Introduction
Lung cancer accounts for 22.7% of all cases of
malignant tumors (1). It is also the
most common cause of mortality among all types of cancer worldwide,
comprising 17.6% of the world total (2,3).
Cavitation is relatively common in lung cancer, occurring in 2–16%
cases. Typically, the cysts have thick walls (4). A cyst is defined as an air-containing
space surrounded by a thin wall (4 mm or less), while a cavity is
defined as a space with a wall at least 5 mm thick. A typical lung
cancer presenting as solitary thin-wall cysts may occasionally
occur (5–7). Accordingly, 45 patients with solitary
thin-wall cystic lung cancer were reviewed, in order to better
understand this type of lung cancer. In the present study, the
solitary thin-wall cystic lung cancer is defined as a cystic lesion
with a wall of less than 4 mm.
Materials and methods
Clinical information on 45 patients (32 males, 13
females; mean age, 55 years; range, 33–78 years) with thin-wall
cystic lung cancer treated in Beijing Shijitan Hospital (Beijing,
China) between 2006 and 2017 was collected. Thin-wall cystic
changes in the lungs could be observed in all patients. Meanwhile,
the diagnosis of this disease was based on radiological findings
and biopsy following surgical resection or bronchoscopic biopsy.
Patients without the above typical imaging features were excluded.
All images and pathology were confirmed by two experts affiliated
with the Beijing Shijitan Hospital, if there was a dispute, a more
experienced expert would be asked to decide. All patients underwent
chest computed tomography (CT), 6 patients underwent positron
emission tomography (PET)/CT (a number of patients did not undergo
PET scans due to the prohibitive cost), 39 patients underwent
pulmonary lobectomy or wedge excision, and four patients were
administered with Iressa (250 mg once a day; reexamined every
month) due to metastasis. CT scans were performed using 64-section
spiral CT with a slice thickness of 1.25 or 1.5 mm for all
patients. These cases were retrospectively analyzed following
review of patient medical records, radiological findings,
pathological changes and treatment strategies. Additionally,
certain patients were telephoned in order to learn about their
recent physical conditions. Telephone consultations were only used
when patients were unable to travel to the hospital. The present
study was conducted in accordance with the institutional policy
regarding the protection of patient confidential information and
was approved by the Research Ethics Committee of Beijing Shijitan
Hospital affiliated to Capital Medical University. All procedures
were performed in accordance with the approved guidelines of
