Introduction
Lymphoma is a type of blood cell tumor that arises
from developing lymphatic cells (1);
it represents 3–4% of all types of cancer, making it the seventh
most common type of cancer (2). The
disease is characterized by painless enlarged lymph nodes (3). There are dozens of subtypes of lymphoma
(4); the two primary categories of
lymphoma are Hodgkin's lymphoma and non-Hodgkin's lymphoma, with
~90% of lymphoma cases non-Hodgkin lymphoma (5). Recently, in the USA, the 5-year survival
rate for non-Hodgkin's lymphoma is 69%, and for the Hodgkin's
lymphoma subtypes it is 85% (6). In
2012, there were 566,000 cases of lymphoma and 305,000 incidences
of mortality worldwide (7). In order
to develop therapies to reduce these numbers, it is important to
investigate effective therapeutic strategies for this disease.
MicroRNAs (miRNAs/miRs) are short non-coding RNAs,
which have previously been demonstrated to serve an important role
in cell proliferation and differentiation (8,9). Notably,
miRNAs have important functions in lymphoma biology. For example,
miR-155 and miR-34a are crucial to the development of B-cell
lymphoma via participation in numerous signaling pathways (nuclear
factor κB, phosphoinositide 3-kinase/protein kinase B and
transforming growth factor β) fundamental to B-cell development
(10). miR-376a has been demonstrated
to serve crucial roles in various diseases, including
hepatocellular carcinoma (11) and
melanoma (12); however, only a small
number of studies have assessed its role in lymphoma (13,14). At
present, the volume of data concerning the functional role of
miRNAs in lymphoma is increasing steadily (15). A previous study by Arribas et
al (13) analyzed the miRNA
profiles of 31 splenic marginal zone lymphoma cases, and identified
that miR-376a was abnormally expressed in lymphoma. However, the
exact mechanism of the effect of miR-376a on lymphoma remains
unclear.
The present study aimed to investigate miR-376a
expression in lymphoma and the associations between aberrantly
expressed miR-376a and cell proliferation or apoptosis at the
cytological and molecular level. The present study may provide
novel insights into the molecular mechanisms underlying lymphoma
development.
Patients and methods
Patients and samples
Between January 2015 and February 2016, a total of
29 patients (20 males and 9 females, age range, 24–44 years, mean
age, 33.6±10.4 years) who were diagnosed with lymphoma in the
Cancer Hospital affiliated to Zhengzhou University, Henan Cancer
Hospital (Zhengzhou, China) were enrolled in the present study.
Among the 29 patients, 5 cases were Hodgkin's lymphoma and 24 cases
were non-Hodgkin's lymphoma. The diagnosis of lymphoma was
pathologically defined according to World Health Organization
classification of lymphoid neoplasms (16). The pathological lymphoid tissues were
collected from patients using resection, extracted and immediately
snap-frozen in liquid nitrogen. Additionally, 12 patients (8 males
and 4 females, age range, 21–42 years, mean age, 30.8±9.6 years)
with benign hyperplastic lymphadenitis were enrolled as controls.
All patients provided written informed consent and the present
study was approved by the Protection of Human Ethics Committee of
the Cancer Hospital affiliated to Zhengzhou University, Henan
Cancer Hospital.
Cell culture and transfection
The human mantle cell lymphoma (MCL) JeKo-1 cell
line (CRL-3006™) and normal lymphocytes
(CRL-2319™), which were obtained from the American Type
Culture Collection (Manassas, VA, USA), were cultured in RPMI-1640
medium (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
supplemented with 10% fetal bovine serum (FBS; Gibco; Thermo Fisher
Scientific, Inc.), 2 mM L-Glutamine, 50 units penicillin and 50
µg/ml streptomycin (Sigma-Aldrich; Merck KGaA).
Cell transfections were performed using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol.
miR-376a mimic, inhibitor and scramble, and small interfering RNA
(siRNA) targeting Forkhead box P2 (FOXP2) were synthesized by
Sangon Biotech Co., Ltd. (Shanghai, China). Cells were transfected
with 50 nM miR-376a mimic, 150 nM miR-376a inhibitor and 4 µg
si-FOXP2 for 24 h prior to subsequent experimentation. The primer
sequences were as follows: miR-376a mimic sense,
5′-GUAGAUUCUCCUUCUAUGAGUA-3′ and anti-sense,
5′-CUCAUAGAAGGAGAAUCUACUU-3′; miR-376a inhibitor
5′-UACUCAUAGAAGGAGAAUCUAC-3′; si-FOXP2 sense,
5′-CGACAGAGACAAUAAGCAACA-3′ and antisense,
5′-UUGCUUAUUGUCUCUGUCGCA-3′.
Cell proliferation assay
Cell proliferation ability was assessed using an MTT
assay. At 24 h after transfection, cells were cultured in RPMI-1640
medium supplemented with 10% FBS. JeKo-1 cells were seeded into the
96-well plates at a density of 5×103 cells/well. At
various time points (24, 36, 48, 72 and 96 h), cells were
centrifuged at 6,000 × g for 5 min at 4°C, and the supernatant was
removed. Next, 20 µl 5 mg/ml MTT solution was added into the
culture medium and cultured for 4 h at room temperature. Finally,
150 µl dimethyl sulfoxide was added to each well to dissolve the
crystals. Following 10 min, the absorbance of cells in each well
was observed at 570 nm using an absorption spectrophotometer
(Olympus Corporation, Tokyo, Japan). All experiments were conducted
independently in triplicate.
Clonogenic assay
A clonogenic assay was performed as described
previously (14), with minor
modifications. Briefly, 24 h following transfection, JeKo-1 cells
were plated in triplicate into the 60-mm tissue culture dishes at a
density of 100 cells/dish. After 14 days of culture in RPMI-1640
containing 10% FBS, cells were fixed with 90% methanol for 15 min
at room temperature, and stained with the Diff-Quick cell staining
kit (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) for 20–40 sec
at room temperature. Colonies were counted under an inverted
microscope (IX83; Olympus Corporation; magnification, ×200), and
colonies consisting of at least 30 cells were counted as a single
colony.
