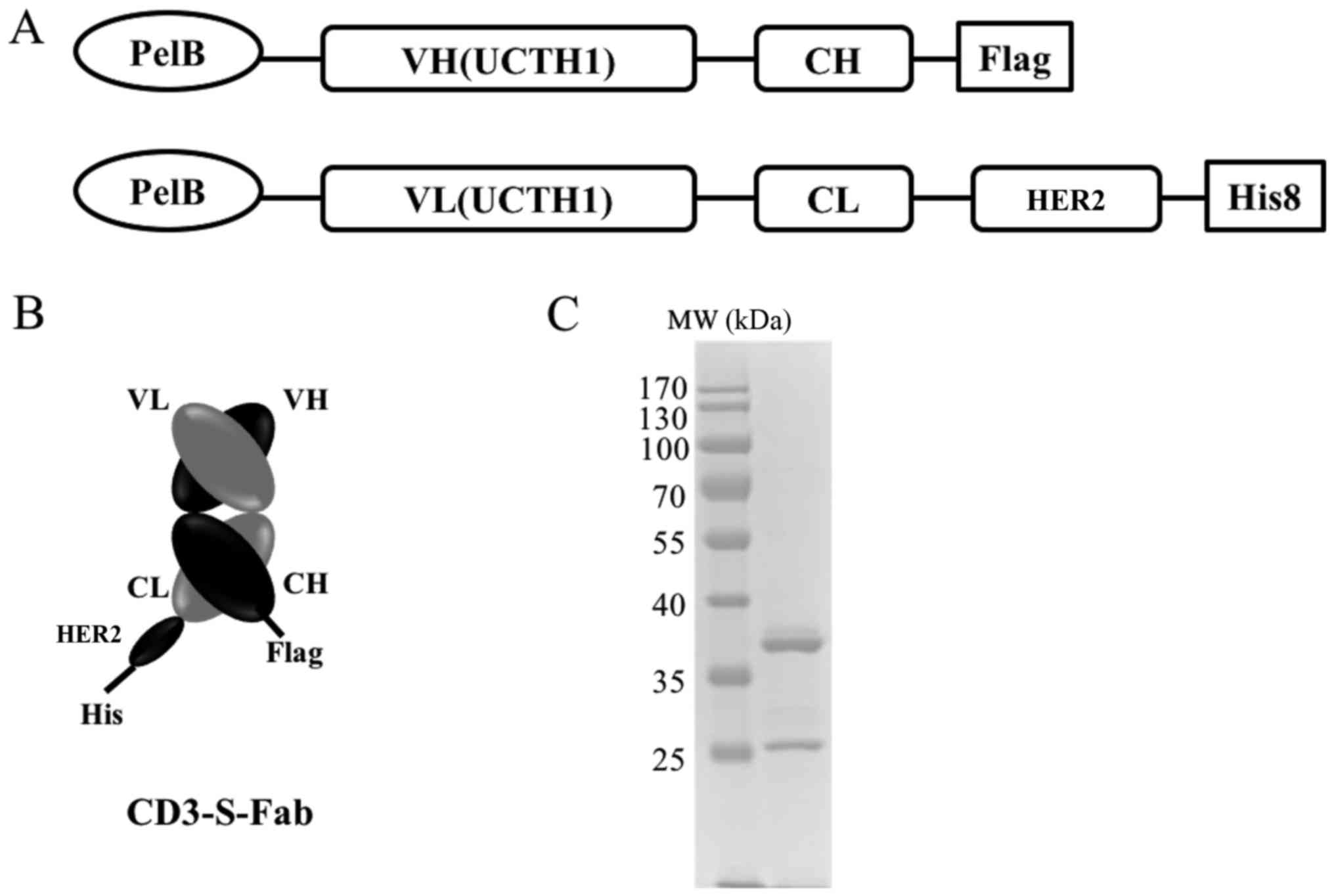
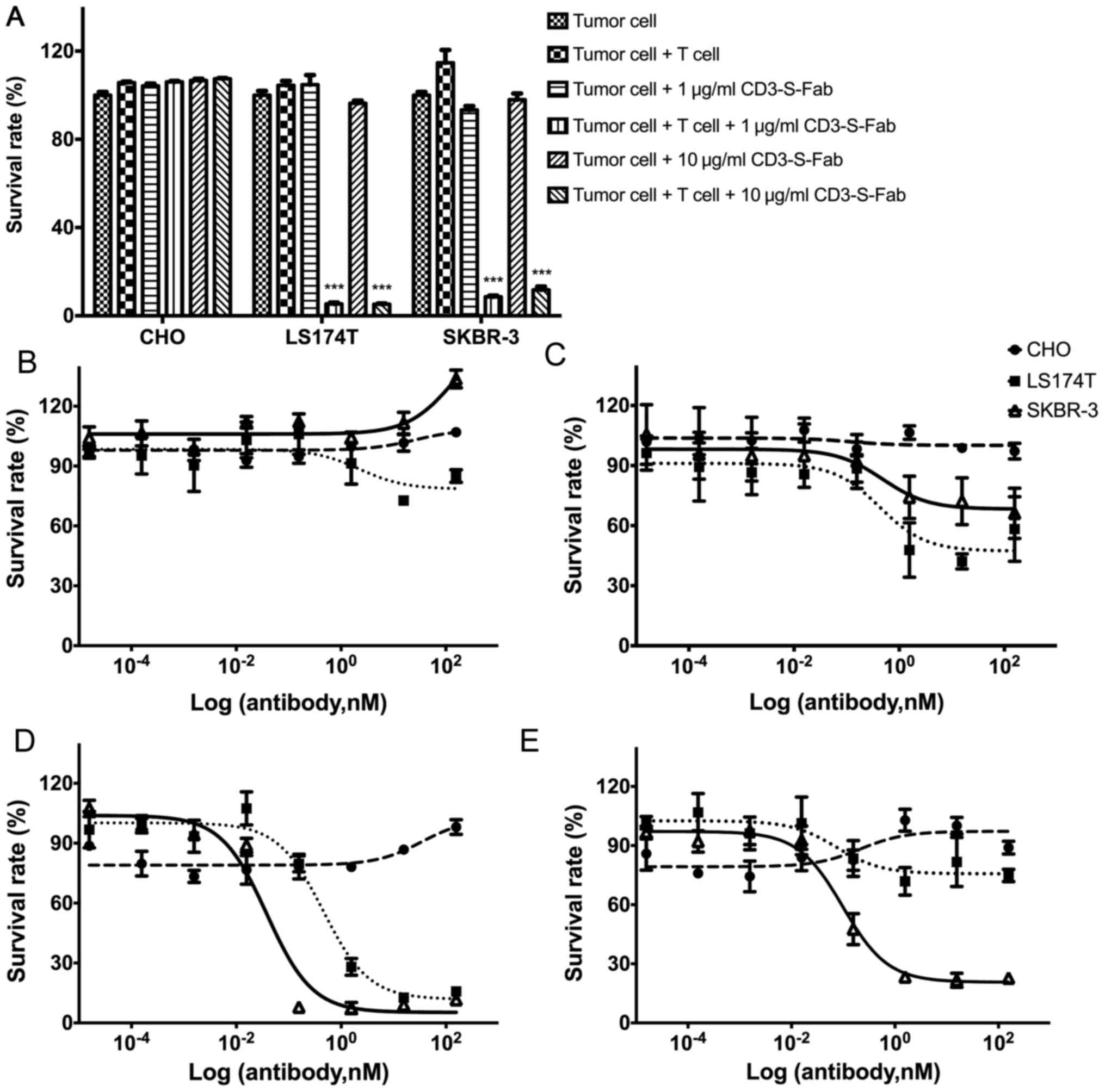
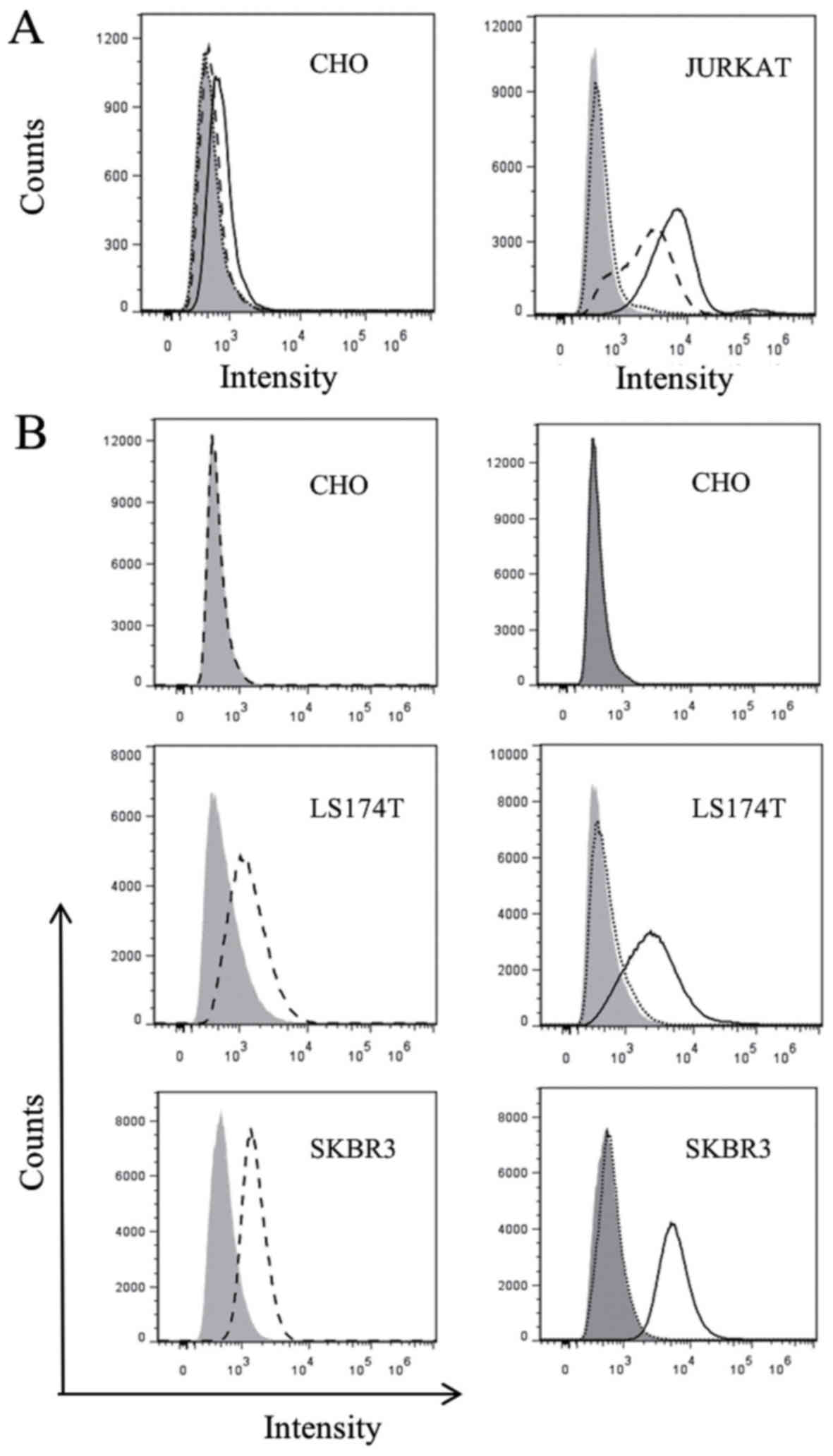
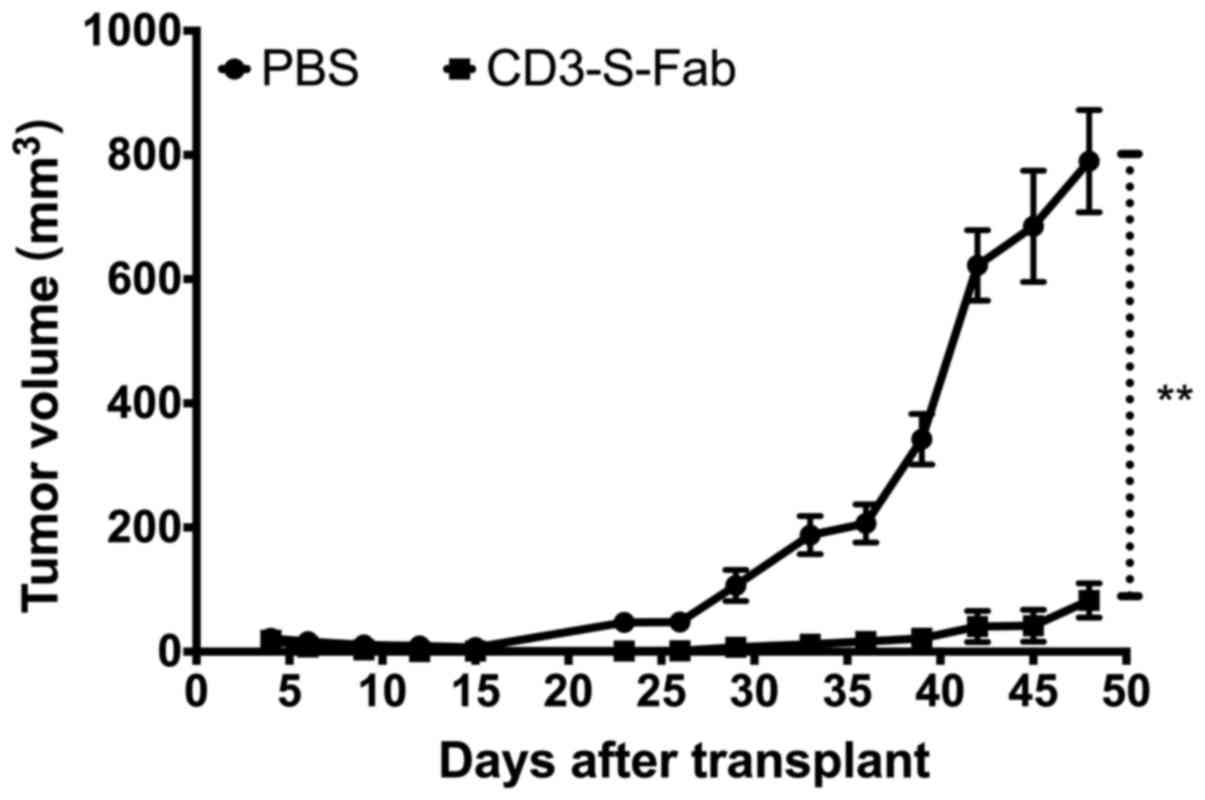
|
1
|
Slamon DJ, Clark GM, Wong SG, Levin WJ,
Ullrich A and McGuire WL: Human breast cancer: Correlation of
relapse and survival with amplification of the HER-2/neu oncogene.