Beijing Shijitan Hospital affiliated to Capital Medical
University.
Results
Clinical characteristics of forty-five
patients
There were 32 male (71.1%) and 13 female (28.9%)
patients enrolled in the present study (Table I). Their ages ranged between 33 and 78
years old, and 55.5% of them were below 60 years of age.
Furthermore, 72.5% of the patients stated that they had no history
of smoking. A total of 23 patients (45.7%) were asymptomatic, and
the others exhibited a cough, sputum or bloody sputum.
 | Table I.Clinical characteristics of 45
patients with thin-walled cystic lung cancer. |
Table I.
Clinical characteristics of 45
patients with thin-walled cystic lung cancer.
| Target | Number (%) |
|---|
| Sex |
|
| Male | 32 (71.1) |
|
Female | 13 (28.9) |
| Age, years |
|
|
30–40 | 2 (4.4) |
|
41–50 | 15 (33.3) |
|
51–60 | 8 (17.8) |
|
>60 | 20 (44.5) |
| Smoking |
|
| No | 31 (72.5) |
| Yes | 14 (27.5) |
| Symptoms |
|
| No
symptoms | 23 (45.7) |
|
Cough | 14 (23.9) |
|
Sputum | 12 (21.7) |
| Blood in
sputum | 5 (8.7) |
| HRCT presentation of
thin-walled cysts |
|
|
Asymmetric thickening of the
wall | 37 (96.8) |
|
Ground-glass shadow | 4 (9.6) |
| Short
spicules | 13 (35.5) |
| Irregular
margins | 20 (51.6) |
|
Separation in cysts | 21 (54.8) |
|
Lobulation | 7 (19.3) |
| Pleural
indentation | 9 (22.6) |
| Blood
vessel convergency | 6 (16.1) |
| Maximum cyst
diameter, cm |
|
<1 | 0 (0.0) |
| 1–2 | 4 (8.9) |
|
2.1–4 | 33 (73.3) |
|
>4 | 8 (17.8) |
| Metastasis |
|
| No | 42 (93.3) |
| Yes | 3 (6.7) |
| Pathology |
|
|
Adenocarcinoma | 43 (93.3) |
| Squamous
cell carcinoma | 2 (6.7) |
| Prognosis |
|
|
Relapse | 1 (2.2) |
| Succumbed
to mortality from the disease | 2 (4.5) |
| Succumbed
to mortality from another disease | 1 (2.2) |
|
Survival | 41 (91.1) |
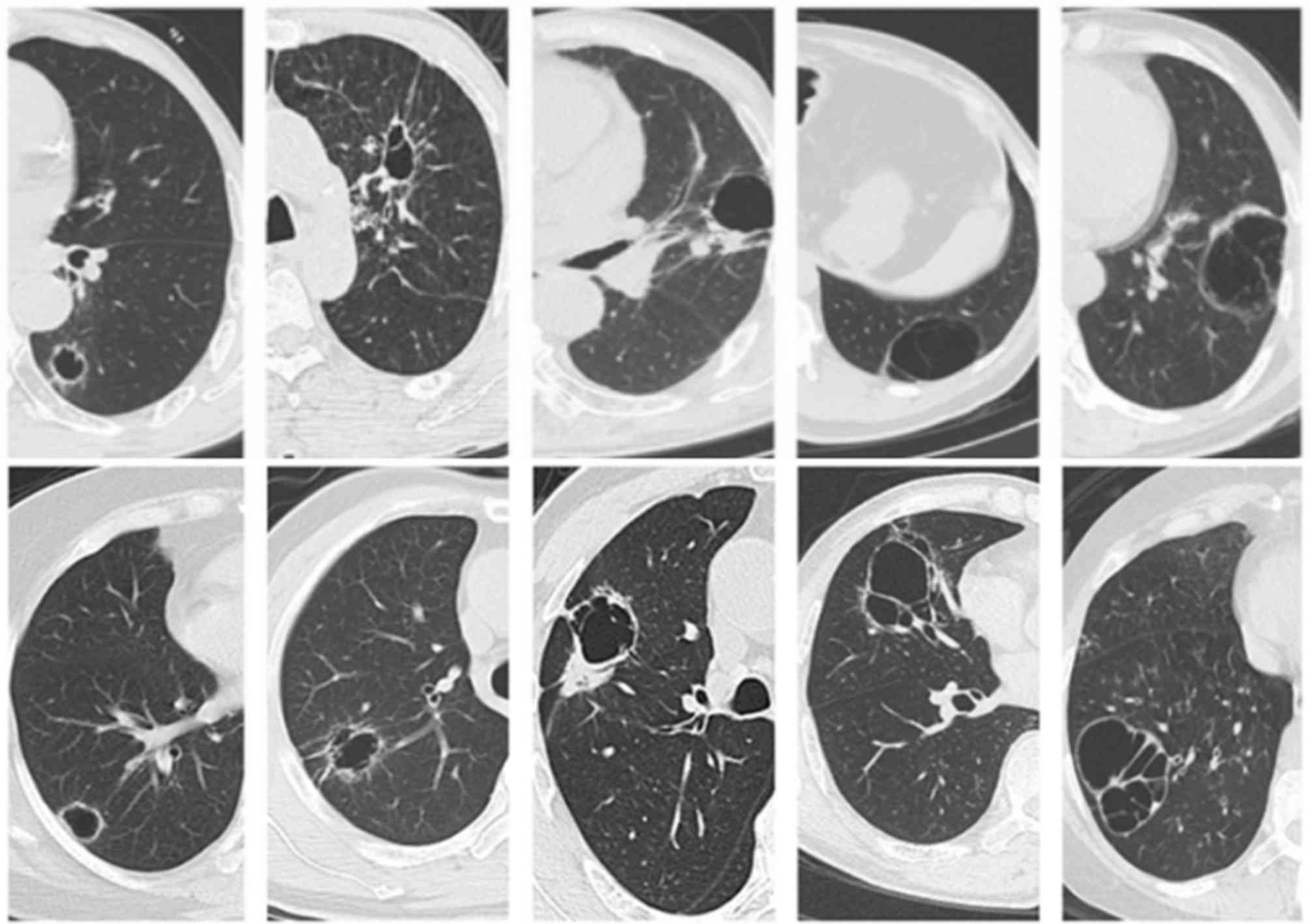
Fig. 1 demonstrates CT
manifestations of thin-wall cystic lung cancer. The primary lesions
occurred at the right lower lobe in 14 patients, at the right
middle lobe in 5 patients, at the right upper lobe in 4 patients,
at the left upper lobe in 10 patients, and at the left lower lobe
in 12 patients. The wall thickness of cysts ranged between 1 and 4
mm. Each of them displayed one or more suspected malignant signs of
lung cancer, including asymmetric thickening (96.8%), separation in
cysts (54.8%), irregular margin (51.6%), small spicules (35.5%),
dragging sensation in the pleura (22.6%), lobulation (19.3%) and
ground-glass opacity (9.6%).
PET/CT scans
A total of 6 patients underwent PET/CT scans, one of
which demonstrated a SUVmax value of 3.9, while the others
exhibited normal values (0.8–1.7). The serum tumor markers (CEA,
8.28 µg/l) were higher than normal (0–5 µg/l) in only one
patient.