Apoptosis assay
Cell apoptosis was detected by flow cytometry. The
number of apoptotic JeKo-1 cells was determined using an Annexin
V-fluorescein isothiocyanate (FITC) cell apoptosis kit (Invitrogen;
Thermo Fisher Scientific, Inc.) according to the manufacturer's
protocol. A total of 24 h following cell transfection, JeKo-1 cells
were cultured in serum-free RPMI-1640 medium. The cells were
harvested and washed 3 times using PBS buffer (pH 7.4), and then
resuspended in the staining buffer. Subsequently, 5 µl
Annexin-V-FITC and 5 µl propidium iodide (PI) were added into the
cells and incubated at room temperature for 10 min. The mixtures
were analyzed using a FACScan flow cytometer (BD Biosciences,
Franklin Lakes, NJ, USA). Annexin V-positive and PI-negative cells
were considered to be apoptotic cells. The apoptotic cells were
analyzed using Cellquest software (version 3.0; BD
Biosciences).
Target prediction
Putative target genes of miR-376a were predicted by
bioinformatics analysis using TargetScan (version 7; http://www.targetscan.org/vert_71/).
Luciferase reporter analysis
Luciferase reporter plasmid, psiCHECK2, containing
the FOXP2 3′-untranslated region (UTR), miR-376a mimic and scramble
were produced and synthesized by Sangon Biotech Co., Ltd. Cell
transfections were performed using Lipofectamine® 2000
(Invitrogen; Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. At 48 h after transfection, luciferase
activities were measured using the Dual-Luciferase Reporter Assay
system (Promega Corporation, Madison, WI, USA). The relative
reporter activity was normalized to Renilla luciferase
activity.
Reverse transcription quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from tissues and JeKo-1
cells using TRIzol® (Life Technologies; Thermo Fisher
Scientific, Inc.) and treated with RNase-free DNase I (Promega
Corporation). The concentration and purity of isolated RNA were
measured with SMA400 UV–VIS (Merinton Instrument, Ltd., Shanghai,
China). cDNA was then generated with the PrimerScript 1st Strand
cDNA Synthesis kit (Invitrogen; Thermo Fisher Scientific, Inc.).
qPCR was performed using a standard SYBR Green I kit (Takara
Biotechnology Co., Ltd., Dalian, China). PCR was performed under
the following conditions: 1 predenaturation cycle for 1 min at
95°C, 36 cycles at 95°C for 15 sec, 60°C for 30 sec and 72°C for 1
min and a final extension at 72°C for 5 min. U6 and GAPDH were used
as the endogenous controls for the normalization of the amount of
miRNA and total mRNA. The expression levels were calculated using
the 2−ΔΔCt method (17).
Sequences of all primers used are summarized in Table I.
 | Table I.Primers used for target
amplification. |
Table I.
Primers used for target
amplification.
| Gene name | Forward primer,
5′-3′ | Reverse primer,
5′-3′ |
|---|
| FOXP2 |
GAAGACAATGGCATTAAACATGGAGG |
GAATAAAGCTCATGAGATTTACCTGTC |
| Cyclin A |
GACTGGTTAGTTGAAGTA |
CTCCATTCTCAGAACTTG |
| Cyclin B |
TAGTTCCATCAGGTATTTGGC |
TAGTTCCATCAGGTATTTGGC |
| Cyclin D2 |
CTGTGTGCCACCGACTTTAAGTT |
GATGGCTGCTCCCACACTTC |
| Bax |
CTGAGCTGACCTTGGAGC |
GACTCCAGCCACAAAGATG |
| Bcl-2 |
CTGGTGGACAACATCGCTCTG |
GGTCTGCTGACCTCACTTGTG |
| GAPDH |
TGGACTCCACGACGTACTCAG |
CGGGAAGCTTGTCATCAATGGAA |
| U6 |
GCTTCGGCAGCACATATACTAA |
AACGCTTCACGAATTTGCGT |
Western blot analysis
The protein expression levels of FOXP2, cyclin A,
cyclin B, cyclin D2 and GAPDH were determined in total cell
extracts from transfected JeKo-1 cells. Total proteins were
extracted using radioimmunoprecipitation assay buffer (Sangon
Biotech Co., Ltd.) and then the cell lysates were centrifuged at
6,000 × g for 10 min at 4°C and separated using 10% SDS-PAGE.
Protein concentration was determined using the bicinchoninic acid
assay (Pierce; Thermo Fisher Scientific, Inc.). Then, proteins (20
µg/per lane) were transferred onto the polyvinylidene difluoride
membranes (EMD Millipore, Billerica, MA, USA) and probed with the
following primary antibodies; FOXP2 (ab16046; dilution 1:1,000),
Bcl2 (ab32124; dilution 1:1,000), Bax (ab32503; dilution 1:1,000),
cyclin A (ab181591; dilution 1:1,000), cyclin B (ab32053; dilution
1:1,000) and cyclin D2 (ab207604; dilution 1:1,000) and GAPDH
(ab8245; dilution 1:5,000) at 4°C overnight. All primary antibodies
were purchased fro Abcam (Cambridge, UK). Protein bands were
blocked using 5% non-fat dry milk in Tris buffered saline (TBS)
supplemented with 0.05% TBS and Tween for 1 h at room temperature.
Membranes were then incubated with peroxidase-conjugated goat
anti-rabbit (ab6721; dilution 1:5,000; Abcam) for 1 h at room
temperature. The immunoreactive protein bands were developed using
the enhanced chemiluminescence kit (Thermo Fisher Scientific,
Inc.).
Statistical analysis
Statistical analyses were performed using SPSS 19.0
statistical software (IBM Corp., Armonk, NY, USA). Data were
expressed as the mean ± standard deviation from 3 independent
experiments. One way analysis of variance followed by post hoc
Tukey's test was used to calculate the difference >2 groups.
Student's t-test was used to calculate the difference between two
groups. P<0.05 was considered to indicate a statistically
significant difference.