Science. 235:177–182. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Press MF, Pike MC, Hung G, Zhou JY, Ma Y,
George J, Dietz-Band J, James W, Slamon DJ, Batsakis JG, et al:
Amplification and overexpression of HER-2/neu in carcinomas of the
salivary gland: Correlation with poor prognosis. Cancer Res.
54:5675–5682. 1994.PubMed/NCBI
|
|
3
|
Daniele L and Sapino A: Anti-HER2
treatment and breast cancer: State of the art, recent patents, and
new strategies. Recent Pat Anticancer Drug Discov. 4:9–18. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yarden Y: Biology of HER2 and its
importance in breast cancer. Oncology. 61 Suppl 2:S1–S13. 2001.
View Article : Google Scholar
|
|
5
|
Ben-Kasus T, Schechter B, Lavi S, Yarden Y
and Sela M: Persistent elimination of ErbB-2/HER2-overexpressing
tumors using combinations of monoclonal antibodies: Relevance of
receptor endocytosis. Proc Natl Acad Sci USA. 106:3294–3299. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Keler T, Graziano RF, Mandal A, Wallace
PK, Fisher J, Guyre PM, Fanger MW and Deo YM: Bispecific
antibody-dependent cellular cytotoxicity of HER2/neu-overexpressing
tumor cells by Fc gamma receptor type I-expressing effector cells.