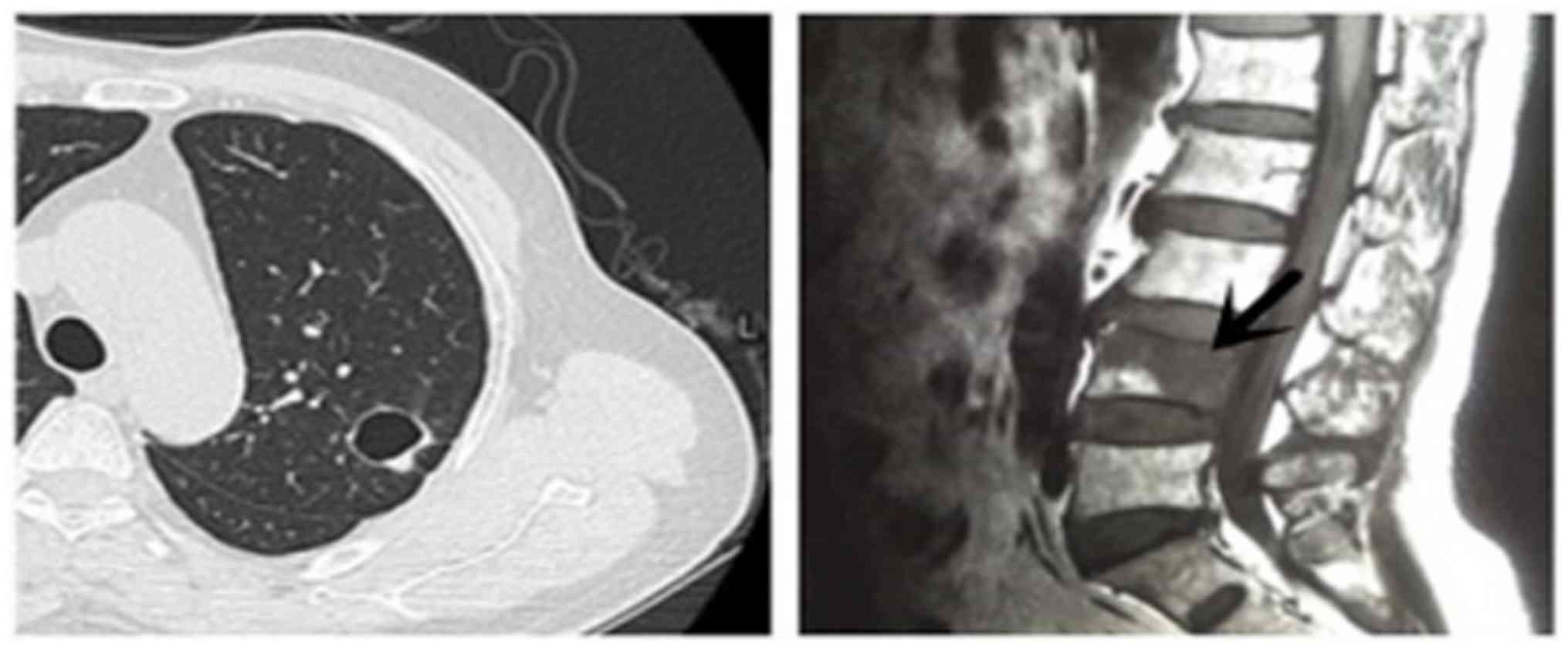
Distant metastasis
Metastasis to the fourth lumbar vertebra was noted
in the whole-body bone scan of one patient (Fig. 2), which was confirmed as metastatic
adenocarcinoma by pathological findings following percutaneous
needle washing (PNW).
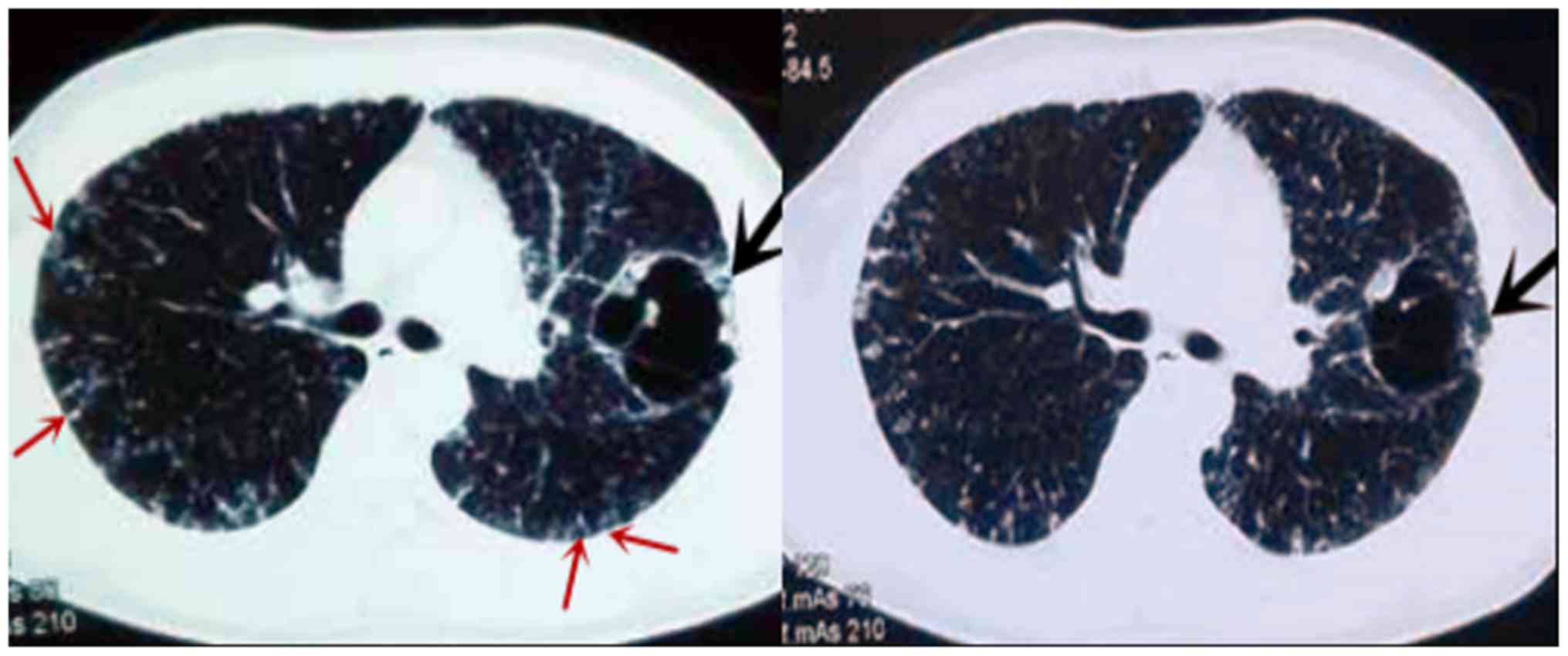
Intrapulmonary metastasis
One 33-year-old patient with a 2-month history of
cough and sputum presented with a large, abnormal cystic lesion in
the left lung and multiple cystic lesions in the two lungs on CT
images (Fig. 3). The diagnosis by CT
and PET/CT was pulmonary bullae. Pleural involvement, repeated
