Introduction
Gastric carcinoma (GC) is a common and very
aggressive malignancy of the digestive system with poor early
diagnosis. Adenocarcinoma is a multifactorial disease that usually
arises from the epithelium and is categorized into two pathological
variants: intestinal and diffuse (1).
Currently, GC therapies are mainly focused on surgical removal,
radiotherapy and chemotherapy. However, GC therapies are often
accompanied by limited efficacy, drug resistance, or even serious
side effects (2). Despite numerous
improvements in GC therapies, GC patient survival is less than 40%
(3). Therefore, identifying highly
effective and safe medicines is necessary.
Targeted molecular therapy for GC has attracted
interest (2). Transmembrane protein
with epidermal growth factor (EGF)-like and two follistatin motifs
2 (TMEFF2) is a potential biomarker for human GC (4). The downregulation of TMEFF2 frequently
occurs in human gastric cancer tissue, and its levels correlate
with tumor aggressiveness and survival time in patients (5). In contrast, upregulated TMEFF2
suppresses tumor progression and may be a potential effective
strategy for preventing human GC. TMEFF2 plays an important role in
regulating the cell cycle, apoptosis, and DNA repair and is thus
directly associated with tumorigenesis in GC cells (4). Therapeutic strategies for GC that target
TMEFF2 include proliferation inhibitors, apoptosis promoters, and
genetic stability regulators (6),
providing possibilities for exploring more effective medicines to
block human GC.
Vitamin C, which is known as a water-soluble hexose
derivative, serves as a natural antioxidant (7). Dietary vitamin C prevents the occurrence
of human GC by increasing mucosal immune responses, eliminating
free radicals, and decreasing the N-nitrosamine content in gastric
juice (8). Epidemiological studies
revealed that vitamin C deficiency in humans is linked to more
severe H. pylori-associated gastritis and a higher risk of
human GC (9). Serum vitamin C levels
may be used to predict GC risk (10).
Higher concentrations of circulating vitamin C decrease GC risk.
TMEFF2 is a transmembrane protein that participates in various
cellular functions by binding to protein tyrosine phosphatase
(SPH-1), a key regulator of gastric carcinogenesis, and is thus
closely related to tumorigenesis in GC cells (6). Evidence suggests that proinflammatory
cytokines, for example, interleukin (IL)-1β and tumor necrosis
factor (TNF)-α, induce TMEFF2 ectodomain shedding via the
activation of the nuclear factor (NF)-κB signaling pathway and thus
lead to protease inactivity (11).
Vitamin C is a highly effective anti-inflammatory agent that
reduces the activation of the NF-κB signaling pathway (12). However, data on the interaction
between serum vitamin C concentrations and TMEFF2 expression are
currently scarce.
Vitamin C prevents the proliferation of normal human
gastric epithelial cells (GES-1) (13) and AGS human gastric adenocarcinoma
cells (9,14), depending in part on its antioxidant
properties. AGS cells are a type of GC cell line. However, the
exact molecular mechanisms are not completely elucidated. TMEFF2
silencing promotes the proliferation of human gastric cancer cells
or normal gastric epithelial cells, such as AGS, MKN28, SGC7901,
MGC803, or GES-1 cells; in contrast, upregulated TMEFF2 inhibits
cell proliferation. However, little is known about the roles of
TMEFF2 in the antiproliferative effects of vitamin C and tyrphostin
AG490 on human gastric cancer cells.
In the present study, fifty patients with GC from
Jiangsu, China, were enrolled. We tested vitamin C concentrations
(µg/ml) in the peripheral blood and mRNA levels of TMEFF2 in
gastric cancer tissue. Our study suggested the existence of a
positive correlation between serum vitamin C concentrations and
TMEFF2 expression in human gastric cancer. In addition, the
siRNA-TMEFF2-induced proliferation of GES-1 and AGS cells was
successfully established. Vitamin C and tyrphostin AG490, which are
known as antiproliferative agents in GC cells, were used for
treatment. Our data indicated the involvement of TMEFF2 in the
antiproliferative effects of vitamin C and AG490 on these two cell
types.
Materials and methods
Plasma vitamin C assay
Fifty patients with GC were recruited. The present
study was approved by Yancheng Third People's Hospital (Jiangsu,
China), and written informed agreement was obtained from each
participant involved in this study. Vitamin C levels (µg/ml) in the
peripheral blood of GC patients were determined using commercially
available kits (www.nijcbio.com), according to the manufacturer's
instructions.
Cell culture and treatment
Cell culture medium was prepared with the following
ingredients (9:1:0.01 (v/v/v)): RPMI-1640 (SH30809.01B; HyClone; GE
Healthcare Life Sciences, Logan, UT, USA); fetal bovine serum
(16000-044, Gibco); and penicillin/streptomycin (100×, P1400-100;
Beijing Solarbio Science & Technology Co., Ltd., Beijing,
China), respectively. The concentrations of penicillin and
streptomycin in the medium were 100 U/ml and 100 mg/ml,
respectively. After preparation, the mixture was stored at 4°C.
GES-1 and AGS cells were cultured in this medium at 37°C under 5%
CO2. Cells in log-phase growth were used in the
following study.
To establish the knockdown of TMEFF2 within GES-1
and AGS cells, Lipofectamine 2000 (11668-019; Invitrogen; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) was used to transfect
cells with siRNA-TMEFF2 according to the manufacturer's protocol.
After transduction for 48 h, the efficacy of stable TMEFF2
silencing was verified by RT-PCR and western blotting. The
siRNA-TMEFF2 sequences targeting TMEFF2 mRNA (GenBank NM_016192.3)
were as follows: 5′-GCUGGAAUUGCUCUGGUUATT-3′ (sense) and
5′-UAACCAGAGCAAUUCCAGCTT-3′ (antisense) at position 567–585;
5′-GGAGACAUCCACCUGUGAUTT-3′ (sense) and 5′-AUCACAGGUGGAUGUCUCCTT-3′
(antisense) at position 880–898; and 5′-GCAGGUGUGAUGCUGGUUATT-3′
(sense) and 5′-UAACCAGCAUCACACCUGCTT-3′ (antisense) at position
1275–1293. In addition, a scrambled siRNA was included as a
negative control (siRNA-NC).
