Introduction
Ependymoma is an uncommon type of primary brain
tumor, accounting for 37% of all brain tumors (1). A rare form of ependymoma known as
tanycytic ependymoma (TE) has been classified as Grade II by the
World Health Organization (WHO) (2).
It was first described and termed by Friede and Pollak in 1978
(3). The majority of TE originates
from the ependymal cells of the brain ventricular wall and is
characterized by elongated spindle cells. Tanycyte cells are
implicated in two types of CNS tumors, namely, tanycytic ependymoma
and astroblastoma (4). They are
elongated spindly bipolar cells that generally present in the
circumventricular organs, particularly in the third ventricle and
central canal of spinal cord (5). It
is common for TE to be misdiagnosed as a nervous system tumor of
the spindle cells (6). The definitive
diagnosis of TE requires pathological analysis, including
histological characteristics. Electron microscopy may contribute to
diagnosis and differential diagnosis. Several cases have been
diagnosed as astrocytoma or subependymoma or hemangioblastoma by
preoperative imaging examination (7).
TE is usually located within the cervical and thoracic (T) spinal
cord; however, on rare occasions it can be located within the filum
terminale. To the best of our knowledge, only 5 cases of TE
associated with the filum terminale have been reported in previous
studies (Table I) (6,8–11); however, in the present study, 8 cases
of TE in the filum terminale were reported. Combined with data from
previous studies, the clinical, radiological and pathological
features of TE in the filum terminale were summarized for diagnosis
and differential diagnosis.
 | Table I.Reported cases of tanycytic ependymoma
arising from filum terminale. |
Table I.
Reported cases of tanycytic ependymoma
arising from filum terminale.
| Author/year | Sex/age, years | Location | Symptom
(duration) | Pathological
findings | Immunopositivity | Treatment | Prognosis
(duration) | (Refs.) |
|---|
| Mohindra et
al, 2008 | F/10 | L2-4 | Severe backache (2
years), paraparesis (1 year) | Elongated cells,
pseudorosettes, microtubules | GFAP, S-100,
vimentin | GTR | No recurrence (1
year) | (8) |
| Shintaku et
al, 2009 | F/55 | L1 | Lumbago and numbness
of right leg (2 months) | Spindle cells,
pleomorphic giant cells, microtubules | GFAP, EMA, S-100 (6
months) | GTR | No recurrence | (9) |
| Radhakrishnan et
al, 2012 | M/44 | T12-L1 | Low backache (3
years), lower limb weakness and uracratia (6 months) | Elongated cells | GFAP, S-100 | GTR | ND | (6) |
| Funayama et
al, 2013 | M/53 | T12-L2 | Numbness of both
legs (3 years), lower backache (1 year) | Elongated cells,
pseudorosettes | GFAP, S-100 | GTR | No recurrence (32
months) | (10) |
| Tomek et al,
2016 | F/69 | T12-L1 | Low backache and
limited mobility (several months) | Elongated cells,
pseudorosettes | GFAP, EMA,
S-100 | GTR | No recurrence (7
months) | (11) |
Materials and methods
The present retrospective study investigated
clinical data from 8 patients who underwent microsurgery for a TE
of the filum terminale between August 2011 and June 2016 at the
Department of Neurosurgery of Beijing Tiantan Hospital (Beijing,
China). All patients had performed preoperative and postoperative
magnetic resonance imaging (MRI) with gadolinium-contrast
enhancement. The relevant clinical data (including patient age,
sex, symptoms, neuroimaging, preoperative diagnosis, surgical
records and follow-up data) were collected via a chart review and
telephone interviews. The diagnosis of TE was confirmed by a review
of the pathology slides at the Beijing Neurosurgical Institute
(Beijing, China). All patients underwent laminectomies to remove
the tumor. The following definitions were used: Gross total
resection (GTR) was defined as the total macroscopic removal of the
tumor; and subtotal resection was defined as the subtotal
macroscopic tumor removal. The estimation of the extent of tumor
removal was validated by reviewing the postoperative MRI.
Pathological examination
All specimens underwent fixation in 4% neutral
formalin (24 h at 4°C), routine dehydration using a alcohol
gradient series (ethanol 95% for 5 min, ethanol 95% for 5 min and
ethanol 80% for 5 min), paraffin-embedding and slicing into 4-µm
sections, and then staining using hematoxylin-eosin (hematoxylin
1%, eosin 0.5%) at room temperature for ~2 h. Immunohistochemical
staining was used for differential diagnoses. Immunohistochemistry
was performed using the indirect immunoperoxidase technique. Bovine
serum albumin (3%) (Beijing Zhongshan Golden Bridge Biotechnology
Co., Ltd.; OriGene Technologies, Beijing, China) was used for
blocking at room temperature for 1 h. Primary antibodies included
pre-diluted monoclonal antibodies against glial fibrillary acidic
protein (GFAP; cat. no. ZM-0118, Beijing Zhongshan Golden Bridge
Biotechnology Co., Ltd.; OriGene Technologies; 1:200), nestin (cat.
no. ZA-0628, 1:200) and epithelial membrane antigen (EMA; cat. no.