pneumothorax and choking sensations in the chest were evident. Due
to these signs, the possibility of malignant lesions was not ruled
out. Therefore, bronchoscopic biopsy was performed, which confirmed
these cystic lesions as intrapulmonary metastatic
adenocarcinoma.
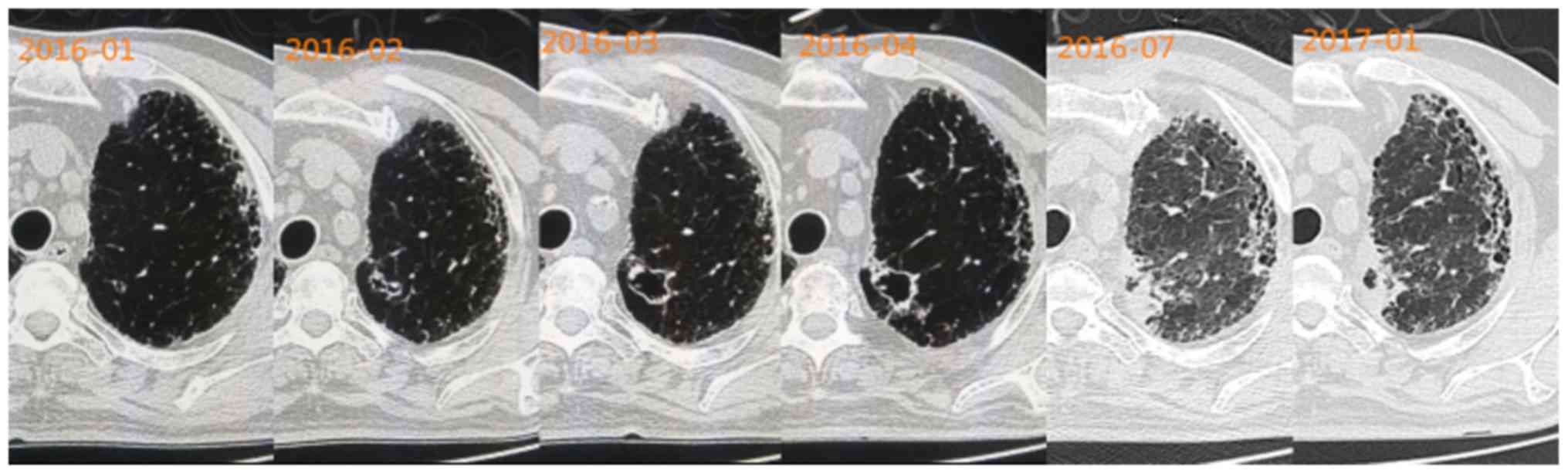
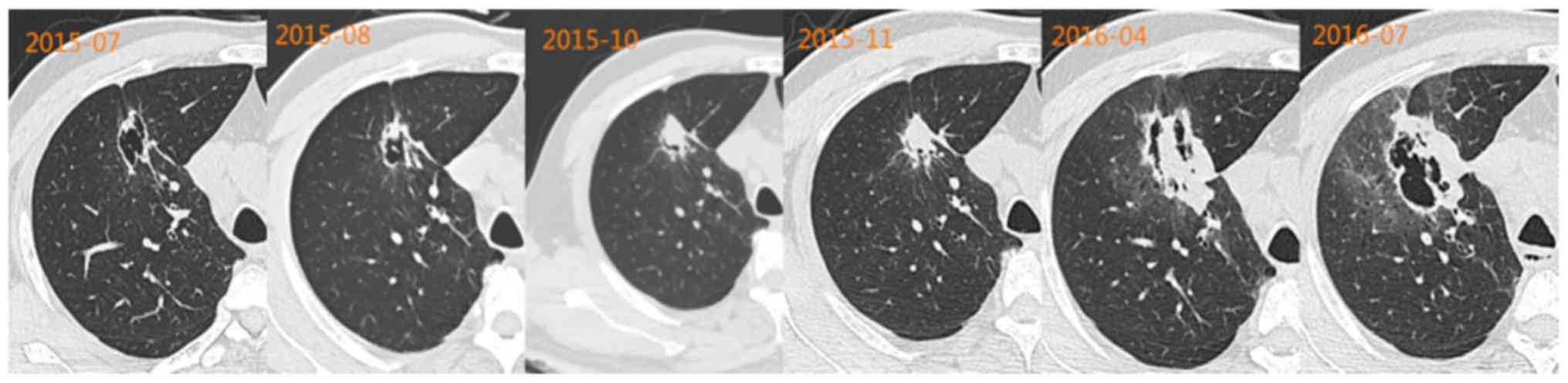
Imaging evolution
One 60-year-old patient who had interstitial lung
disease presented with a small cystic lesion on CT images in
January 2016. A chest CT performed after a month revealed that the
cystic lesion had become larger, and its wall had become slightly
irregularly thickened, compared with the previous CT findings.