To study the effects of vitamin C on the
proliferation of GES-1 and AGS cells, cells in log-phase growth
were treated with vitamin C (an L form of ascorbic acid, A7506;
Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) at concentrations of
10−9, 10−8, 10−7 and
10−6 mol/l and then cultured as described above.
To study whether the antioxidants vitamin C, vitamin
E, N-acetyl cysteine (NAC), resveratrol, and glutathione (GSH)
could regulate TMEFF2 expression, GES-1 and AGS cells were treated
separately with 20 µmol/l vitamin C, 20 µmol/l vitamin E (CAS no:
14638-18-7; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany), 5
mmol/l NAC (CAS no: 616-91-1; Sigma-Aldrich; Merck KGaA), 50 µmol/l
resveratrol (CAS no: 501-36-0, Sigma, USA), and 40 µg/l GSH (CAS
no: 70-18-8; Shanghai Aladdin Bio-Chem Technology Co., Ltd.) and
then cultured as described above.
To study the effects of vitamin C and AG490 on the
proliferation of siRNA-TMEFF2-transfected GES-1 and AGS cells,
after transduction for 48 h, the cells were treated separately with
vitamin C (10−7 and 10−6 mol/l) and 30 µmol/l
AG490 (Sigma-Aldrich; Merck KGaA) and then cultured as described
above.
Cell proliferation assay
A Cell Counting Kit-8 (CCK-8) kit (CP002, SAB) was
used to assess cell viability. Cells with or without siRNA-TMEFF2
transfection were fixed in a 96-well plate (3×103
cells/well) in 100 µl of culture medium. After treatment, 10 µl of
CCK-8 solution was added to each well at 0, 24, 48 or 72 h and
incubated for an additional 1 h. The optical density (OD) was
detected via a microplate reader (Thermo Fisher Scientific, Inc.)
at 450 nm.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
To assess the mRNA expression of TMEFF2 in gastric
cancer tissue, as well as in GES-1 and AGS cells, RT-qPCR was
conducted following the previously reported procedure (6). Primers targeting the mRNA sequence of
TMEFF2 (GenBank NM_001305134.1) at position 851–1089 were designed
as follows: 5′-CATGAAGGCTCTGGAGAAAC-3′ (forward) and
5′-CATCGACCCAAAGACATGAC-3′ (reverse). Primers targeting the mRNA
sequence of glyceraldehyde-3-phosphate dehydrogenase (GAPDH)
(GenBank NM_0,012,56799.1) at position 1065–1174 were designed as
follows: 5′-CACCCACTCCTCCACCTTTG-3′ (forward) and
5′-CCACCACCCTGTTGCTGTAG-3′ (reverse). The 2−∆∆Cq method
was used for relative quantification (15).
Western blot analysis
Western blotting methods were conducted as
previously reported (6). The
following antibodies were adopted: A TMEFF2 antibody (1 µg/ml,
Ab50002; Abcam), a PCNA antibody (1:1,000 dilution, cat. no: 13110,
CST), a STAT3 antibody (1:100 dilution, Ab50761; Abcam), a p-STAT3
antibody (1:200,000 dilution, cat. no: Ab76315, Abcam), and a GAPDH
antibody (1:2,000 dilution, cat. no: 5174; Cell Signaling
Technology, Inc., Danvers, MA, USA).
Statistical analysis
Triplicate experiments were conducted. The data are
presented as the mean (n=3) ± standard error of the mean. Pearson's
correlation coefficients (r) and linear regression were carried out
to analyze the correlation between serum vitamin C levels (µg/ml)
and mRNA levels of TMEFF2 in gastric cancer tissue. One-way
analysis of variance with Tukey's post hoc test was employed to
assess differences sing GraphPad Prism v7.00 (GraphPad Software,
Inc., La Jolla, CA, USA). P<0.05 was considered to indicate a
statistically significant difference.
Results
Positive correlation between vitamin C
and TMEFF2 in GC patients
To study the association between vitamin C and
TMEFF2 in the development of human GC, fifty GC patients from
Jiangsu, China, were recruited. Vitamin C concentrations in the
peripheral blood and the mRNA levels of TMEFF2 in gastric tissue
from GC patients were measured. The serum vitamin C levels of
healthy people living in Wuwei City, a GC endemic region in China,
ranged from 2.9–8.5 µg/ml (16). In
Europe, the mean concentrations of plasma vitamin C in patients of
different ages with GC ranged from 31.4–36.6 µmol/ml (5.5–6.4
µg/ml) but were 30.5–35.0 µmol/ml (5.4–6.2 µg/ml) in the
corresponding group of healthy people (17). In our present study, the vitamin C
levels (µg/ml) in peripheral blood from fifty GC patients ranged
from 1.94–7.67 µg/ml. In addition, RT-PCR was performed to measure
the mRNA levels of TMEFF2 in gastric tissue from GC patients.
