Introduction
Gastric cancer (GC) is the fourth most common cancer
type and the second leading cause of cancer-induced mortality
worldwide (1). Lymph node (LN)
metastasis is the primary spread route and considered the strongest
prognostic indicator of GC (2). Even
for patients with early stage GC, the frequency of LN metastasis is
estimated to be up to 20% (1).
Adequate surgical resection is considered the most
favorable therapeutic option for GC (3–5). For this
procedure, complete resection of the primary cancer with tumor-free
surgical margins of ≥4 cm and adequate lymphadenectomy are
required. D2 lymphadenectomy [perigastric (D1) plus coeliac artery
and its branches] is generally recommended, based on associated
clinical trials demonstrating the survival advantages of this
surgery type (1,6); however, the postsurgical morbidity and
mortality outcomes are not always acceptable if the procedures are
not performed by experienced surgeons. Additionally, surgeons in
Western countries, including USA, UK and the Netherlands prefer
using conservative D0 or D1 resection or modified D2 resection
(without routine pancreatectomy and splenectomy) due to the
negative results of two earlier randomized trials comparing D1 and
D2 resection from the UK and the Netherlands (1,6,7). These differences in lymphadenectomy are
primarily attributable to respective history-based clinical
experience and comprehensive evaluation of the benefits and risks
of the procedure.
Precise resection of the LN with metastasis, while
sparing healthy nodes, should present the most reliable criteria.
This approach not only allows the removal of tumor cells in
vivo as thoroughly as possible, in order to prevent relapse,
but also avoids excessive lymphadenectomy, which notably affects
the benefits to patients; however, the majority of LN metastasis
patterns of GC remain unknown. Previous studies indicated that LN
metastasis patterns in GC are notably complex, due to aberrant
lymphatic drainage patterns with frequent occurrence of skip LN
metastasis (8,9). Micrometastases or isolated tumor cells
in the LN increase the challenge of treating GC (2).
Limited preclinical studies have focused on LN
metastasis in GC to date (10–12).
Furthermore, previous studies only reported the frequency of LN
metastasis and did not highlight the anatomic positions of
metastatic LNs. Clarification of the mechanisms underlying LN
metastasis of GC may also be of significant value in directing
clinical practice.
In the present study, an orthotopic human GC model
was established in nude mice with luciferase-expressing NCI-N87
human GC cells. Cells were carefully injected into the subserosa of
the gastric body, following which tumors formed and grew. Tumor
development and metastasis in the LN and other major organs were
monitored with the aid of sensitive bioluminescence imaging. The
patterns of LN metastasis at four anatomic positions in the
abdominal cavity were further characterized.
Materials and methods
Cell line and mouse experiments
The luciferase-expressing human GC cell line
NCI-N87-Luc was obtained from Shanghai Biomodel Organisms Center
Co., Ltd. (Shanghai, China). Cells were cultured in Dulbecco's
modified Eagle's medium (DMEM) supplemented with 10% fetal bovine
serum and antibiotics (100 mg/ml streptomycin and 100 U/ml of
penicillin) (all from (Thermo Fisher Scientific, Inc., Waltham, MA,
USA) at 37°C in a humidified incubator with 5% CO2.
A total of 100 female BALB/c nude mice (4–5 weeks
old, ~20 g) were provided by the Shanghai Laboratory Animal Center
(Chinese Academy of Sciences, Shanghai, China). The mice were fed
in a specific pathogen-free environment at room temperature, with
free access to food and water and 12:12 h light/dark cycle. The
animal experiment designed in the present study was approved by the
ethical committee of Shanghai Jiao Tong University School of
Medicine (Shanghai, China).
Orthotopic injection of NCI-N87-Luc
cells
The orthotopic injection technique was performed
using a previously reported method (13). The mouse abdomen was opened via a
midline incision under aseptic conditions, and the stomach was
exteriorized. A volume of 40 µl DMEM containing 5×106
NCI-N87-Luc cells was injected into the subserous layer of the
middle of the stomach using a 100 µl syringe and 30G needle. A
cotton swab was pressed against the injection site for ≥20 sec to
prevent tumor cell leakage into the peritoneal cavity.
Subsequently, the stomach was returned to the peritoneal cavity,
and peritoneum and skin sutured sequentially. The feasibility of
the injection method was confirmed using trypan blue as an
indicator, which demonstrated an accurate location of the injected
dye in the subserosa of the gastric wall.