Close clinical follow-up on a monthly basis was recommended for the
patient. Over the following two months, the cystic lesion grew
larger than previously and seemed to be uniform in size, but its
wall became irregularly thickened with certain malignant
observations, including irregular margin, small spicules, signs of
pleural indentation, ground-glass opacity and clear nodules. This
cystic lesion was suspected to be a malignant lesion. Finally,
imaging examinations revealed that the cystic lesion was getting
smaller and the wall was getting thicker (Fig. 4).
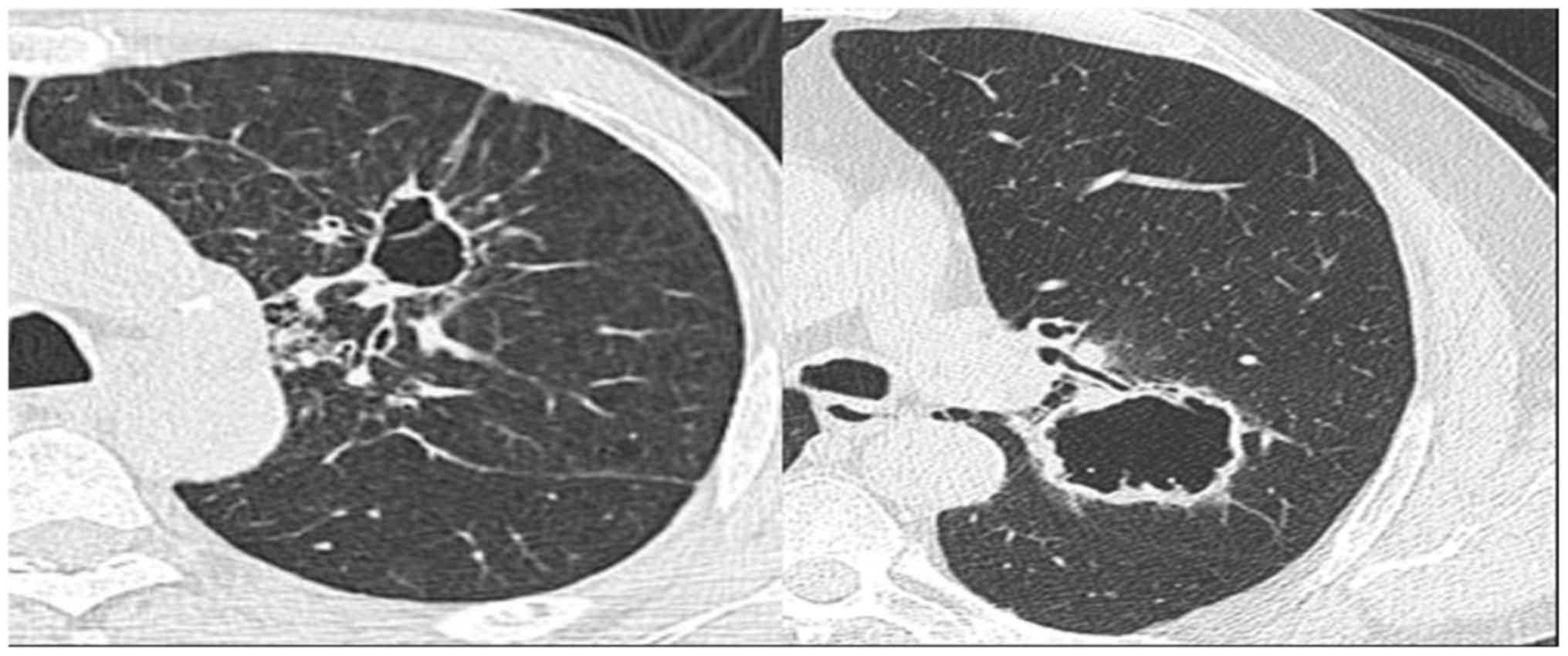
Imaging evolution
A cystic lesion was identified in the right upper
lung of a 51-year-old patient, who had a long history of smoking
and occasional bloody sputum, when he was admitted to hospital for
fracture. PET/CT revealed a highly metabolic lesion with multiple
highly metabolic lymph nodes in the right hilum. Pathology revealed
a poorly differentiated adenocarcinoma with multiple lymph node
metastases, and the patient received Iressa (250 mg once a day).