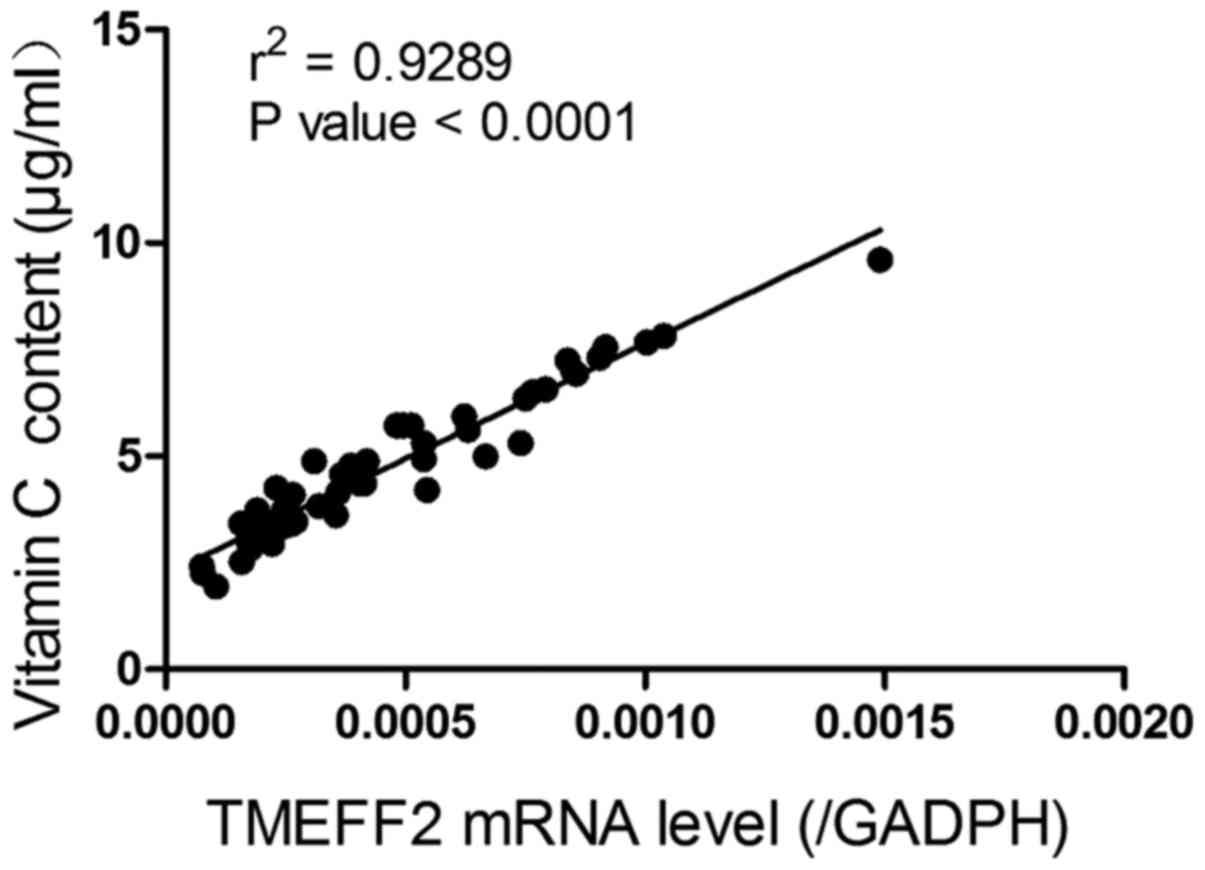
Pearson's r and linear regression were carried out to analyze
correlations. Fig. 1 reveals a
positive correlation between vitamin C content and TMEFF2 mRNA
levels in GC patients (r2=0.9289, P<0.0001).
Effect of vitamin C on cell
proliferation in the GES-1 and AGS cell lines
GES-1 and AGS cells were treated with different
concentrations of vitamin C (10−9, 10−8,
10−7 and 10−6 mol/l) (Fig. 2). At 0, 24, 48 and 72 h, CCK-8 was
performed to detect cell viability. In Fig. 2A and C, with vitamin C treatment,
significantly decreased cell viability was observed in a
dose-dependent manner, with minimum values observed at
10−6 mol/l, confirming the inhibitory effects of vitamin
C on these two cell lines. We selected two optimal concentrations
(10−7 and 10−6 mol/l) of vitamin C to treat
GES-1 and AGS cells for 48 h, and protein levels of proliferating
cell nuclear antigen (PCNA), which is known as a proliferation
marker, and the signal transducer and activator of transcription 3
(STAT3) pathway were then evaluated. As shown in Fig. 2B and D, vitamin C had almost no effect
on total STAT3 levels but decreased the levels of
phosphorylated-STAT3 (p-STAT3) and PCNA in a dose-dependent manner,
further confirming the antiproliferative effect of vitamin C at the
molecular level.
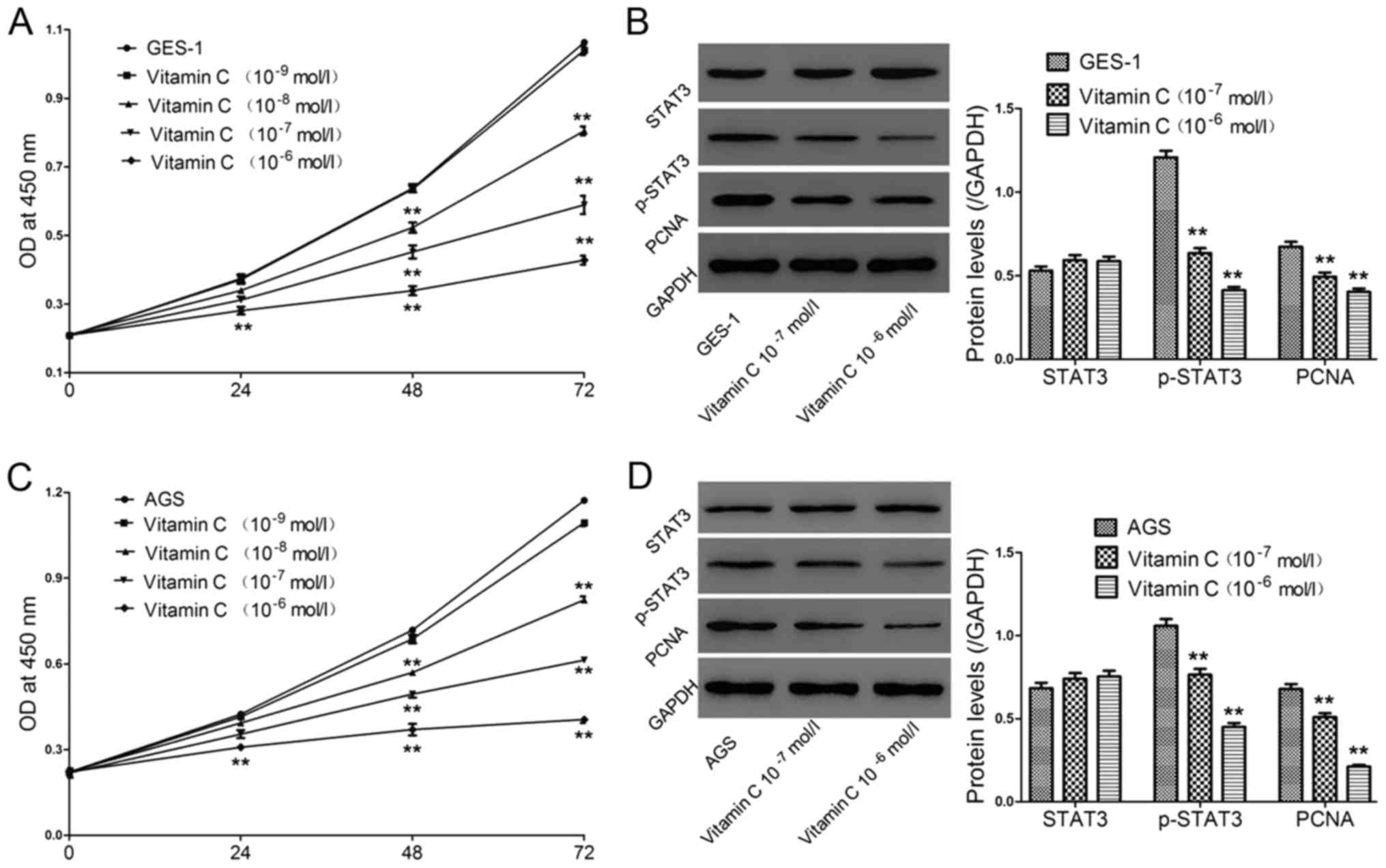 | Figure 2.Effect of vitamin C on cell
proliferation in the GES-1 and AGS cell lines. Cells were treated