ZM-0095, 1:200), Ki-67 antigen (cat. no. ZM-0165, 1:200), which
were incubated for 12 h at 4°C and subsequently incubated with the
HRP-conjugated mouse anti-human IgG (cat. no. SPN-9001; Beijing
Zhongshan Golden Bridge Biotechnology Co., Ltd.; OriGene
Technologies; 1:100) for 2 h at room temperature. The SuperPicture™
3rd Gen IHC Detection kit (Invitrogen; Thermo Fisher Scientific,
Inc., Waltham, MA, USA) was used to evaluate staining, according to
the manufacturer's protocol. For antigen retrieval, slides were
boiled in EDTA buffer (pH 8.0; cat. no. ZLI-9066; Beijing Zhongshan
Golden Bridge Biotechnology Co., Ltd.; OriGene Technologies; Tris
30.27 g, EDTA 1.461 g and H2O 500 ml) under high
pressure. Slides were counterstained with hematoxylin (1%) at room
temperature for ~2 h. An appropriate negative control (1 mmol/l
PBS) was used. Quantitative evaluation of Ki-67 was obtained by
calculating the percentage of Ki-67 positive nuclei in 100 tumor
cells from the microscopic field (light microscope; 6 fields
analyzed; magnification, ×100; Beijing Konghai Science and
Technology Development Co., Ltd., Beijing, China) with the highest
density of labeled nuclei.
Follow-up data for all patients were obtained
through individual office visits or telephone interviews performed
every 6 months. Modified Japanese Orthopedic Association (JOA)
scores (Table II) were used to
assess neurological function. All patients were reassessed in June
2016 as a last clinical follow-up.
 | Table II.Modified Japanese Orthopedic
Association scale (total score out of 17 points). |
Table II.
Modified Japanese Orthopedic
Association scale (total score out of 17 points).
| Section | Score, points |
|---|
| Motor function of
upper extremity |
|
| Unable
to feed oneself | 0 |
| Unable
to use knife and fork; able to eat with a spoon | 1 |
| Able to
use knife and fork with much difficulty | 2 |
| Able to
use knife and fork with slight difficulty | 3 |
|
Normal | 4 |
| Motor function of
lower extremity |
|
| Unable
to walk | 0 |
| Can
walk on flat floor with walking aid | 1 |
| Can
walk up and/or down stairs with handrail | 2 |
| Lack of
stability and smooth gait | 3 |
|
Normal | 4 |
| Sensory function of
upper extremity |
|
| Severe
sensory loss or pain | 0 |
| Mild
sensory loss | 1 |
|
Normal | 2 |
| Sensory function of
lower extremity |
|
| Severe
sensory loss or pain | 0 |
| Mild
sensory loss | 1 |
|
Normal | 2 |
| Sensory function of
trunk extremity |
|
| Severe
sensory loss or pain | 0 |
| Mild
sensory loss | 1 |
|
Normal | 2 |
| Bladder
function |
|
| Unable
to void | 0 |
| Marked
difficulty in micturition (retention) | 1 |
|
Difficulty in micturition
(frequency, hesitation) | 2 |
|
Normal | 3 |
Results
Clinical presentation
The clinical features of the 8 patients are