Bioluminescence imaging of orthotopic
tumor growth and metastasis
Female BALB/c nude mice (4–5 weeks old) were used
for animal studies. At 4, 6, 8 and 10 weeks after tumor cell
injection, mice (n=5) were intraperitoneally injected with 200 µl
D-luciferin solution (150 mg/kg body weight, J&K Scientific
Ltd, Shanghai, China). Subsequently, mice were anesthetized with 2%
isoflurane, and at 8 min after D-luciferin injection, placed in the
Xenogen IVIS 200 (PerkinElmer, Inc., Waltham, MA, USA) chamber with
right lateral recumbency for bioluminescence imaging of the
orthotopic NCI-N87-Luc tumor according to the manufacture's
protocols. Body weights of mice were recorded weekly throughout the
test. To detect metastasis in LN and other major organs at
different time points, mice (n=5) were sacrificed by cervical
vertebra dislocation between week 3 and 10. LNs at four anatomical
positions in the abdomen (14),
including gastric LN (GLN), pancreaticoduodenal LN (PLN),
mesenteric LN (MLN) and lumbar LN (LuLN), and other major organs,
including the heart, liver, spleen, lung and kidney, were excised
for ex vivo bioluminescence imaging. A metastasis heat map
was generated using GraphPad Prism 7 software (GraphPad Software,
Inc., La Jolla, CA, USA).
Histopathological assay
Excised LN and organs were fixed in 4%
paraformaldehyde at room temperature overnight, embedded in
paraffin and cut into 4 µm sections. Subsequently the sections were
stained with hematoxylin for 3 min and eosin for 2 min at room
temperature. The sections were observed and photographed under
upright metallurgical microscope with digital image capturing
system (DP50; magnification, ×40 or ×200; Olympus Cooperation,
Tokyo, Japan).
Statistical analysis
Data analysis was conducted with the GraphPad Prism
7.0 software (GraphPad Software, Inc., La Jolla, CA, USA). The
quantified time-dependent bioluminescence signal of the tumors and
also the mouse body weight are presented as mean ± standard
deviation.
Results
Establishment and characterization of
an orthotopic model of GC
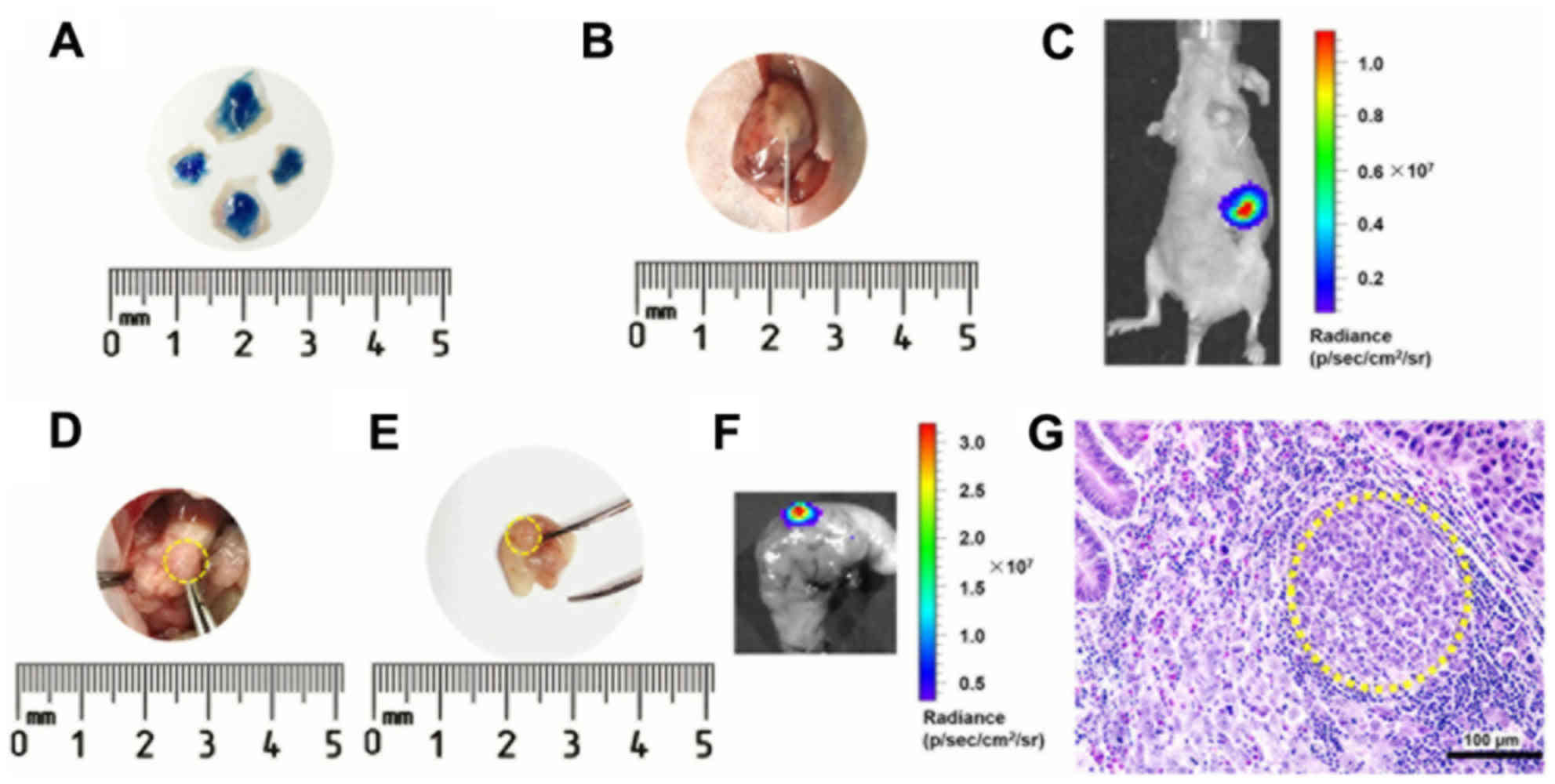
Trypan blue dye was used to confirm the reliability
of the injection technique (Fig. 1A).