Patients were followed up since 2015. Imaging examinations revealed
an increasingly smaller cyst and increasing parenchyma. In the
final imaging scans, formation of a new cavity was noted (Fig. 5).
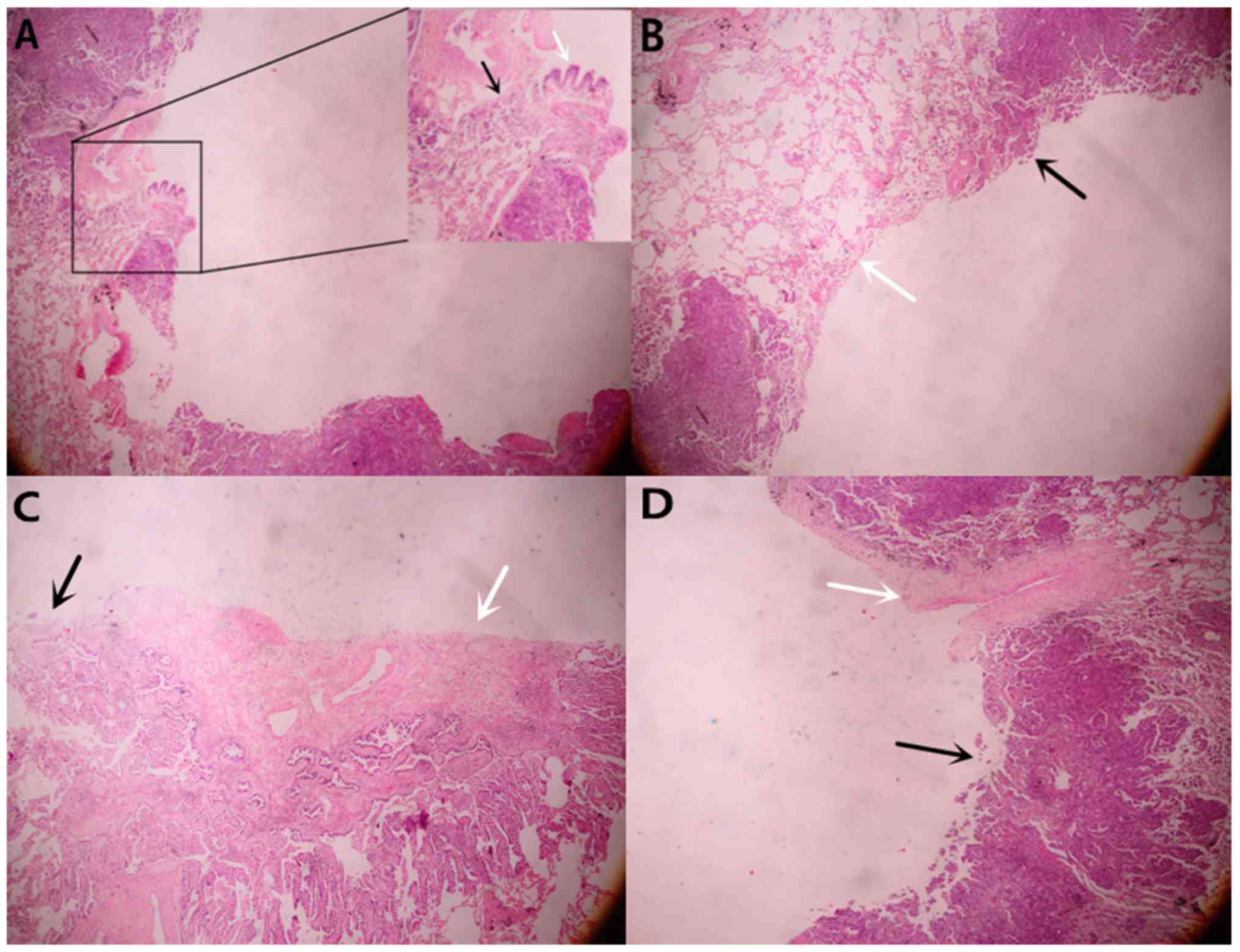
The pathological results confirmed the diagnosis of
adenocarcinoma in the 43 patients. Two patients had squamous cell
carcinoma, and also displayed cysts on the images (Fig. 6). Tumor cells (black arrow) destroyed
the wall of bronchi (white arrow) (Fig.
7A). The entire cavity wall was covered with tumor cells in
patients who underwent surgery. The black arrow represents area of
the wall covered with tumor cells and the white arrow represents
the area of the wall not covered with tumor cells (Fig. 7B). Hyperplastic fibrous tissue was
observed inside the cyst (Fig. 7C).
The blood vessels (indicated by the white arrow) blocked the
proliferation of tumor cells (Fig.
7D).
Follow-up results
Follow-up information was available for 37 patients.