with vitamin C at concentrations of 10−9,
10−8, 10−7 and 10−6 mol/l and (A)
GES-1 and (C) AGS cell viability was assessed by the Cell Counting
Kit-8 method at 0, 24, 48 and 72 h. In addition, (B) GES-1 and (D)
AGS cells were treated with vitamin C at concentrations of
10−7 and 10−6 mol/l for 48 h, and the
expression levels of p-STAT3 and PCNA were quantified via western
blot analysis. **P<0.01 vs. GES-1 or AGS cells only. STAT3,
signal transducer and activator of transcription 3; p-,
phosphorylated; PCNA, proliferating cell nuclear antigen; OD,
optical density. |
Vitamin C promotes TMEFF2 expression
in the GES-1 and AGS cell lines
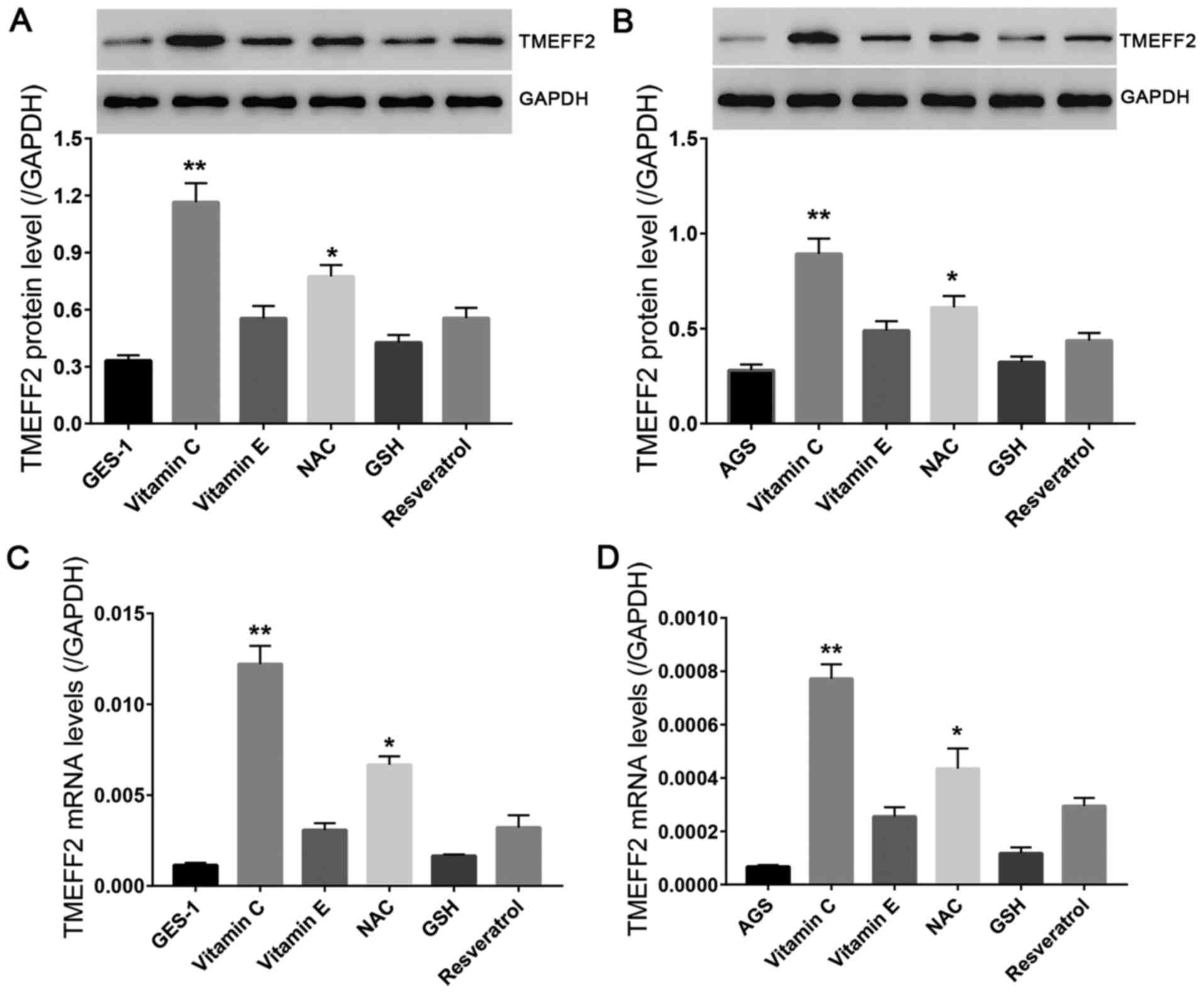
In the present study, we assessed the effects of
vitamin C and several other antioxidants on the protein and mRNA
levels of TMEFF2 in GES-1 and AGS cells. Cells were treated
separately with vitamin C (20 µmol/l), vitamin E (20 µmol/l), NAC
(5 mmol/l), GSH (40 µg/l), and resveratrol (50 µmol/l). After
treatment for 48 h, the protein and mRNA levels of TMEFF2 in the
two cell lines were determined. As shown in Fig. 3, vitamin C and NAC significantly
increased the protein and mRNA levels of TMEFF2, with the strongest
effect observed in the vitamin C-treated group, suggesting a
positive correlation between vitamin C and TMEFF2 at the molecular
level. Unfortunately, vitamin E, resveratrol, and GSH enhanced the
expression of TMEFF2 slightly or moderately.