Results
miR-376a is downregulated and FOXP2 is
upregulated in lymphoma tissues and cells
The expression levels of miR-376a and FOXP2 in
lymphoma tissues were measured in 29 patients with lymphoma and in
lymphoid tissues from 12 patients with benign hyperplastic
lymphadenitis by RT-qPCR and western blot analysis. Additionally,
miR-376a expression levels were also detected in lymphoma B cells
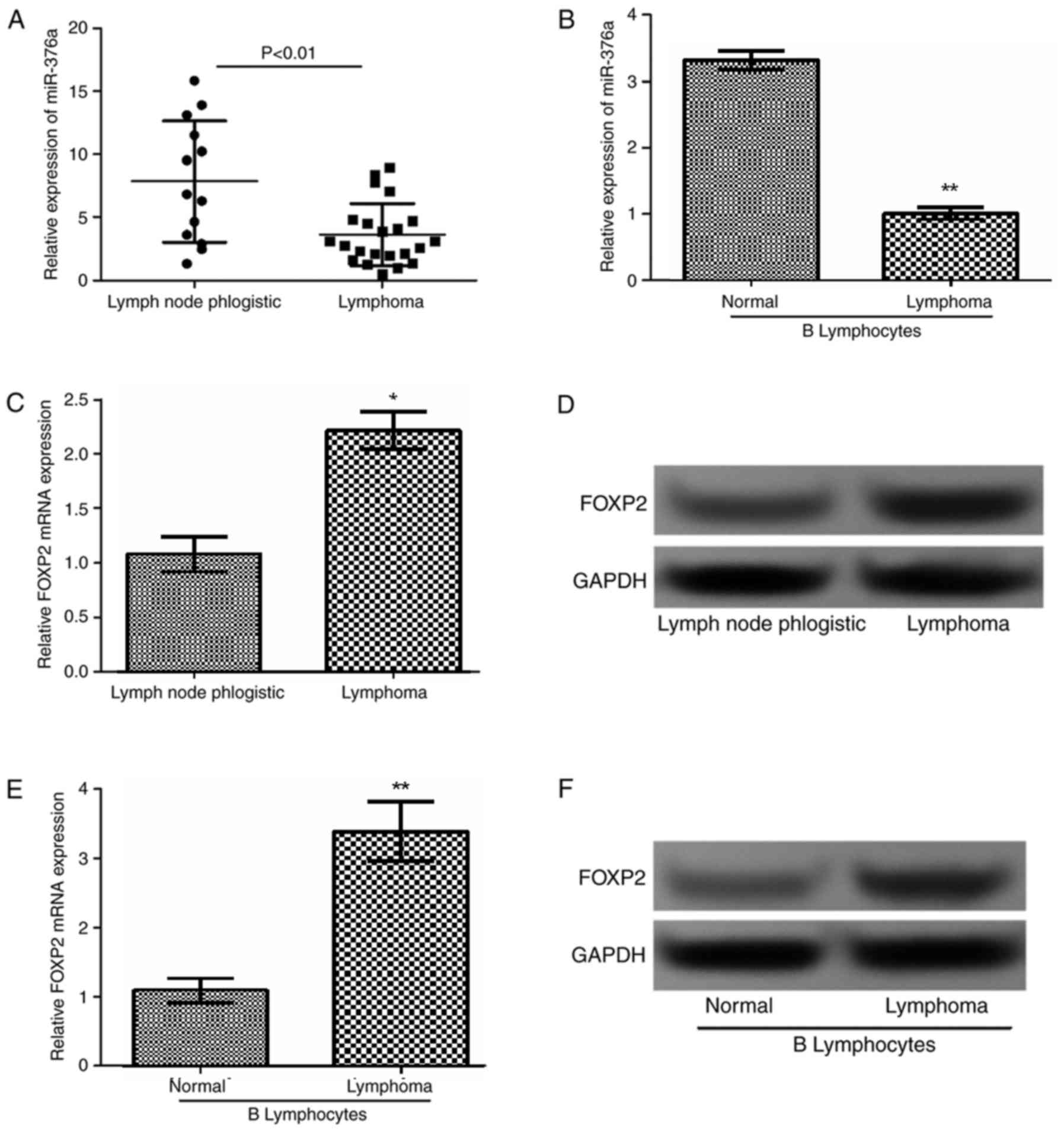
and normal B lymphocytes. As demonstrated in Fig. 1A and B, miR-376a was downregulated in
lymphoma tissues and cells compared with the corresponding controls
(P<0.01). Additionally, as revealed in Fig. 1C-F, FOXP2 expression in lymphoma
tissues and cells was significantly increased compared with that in
normal tissues (P<0.01) and cells (P<0.05).
miR-376a overexpression reduces cell
proliferation in B lymphocytes
The associations between miR-376a and lymphoma were
analyzed at a cytological level. The MCL JeKo-1 cell line was
transfected with miR-376a mimic, inhibitor and scramble plasmids.