summarized in Table III. The mean
age of patients at the time of surgery was 40.5 years (range, 24–62
years). This series consisted of 1 male and 7 females. The duration
of symptoms ranged from 2 weeks to 10 years. The most common
clinical symptoms were pain and numbness of the waist and crura in
4 patients, and double crura in the other 4 patients. The mean
preoperative JOA score was 12.6 (range, 10–14).
 | Table III.Characteristics of 8 patients with
tanycytic ependymoma of the filum terminale. |
Table III.
Characteristics of 8 patients with
tanycytic ependymoma of the filum terminale.
|
|
|
|
| MRI findings |
|
| Modified JOA
scores |
|
|---|
|
|
|
|
|
|
|
|
|
|
|---|
| Case no. | Sex/age, years | Symptom
(duration) | Site | T1WI | T2WI | Enhancement | Preoperative
diagnosis | Treatment | Pre- | Post- | Last-FU | Prognosis |
|---|
| 1 | F/26 | Low backache and
numbness of both legs (3 years) | L2-4 | Iso-hypo | Iso | Markedly;
homogeneous | Schwannoma | GTR | 13 | 15 | 17 | No recurrence (1
year) |
| 2 | F/62 | Low backache (1
year), numbness of left leg (4 months) | L1-2 | Iso | Iso-hypo | Markedly;
heterogeneous | Schwannoma | GTR | 11 | 13 | 16 | No recurrence (17
months) |
| 3 | F/61 | Intermittent right
leg ache (10 years) | L2-3 | Iso | Hyper | Markedly;
homogeneous | Meningioma | GTR | 13 | 13 | 17 | No recurrence (2
years) |
| 4 | F/52 | Left leg ache (2
months) | L2-3 | Iso-hypo | Iso-hyper | Mildly;
heterogeneous | Schwannoma | STR | 14 | 14 | 16 | No recurrence (33
months) |
| 5 | F/49 | Pain and numbness
of right leg (10 years), numbness of left leg (1 year) | L2 | Iso-hyper | Iso | Markedly;
homogeneous | Meningioma | GTR | 13 | 15 | 17 | No recurrence (3
years) |
| 6 | M/25 | Numbness of buttock
and both legs (1 year) | L2-3 | Iso-hyper | Iso | Mildly;
homogeneous | Cholesteatoma | GTR | 13 | 15 | 16 | No recurrence (58
months) |
| 7 | F/24 | Pain of both legs
(1 month), uracratia (10 days) | L1-2 | Iso | Iso | Mildly;
homogeneous | Meningioma | GTR | 10 | 14 | 17 | No recurrence (14
months) |
| 8 | F/25 | Pain of waist and
both legs (2 weeks) | T12-L1 | Iso | Iso | Mildly;
homogeneous | Schwannoma | GTR | 14 | 14 | 17 | No recurrence (22
months) |
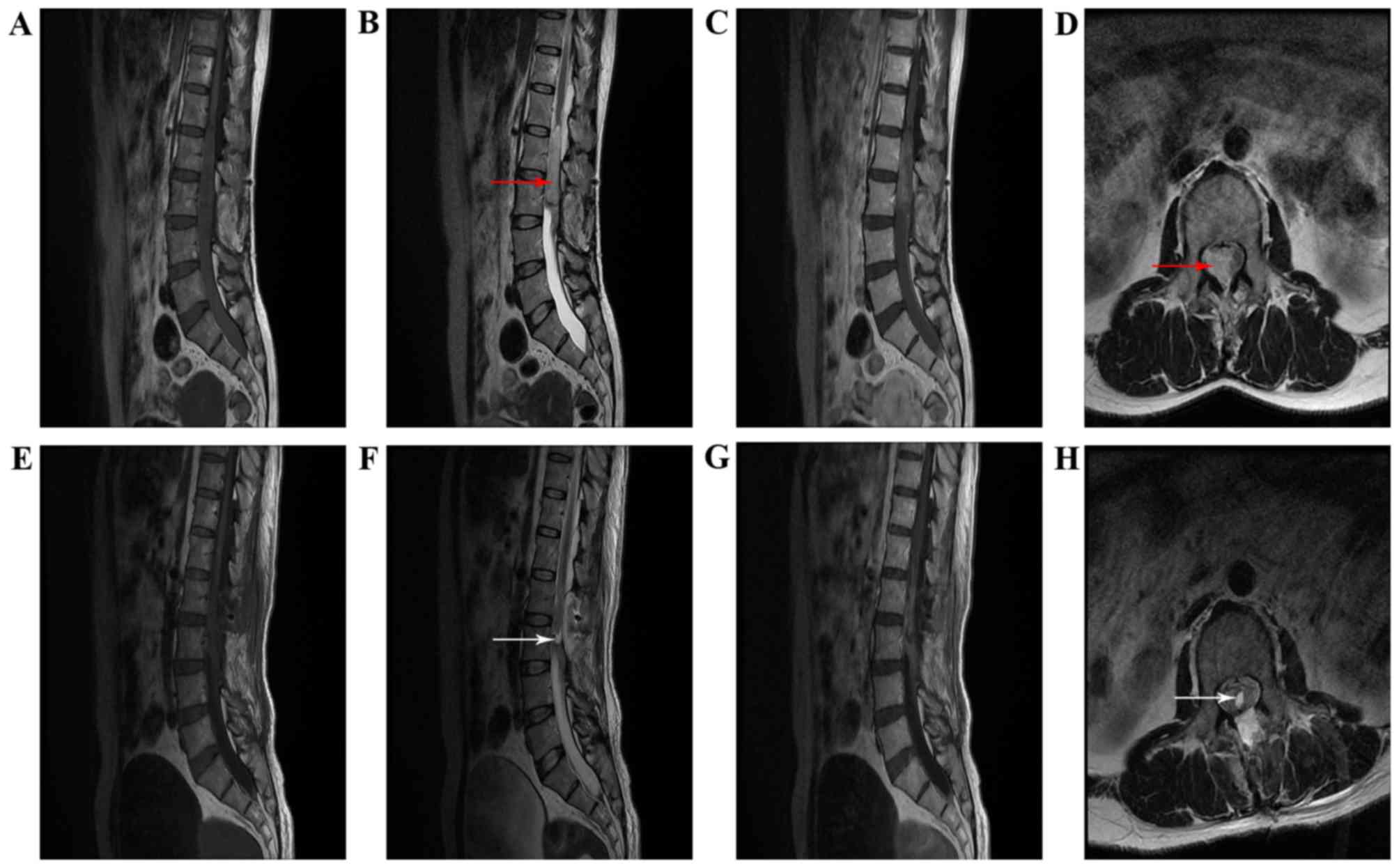
Radiological findings
Preoperative MRI results were available for all
patients (Table III). On the MRI
scans, lesions were oval or round in shape with well-defined
boundaries and to the lesions were close to the spinal cord or
cauda equina. On T1-weighted images (T1WI), lesions were isointense
in 4 cases, iso- to hyperintense in 2 cases, and iso- to
hypointense in 2 cases. On T2-weighted images (T2WI), the lesion
was isointense in 5 cases, iso- to hypointense in 1 case, iso- to
hyperintense in 1 cases, and hyperintense in 1 case.