Following the NCI-N87-Luc cell injection, a small white plaque was
observed under the serosa of the gastric body (Fig. 1B). The cells formed an orthotopic
tumor, which was detected via sensitive bioluminescence imaging
(Fig. 1C). Tumors on the gastric wall
were observed following opening the abdomen (Fig. 1D) or directly from the resected
stomach (Fig. 1E). Ex vivo
bioluminescence imaging highlighted the tumor position in the
stomach (Fig. 1F). Histopathological
assays further confirmed tumor formation and anatomical location of
subserosa (Fig. 1G). All mice
receiving this treatment grew tumors on the stomach, establishing a
100% success rate of the model.
Bioluminescence imaging of the growth
of orthotopic gastric tumors
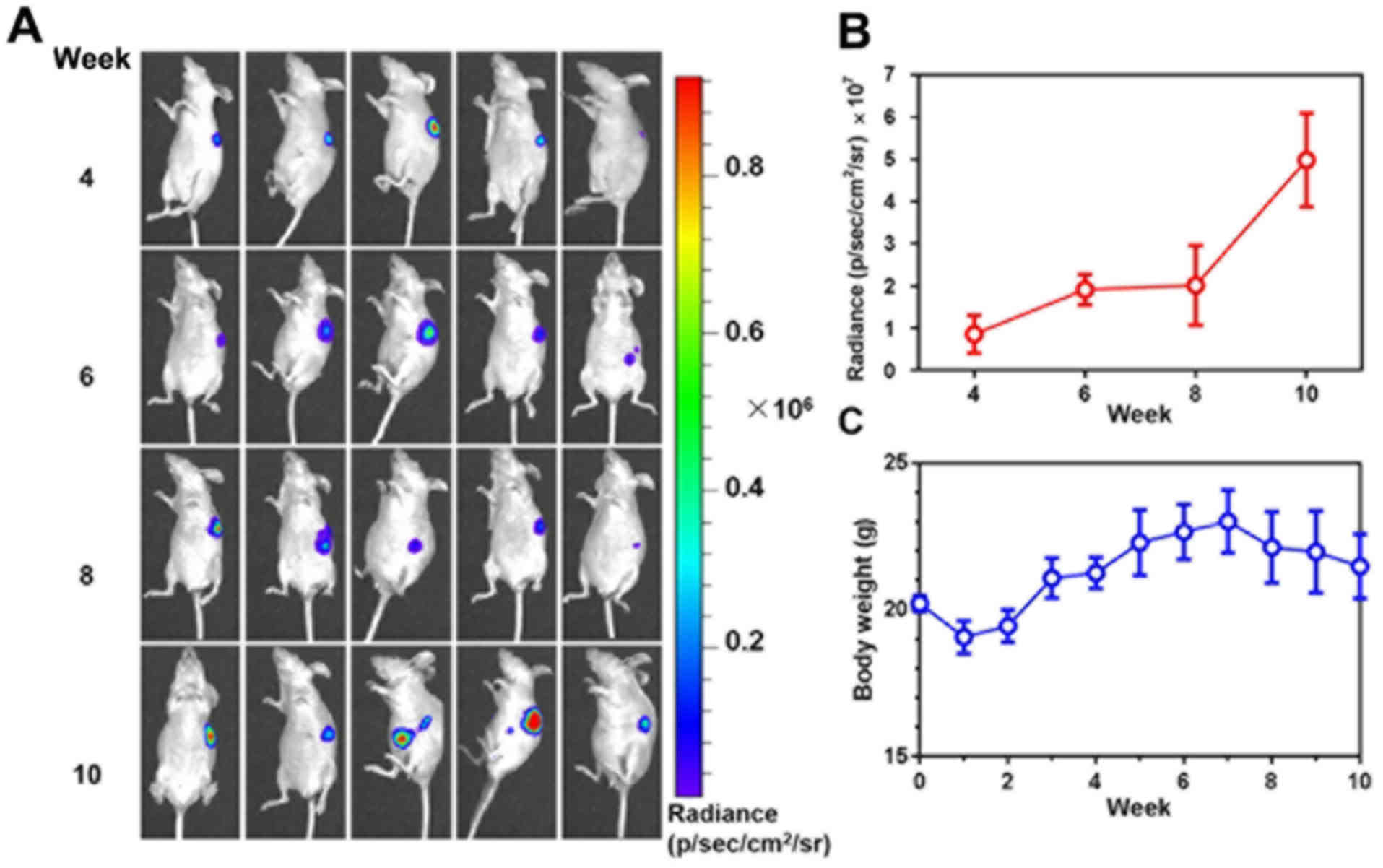
The growth of stomach tumors was monitored via
bioluminescence imaging (Fig. 2A).