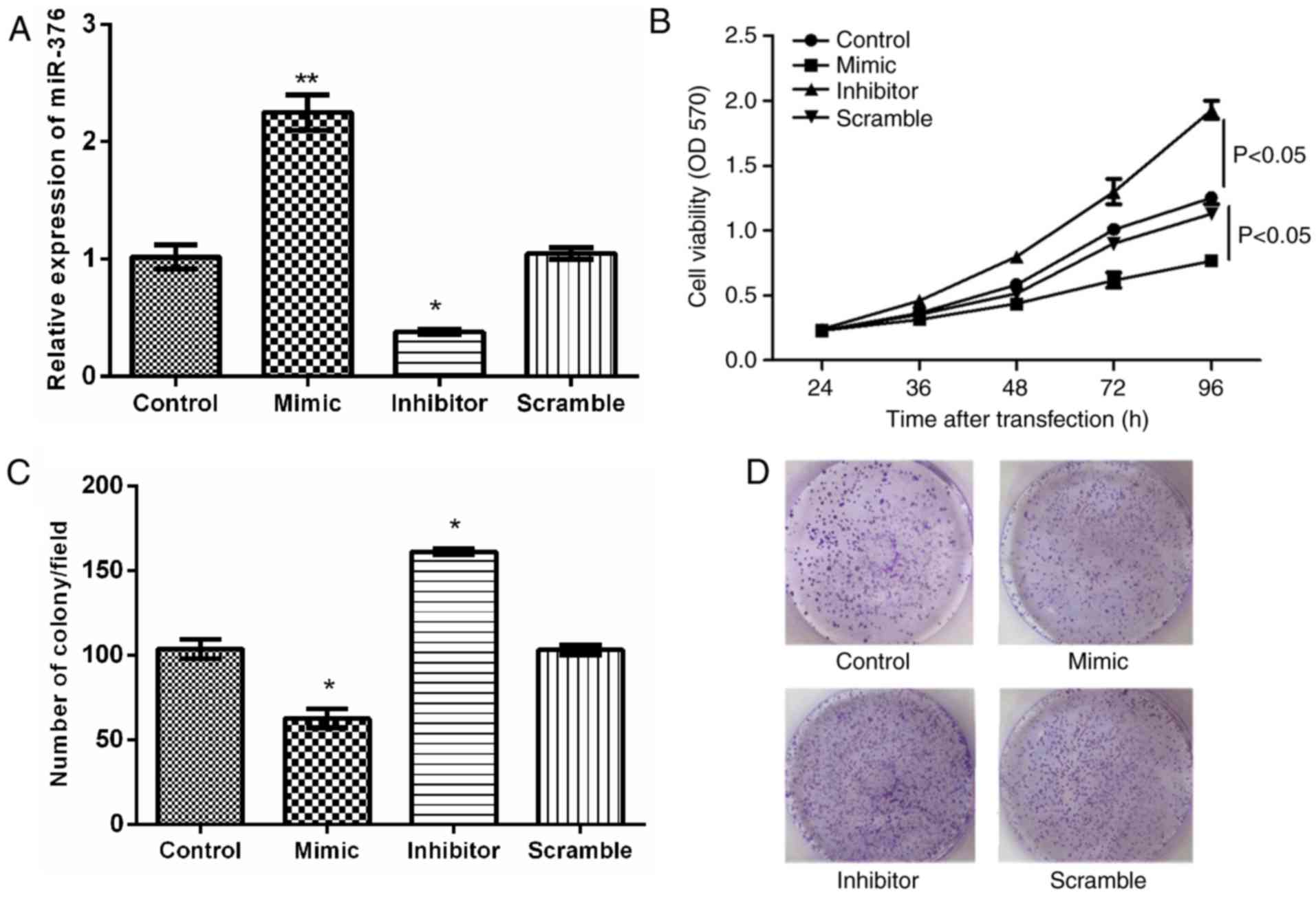
The expression levels of miR-376a in JeKo-1 cells following
transfection are depicted in Fig. 2A.
To validate the effect of miR-376a on JeKo-1 cell proliferation,
MTT and clonogenic assays were used. As indicated in Fig. 2B-D, miR-376a overexpression
significantly inhibited cell proliferation and colony formation
compared with the control group (P<0.05).
miR-376a overexpression induces cell
growth arrest by downregulating cyclin D2 and cyclin A
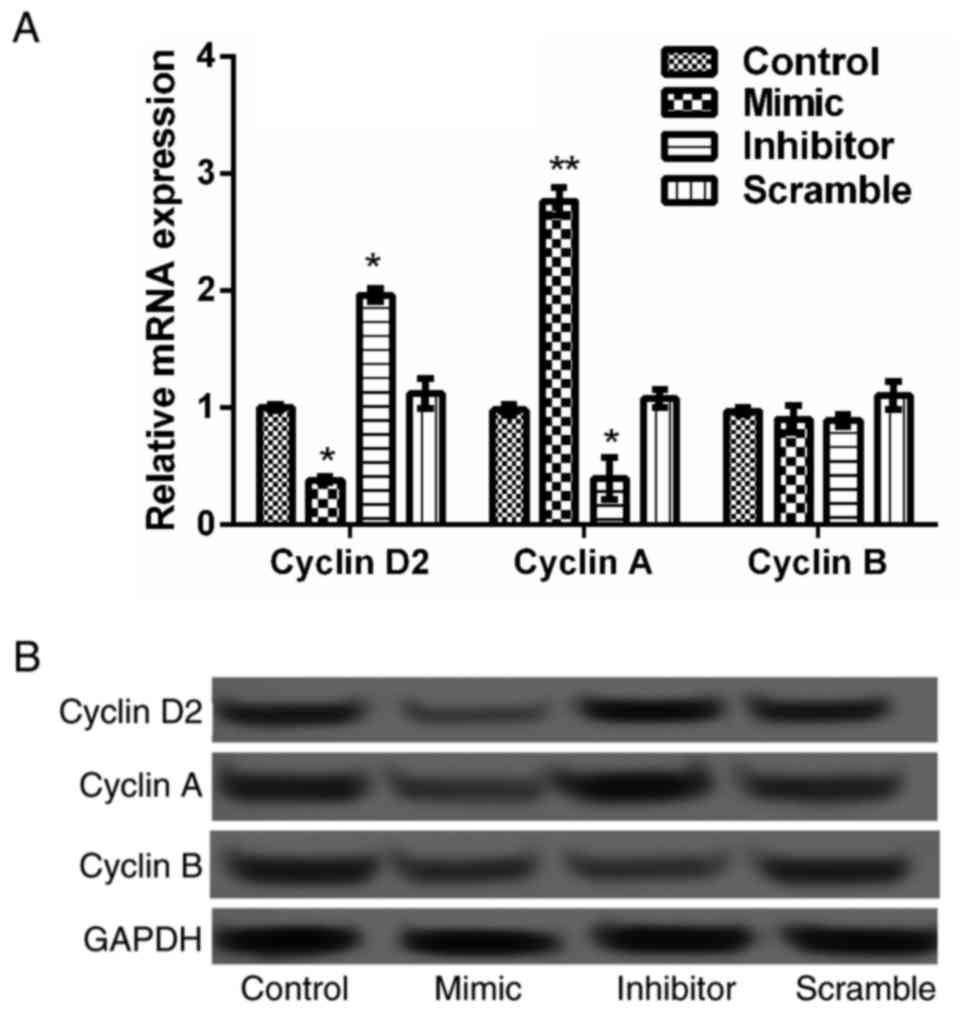
A previous study suggested that cyclin
overexpression was a cause of excessive cell proliferation
(18). Therefore, the expression
levels of cyclin D2, cyclin A and cyclin B were investigated in
transfected JeKo-1 cells. As indicated in Fig. 3, the expression levels of cyclin D2
and cyclin A were significantly increased in the miR-376a inhibitor
group compared to the scramble group (P<0.05), and significantly
decreased in the miR-376a mimic group compared with the control
group (P<0.05 and P<0.01, respectively). For the expression
of cyclin B, no significant difference was identified among the
four groups. This result indicated that miR-376a overexpression
could inhibit the proliferation of lymphoma B cells by
downregulating expression of cyclin D2 and cyclin A.