Contrast-enhanced T1WI revealed 3 cases with markedly homogeneous
enhancement, 1 case with markedly heterogeneous enhancement, 3
cases with mild homogeneous enhancement and 1 case with mild
heterogeneous enhancement. According to the preoperative MRI, no
cases were diagnosed as ependymoma. The differential diagnosis
included meningioma (3 cases, 37.5%), schwannoma (4 cases, 50%) and
cholesteatoma (1 case, 12.5%). Pre- and postoperative MRI for 2
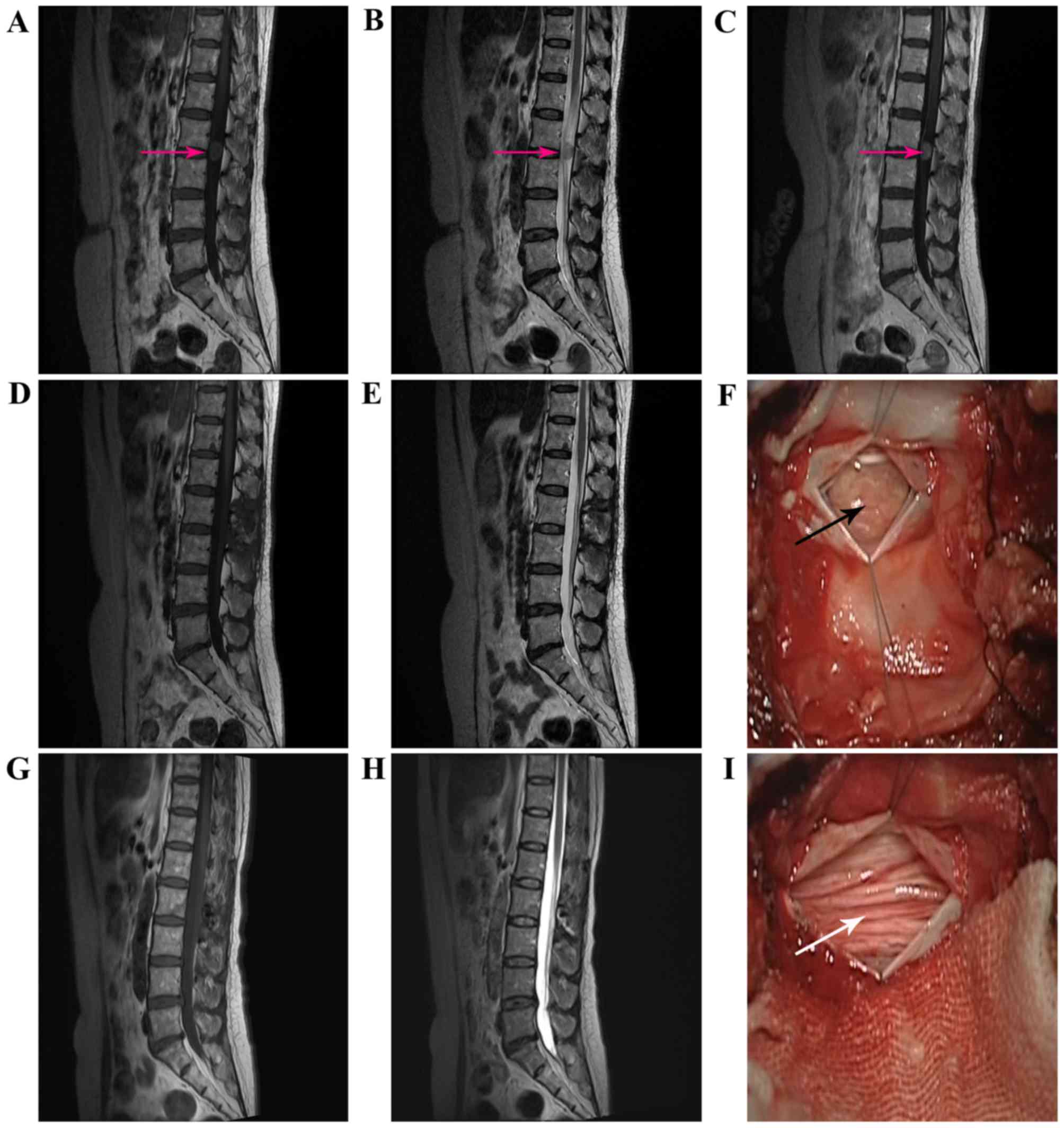
patients (cases 3 and 4) are illustrated in Figs. 1 and 2.