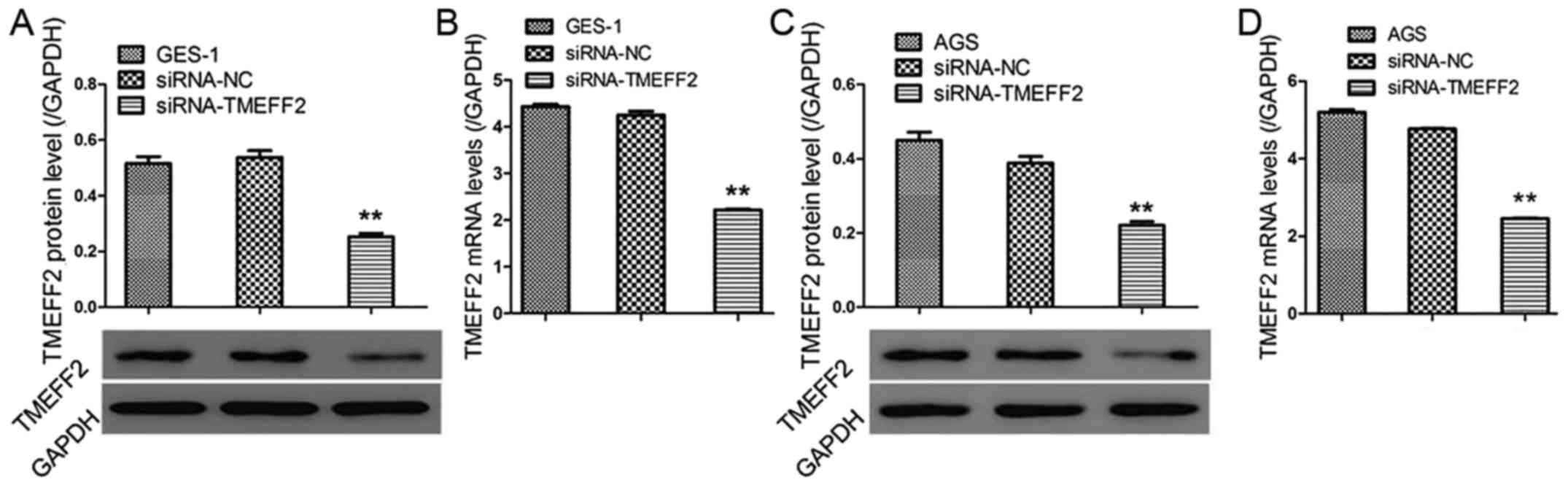
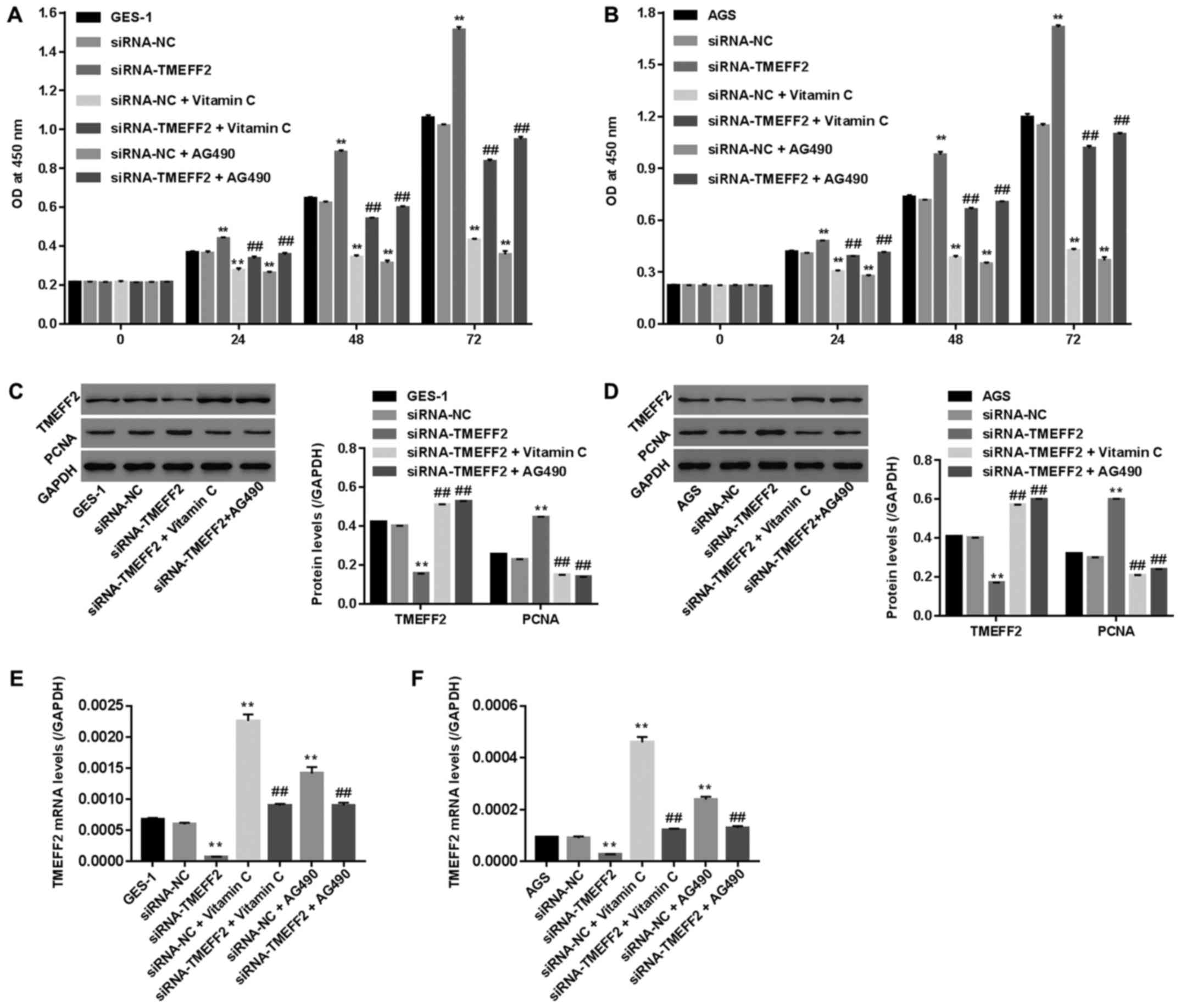
Successful establishment of a
pro-proliferative effect in GES-1 and AGS cells after siRNA-TMEFF2
treatment
Fig. 4 shows
significantly decreased protein and mRNA levels of TMEFF2 in GES-1
and AGS cells, demonstrating the successful establishment of TMEFF2
silencing in the two cell lines. In parallel, cell viability and
protein levels of PCNA were also assessed in
siRNA-TMEFF2-transfected cells. Our data suggested that the
knockdown of TMEFF2 significantly inhibited cell viability and
downregulated PCNA levels (Fig. 5),