miR-376a overexpression induces
apoptosis in B lymphocytes
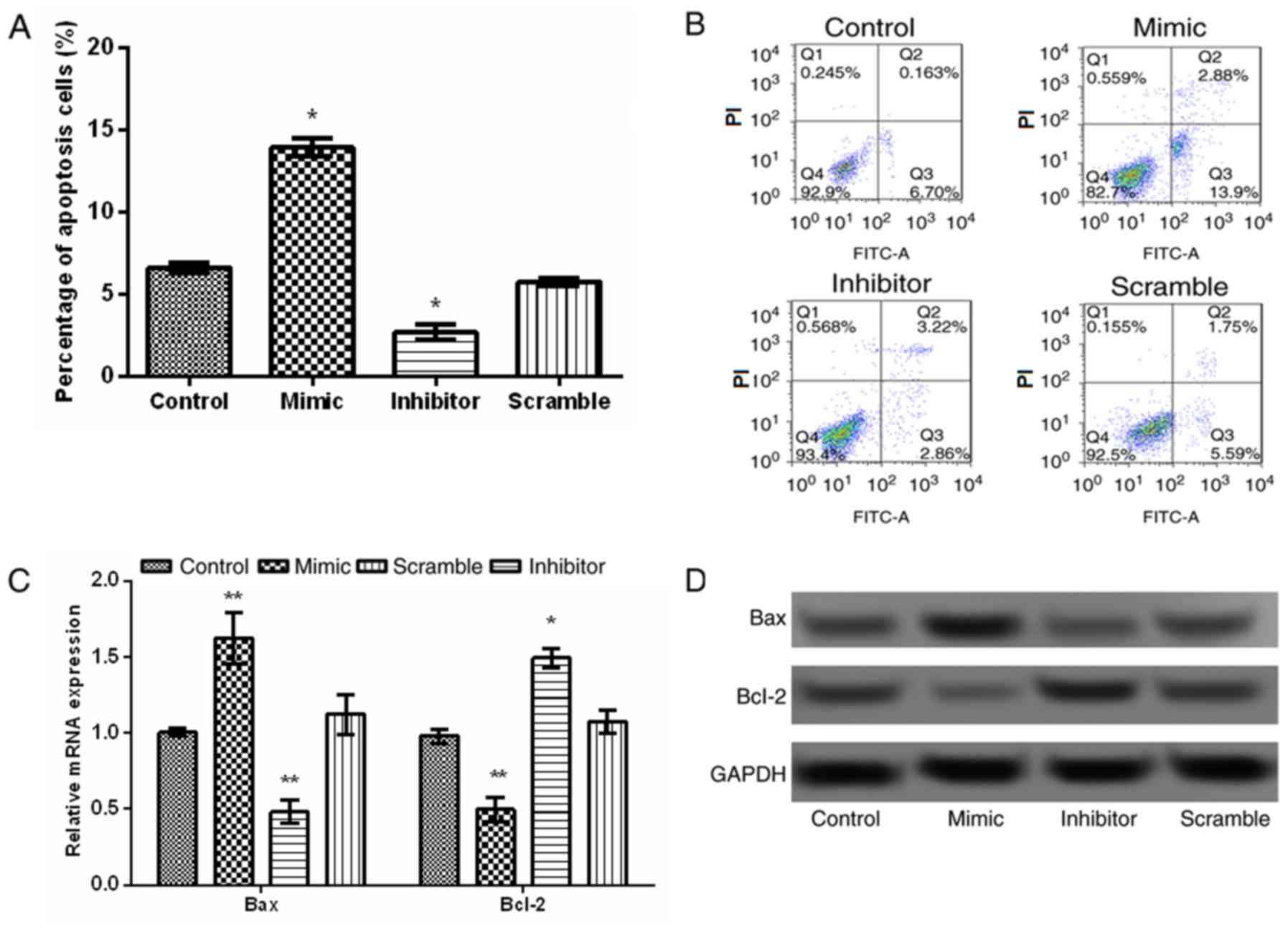
To investigate the effect of miR-376a on apoptosis,
flow cytometry was performed. Additionally, the expression levels
of apoptosis-associated proteins, including Bcl-2 and Bax in JeKo-1
and transfected JeKo-1 cells were detected by RT-qPCR and western
blot analysis. The result of flow cytometry analysis indicated that
the overexpression of miR-376a markedly increased the percentage of
apoptotic cells, compared with the control group, whereas miR-376a
suppression significantly inhibited the percentage of apoptotic
cells, compared with the scramble group (both P<0.05; Fig. 4A and B). The expression levels of Bax
and Bcl-2 are depicted in Fig. 4C and
D. The expression of Bax increased significantly, whereas that
of Bcl-2 decreased significantly in the miR-376a mimic group
compared with the control group (P<0.01), additionally
indicating the pro-apoptotic effect of miR-376a overexpression.
miR-376a overexpression induces
apoptosis by targeting FOXP2
FOXP2 was identified to be a potential target gene
of miR-376a by bioinformatic prediction (Fig. 5A). Analysis of the relative luciferase
activities of FOXP2 identified that the relative luciferase
activity of the reporter containing the wild-type 3′-UTR of FOXP2
was significantly reduced in the miR-376a-overexpression group
compared with the control (P<0.05; Fig. 5B). Concurrently, RT-qPCR and western
blot analysis identified that the relative expression levels of
FOXP2 significantly decreased when miR-376a was overexpressed
compared with the control group and significantly increased when
miR-376a was suppressed compared with the scramble group (both
P<0.05; Fig. 5C), indicating that
miR-376a may negatively regulate the expression of FOXP2.