The patient with metastasis to the fourth lumbar vertebrae has, to
the present date, maintained a healthy condition through oral
intake of Iressa (250 mg once a day), which has been taken for the
past year. One patient succumbed to mortality as a result of tumor
recurrence four years after surgery. One patient succumbed to
mortality as a result of intestinal obstruction, but this was not
associated with the primary lung cancer. One patient was suspected
to experience tumor recurrence half a year after surgery. The other
patients had no signs of recurrence and no symptoms following
surgery. The one who survived the longest survived for 8 years
after surgery.
Discussion
Cavities are frequently observed manifestations of a
wide variety of pathological processes involving the lung.
Cavitation detected by CT has been reported in up to 22% of primary
lung cancer cases (8), but lung
cancer presenting as a thin-wall cyst is unusual and may easily
mislead clinicians to diagnoses of other benign diseases, including
infection, tuberculosis, and emphysema. Pulmonary tuberculosis
generally presents sub-acutely, with weeks to months of a
productive cough, fever, night sweats and weight loss. The chest
radiograph typically reveals pulmonary infiltrates in the apical
and posterior segments of the upper lobe or the superior segment of
the lower lobe, often with cavitation (8). Infection often presents acutely, with
fever, cough and an increased neutrophil ratio. The cavitary lesion
observed in CT scans may shrink or disappear following the
administration of antibiotics. Patients with pulmonary emphysema
often have an identified history of bullae. In the present study,
one patient with the above malignant findings was recommended
reexamination after one month. In the following two months, the
cystic lesion became larger than previously and finally was
confirmed to be adenocarcinoma. Patients with new cystic lesions in
the lung may take monthly reexaminations in the first three months.
If cystic lesions persist, patients may receive follow-up CT scans
for a longer period. Progressive wall thickening of a cystic
airspace or the appearance of a nodule abutting a cystic airspace
should raise suspicion of lung cancer (9).
Watanabe et al (10) designated cavities as being either
thick-walled (≥4 mm) or thin-walled (<4 mm), and revealed that
the thick-wall cavities may negatively affect prognosis. In the
present study, however, all the patients presented with thin-wall
(<4 mm) cysts and three of them exhibited distant metastasis. In
the first patient, metastasis to the fourth lumbar spinal vertebra
was identified by whole-body bone scan, which was confirmed as
metastatic adenocarcinoma from the lung by the pathological
findings following PNW. Pathological findings of the second patient
following bronchoscopic biopsy confirmed these cystic lesions as
intrapulmonary metastatic adenocarcinoma. Pathology confirmed that
the third patient exhibited poorly differentiated adenocarcinoma
with multiple lymph node metastases, meaning that they had missed
the optimum time for surgery. Therefore, thin-wall cystic lung
cancer requires more attention. Missing the optimum opportunity for
surgical treatment results in a markedly diminished quality of
life. Two patients who had missed the optimum time for surgical
treatment were followed up and it was revealed that the cyst was
getting smaller and the parenchyma was increasing in size.
Thin-wall cystic lung cancer is most common in
adenocarcinomas. Xue et al (11) reported that thin-wall cystic lesions
were detected in 15/18 patients with moderately- or
well-differentiated adenocarcinoma. Qi et al (12) also reported 16 cases of
adenocarcinoma. In the present study, a 57-year-old man presented
with primary thin-wall cystic squamous cell carcinoma. Initially he
was diagnosed with tuberculosis cysts, but a bronchoscopic biopsy
identified it as poorly-differentiated squamous cell carcinoma. The
lesion displayed the suspected malignant signs of lung cancer,
including asymmetric thickening of the wall, short spicules,
lobulation and irregular margins. However, no necrosis was observed
inside the lesion. This case indicates that thin-wall cystic
lesions are not limited to adenocarcinoma.
The follow-up results were retrospectively analyzed.
Only one patient died of recurrent tumor four years after surgery.
One patient died of another disease. One patient was suspected to
have tumor recurrence half a year after surgery. Early surgical
treatment for primary thin-wall cystic lung cancer may, in
practice, obtain a good outcome.
A check-valve mechanism is widely accepted (13–16). The
check-valve is difficult to observe in pathological sections. Out
of 45 patients in the present study, tumor cell infringed
bronchiolar walls were only observed in 3 patients, which may alter
bronchial structure and lead to obstruction and collapse of the
trachea. We hypothesized that with air in the trachea, enclosure
will be formed during expiration, forming a one-way flutter valve.
The hyperplastic fibrous tissue maintains a certain tension within
the cyst.
In conclusion, the results of the present study
indicated that patients who present with new cystic lesions in the
lung may take monthly reexaminations in the first three months. The
malignant radiographic signs may be valuable indications for early
accurate diagnosis. This type of lung cancer is not limited to
adenocarcinoma. A total of 6 patients underwent PET/CT scans, 5 of
which exhibited normal values. Due to the small number of patients,
the results of the present study could not be statistically
analyzed. A total of 41 of these patients survived following
surgery. However, metastasis may occur in thin-wall cystic lung
cancer. If the patients missed the optimum time for surgical
treatment, their quality of life may be markedly reduced. In the
present study, the imaging features, diagnosis, pathology,
metastasis and prognosis of the disease are systematically
described. Prognosis and confirmed metastasis in pathology have not
been previously reported. The present study will permit an improved
understanding of the disease. To begin with, 45 cases of thin-wall
cystic lung cancer were assessed in the present study, a number
that is greater than that in aforementioned studies (11–13,15,16).