Quantification of bioluminescence intensity demonstrated the
time-dependent tumor growth until week 10 after tumor cell
injections (Fig. 2B). During the same
time-period, the body weight of the mice increased in the first 7
weeks. However, subsequent to week 7 the body weight of the mice
declined (Fig. 2C), suggesting the
involvement of harmful gastric tumors in the deterioration of the
mice's health.
LN metastasis in the abdomen
Firstly, the four anatomical positions of LNs (GLN,
PLN, MLN, and LuLN) were identified in the abdomen (Fig. 3A-C). H&E staining further
confirmed the presence of LNs, and not adipose tissues or omentum,
at these positions (Fig. 3D).
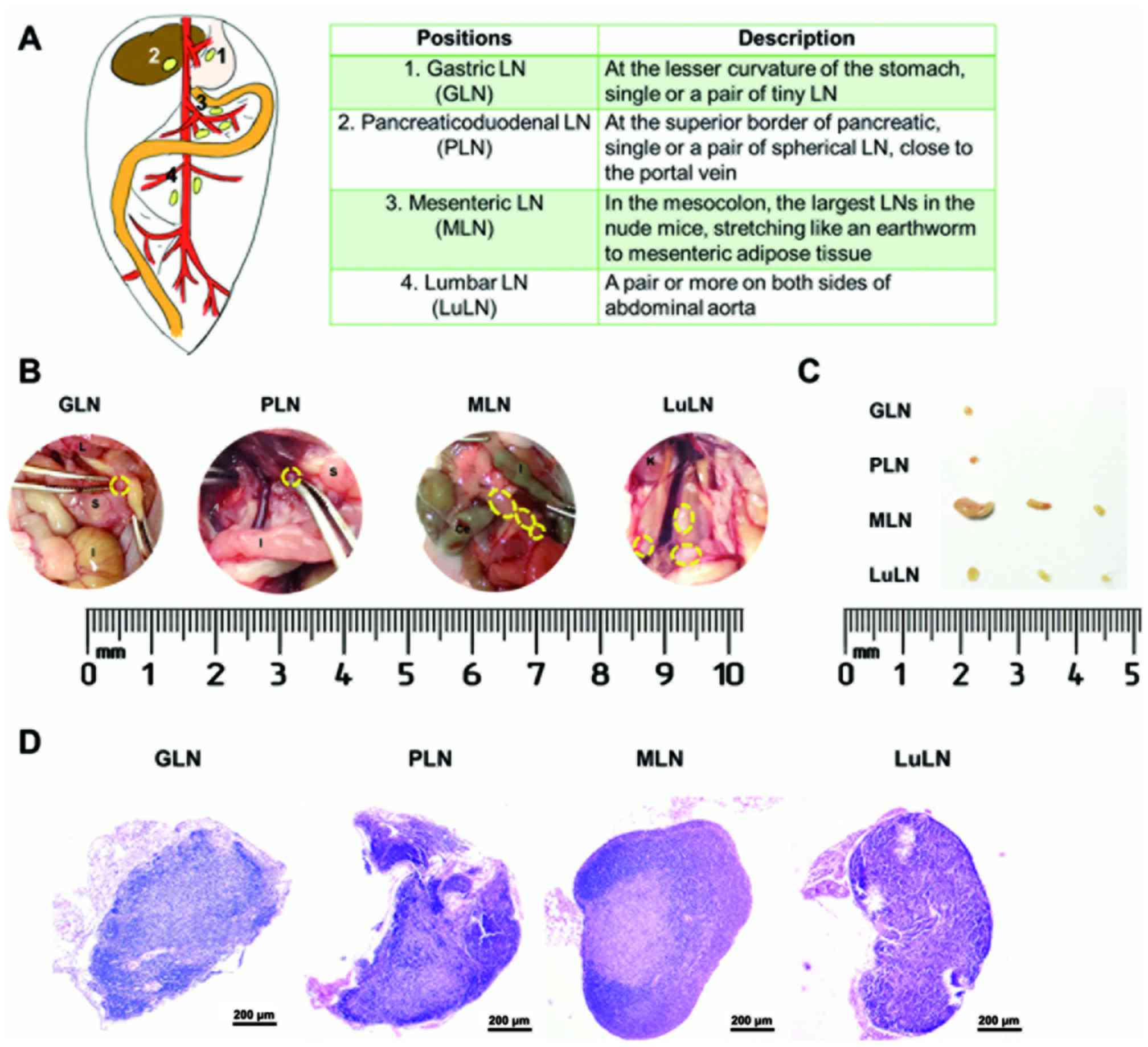 | Figure 3.Identification of LNs at different
anatomical position in the abdomen. (A) Schematic anatomical
positions of the LNs (GLN, PLN, MLN and LuLN) detected in the
present study. At 10 weeks after tumor cell injection, the LNs at
various positions were indicated following (B) exploratory
laparotomy, and (C) then were excised and arranged on clean paper.