Subsequently, a siRNA targeting FOXP2 was transfected into JeKo-1
cells. As indicated in Fig. 5D, the
expression of FOXP2 decreased significantly in the FOXP2-siRNA
group compared with the control group (P<0.01). Additionally,
the apoptosis assay identified that, compared with the
miR-376a-overexpression group, the percentage of apoptosis cells
significantly increased in the FOXP2 siRNA/miR-376a-overexpression
group (P<0.05; Fig. 5E and F),
indicating that miR-376a overexpression induced apoptosis by
targeting FOXP2.
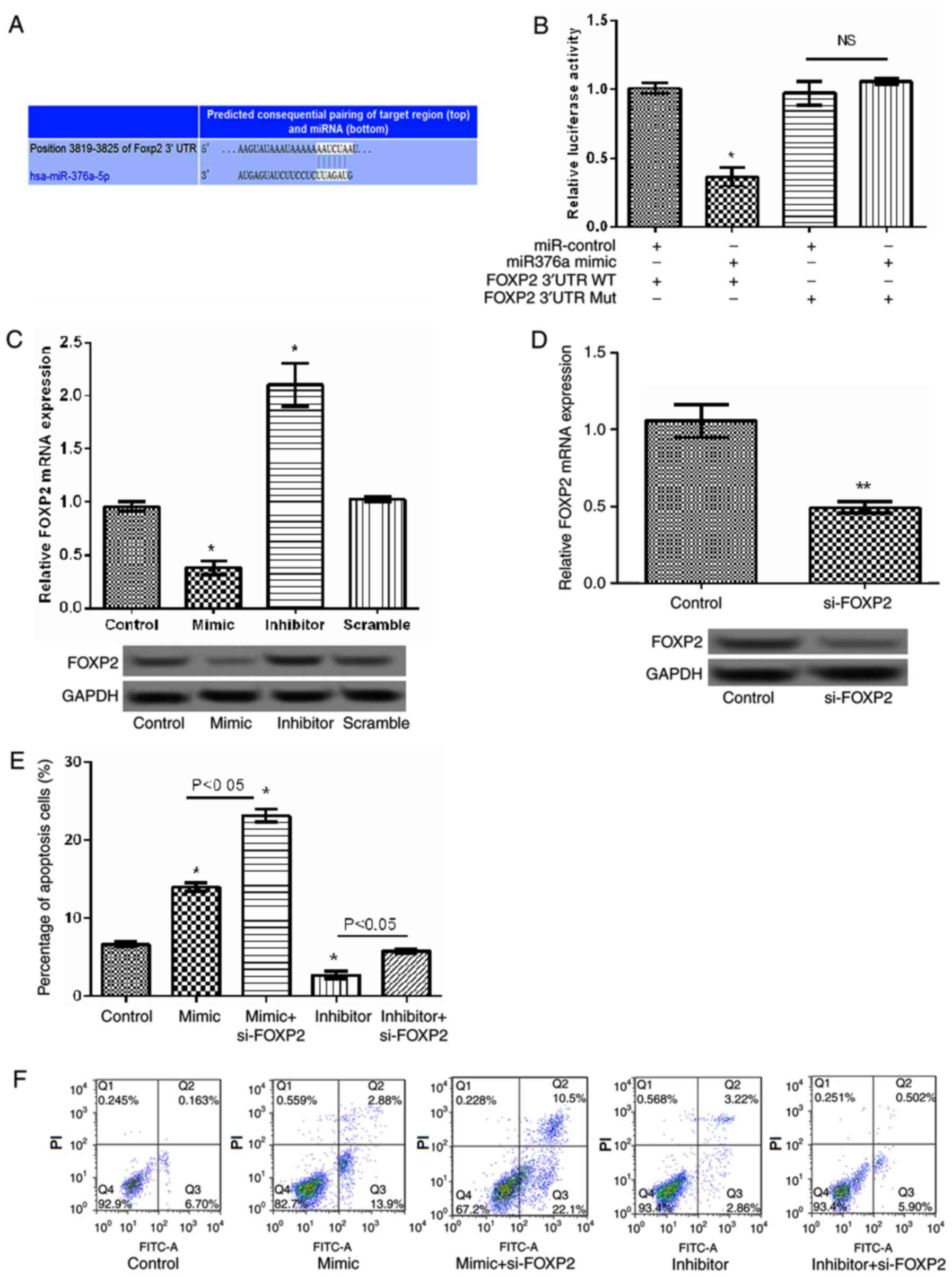 | Figure 5.miR-376a regulated cell apoptosis by
targeting FOXP2 in lymphoma. (A) The gene sequences of FOXP2
regulated by miR-376a. (B) The relative luciferase activities in
wild-type 3′-UTR of FOXP2 and mutant FOXP2 3′-UTR in transfected
cells. (C) The relative mRNA and protein levels of FOXP2 in
transfected cells detected by RT-qPCR and western blot analysis.
(D) The relative mRNA and protein expression of FOXP2 following
cell transfection with si-FOXP2 detected by RT-qPCR and western
blot analysis. (E) Percentage of apoptotic cells. (F)
Representative images of the apoptosis assay following cell
transfection with miR-376a overexpression vectors and si-FOXP2.
Data were expressed as mean ± standard deviation (SD). *P<0.05
and **P<0.01 vs. control. miR-376a, microRNA-376a; RT-qPCR,
reverse transcription quantitative polymerase chain reaction;
FOXP2, forkhead box protein P2; 3′-UTR, 3′-untranslated region;
si-FOXP2, small interfering RNA targeting FOXP2; FITC, fluorescein
isothiocyanate; PI, propidium iodide; WT, wild type; Mut, mutant;
NS, not significant. |
Discussion
The present study identified that miR-376a was
downregulated in lymphoma tissues and cells compared with the
corresponding controls. FOXP2 was a target gene of miR-376a, which
was upregulated in lymphoma. Additional experiments identified that
miR-376a overexpression inhibited cell proliferation and induced
cell apoptosis by regulating FOXP2 expression.