Cancer Res. 57:4008–4014. 1997.PubMed/NCBI
|
|
7
|
Vasconcellos FA, Aleixo PB, Stone SC,
Conceicao FR, Dellagostin OA and Aleixo JA: Generation and
characterization of new HER2 monoclonal antibodies. Acta Histochem.
115:240–244. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Vaneycken I, Devoogdt N, Van Gassen N,
Vincke C, Xavier C, Wernery U, Muyldermans S, Lahoutte T and
Caveliers V: Preclinical screening of anti-HER2 nanobodies for
molecular imaging of breast cancer. FASEB J. 25:2433–2446. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hicks DG and Kulkarni S: HER2+ breast
cancer: Review of biologic relevance and optimal use of diagnostic
tools. Am J Clin Pathol. 129:263–273. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ranson M and Sliwkowski MX: Perspectives
on anti-HER monoclonal antibodies. Oncology. 63 Suppl 1:S17–S24.
2002. View Article : Google Scholar
|
|
11
|
Hudis CA: Trastuzumab-mechanism of action
and use in clinical practice. N Engl J Med. 357:39–51. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Spector NL and Blackwell KL: Understanding
the mechanisms behind trastuzumab therapy for human epidermal
growth factor receptor 2-positive breast cancer. J Clin Oncol.
27:5838–5847. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Valabrega G, Montemurro F and Aglietta M:
Trastuzumab: Mechanism of action, resistance and future
perspectives in HER2-overexpressing breast cancer. Ann Oncol.
18:977–984. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Junttila TT, Parsons K, Olsson C, Lu Y,
Xin Y, Theriault J, Crocker L, Pabonan O, Baginski T, Meng G, et
al: Superior in vivo efficacy of afucosylated trastuzumab in the
treatment of HER2-amplified breast cancer. Cancer Res.
70:4481–4489. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Arteaga CL, Sliwkowski MX, Osborne CK,
Perez EA, Puglisi F and Gianni L: Treatment of HER2-positive breast
cancer: Current status and future perspectives. Nat Rev Clin Oncol.
9:16–32. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Benchetrit F, Gazagne A, Adotevi O,
Haicheur N, Godard B, Badoual C, Fridman WH and Tartour E:
Cytotoxic T lymphocytes: Role in immunosurveillance and in
immunotherapy. Bull Cancer. 90:677–685. 2003.PubMed/NCBI
|
|
17
|
Nagorsen D, Bargou R, Ruttinger D, Kufer
P, Baeuerle PA and Zugmaier G: Immunotherapy of lymphoma and
leukemia with T-cell engaging BiTE antibody blinatumomab. Leuk
Lymphoma. 50:886–891. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Junttila TT, Li J, Johnston J,
Hristopoulos M, Clark R, Ellerman D, Wang BE, Li Y, Mathieu M, Li
G, et al: Antitumor efficacy of a bispecific antibody that targets
HER2 and activates T cells. Cancer Res. 74:5561–5571. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Baeuerle PA and Reinhardt C: Bispecific
T-cell engaging antibodies for cancer therapy. Cancer Res.
69:4941–4944. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schlereth B, Fichtner I, Lorenczewski G,
Kleindienst P, Brischwein K, da Silva A, Kufer P, Lutterbuese R,
Junghahn I, Kasimir-Bauer S, et al: Eradication of tumors from a
human colon cancer cell line and from ovarian cancer metastases in
immunodeficient mice by a single-chain Ep-CAM-/CD3-bispecific
antibody construct. Cancer Res. 65:2882–2889. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Taki S, Kamada H, Inoue M, Nagano K, Mukai
Y, Higashisaka K, Yoshioka Y, Tsutsumi Y and Tsunoda S: A novel
bispecific antibody against human CD3 and ephrin receptor A10 for
breast cancer therapy. PLoS One. 10:e01447122015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dreier T, Lorenczewski G, Brandl C,
Hoffmann P, Syring U, Hanakam F, Kufer P, Riethmuller G, Bargou R
and Baeuerle PA: Extremely potent, rapid and
costimulation-independent cytotoxic T-cell response against
lymphoma cells catalyzed by a single-chain bispecific antibody. Int
J Cancer. 100:690–697. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Oak E and Bartlett NL: Blinatumomab for
the treatment of B-cell lymphoma. Expert Opin Investig Drugs.