The LNs were indicated with yellow circles. (D) Hematoxylin and
eosin staining of the resected LNs at different positions. L,
liver; S, stomach; I, intestine; Ce, cecum; K, kidney; LN, lymph
nodes; GLN, gastric LN; PLN, pancraticoduodenal LN; MLN, mesenteric
LN; LuLN, lumbar LN. |
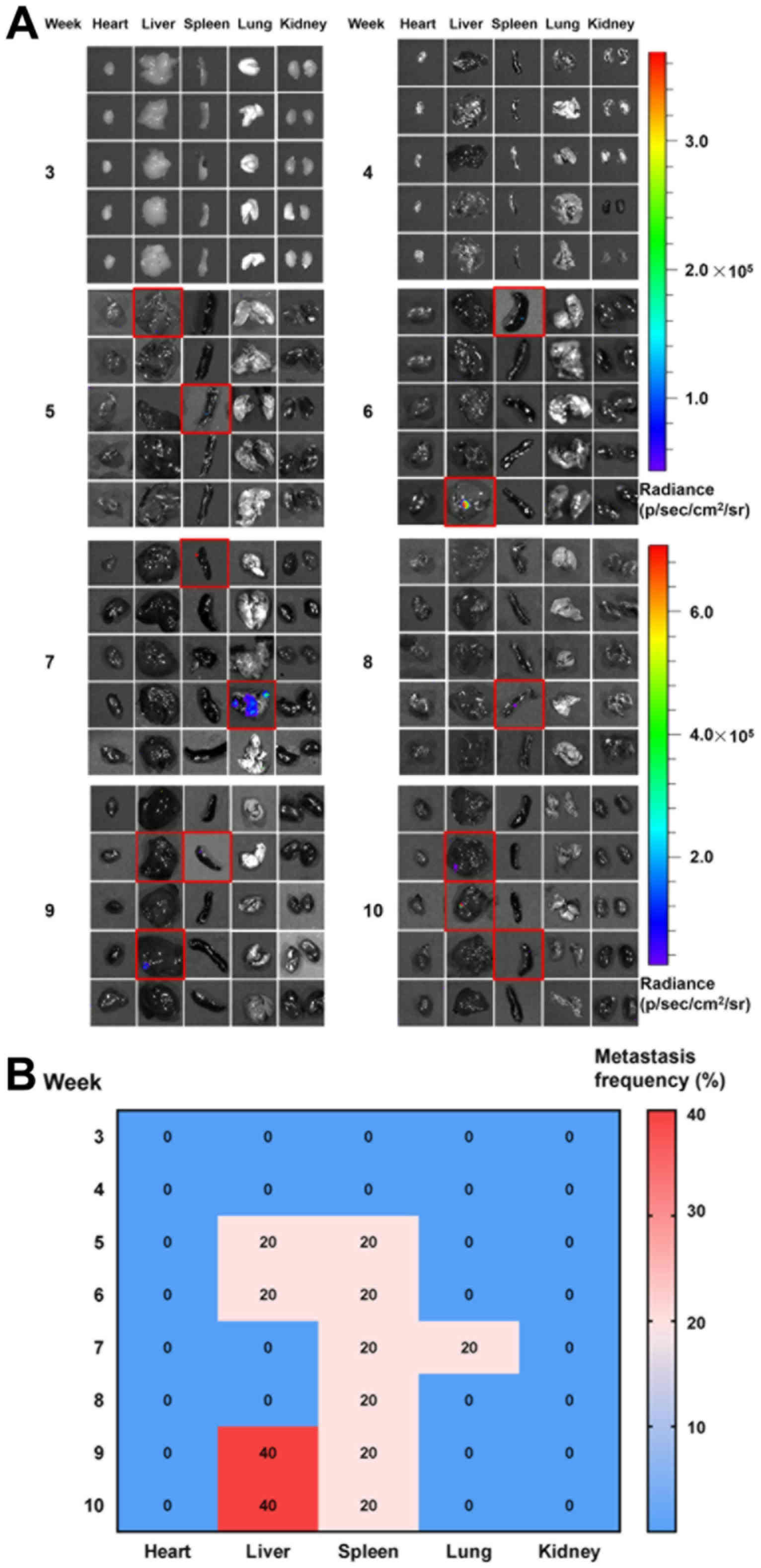
From week 3, LNs of mice (n=5) were resected for
ex vivo bioluminescence imaging to investigate the
metastasis patterns (Fig. 4A). The
overall LN metastasis patterns from weeks 3–10 were summarized in a
heat map (Fig. 4B). Histopathological
examination with bioluminescence imaging further confirmed the
presence of metastatic tumor cells in the LNs (Fig. 4C). This demonstrated that no
metastasis occurred in the LN at week 3 and 4. At week 5, GLN of
one mouse was positive for metastasis, based on the bioluminescence
signal. At week 6, metastasis was observed at PLN, MLN and LuLN in
one mouse, indicating distant spreading. From weeks 7–9, metastasis
occurred in a greater number of mice and spread into LNs at various
anatomical positions. At week 10, all tested mice exhibited
positive bioluminescence signals in LNs. It is also notable that
the metastasis frequency for GLN, MLN and LuLN decreased from 40%
at week 7 to 20% at week 8. This phenomenon may be ascribed to the
individual differences of the mice at each week, which may be
avoided if a greater number of animals (n>5) were examined at
each time point. Additionally, it can be concluded, though this
observation, that the metastasis frequency for GLN, MLN and LuLN
varied in the range of 20–40% in this duration.