Numerous studies have revealed that miRNAs are
potential biomarkers for various types of human cancer (19–21). In
the present study, miR-376a was identified to be downregulated in
lymphoma, which was in agreement with the results of a previous
study by Arribas et al (13).
The authors identified that miR-376a was also downregulated in
splenic marginal zone lymphoma. miR-376a has also been identified
to be downregulated in hepatocellular carcinoma (11). To confirm the biological function of
miR-376a in lymphoma, the effects of miR-376a on JeKo-1 cell
proliferation and apoptosis were investigated. The results of this
investigation demonstrated that miR-376a inhibited JeKo-1 cell
proliferation and induced apoptosis. Previous studies have
described the roles of miR-376a in different cancer cell lines; for
example, Zhang et al (22)
suggested that miR-376a was a potential tumor suppressor in human
epithelial ovarian cancer. In accordance with previous studies
(11,13,22), the
findings of the present study indicate that miR-376a suppresses
cell proliferation and induced apoptosis in lymphoma.
Previous studies have highlighted the oncogenic
nature of abnormal cell cycle in the tumorigenesis (23–25).
Cellular proliferation allows for the orderly progression of cells
through the cell cycle, which is regulated by certain cell cycle
regulators, including cyclins and cyclin-dependent kinases
(26,27). Abnormalities in expression levels of
these genes/proteins have been demonstrated in several tumor types
(28,29). For example, high expression levels of
cyclin D2 have been observed in testicular and ovarian tumors
(30). The data presented in the
present study identified that the expression of cyclin D2 and
cyclin A significantly increased when miR-376a was suppressed,
whereas they were decreased significantly when miR-376a was
overexpressed, additionally indicating that miR-376a suppression
may promote cell proliferation via regulating the cell
cycle-associated proteins of cyclin D2 and A in lymphoma.
Abnormal or unwanted cells will be removed during
organism development (31). The
failure of cells to undergo apoptosis is crucial for the initiation
and progression of cancer (32). The
Bcl-2 family proteins, which exhibit either pro-apoptotic (Bax) or
anti-apoptotic (Bcl-2) activities, have been studied extensively
due to their importance in the regulation of apoptosis and
tumorigenesis (33). In the present
study, to investigate the effect of miR-376a on apoptosis, the
expression levels of Bax and Bcl-2 were detected in transfected
JeKo-1 cells. It was observed that Bax expression increased
significantly in the miR-376a mimic group whereas Bcl-2 expression
decreased significantly compared with the control group
(P<0.01), indicating that miR-376a suppression may reduce the
rate of cell apoptosis by regulating Bax and Bcl-2 levels.
FOXP2 is a member of the FOXP family of
transcription factors, which has been implicated in regulating
language development and developmental neurogenesis in humans
(34,35). Notably, this family has also been
implicated in various types of human cancer. For example, FOXP1 has
been suggested to serve a role in prostate cancer (36), FOXP3 is markedly expressed in lymphoma
(37), and FOXP4 is downregulated in
kidney cancer (38). The role of
FOXP2 in cancer has not been widely studied, although Campbell
et al (39) identified that
FOXP2 was overexpressed in lymphoma. In concordance with the study
by Campbell et al (39), the
present study also identified FOXP2 to be overexpressed in
lymphoma. Therefore, whether miR-376a was regulated cell apoptosis
by targeting FOXP2 was additionally verified by transfecting JeKo-1
cells with miR-376a mimics and FOXP2 siRNA. The data generated by
the present study demonstrated that FOXP2 expression was
significantly decreased when miR-376a was overexpressed, and
significantly increased when miR-376a expression was suppressed,
indicating that miR-376a serves a regulatory effect of on FOXP2.
Furthermore, quantification of apoptosis identified that FOXP2
suppression combined with miR-376a overexpression exhibited the
most marked pro-apoptotic effect on lymphoma cells, indicating that
miR-376a overexpression induced cell apoptosis by targeting FOXP2
in lymphoma.
In conclusion, the present study indicates that
miR-376a expression is downregulated in lymphoma, whereas that of
FOXP2 is upregulated. Overexpression of miR-376a inhibited cell
proliferation and induced apoptosis by targeting FOXP2 in lymphoma.
Therefore, miR-376a and FOXP2 may be used as biomarkers of
lymphoma. However, additional experimental studies will be required
to investigate the roles of miR-376a in the progression of
lymphoma.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Scott DW and Gascoyne RD: The tumour
microenvironment in B cell lymphomas. Nat Rev Cancer. 14:517–534.
2014. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Marcus EBR, Sweetenham JW and Williams ME:
Pathology, diagnosis and treatment. Cambridge University Press;
2013, View Article : Google Scholar
|
|
3
|
Kuna T, John O, Kobler P and Filipovićzore
I: A finding of diffuse cellular non-Hodgkin lymphoma in the oral
cavity-case presentation Dg: Diffuse giant cell Non-Hodgkin
lymphoma B-immunophenotype. Acta Stomatol Croatica. 38:2792004.
|
|
4
|
Bardia A and Seifter E: Johns Hopkins
Patients' Guide to Lymphoma. Shockney L and Shapiro GR: Jones and
Bartlett; Sudbury, MA, USA: 2011
|
|
5
|
Armitage JO, Gascoyne RD, Lunning MA and
Cavalli F: Non-Hodgkin lymphoma. Lancet. 390:298–310. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Surveillance E and Program ER: SEER stat
fact sheets: Non-Hodgkin Lymphoma. 2014.
|
|
7
|
Stewart BW and Wild CP: World Cancer
Report 2014. IARC Publications; Lyon, France: 2015
|
|
8
|
Paydas S, Acikalin A, Ergin M, Celik H,
Yavuz B and Tanriverdi K: Micro-RNA (miRNA) profile in Hodgkin
lymphoma: Association between clinical and pathological variables.
Med Oncol. 33:342016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Healy NA, Heneghan HM, Miller N, Osborne
CK, Schiff R and Kerin MJ: Systemic mirnas as potential biomarkers
for malignancy. Int J Cancer. 131:2215–2222. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Musilova K and Mraz M: MicroRNAs in B-cell
lymphomas: How a complex biology gets more complex. Leukemia.