Surgical and pathological
findings
All patients underwent a posterior laminectomy
approach, with GTR performed in 7 patients. Following opening the
dura mater, the lesions were located in the cauda equina and
appeared to arise from nerve roots or TE. Intraoperatively, the
tumors typically appeared as moderate consistency, reddish-gray,
solid masses. The tumors were closely adhered to the cauda equina,
with moderate blood supply and a median intraoperative blood loss
of 100 ml. In the fourth patient, total resection of the tumor
failed due to its rich blood supply and close adhesion to the cauda
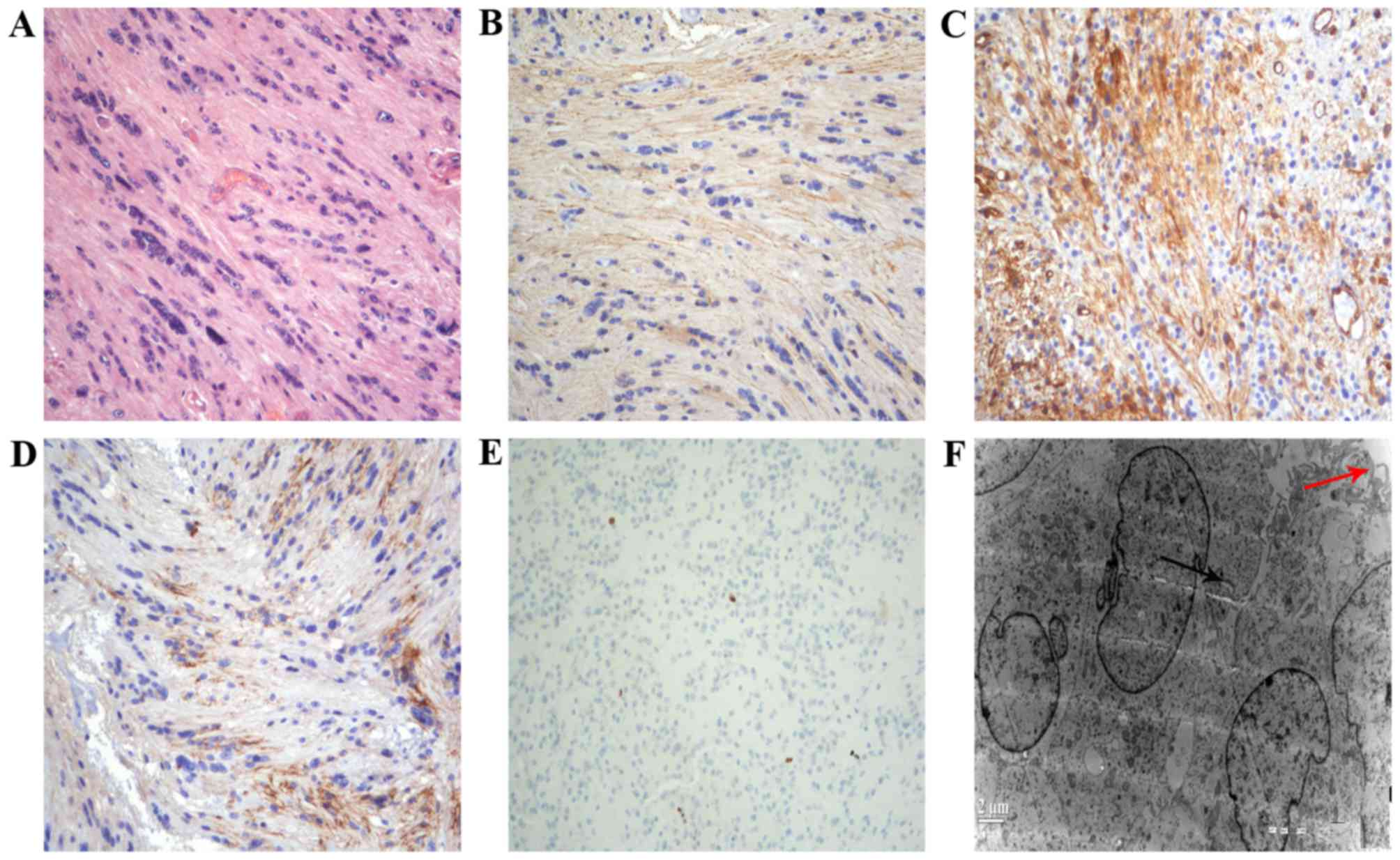
equina. Histological examination of the tumor with hematoxylin and
eosin staining revealed spindle cells with oval and elongated
nuclei, termed tanycytes. Immunohistochemical staining demonstrated
strong, positive staining of glial fibrillary acidic protein
(GFAP), nestin and epithelial membrane antigen (EMA). The mindbomb
homolog 1, also known as Ki-67 antigen, labeling index was 0.5–5%,
which indicated low proliferative activity. Ultrastructural
examination demonstrated spindle cells arranged in bundles with
scant extracellular matrix. Intercellular junctions, numerous
slender surface microvilli and microvilli-lined lumina were
observed (Fig. 3).
Postoperative evaluation
Following surgery, 5 patients experienced immediate
improvement and 3 patients remained unchanged from their
preoperative condition. All patients were postoperatively treated
with neurotrophic drugs (Methylcobalamin, 1.5 mg, 1 tablet 3 times
daily). In the 2-week and 3-month follow-up MRI, there was no
evidence of tumor recurrence. During a mean follow-up period of 27
months, neurological status had markedly improved in 7 patients and
was stable in 1 patient, compared with their postoperative
neurological deficits, including pain or loss of sensation in the
legs and uracratia (urinary incontinence, i.e., loss of urinary
control ability). However, a number of patients complained of
sensory deficit (2 cases, 25%) and sphincter dysfunction (1 case,
12.5%). During the final assessment, the mean JOA score was 16.6
(range, 16–17) for the 8 patients. The final mean JOA score was
markedly improved compared with the preoperative mean JOA score.
Surgical outcomes and assessment of neurological functions are
summarized in Table III.