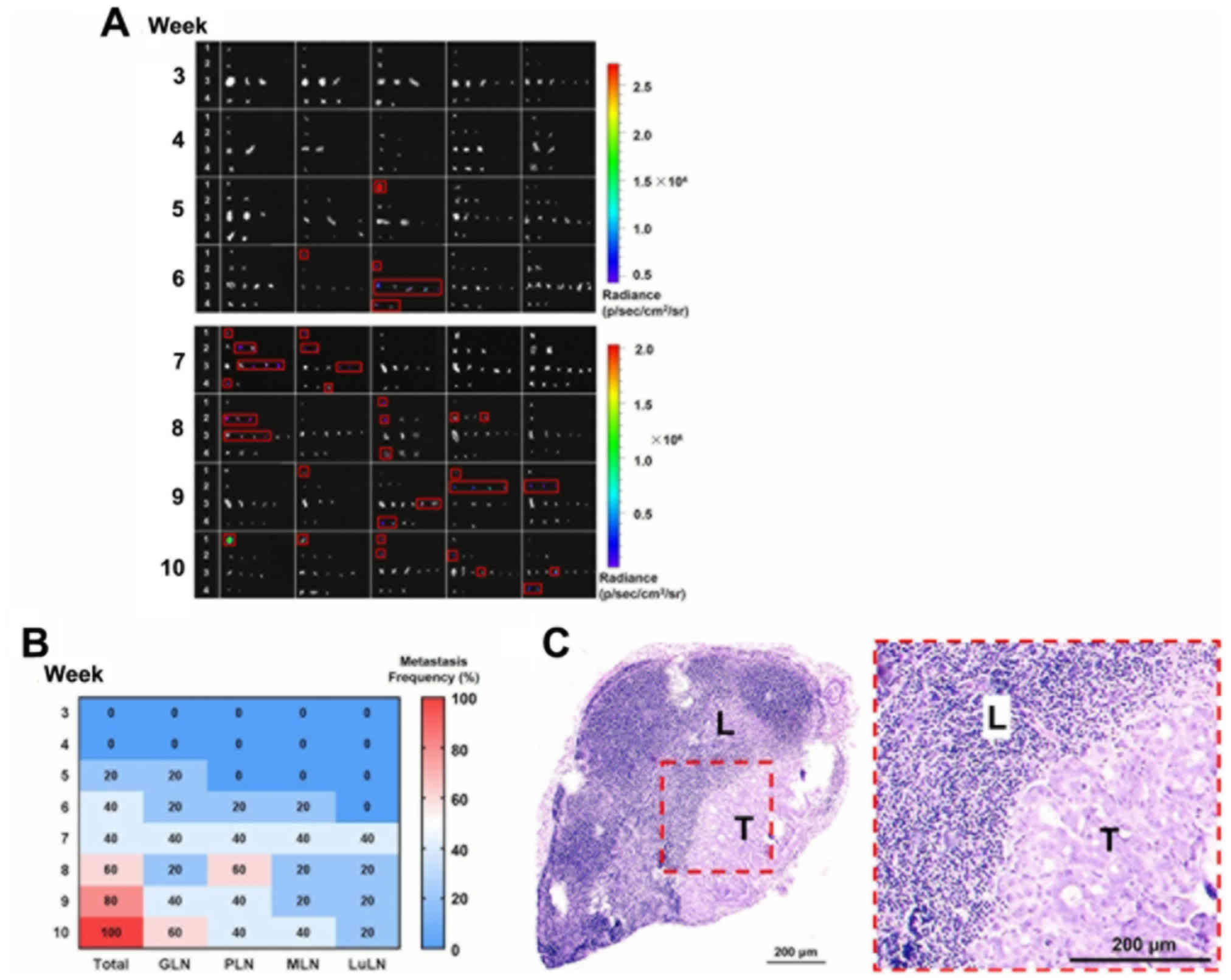 | Figure 4.LN metastasis of orthotopic gastric
cancer model. From weeks 3–10, mice (n=5 for each week) were
sacrificed, and the LNs (1, GLN; 2, PLN; 3, MLN; and 4, LuLN) were
resected for metastasis detection. Note that the images presented
for each week are not from the same mouse. (A) LNs metastasis
detected using the bioluminescence imaging. Each black rectangle
indicates the LNs of one mouse. The red rectangles indicated the
identified metastatic LNs. (B) The time-dependent metastasis
frequencies of LNs at different sites were summarized in the heat
map. (C) Hematoxylin and eosin staining of the resected metastatic
GLN after 6 weeks. L, lymphocyte; T, tumor cells; LN, lymph nodes;
GLN, gastric LN; PLN, pancraticoduodenal LN; MLN, mesenteric LN;
LuLN, lumbar LN. |
Major organ metastasis
Organ metastasis was observed at week 5 (the same
time as LN metastasis). One mouse exhibited metastasis to the liver
and one to the spleen (Fig. 5);
however, the bioluminescence signals detected were weak, indicating
the metastasis of only a small number of tumor cells. From weeks
6–10, the spleen and liver remained the primary metastatic organs.