29:1004–1017. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zheng Y, Yin L, Chen H, Yang S, Pan C, Lu
S, Miao M and Jiao B: miR-376a suppresses proliferation and induces
apoptosis in hepatocellular carcinoma. FEBS Lett. 586:2396–2403.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zehavi L, Avraham R, Barzilai A, Bar-Ilan
D, Navon R, Sidi Y, Avni D and Leibowitz-Amit R: Silencing of a
large microRNA cluster on human chromosome 14q32 in melanoma:
Biological effects of mir-376a and mir-376c on insulin growth
factor 1 receptor. Mol Cancer. 11:442012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Arribas AJ, Gómez-Abad C, Sánchez-Beato M,
Martinez N, Dilisio L, Casado F, Cruz MA, Algara P, Piris MA and
Mollejo M: Splenic marginal zone lymphoma: Comprehensive analysis
of gene expression and miRNA profiling. Mod Pathol. 26:889–901.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yu X, Li Z and Liu J: MiRNAs in primary
cutaneous lymphomas. Cell Prolif. 48:271–277. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lawrie CH: MicroRNAs and lymphomagenesis:
A functional review. Br J Haematol. 160:571–581. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Swerdlow SH, Campo E, Pileri SA, Harris
NL, Stein H, Siebert R, Advani R, Ghielmini M, Salles GA, Zelenetz
AD and Jaffe ES: The 2016 revision of the World Health Organization
classification of lymphoid neoplasms. Blood. 127:2375–2390. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sicinski P, Donaher JL, Geng Y, Parker SB,
Gardner H, Park MY, Robker RL, Richards JS, McGinnis LK, Biggers
JD, et al: Cyclin D2 is an FSH-responsive gene involved in gonadal
cell proliferation and oncogenesis. Nature. 384:470–474. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Heneghan HM, Miller N, Lowery AJ, Sweeney
KJ, Newell J and Kerin MJ: Circulating microRNAs as novel minimally
invasive biomarkers for breast cancer. Ann Surg. 251:499–505. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang J, Chen J, Chang P, LeBlanc A, Li D,
Abbruzzesse JL, Frazier ML, Killary AM and Sen S: MicroRNAs in
plasma of pancreatic ductal adenocarcinoma patients as novel
blood-based biomarkers of disease. Cancer Prev Res (Phila).
2:807–813. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Garzon R, Calin GA and Croce CM: MicroRNAs
in cancer. Ann Rev Med. 60:167–179. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang L, Volinia S, Bonome T, Calin GA,
Greshock J, Yang N, Liu CG, Giannakakis A, Alexiou P, Hasegawa K,
et al: Genomic and epigenetic alterations deregulate microRNA
expression in human epithelial ovarian cancer. Proc Natl Acad Sci
USA. 105:7004–7009. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Diaz-Moralli S, Tarrado-Castellarnau M,
Miranda A and Cascante M: Targeting cell cycle regulation in cancer
therapy. Pharmacol Ther. 138:255–271. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Malumbres M and Barbacid M: Cell cycle,
CDKs and cancer: A changing paradigm. Nat Rev Cancer. 9:153–166.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Stewart ZA, Westfall MD and Pietenpol JA:
Cell-cycle dysregulation and anticancer therapy. Trends Pharmacol
Sci. 24:139–145. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cordon-Cardo C: Mutations of cell cycle
regulators. Biological and clinical implications for human
neoplasia. Am J Pathol. 147:545–560. 1995.PubMed/NCBI
|
|
27
|
Hunter T and Pines J: Cyclins and cancer
II: Cyclin D and CDK inhibitors come of age. Cell. 79:573–582.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bartkova J, Lukas J, Strauss M and Bartek
J: The PRAD-1/cyclin D1 oncogene product accumulates aberrantly in
a subset of colorectal carcinomas. Int J Cancer. 58:568–573. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Motokura T and Arnold A: Cyclins and
oncogenesis. Biochim Biophys Acta. 1155:63–78. 1993.PubMed/NCBI
|
|
30
|
Surhone LM, Tennoe MT, Henssonow SF,
Macromolecule and Acid N: Cyclin D2. 2011.
|
|
31
|
Thompson CB: Apoptosis in the pathogenesis
and treatment of disease. Science. 267:1456–1462. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Fuchs Y and Steller H: Programmed cell
death in animal development and disease. Cell. 147:742–758. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Youle RJ and Strasser A: The BCL-2 protein
family: Opposing activities that mediate cell death. Nat Rev Mol
Cell Biol. 9:47–59. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tsui D, Vessey JP, Tomita H, Kaplan DR and
Miller FD: FoxP2 regulates neurogenesis during embryonic cortical
development. J Neurosci. 33:244–258. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Vernes SC, Oliver PL, Spiteri E, Lockstone
HE, Puliyadi R, Taylor JM, Ho J, Mombereau C, Brewer A, Lowy E, et
al: Foxp2 regulates gene networks implicated in neurite outgrowth
in the developing brain. PLoS Genetics. 7:e10021452011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Taylor BS, Schultz N, Hieronymus H,
Gopalan A, Xiao Y, Carver BS, Arora VK, Kaushik P, Cerami E, Reva
B, et al: Integrative genomic profiling of human prostate cancer.
Cancer Cell. 18:11–22. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bignone PA and Banham AH: FOXP3+
regulatory T cells as biomarkers in human malignancies. Expert Opin
Biol Ther. 8:1897–1920. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Teufel A, Wong EA, Mukhopadhyay M, Malik N
and Westphal H: FoxP4, a novel forkhead transcription factor.
Biochim Biophys Acta. 1627:147–152. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Campbell AJ, Lyne L, Brown PJ, Launchbury
RJ, Bignone P, Chi J, Roncador G, Lawrie CH, Gatter KC, Kusec R and
Banham AH: Aberrant expression of the neuronal transcription factor
FOXP2 in neoplastic plasma cells. Br J Haematol. 149:221–230. 2010.
View Article : Google Scholar : PubMed/NCBI
|