Discussion
Table I depicts a
summary of the 5 reported cases of TE located in the filum
terminale (6,8–11). Of the
5 patients, 3 were female and 2 were male, 4 patients were adults
ageing from 44–69 years old. The remaining patient was a
10-year-old female child, which was reported by Mohindra et
al in 2008 (8); the youngest
patient with TE that has been reported, to the best of our
knowledge. The predominant symptoms experiences by patients
included backache, pain and numbness in the lower extremities. All
5 patients received complete surgical resection and postoperative
pathology demonstrated that there were notable long spindle cells
and pseudorosettes in the tumor, and immunohistochemical staining
indicated that the tumor was positive for GFAP, EMA and S-100. With
the exception of 1 case in which the prognosis of 1 patient
(6) was not described, no recurrence
was identified in the remaining 4 patients during the follow-up
period.
TE may occur at any age, however the pathogenic
sites vary for different ages. Pathogenic sites in children and
adolescents are commonly in the cerebral cranium, whilst the
majority of pathogenic sites in middle aged and elderly patients
are in the spinal cord (11). With TE
and spine as keywords, a search of the PUBMED database returned 27
articles (January 10th, 2017; http://www.ncbi.nlm.nih.gov/pmc/), in which 34 cases
of spinal TE were reported in total (2,3,6–30). By
reviewing the material of these 34 cases of patients with spinal
TE, it was revealed that the ages of the patients ranged from 10–76
years, with an average age of 40 years, and that the sex ratio of
females to males was 1.25:1. The pathogenic sites were mainly in
the cervical spinal cord (15 cases) and T cord (14 cases), whilst
the remaining 5 cases occurred in the cauda equine and filum
terminale. Combining the data from the present study and the 5
previous cases, it was identified that 10/13 patients with TE of
the filum terminale were female. The sex ratio of females to males
was 10:3, which was greater than the sex ratio of all patients with
spinal TE, indicating that TE of the filum terminale mostly
affected females. The clinical manifestations of TE were associated
with the pathogenic site of the tumor. For patients exhibiting TE
in the filum terminale, the predominant clinical manifestations
were backache, pain and numbness in the lower extremities, among
others. In terms of the MRI scan, the tumor generally displayed
isointense T1 signals and iso- or hyperintense T2 signals whose
boundaries were relatively clear and would be strengthened
significantly on contrast MRI scans.
The tissue origin of ependymoma has always been
controversial (31), however the
general opinion is that it originates in the radial glias, namely
the tanycytes of ependyma and the precursor cells of neurogliocytes
(32,33). Ependymal cells, also termed
periventricular cells, have long cell processes extending to the
ependymal surface (28). In 1978,
Friede and Pollak (3) first proposed
TE, reporting6cases according to the characteristics of tumor cells
similar to tanycytes. Previous studies on this particular tumor
type suggested that it originated in the ependymal cells, and it
has been classified as a subtype of ependymoma in the WHO
classification of Tumors of the Central Nervous System in 2000
(7,26,34). The
genetic characteristics of TE remain unclear; however, it has been
reported in the literature that 2 cases were associated with
neurofibromatosis type 2 and its tumor suppressor gene
neurofibromin 2, which was inactivated; 1 case was potentially
caused by family genetic ependymoma (16,17,35).
The majority of TE diagnoses are based on its
histological characteristics, notably the tanycyte and
immunohistochemical characteristics (32). Electron microscopy is vital in
diagnosing TE, due to the difficulty in differentiating it from
similar tumors (8). The majority of
the cases in this group were initially diagnosed as schwannoma or
spinal meningioma by a preoperative imaging examination, and were
postoperatively confirmed as misdiagnoses. In pathological
diagnosis, improving the understanding of the histological
morphology of this tumor is required. Schwannoma is a common spinal
canal tumor, which is easy to confuse with TE due to its similar
histological morphology of elongated cytoplasmic processes and
absent atypia in nuclei. Schwannoma, in the spinal canal, always
occurs extra-medullary spaces with well-defined boundaries.
Histologically, its thick wall and hyalinized blood vessels are
also important in differentiating these two tumors; in addition,
schwannoma typically has an Antoni type A and B pattern. In routine
pathological examination, a definite diagnosis can be achieved by
immunohistochemically labeling for GFAP and S-100 proteins
(36). Spinal meningioma is another
example of a tumor that TE can be misidentified as. This tumor
originates from arachnoid granulations, the majority of which are
located subdurally-extra-medullarily, notably in the T vertebra;
additionally, dural tail sign is occasionally present in spinal
meningioma. This type of tumor is mainly composed of dural cells
arranged in patches, or nest-like bulks, of different sizes, with a
small amount of mesenchyme between the nest-like bulks. Dural cells
are characterized by the following: A large volume; obscure
boundaries presenting chimerism; abundant cytoplasm; large nuclei
in round or oval shape; and clear nuclear membranes (11,37). The
final example of a tumor that TE can be misidentified as is
subependymoma. This type of tumor, previously termed subependymal
astrocytoma, frequently affects middle-aged and elderly people,
with the majority of cases located in the fourth ventricle and
lateral ventricles. Histologically, its clustered nuclei are
embedded in a dense matrix of glial fibers and, along with the
formation of numerous follicular cavities, these can be used as
identifying factors in differentiating subependymoma from
ependymoma (6,7).