Lung metastasis was only observed in one mouse at week 7. It also
demonstrated a liver metastasis frequency of 20% at week 5 and 6,
which reduced to 0% at week 7 and 8. It is notable that the
observations at each week were from different mice. If a greater
number of animals (n>5) were included for each week, the
influence of animal individual differences on the metastasis
frequency may be avoided. These observations also indicated that
the liver metastasis were not frequent in the first 8 weeks in the
present model.
Discussion
Treatment of GC primarily depends on the management
of LN metastasis; however, unique metastasis patterns, such as skip
LN metastasis, cause a notable challenge to effective treatments.
Micrometastasis and isolated tumor cells in regional LNs also pose
a serious threat to prognosis. The complex profile of LN metastasis
of GC remains largely unknown, which is partially responsible for
the controversy regarding the range of lymphadenectomy. Previous
preclinical studies have been limited to the time-dependent
frequency of LN metastasis in GC (12,13,15).
However, to the best of our knowledge, no studies have been
conducted regarding LN metastasis patterns at specific anatomical
positions, which may be beneficial for a surgeon to perform precise
surgery.
In the present study, an orthotopic GC model was
established via injection of NCI-N87-Luc cells into the subserosa
of the gastric body of nude mice. The present method appeared to be
reliable and reproducible, resulting in a tumor formation rate of
100%. Additionally, the present procedure could overcome the risk
of tumor cells shedding into the peritoneal cavity, which occurs
with other methods, including fixing the donor tumor fragment into
the recipient gastric wall using tissue adhesive (15). The metastasis was identified using the
sensitive bioluminescence imaging, which can detect 100 tumor cells
in vitro (data not shown) and avoid the false negative
results when performing traditional serial sectioning and
histopathological examinations (2).
It demonstrated that there was no detectable metastasis at week 3
and 4 in LNs and the major organs (Figs.
4 and 5), indicating that the
metastasis was probably from the primary GC, which requires a long
time for the tumor cells to colonize and form detectable foci.
The time-dependent metastasis patterns in the four
LN positions (GLN, PLN, MLN and LuLN) in the abdomen were
characterized. As expected, the frequency of metastasis increased
with time following tumor cell injection. Notably, skip metastasis
was observed in mice at the later stages (weeks 8–10) of the
experiment period, demonstrating a discontinuous spread of
malignancy in the LN, providing further evidence of the complexity
of LN metastasis in GC.
The frequency and extent of metastasis in major
organs other than LN was significantly reduced, which may be
partially attributed to the well-differentiated property of NCI-N87
cells. Further studies are warranted to investigate the metastatic
behavior of orthotopic poorly-differentiated gastric tumor types,
including the AGS (16,17) and MKN-45 (18,19) cell
lines, or tumor types with diverse molecular classifications,
including HER2-positive cancer types (1,20).
In conclusion, GC metastasis in LNs and major organs
was characterized with the aid of an established orthotopic mouse
model. Time-dependent metastasis patterns at different anatomical
positions in the abdomen are presented. The present model with
characterized LN and organ metastasis may be effectively used for
preclinical GC research regarding the metastatic mechanism and drug
development.
Acknowledgements
We thank Dr Jing Zhou (Department of Experimental
Animal Science, Shanghai Jiao Tong University School of Medicine)
for assisting with IVIS imaging testing. We also thank Dr Lin Zheng
(Department of Pathology, Shanghai Jiao Tong University School of
Medicine) for assisting with the histopathological assay.
Funding
The present study is supported by National Natural
Science Foundation of China (grant nos. 81401958, 81572998 and
81773274), Shanghai Municipal Commission of Health and Family
Planning (grant no. ZK2015A25), Shanghai Changning Commission of
Health and Family Planning (grant no. YXMZK004) and Shanghai
Municipal Science and Technology Commission (grant nos. 14ZR1431700
and 16520710700), ‘Shu Guang’ Program of Shanghai Education
Development Foundation and Shanghai Municipal Education Commission
(grant no. 16SG13), Shanghai Collaborative Innovation Center for
Translational Medicine (grant no. TM201731).
Availability of data and materials
The datasets used and analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
HF, PS and CF conceived and designed the
experiments. HF, YZ, HL, XD, SY and QL performed the experiments.
HF and CF collected and analyzed the data. FM and HZC provided
suggestions and technical support on the project. HF and CF wrote
the manuscript. PS and CF supervised the project. All authors read
and approved the final manuscript.