demonstrating the antiproliferative effect of TMEFF2 silencing on
GES-1 and AGS cells.
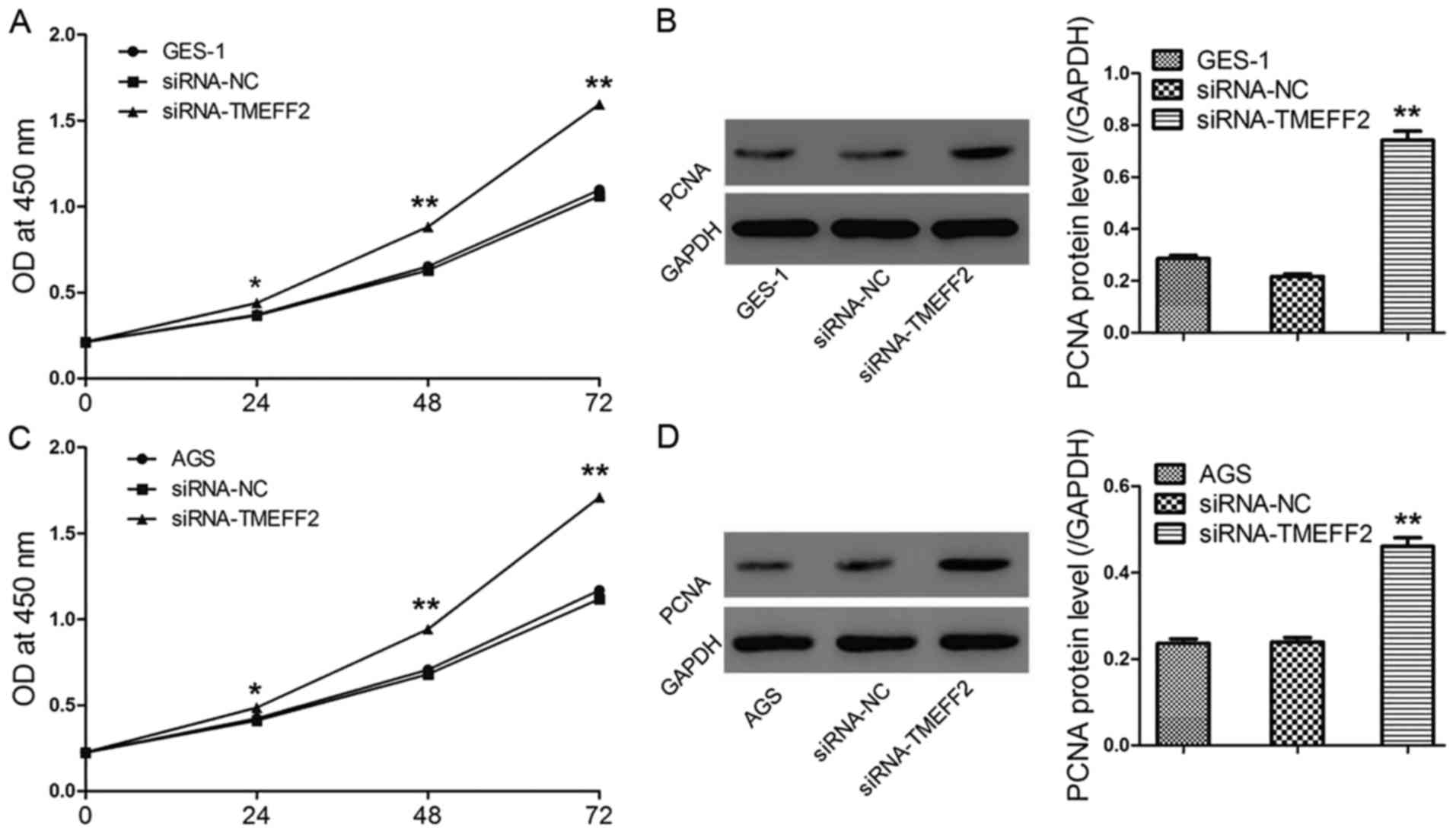
Upregulated TMEFF2 expression is directly involved
in the antiproliferative effects of vitamin C and AG490 on GES-1
and AGS cells. To explore whether upregulated TMEFF2 was involved
in the antiproliferative effect of vitamin C or AG490 on GES-1 and
AGS cells, siRNA-TMEFF2-transfected cells were treated with vitamin
C (10−6 mol/l) or AG490 (30 µmol/l) for 48 h. Cell
viability, mRNA levels of TMEFF2, and protein levels of TMEFF2 and
PCNA were assessed. As shown in Fig.
