M | 81 | M-MVR | T4a | III | 7 m | Virchow lymph
node | 2 y 10 m
deceased |
| M | 63 | M-MVR | T4b | III | 3 m | Liver, lung,
peritoneum | 6 m deceased |
Discussion
Previously, we reported that SPS was safe, feasible
and could provide perioperative and short-term oncological outcomes
equal to those of multi-port surgery (15,16). S-MVR
is a somewhat challenging technique, and we consider that
maintaining the regional oncologic outcome is the most important
factor for surgical treatment. In our series, the R0 resection rate
of the primary tumor was 100% in the S-MVR group, and similar with
the M-MVR group. No gross or microscopic positive resection margins
were observed in the S-MVR group. The median number of harvested
lymph nodes was 30 in the S-MVR group, and did not differ compared
with the M-MVR group, which exceeded that of other reports
(7–9).
To the best of our knowledge, this is the first study to report the
clinical outcomes of S-MVR for locally advanced left colon
cancer.
In this study, we showed that S-MVR and M-MVR do not
differ significantly in terms of blood loss, operative time and
number of harvested lymph nodes. The operative time in the present
study was comparable with that of other reports (7–9). S-MVR was
performed successfully in 78.6% of patients. Some studies of SPS
have reported that the conversion rate to MPS or open surgery
ranged from 1.4 to 10.9% (11,12,14).
Previous reports have demonstrated the safety and feasibility of
SPS in colorectal cancer, but their inclusion criteria yielded
highly selected patients (with early cancer or small tumors)
(11,12). The EAES consensus and SAGES guidelines
do not necessary recommend laparoscopic en bloc resection for
locally advanced adherent colon cancers (5,6). However,
our results with S-MVR for advanced primary colon cancer had high
reliability in terms of oncological clearance, blood loss,
operative time and a successful completion rate regarding the
regional oncologic aspect.
In this study, the rate of pathological tumor
invasion of adjacent organs or structures (pT4b) was 35.7% in the
SPS group, and did not differ compared with the MPS group
(P=1.000). A previous review reported that pathological true
infiltration is present in 54.1% of patients who underwent
multivisceral resection for colorectal cancer, whereas peritumoral
adhesion can be caused by an inflammatory reaction in the remaining
patients (20). Because it is very
difficult to judge whether the adhesion is due to true tumor
invasion or an inflammatory reaction intraoperatively, it is
important to resect the primary colon cancer en bloc with adjacent
organ, preventing exposure and dissemination of cancer cells.
We showed that the overall postoperative
complication rate in the SPS group was 28.6%, and did not differ
compared with the MPS group (P=0.450). None of the patients in the
SPS group experienced perioperative death. Previous studies of
laparoscopic multivisceral resection have reported that the
morbidity ranged from 21 to 33% (7–9), and our
series was comparable with those of the previous studies. The
incidence of complications in the MPS group in the present study
was higher compared with that of other reports (7–9). Even
minor complications that might have little or no influence on
postoperative hospital stay or mortality were counted.
Nevertheless, we understand that the rate of complications is
within an acceptable range. Regarding the length of hospital stay,
recent studies in Western countries indicate a median or mean
postoperative hospital stay of 5 to 10 days, whereas the median
hospital stay was 11 days in the SPS group and 18 days in the MPS
group in this study. The reason of longer hospital stay is that the
Japanese health insurance system, which maintains low medical
costs, has influenced this result.
Our perioperative outcomes of single-port
multivisceral resection might be explained by two factors. First,
we selected the patients carefully according to whether S-MVR was
possible. In this study, none of patients underwent total
cystectomy and urinary diversion. Laparoscopic total cystectomy and
intracorporeal urinary diversion are technically challenging under
robotic assistance, and many surgeons are still adopting the hybrid
approach when performing the cystectomy using robotic assistance
and completing the urinary diversions extracorporeally (21). We consider that open surgery should be
chosen if total cyctectomy and urinary diversion are needed.