Currently, the primary treatment method for TE is
complete surgical resection of the tumor (2,38). For
patients undergoing complete surgical resection, postoperative
radiotherapy is not necessary (11,26). For
patients with postoperative recurrence, the treatments used are
surgical resection with postoperative radiotherapy (7,30). Of the
patients investigated in the present study group, only 1 patient
did not receive complete surgical resection of the tumor as the
tumor had adhered firmly to the spinal cord and wrapped around the
cauda equina. In order to decrease postoperative complications, a
partial resection was opted for instead of a complete surgical
resection. Therefore, the treatment principle for TE in the filum
terminale is to carefully separate tumor boundaries
intraoperatively, spare no effort to protect the cauda equina and
strive to achieve complete surgical resection. The prognosis of TE
is associated with the patient age, tumor site and the extent of
surgical resection. It has been reported that the 5-year and
10-year survival rates for TE are improved compared with those with
other subtypes of ependymoma (33,39).
In conclusion, the current case series and
literature review suggest that cauda equina TE should be taken into
consideration when the tumors exhibit enhancement on MRI. TE is the
rarest variant of ependymoma and its diagnosis largely depends on
histopathological examination. Neurosurgeons should be aware of
this lesion entity. Fortunately, cauda equina TE usually has a
favorable prognosis, and GTR or STR are expected whenever
possible.
Glossary
Abbreviations
Abbreviations:
|
TE
|
tanycytic ependymoma
|
|
WHO
|
World Health Organization
|
|
MRI
|
magnetic resonance imaging
|
|
JOA
|
Japanese Orthopedic Association
|
|
GTR
|
gross total resection
|
|
GFAP
|
glial fibrillary acidic protein
|
|
EMA
|
epithelial membrane antigen
|
|
T
|
thoracic
|
References
|
1
|
Gilbert MR, Ruda R and Soffietti R:
Ependymomas in adults. Curr Neurol Neurosci Rep. 10:240–247. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cepeda S, Hernández-Laín A, Munarriz PM,
González Martinez MA and Lagares A: Spinal tanycytic ependymoma
associated with neurofibromatosis type 2. Clin. Neuropathol.
33:311–314. 2014.
|
|
3
|
Friede RL and Pollak A: The cytogenetic
basis for classifying ependymomas. J Neuropathol Exp Neurol.
37:103–118. 1978. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ragel BT, Townsend JJ, Arthur AS and
Couldwell WT: Intraventricular tanycytic ependymoma: Case report
and review of the literature. J Neurooncol. 71:189–193. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu Z, Li J, Liu Z, Wang Q, Famer P, Mehta
A, Chalif D, Wang Y and Li JY: Supratentorial cortical ependymoma:
Case series and review of the literature. Neuropathology.
34:243–252. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Radhakrishnan N, Nair NS, Hingwala DR,
Kapilamoorthy TR and Radhakrishnan VV: Tanycytic ependymoma of
filum terminale: A case report. Clin Neur Neurosurg. 114:169–171.
2012. View Article : Google Scholar
|
|
7
|
Ydel Ortiz M, Pérez Berenguer JL, Acosta
Mercado J, Polo M, de Jesús-Garces O and Vega IE: Tanycytic
ependymoma in a 76-year-old Puerto Rican male. Int J Clin Exp
Pathol. 7:7789–7794. 2014.PubMed/NCBI
|
|
8
|
Mohindra S, Bal A and Singla N: Pediatric
tanycytic ependymoma of the cauda equina: Case report and review of
the literature. J Child Neurol. 23:451–454. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shintaku M and Sakamoto T: Tanycytic
ependymoma of the filum terminale with pleomorphic giant cells.
Brain Tumor Pathol. 26:79–82. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Funayama T, Sakane M, Yoshizawa T,
Takeuchi Y and Ochiai N: Tanycytic ependymoma of the filum
terminale associated with multiple endocrine neoplasia type 1:
First reported case. Spine J. 13:e49–e54. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tomek M, Jayajothi A, Brandner S,
Jaunmuktane Z, Lee CH and Davagnanam I: Imaging features of spinal
tanycytic ependymoma. Neuroradiol J. 29:61–65. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Spaar FW, Blech M and Ahyai A: DNA-flow
fluorescence-cytometry of ependymomas. Report on ten surgically
removed tumours. Acta Neuropathol. 69:153–160. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Langford LA and Barré GM: Tanycytic
ependymoma. Ultrastruct Pathol. 21:135–142. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dvoracek MA and Kirby PA: Intraoperative
diagnosis of tanycytic ependymoma: Pitfalls and differential
diagnosis. Diagn Cytopathol. 24:289–292. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kawano N, Yagishita S, Oka H, Utsuki S,
Kobayashi I, Suzuki S, Tachibana S and Fujii K: Spinal tanycytic
ependymomas. Acta Neuropathol. 101:43–48. 2001.PubMed/NCBI
|
|
16
|
Kobata H, Kuroiwa T, Isono N, Nagasawa S,
Ohta T and Tsutsumi A: Tanycytic ependymoma in association with
neurofibromatosis type 2. Clin Neuropathol. 20:93–100.