24:715–724. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Haense N, Atmaca A, Pauligk C, Steinmetz
K, Marmé F, Haag GM, Rieger M, Ottmann OG, Ruf P, Lindhofer H and
Al-Batran SE: A phase I trial of the trifunctional anti HER2 × anti
CD3 antibody ertumaxomab in patients with advanced solid tumors.
BMC Cancer. 16:4202016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Vaishampayan U, Thakur A, Rathore R,
Kouttab N and Lum LG: Phase I study of Anti-CD3 × Anti-HER2
bispecific antibody in metastatic castrate resistant prostate
cancer patients. Prostate Cancer. 2015:2851932015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cao Y, Axup JY, Ma JS, Wang RE, Choi S,
Tardif V, Lim RK, Pugh HM, Lawson BR, Welzel G, et al: Multiformat
T-cell-engaging bispecific antibodies targeting human breast
cancers. Angew Chem Int Ed Engl. 54:7022–7027. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhou Y, Gou LT, Guo ZH, Liu HR, Wang JM,
Zhou SX, Yang JL and Li XA: Fully human HER2/cluster of
differentiation 3 bispecific antibody triggers potent and specific
cytotoxicity of T lymphocytes against breast cancer. Mol Med Rep.
12:147–154. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li A, Xing J, Li L, Zhou C, Dong B, He P,
Li Q and Wang Z: A single-domain antibody-linked Fab bispecific
antibody HER2-S-Fab has potent cytotoxicity against HER2-expressing
tumor cells. AMB Express. 6:322016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li L, He P, Zhou C, Jing L, Dong B, Chen
S, Zhang N, Liu Y, Miao J, Wang Z and Li Q: A novel bispecific
antibody, S-Fab, induces potent cancer cell killing. J Immunother.
38:350–356. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Vincke C, Loris R, Saerens D,
Martinez-Rodriguez S, Muyldermans S and Conrath K: General strategy
to humanize a camelid single-domain antibody and identification of
a universal humanized nanobody scaffold. J Biol Chem.
284:3273–3284. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shalaby MR, Shepard HM, Presta L,
Rodrigues ML, Beverley PC, Feldmann M and Carter P: Development of
humanized bispecific antibodies reactive with cytotoxic lymphocytes
and tumor cells overexpressing the HER2 protooncogene. J Exp Med.
175:217–225. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
von Roman Freiherr M, Koller A, von Rüden
D and Berensmeier S: Improved extracellular expression and
purification of recombinant Staphylococcus aureus protein A.
Protein Expr Purif. 93:87–92. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yoon SH, Kim SK and Kim JF: Secretory
production of recombinant proteins in Escherichia coli. Recent Pat
Biotechnol. 4:23–29. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kwong KY and Rader C: E. coli expression
and purification of Fab antibody fragments. Curr Protoc Protein Sci
Chapter 6. Unit 6.10. 2009. View Article : Google Scholar
|
|
35
|
Skrlj N, Serbec VC and Dolinar M:
Single-chain Fv antibody fragments retain binding properties of the
monoclonal antibody raised against peptide P1 of the human prion
protein. Appl Biochem Biotechnol. 160:1808–1821. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
So EC, Sallin MA, Zhang X, Chan SL, Sahni
L, Schulze DH, Davila E, Strome SE and Jain A: A high throughput
method for enrichment of natural killer cells and lymphocytes and
assessment of in vitro cytotoxicity. J Immunol Methods. 394:40–48.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Busch R, Cesar D, Higuera-Alhino D, Gee T,
Hellerstein MK and McCune JM: Isolation of peripheral blood CD4(+)
T cells using RosetteSep and MACS for studies of DNA turnover by
deuterium labeling. J Immunol Methods. 286:97–109. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rozan C, Cornillon A, Petiard C, Chartier
M, Behar G, Boix C, Kerfelec B, Robert B, Pèlegrin A, Chames P, et
al: Single-domain antibody-based and linker-free bispecific
antibodies targeting FcγRIII induce potent antitumor activity
without recruiting regulatory T cells. Mol Cancer Ther.