6, the knockdown of TMEFF2 significantly promoted cell
viability, decreased the expression of TMEFF2 and increased the
expression of PCNA, and these changes could be obviously reversed
with additional vitamin C or AG490 treatment. Vitamin C exerted
effects similar to those of AG490; cell viability and PCNA
expression were significantly reduced and basal TMEFF2 expression
was remarkably increased compared with the values of the
siRNA-TMEFF2 group, demonstrating that vitamin C and AG490
inhibited cell proliferation by increasing TMEFF2 levels.
Discussion
Multiple steps and multiple factors contribute to
the occurrence of GC. TMEFF2 dysregulation is involved in human GC
and is an auxiliary indicator for the early diagnosis of GC
(6). Vitamin C exerts protective
effects on human gastric tissue and is a potential chemopreventive
drug for human GC (18). In 2005, Luo
et al (19) reported that the
average levels of serum vitamin C in 293 healthy people were
5.74±2.79 mg/l (µg/ml); however, in this study, the peripheral
blood levels of vitamin C in fifty GC patients were 2–10 µg/ml,
suggesting a statistically nonsignificant difference in serum
vitamin C content between healthy people and GC patients; thus,
there may be multiple factors that contribute to the GC process.
TMEFF2 is inactivated by proinflammatory cytokines (11), whereas vitamin C is an effective
anti-inflammatory agent (12).
However, the interaction between serum vitamin C concentrations and
TMEFF2 expression remained poorly understood. In our present study,
we tested the interaction between vitamin C content in the
peripheral blood and TMEFF2 mRNA levels in gastric cancer tissue,
and the result was statistically significant. In addition, we found
that vitamin C increased the protein and mRNA levels of TMEFF2 in a
dose-dependent manner in GES-1 and AGS cells, revealing a positive
correlation between vitamin C and TMEFF2 at the molecular level and
suggesting that enhanced transcription and translation of TMEFF2
mRNA was the underlying biological mechanism. Our data indicated
that serum vitamin C content may be a predictor for TMEFF2 levels
in gastric tissue from GC patients.
Evidence suggests that vitamin C significantly
prevents the proliferation of human SGC-7901 gastric adenocarcinoma
cells at concentrations of 10−4 to 10−8 mol/l
(20). Other antioxidants, such as
N-acetyl cysteine (NAC) and resveratrol, have been demonstrated to
exert antiproliferative effects on GES-1 or AGS cells at mM
concentrations (21,22), demonstrating less sensitivity than
vitamin C. In our present study, human GES-1 and AGS cells were
treated with vitamin C at doses of 10−9,
10−8, 10−7 and 10−6 mol/l. We
confirmed obvious inhibitory effects of vitamin C on the
proliferation of GES-1 and AGS cells at doses of 10−8,
10−7 and 10−6 mol/l after 24, 48 and 72 h.
PCNA, which serves as a proliferation marker, is widely used to
measure cell proliferation. Vitamin C increased the protein
expression of PCNA in nitrofen-stimulated human pneumocytes
(23). However, whether PCNA could be
regulated by vitamin C in GES-1 and AGS cells remained largely
unknown. In our present study, we found that vitamin C
significantly decreased PCNA expression, suggesting the involvement
of PCNA in the antiproliferative effects of vitamin C. Moreover,
the activation of the STAT3 pathway is widely implicated in the
growth and survival of human gastric cancer cells. The generation
of reactive oxygen species (ROS) is required for STAT3 activation.
Vitamin C, as a powerful antioxidant reagent, abrogates STAT3
activation in COS-7 cells (24).
However, little is known about whether vitamin C acts on the STAT3
pathway in GES-1 and AGS cells. Our data suggested that vitamin C
decreased p-STAT3 expression but had no effect on the expression of
total STAT3, indicating that blocking the STAT3 signaling pathway
was the underlying mechanism in this process.
TMEFF2 is downregulated in human gastric cancer
cells (6). The basal mRNA level of
TMEFF2 was much lower in AGS cells than that in GES-1 cells
(6), which was further confirmed in
our study. TMEFF2 overexpression in gastric cancer cells inhibits
cell proliferation (4). In our
present study, the siRNA-mediated knockdown of TMEFF2 was
established. We confirmed that TMEFF2 silencing induced
proliferation in GES-1 and AGS cells by obviously promoting cell
viability and increasing PCNA expression. More importantly, with
additional vitamin C treatment (10−6 mol/l), TMEFF2
expression was enhanced above basal levels, and cell proliferation
was significantly reduced, demonstrating the upregulation of TMEFF2
in response to the antiproliferative effect of vitamin C in GES-1
and AGS cells. Vitamin C exerts its antiproliferative effect on
human gastric cancer cells as an antioxidant. In this study, we
further assessed the effects of other antioxidants (vitamin E, NAC,
resveratrol, and GSH) on the protein and mRNA levels of TMEFF2 in
GES-1 and AGS cells. We found that NAC remarkably upregulated
TMEFF2 expression (P<0.05), whereas vitamin E, GSH, and
resveratrol increased TMEFF2 expression slightly or moderately.