Secondly, the present study demonstrated that squamous cell
carcinoma may also manifest as a thin-wall cystic lung cancer.
Thirdly, the metastases were confirmed by pathology. Finally, the
development of thin-wall cystic lung cancer was observed in imaging
in the present study. Future studies should incorporate more cases
for data analysis.
Acknowledgements
Not applicable.
Funding
The present study was supported by Beijing
Outstanding Young Talent Fund (grant no. 2014000021469G253) and the
National Natural Science Fund Youth Project (grant no. 81700007).
Additionally, the study was supported by a special fund from the
Railway Head Corporation (grant no. J2015C001-B).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
HD is responsible for writing this paper and
collecting data. CCW and JG are responsible for the pathology and
image review, and JYZ, JZ, SZ, HJ, XLC, DXW, LP, YW and XYX were
responsible for the collection of data.
Ethics statement and consent to
participate
The present study was conducted in compliance with
the institutional policy regarding the protection of patient
confidential information and was approved by the Research Ethics
Committee of Beijing Shijitan Hospital affiliated to Capital
Medical University. All procedures were performed in accordance
with the approved guidelines of Beijing Shijitan Hospital
affiliated to Capital Medical University.
Consent for publication
Patients provided written informed consent for the
publication of their data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chen Z: Third national retrospect
port-check of death-causation. 1st edition. Peking Union Medical
College Press; Beijing, China: pp. 153–154. 2008
|
|
2
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Woodring JH: Pitfalls in the radiologic
diagnosis of lung cancer. AJR Am J Roentgenol. 154:1165–1175. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Matsuoka T, Fukamitsu G, Onoda M, Uesugi
N, Kawano K and Katou T: Synchronous multiple lung cancer including
a lesion with a thin-wall cavity; report of a case. Kyobu Geka.
63:164–167. 2010.(In Japanese). PubMed/NCBI
|
|
6
|
Isobe K, Hata Y, Iwata M, Ishida F,
Kaburaki K, Gocho K, Kobayashi M, Sakaguchi S, Satou D, Sano G, et
al: An autopsied case of mucinous bronchioloalveolar carcinoma
associated with multiple thin-wall cavities. Nihon Kokyuki Gakkai
Zasshi. 47:512–517. 2009.(In Japanese). PubMed/NCBI
|
|
7
|
Sekine A, Hagiwara E, Ogura T, Sato T,
Shinohara T, Baba T, Endo T, Sogo Y, Nishihira R, Komatsu S, et al:
A case of lung adenocarcinoma with gradual enlargement of thin-wall
cavity causing pneumothorax. Nihon Kokyuki Gakkai Zasshi.
46:552–557. 2008.(In Japanese). PubMed/NCBI
|
|
8
|
Gadkowski LB and Stout JE: Cavitary
pulmonary disease. Clin Microbiol Rev. 21:305–333. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mascalchi M, Attinà D, Bertelli E,
Falchini M, Vella A, Pegna AL, Ambrosini V and Zompatori M: Lung
cancer associated with cystic airspaces. J Comput Assist Tomogr.
39:102–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Watanabe Y, Kusumoto M, Yoshida A,
Shiraishi K, Suzuki K, Watanabe SI and Tsuta K: Cavity wall
thickness in solitary cavitary lung adenocarcinomas is a prognostic
indicator. Ann Thorac Surg. 102:1863–1871. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xue X, Wang P, Xue Q, Wang N, Zhang L, Sun
J, Wang K, Yang B and Wang J: Comparative study of solitary
thin-wall cavity lung cancer with computed tomography and
pathological findings. Lung Cancer. 78:45–50. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Qi Y, Zhang Q, Huang Y and Wang D:
Manifestations and pathological features of solitary thin-wall
cavity lung cancer observed by CT and PET/CT imaging. Oncol Lett.
8:285–290. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sugimoto Y, Semba H, Fujii S, Furukawa E
and Kurano R: Clinical analysis of primary lung cancer with a
thin-wall cavity to explain the mechanism of thin-wall cavity
formation. Nihon Kokyuki Gakkai Zasshi. 45:460–464. 2007.(In
Japanese). PubMed/NCBI
|
|
14
|
Kim TS, Koh WJ, Han J, Chung MJ, Lee JH,
Lee KS and Kwon OJ: Hypothesis on the evolution of cavitary lesions
in nontuberculous mycobacterial pulmonary infection: Thin-section
CT and histopathologic correlation. Am J Roentgenol. 184:1247–1252.
2005. View Article : Google Scholar
|
|
15
|
Lu M, Zhu X, Liu C, Cao B, Yao W and Chen
Y: Squamous cell carcinoma presenting as a refilled thin-wall
cavity in lung: A case report. Clin Respir J. 10:520–523. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Iwata T, Nishiyama N, Nagano K, Izumi N,
Tsukioka T, Hanada S, Kimura T, Kudoh S, Hirata K and Suehiro S:
Squamous cell carcinoma presenting as a solitary growing cyst in
lung: A diagnostic pitfall in daily clinical practice. Ann Thorac
Cardiovasc Surg. 15:174–177. 2009.PubMed/NCBI
|