Second, we have experience in carrying out a large number of
single-port laparoscopic colorectal surgeries. Therefore, the
frequency of single-port multivisceral resections is increasing
gradually at our department. It is very important to select optimal
cases and perform single-port laparoscopic colorectal surgeries
adequately for successful single-port multivisceral resection.
We showed that the pathological R0 resection rate
was 100% in the SPS group. High vascular ligation and a non-touch
technique were maintained in this series. None of patients in the
SPS group had regional lymph nodes recurrence. However, 4 patients
had distant metastases in the SPS group. Cukier et al
reported that 3 year overall survival and disease-free survival
rate was 85.9 and 73.7%, respectively (22). López-Cano et al reported that
5-year overall disease-free survival rate was 48% (23). Luna-Perez et al reported that
5-year overall survival rate was 45% over a median follow-up period
of 36.8 months (24). Our oncological
outcome was comparable with those of the previous studies
regardless of operative approach.
The main limitations of this study are that it was
carried out at a single institution and that it was very small and
retrospective in nature. Therefore, it contains selection bias and
an inability to match the backgrounds of patients between groups.
In addition, our total patient population may be typical for Japan
and not be applicable to the average European or United States
population. However, we believe that when patients are selected
appropriately, the single-port multivisceral resection has no
adverse effects on perioperative and short-term oncological
outcomes. More studies are needed to validate our results and prove
the true value of the single-port multivisceral resection.
To the best of our knowledge his is the first report
of S-MVR for left colon cancer invading or adhering to neighboring
organs. S-MVR in locally advanced left colon cancer is safe and
feasible in appropriately selected patients.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
MTe drafted the paper and analyzed and interpreted
data. MO, YS, KK and MTa made substantial contributions to the
acquisition of the data. HA made substantial contributions to the
concept and design of the study.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Osaka Police Hospital.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lacy AM, García-Valdecasas JC, Delgado S,
Castells A, Taurá P, Piqué JM and Visa J: Laparoscopy-assisted
colectomy versus open colectomy for treatment of non-metastatic
colon cancer: A randomised trial. Lancet. 359:2224–2229. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Milsom JW, Böhm B, Hammerhofer KA, Fazio
V, Steiger E and Elson P: A prospective, randomized trial comparing
laparoscopic versus conventional techniques in colorectal cancer
surgery: A preliminary report. J Am Coll Surg. 187:46–54,
Discussion 54–55. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Leung KL, Kwok SP, Lam SC, Lee JF, Yiu RY,
Ng SS, Lai PB and Lau WY: Laparoscopic resection of rectosigmoid
carcinoma: Prospective randomised trial. Lancet. 363:1187–1192.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Clinical Outcomes of Surgical Therapy
Study Group, . Nelson H, Sargent DJ, Wieand HS, Fleshman J, Anvari
M, Stryker SJ, Beart RW Jr, Hellinger M, Flanagan R Jr, et al: A
comparison of laparoscopically assisted and open colectomy for
colon cancer. N Engl J Med. 350:2050–2059. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Veldkamp R, Gholghesaei M, Bonjer HJ,
Meijer DW, Buunen M, Jeekel J, Anderberg B, Cuesta MA, Cuschierl A,
Fingerhut A, et al: Laparoscopic resection of colon cancer:
Consensus of the European Association of Endoscopic Surgery (EAES).
Surg Endosc. 18:1163–1185. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zerey M, Hawver LM, Awad Z, Stefanidis D,
Richardson W and Fanelli RD: Members of the SAGES Guidelines
Committee: SAGES evidence-based guidelines for the laparoscopic
resection of curable colon and rectal cancer. Surg Endosc. 27:1–10.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kim KY, Hwang DW, Park YK and Lee HS: A
single surgeon's experience with 54 consecutive cases of
multivisceral resection for locally advanced primary colorectal
cancer: Can the laparoscopic approach be performed safely? Surg
Endosc. 26:493–500. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nagasue Y, Akiyoshi T, Ueno M, Fukunaga Y,
Nagayama S, Fujimoto Y, Konishi T, Nagasaki T, Nagata J, Mukai T,
et al: Laparoscopic versus open multivisceral resection for primary
colorectal cancer: Comparison of perioperative outcomes. J
Gastrointest Surg. 17:1299–1305. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bretagnol F, Dedieu A, Zappa M, Guedj N,
Ferron M and Panis Y: T4 colorectal cancer: Is laparoscopic
resection contraindicated? Colorectal Dis. 13:138–143. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Remzi FH, Kirat HT, Kaouk JH and Geisler
DP: Single-port laparoscopy in colorectal surgery. Colorectal Dis.