12:1481–1491. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Choi JH and Lee SY: Secretory and
extracellular production of recombinant proteins using Escherichia
coli. Appl Microbiol Biotechnol. 64:625–635. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fu XY: Extracellular accumulation of
recombinant protein by Escherichia coli in a defined medium. Appl
Microbiol Biotechnol. 88:75–86. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Scott AM, Wolchok JD and Old LJ: Antibody
therapy of cancer. Nat Rev Cancer. 12:278–287. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Osada T, Patel SP, Hammond SA, Osada K,
Morse MA and Lyerly HK: CEA/CD3-bispecific T cell-engaging (BiTE)
antibody-mediated T lymphocyte cytotoxicity maximized by inhibition
of both PD1 and PD-L1. Cancer Immunol Immunother. 64:677–688. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Karagiannis P, Singer J, Hunt J, Gan SK,
Rudman SM, Mechtcheriakova D, Knittelfelder R, Daniels TR, Hobson
PS, Beavil AJ, et al: Characterisation of an engineered trastuzumab
IgE antibody and effector cell mechanisms targeting
HER2/neu-positive tumour cells. Cancer Immunol Immunother.
58:915–930. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lambertini M, Ponde NF, Solinas C and de
Azambuja E: Adjuvant trastuzumab: A 10-year overview of its
benefit. Expert Rev Anticancer Ther. 17:61–74. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Xin Y, Guo WW, Huang Q, Zhang P, Zhang LZ,
Jiang G and Tian Y: Effects of lapatinib or trastuzumab, alone and
in combination, in human epidermal growth factor receptor
2-positive breast cancer: A meta-analysis of randomized controlled
trials. Cancer Med. 5:3454–3463. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Malenfant SJ, Eckmann KR and Barnett CM:
Pertuzumab: A new targeted therapy for HER2-positive metastatic
breast cancer. Pharmacotherapy. 34:60–71. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zazo S, Gonzalez-Alonso P, Martin-Aparicio
E, Chamizo C, Cristóbal I, Arpí O, Rovira A, Albanell J, Eroles P,
Lluch A, et al: Generation, characterization, and maintenance of
trastuzumab-resistant HER2+ breast cancer cell lines. Am J Cancer
Res. 6:2661–2678. 2016.PubMed/NCBI
|
|
48
|
James ND, Atherton PJ, Jones J, Howie AJ,
Tchekmedyian S and Curnow RT: A phase II study of the bispecific
antibody MDX-H210 (anti-HER2 × CD64) with GM-CSF in HER2+ advanced
prostate cancer. Br J Cancer. 85:152–156. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhu Z and Carter P: Identification of
heavy chain residues in a humanized anti-CD3 antibody important for
efficient antigen binding and T cell activation. J Immunol.
155:1903–1910. 1995.PubMed/NCBI
|
|
50
|
Loffler A, Kufer P, Lutterbüse R, Zettl F,
Daniel PT, Schwenkenbecher JM, Riethmuller G, Dörken B and Bargou
RC: A recombinant bispecific single-chain antibody, CD19 × CD3,
induces rapid and high lymphoma-directed cytotoxicity by
unstimulated T lymphocytes. Blood. 95:2098–2103. 2000.PubMed/NCBI
|
|
51
|
Qasemi M, Behdani M, Shokrgozar MA,
Molla-Kazemiha V, Mohseni-Kuchesfahani H and Habibi-Anbouhi M:
Construction and expression of an anti-VEGFR2 Nanobody-Fc
fusionbody in NS0 host cell. Protein Expr Purif. 123:19–25. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mergulhao FJ, Summers DK and Monteiro GA:
Recombinant protein secretion in Escherichia coli. Biotechnol Adv.
23:177–202. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Khushoo A, Pal Y, Singh BN and Mukherjee
KJ: Extracellular expression and single step purification of
recombinant Escherichia coli L-asparaginase II. Protein Expr Purif.
38:29–36. 2004. View Article : Google Scholar : PubMed/NCBI
|