Ethics approval and consent to
participate
The animal experiments designed in this study were
approved by the ethical committee of Shanghai Jiao Tong University
School of Medicine.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Van Cutsem E, Sagaert X, Topal B,
Haustermans K and Prenen H: Gastric cancer. Lancet. 388:2654–2664.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jeuck TL and Wittekind C: Gastric
carcinoma: Stage migration by immunohistochemically detected lymph
node micrometastases. Gastric Cancer. 18:100–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Songun I, Putter H, Kranenbarg EM, Sasako
M and van de Velde CJ: Surgical treatment of gastric cancer:
15-year follow-up results of the randomised nationwide Dutch D1D2
trial. Lancet Oncol. 11:439–449. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ajani JA, D'Amico TA, Almhanna K, Bentrem
DJ, Chao J, Das P, Denlinger CS, Fanta P, Farjah F, Fuchs CS, et
al: Gastric cancer, version 3.2016, NCCN clinical practice
guidelines in oncology. J Natl Compr Canc Netw. 14:1286–1312. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Smyth EC, Verheij M, Allum W, Cunningham
D, Cervantes A and Arnold D; ESMO, ; Guidelines Committee, :
Gastric cancer: ESMO Clinical Practice Guidelines for diagnosis,
treatment and follow-up. Ann Oncol. 27 Suppl 5:v38–v49. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shen L, Shan YS, Hu HM, Price TJ, Sirohi
B, Yeh KH, Yang YH, Sano T, Yang HK, Zhang X, et al: Management of
gastric cancer in Asia: Resource-stratified guidelines. Lancet
Oncol. 14:e535–e547. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ajani JA, Lee J, Sano T, Janjigian YY, Fan
D and Song S: Gastric adenocarcinoma. Nat Rev Dis Primers.
3:170362017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kim DH, Choi MG, Noh JH, Sohn TS, Bae JM
and Kim S: Clinical significance of skip lymph node metastasis in
gastric cancer patients. Eur J Surg Oncol. 41:339–345. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Choi YY, An JY, Guner A, Kang DR, Cho I,
Kwon IG, Shin HB, Hyung WJ and Noh SH: Skip lymph node metastasis
in gastric cancer: Is it skipping or skipped? Gastric Cancer.
19:206–215. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Qiao R, Liu C, Liu M, Hu H, Liu C, Hou Y,
Wu K, Lin Y, Liang J and Gao M: Ultrasensitive in vivo detection of
primary gastric tumor and lymphatic metastasis using upconversion
nanoparticles. ACS Nano. 9:2120–2129. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Furukawa T, Fu X, Kubota T, Watanabe M,
Kitajima M and Hoffman RM: Nude mouse metastatic models of human
stomach cancer constructed using orthotopic implantation of
histologically intact tissue. Cancer Res. 53:1204–1208.
1993.PubMed/NCBI
|
|
12
|
Fujihara T, Sawada T, Hirakawa K, Chung
YS, Yashiro M, Inoue T and Sowa M: Establishment of lymph node
metastatic model for human gastric cancer in nude mice and analysis
of factors associated with metastasis. Clin Exp Metastasis.
16:389–398. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hackl C, Man S, Francia G, Milsom C, Xu P
and Kerbel RS: Metronomic oral topotecan prolongs survival and
reduces liver metastasis in improved preclinical orthotopic and
adjuvant therapy colon cancer models. Gut. 62:259–271. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Van den Broeck W, Derore A and Simoens P:
Anatomy and nomenclature of murine lymph nodes: Descriptive study
and nomenclatory standardization in BALB/cAnNCrl mice. J Immunol
Methods. 312:12–19. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bhargava S, Hotz B, Buhr HJ and Hotz HG:
An orthotopic nude mouse model for preclinical research of gastric
cardia cancer. Int J Colorectal Dis. 24:31–39. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yin J, Cui Y, Li L, Ji J and Jiang WG:
Overexpression of EPHB4 is associated with poor survival of
patients with gastric cancer. Anticancer Res. 37:4489–4497.
2017.PubMed/NCBI
|
|
17
|
Belkhiri A, Dar AA, Zaika A, Kelley M and
El-Rifai W: t-Darpp promotes cancer cell survival by up-regulation
of Bcl2 through Akt-dependent mechanism. Cancer Res. 68:395–403.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Guo B, Zhao Z, Wang Z, Li Q, Wang X, Wang
W, Song T and Huang C: MicroRNA-302b-3p suppresses cell
proliferation through AKT pathway by targeting IGF-1R in human
gastric cancer. Cellular Physiol Biochem. 42:1701–1711. 2017.
View Article : Google Scholar
|
|
19
|
Burgermeister E, Xing X, Röcken C, Juhasz
M, Chen J, Hiber M, Mair K, Shatz M, Liscovitch M, Schmid RM and
Ebert MP: Differential expression and function of caveolin-1 in
human gastric cancer progression. Cancer Res. 67:8519–8526. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Boku N: HER2-positive gastric cancer.
Gastric Cancer. 17:1–12. 2014. View Article : Google Scholar : PubMed/NCBI
|