2001.PubMed/NCBI
|
|
17
|
Ueki K, Sasaki T, Ishida T and Kirino T:
Spinal tanycytic ependymoma associated with neurofibromatosis type
2-case report. Neurol Med Chir (Tokyo). 41:513–516. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Boccardo M, Telera S and Vitali A:
Tanycytic ependymoma of the spinal cord. Case report and review of
the literature. Neurochirurgie. 49:605–610. 2003.PubMed/NCBI
|
|
19
|
Ito T, Ozaki Y, Nakagawara J, Nakamura H,
Tanaka S and Nagashima K: A case of cervicomedullary junction
tanycytic ependymoma associated with marked cyst formation. Brain
Tumor Pathol. 22:29–33. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sato K, Kubota T, Ishida M and Handa Y:
Spinal tanycytic ependymoma with hematomyelia-case report. Neurol
Med Chir (Tokyo). 45:168–171. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shintaku M, Nagata N and Itoh H: Tanycytic
ependymoma of the spinal cord with anaplastic cytological features.
Brain Tumor Pathol. 26:7–10. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Du J, Zhou X, Tang Q, Ma H, Zhou H, Wang
J, Lu Z and Yin H: Tanycytic ependymoma: Two case reports and
review of the literature. Comp Clin Pathol. 18:449–453. 2009.
View Article : Google Scholar
|
|
23
|
Lim BS, Park SQ, Chang UK and Kim MS:
Spinal cord tanycytic ependymoma associated with neurofibromatosis
type 2. J Clin Neurosci. 17:922–924. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ishihama H, Nakamura M, Funao H, Ishii K,
Matsumoto M, Toyama Y and Chiba K: A rare case of spinal dumbbell
tanycytic ependymoma. Spine (Phila Pa 1976). 36:E612–E614. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tosun O, Turkoglu OF, Ozmen EK, Onursever
A and Arslan H: Spinal tanycytic ependymoma with diffusion
restriction on MRI. Acta Neurol Belg. 112:77–80. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Krisht KM and Schmidt MH: Tanycytic
ependymoma: A challenging histological diagnosis. Case Rep Neurol
Med. 2013:1707912013.PubMed/NCBI
|
|
27
|
Kuga Y, Ohnishi H, Kodama Y, Takakura S,
Hayashi M, Yagi R, Matsushima K, Okamoto K, Taomoto K and Takahashi
H: Cerebral and spinal cord tanycytic ependymomas in a young adult
with a mutation in the NF2 gene. Neuropathology. 34:406–413.
2014.PubMed/NCBI
|
|
28
|
Furlan JC, Chui MH, Croul SE and Kongkham
P: Mystery case: Tanycytic ependymoma of the conus medullaris: A
rare cause of low back pain. Neurology. 82:e212–e213. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Maugeri R, Giugno A, Graziano F, Visocchi
M, Giller C and Iacopino DG: Delayed chronic intracranial subdural
hematoma complicating resection of a tanycytic thoracic ependymoma.
Surgical Neurol Int. 7 Suppl 1:S20–S22. 2016. View Article : Google Scholar
|
|
30
|
Singla N, Kapoor A, Radotra BD and
Chatterjee D: Tanycytic ependymoma of cervical cord presenting with
spontaneous intratumoral hemorrhage. Spine J. 16:e733–e734. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rigante L, Novello M, Massimi L and
Caldarelli M: A cortical cystic epileptogenic lesion: Tanycytic
ependymoma. Acta Neurol Belg. 113:523–525. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Divito A, Keller JT, Hagen M and
Zuccarello M: Vestibular schwannoma or tanycytic ependymoma:
Immunohistologic staining reveals. Surg Neurol Int. 5:1582014.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Agarwal S, Stevenson ME, Sughrue ME,
Wartchow EP, Mierau GW and Fung KM: Features of intraventricular
tanycytic ependymoma: Report of a case and review of literature.
Int J Clin Exp Pathol. 7:3399–3407. 2014.PubMed/NCBI
|
|
34
|
Radner H, Blümcke I, Reifenberger G and
Wiestler OD: The new WHO classification of tumors of the nervous
system 2000. Pathologe. 23:260–283. 2002.(In German). View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Dimopoulos VG, Fountas KN and Robinson JS:
Familial intracranial ependymomas. Report of three cases in a
family and review of the literature. Neurosurg Focus.
20:E82006.PubMed/NCBI
|
|
36
|
Erdogan E, Ongürü O, Bulakbasi N, Baysefer
A, Gezen F and Timurkaynak E: Schwannoma of the lateral ventricle:
Eight-year follow-up and literature review. Minim Invasive
Neurosurg. 46:50–53. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shukla S, Malhotra KP, Awasthi NP, Husain
N and Singh SK: Intraventricular tanycytic ependymoma: An uncommon
fibrillary variant. Neurol India. 62:200–201. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kambe A, Kurosaki M, Watanabe T and
Nakazato Y: Pediatric supratentorial cortical tanycytic ependymoma
associated with absence seizures. Clin Neuropathol. 33:308–310.
2014.PubMed/NCBI
|
|
39
|
Zhang S, Wang X, Zhang Z and Chen Y:
Tanycytic ependymoma arising from the right lateral ventricle: A
case report and review of the literature. Neuropathology.
28:427–432. 2008. View Article : Google Scholar : PubMed/NCBI
|