10:823–826. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Takemasa I, Uemura M, Nishimura J,
Mizushima T, Yamamoto H, Ikeda M, Sekimoto M, Doki Y and Mori M:
Feasibility of single-site laparoscopic colectomy with complete
mesocolic excision for colon cancer: A prospective case-control
comparison. Surg Endosc. 28:1110–1118. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Champagne BJ, Papaconstantinou HT, Parmar
SS, Nagle DA, Young-Fadok TM, Lee EC and Delaney CP:
Single-incision versus standard multiport laparoscopic colectomy: A
multicenter, case-controlled comparison. Ann Surg. 255:66–69. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Poon JT, Cheung CW, Fan JK, Lo OS and Law
WL: Single-incision versus conventional laparoscopic colectomy for
colonic neoplasm: A randomized, controlled trial. Surg Endosc.
26:2729–2734. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim SJ, Ryu GO, Choi BJ, Kim JG, Lee KJ,
Lee SC and Oh ST: The short-term outcomes of conventional and
single-port laparoscopic surgery for colorectal cancer. Ann Surg.
254:933–940. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tei M, Wakasugi M and Akamatsu H:
Comparison of short-term surgical results of single-port and
multi-port laparoscopic rectal resection for rectal cancer. Am J
Surg. 210:309–314. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tei M, Wakasugi M and Akamatsu H:
Comparison of the perioperative and short-term oncological outcome
after single- or multiport surgery for colorectal cancer.
Colorectal Dis. 17:O141–O147. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yun JA, Yun SH, Park YA, Huh JW, Cho YB,
Kim HC and Lee WY: Oncologic outcomes of single-incision
laparoscopic surgery compared with conventional laparoscopy for
colon cancer. Ann Surg. 263:973–978. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dindo D, Demartines N and Clavien PA:
Classification of surgical complications: A new proposal with
evaluation in a cohort of 6336 patients and results of a survey.
Ann Surg. 240:205–213. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tei M, Wakasugi M, Omori T, Ueshima S,
Tori M and Akamatsu H: Single-port laparoscopic colectomy is safe
and feasible in patients with previous abdominal surgery. Am J
Surg. 209:1007–1012. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mohan HM, Evans MD, Larkin JO, Beynon J
and Winter DC: Multivisceral resection in colorectal cancer: A
systematic review. Ann Surg Oncol. 20:2929–2936. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sim A, Balbay MD, Todenhöfer T,
Aufderklamm S, Halalsheh O, Mischinger J, Böttge J, Rausch S, Bier
S, Stenzl A, et al: Robot-assisted radical cystectomy and
intracorporeal urinary diversion-safe and reproducible? Cent
European J Urol. 68:18–23. 2015.PubMed/NCBI
|
|
22
|
Cukier M, Smith AJ, Milot L, Chu W, Chung
H, Fenech D, Herschorn S, Ko Y, Rowsell C, Soliman H, et al:
Neoadjuvant chemoradiotherapy and multivisceral resection for
primary locally advanced adherent colon cancer: A single
institution experience. Eur J Surg Oncol. 38:677–682. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
López-Cano M, Mañas MJ, Hermosilla E and
Espín E: Multivisceral resection for colon cancer: Analysis of
prognostic factors. Dig Surg. 27:238–245. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Luna-Pérez P, Rodríguez-Ramírez SE, De la
Barrera MG, Zeferino M and Labastida S: Multivisceral resection for
colon cancer. J Surg Oncol. 80:100–104. 2002. View Article : Google Scholar : PubMed/NCBI
|