Tyrphostin AG490, as an inhibitor of STAT3, is
widely used to produce an antiproliferative response in human
normal gastric epithelial or GC cells. High doses of AG490 (greater
than 20 µmol/l) significantly inhibit cell growth in GES-1 and AGS
cells (25). TMEFF2 and STAT3 are
mutually regulated and negatively correlated (5). However, whether TMEFF2 was involved in
the antiproliferative effect of AG490 remained unknown. To further
substantiate the hypothesis that the upregulation of TMEFF2 could
represent an effective strategy to prevent the proliferation of
GES-1 and AGS cells, siRNA-TMEFF2-transfected cells were treated
with AG490 (30 µmol/l). AG490 exerted an effect similar to that of
vitamin C, and cell viability and PCNA protein levels were
dramatically reduced, while basal TMEFF2 expression was obviously
increased, suggesting that AG490 inhibited cell proliferation by
increasing TMEFF2 levels and verifying that the upregulation of
TMEFF2 could represent an effective strategy for preventing
tumorigenesis in GES-1 and AGS cells.
In brief, our data revealed a positive correlation
between serum vitamin C levels (µg/ml) and mRNA levels of TMEFF2 in
gastric cancer tissue. In addition, vitamin C inhibited the
proliferation of GES-1 and AGS cells by downregulating the
expression of PCNA and blocking the STAT3 signaling pathway. Our
data also revealed that the upregulation of TMEFF2 expression was
involved in the antiproliferative effects of vitamin C and AG490 on
GES-1 and AGS cells, further substantiating the role of TMEFF2 as a
target for exploring novel drugs that block human GC.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used in the present study are available
from the corresponding author on reasonable request.
Authors' contributions
AW and LW designed the study. HH and JX performed
the experiments. WJ analyzed the data and wrote the paper. AW
reviewed and edited the manuscript. All authors have read and
approved the manuscript.
Ethics approval and consent to
participate
The present study was approved by Yancheng Third
People's Hospital (Jiangsu, China), and written informed consent
was obtained from each participant involved in this study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wesołowska M, Pawlik P and Jagodziński PP:
The clinicopathologic significance of estrogen receptors in human
gastric carcinoma. Biomed Pharmacother. 83:314–322. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhang Z: Advances in study on molecular
targeted therapy for advanced gastric cancer. Chin J Gastroenterol.
17:251–254. 2012.
|
|
3
|
Jang BG and Kim WH: Molecular pathology of
gastric carcinoma. Pathobiology. 78:302–310. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sun T, Du W, Xiong H, Yu Y, Weng Y, Ren L,
Zhao H, Wang Y, Chen Y, Xu J, et al: TMEFF2 deregulation
contributes to gastric carcinogenesis and indicates poor survival
outcome. Clin Cancer Res. 13:4689–4704. 2015.
|
|
5
|
Sun TT, Tang JY, Du W, Zhao HJ, Zhao G,
Yang SL, Chen HY, Hong J and Fang JY: Bidirectional regulation
between TMEFF2 and STAT3 may contribute to Helicobacter
pylori-associated gastric carcinogenesis. Int J Cancer.
136:1053–1064. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tiantian S: The role of TMEFF2 and long
non-coding RNA gasi in gastric carcinoma. Shanghai Jiao Tong Univ.
190:2015.
|
|
7
|
Wang Y, Chen K, Liu X and Qiyuan H:
Research progress of oxymatrine and vitamin C in inhibiting the
proliferation of gastric carcinoma cells. China Med Herald.
14:52–55. 2017.
|
|
8
|
Jakszyn P, Agudo A, Lujanbarroso L,
Bueno-de-Mesquita HB, Jenab M, Navarro C, Palli D, Boeing H, Manjer
J, Numans ME, et al: Dietary intake of heme iron and risk of
gastric cancer in the European prospective investigation into
cancer and nutrition study. Int J Cancer. 130:2654–2663. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nagappan A, Park HS, Park KI, Kim JA, Hong
GE, Kang SR, Zhang J, Kim EH, Lee WS, Won CK and Kim GS: Proteomic
analysis of differentially expressed proteins in vitamin C-treated
AGS cells. BMC Biochem. 14:242013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jenab M, Riboli E, Ferrari P, Sabate J,
Slimani N, Norat T, Friesen M, Tjønneland A, Olsen A, Overvad K, et
al: Plasma and dietary vitamin C levels and risk of gastric cancer
in the European Prospective Investigation into Cancer and Nutrition
(EPIC-EURGAST). Carcinogenesis. 27:2250–2257. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin H, Wada K, Yonezawa M, Shinoki K,
Akamatsu T, Tsukui T and Sakamoto C: Tomoregulin ectodomain
shedding by proinflammatory cytokines. Life Sci. 73:1617–1627.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cárcamo JM, Pedraza A, Bórquez-Ojeda O and
Golde DW: Vitamin C suppresses TNF alpha-induced NF kappa B
activation by inhibiting I kappa B alpha phosphorylation.
Biochemistry. 41:12995–13002. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang Y, Yuan L, Yao C, Ding L, Li C, Fang
J, Sui K, Liu Y and Wu M: A combined toxicity study of zinc oxide
nanoparticles and vitamin C in food additives. Nanoscale.
6:15333–15342. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nagappan A, Park KI, Park HS, Kim JA, Hong
GE, Kang SR, Lee DH, Kim EH, Lee WS, Won CK and Kim GS: Vitamin C
induces apoptosis in AGS cells by down-regulation of 14-3-3σ via a
mitochondrial dependent pathway. Food Chem. 135:1920–1928. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Luo HZ, Mi DH, Jing TZ, Xu Q, Yang WS,
Yang GQ, Wang SZ, Liu GH and Su CY: Risk factors of gastric cancer
in Wuwei city-an endemic region of gastric cancer. Ai Zheng.
24:563–566. 2005.(In Chinese). PubMed/NCBI
|
|
17
|
Duell EJ, Lujan-Barroso L, Llivina C,
Muñoz X, Jenab M, Boutron-Ruault MC, Clavel-Chapelon F, Racine A,
Boeing H, Buijsse B, et al: Vitamin C transporter gene (SLC23A1 and
SLC23A2) polymorphisms, plasma vitamin C levels and gastric cancer
risk in the EPIC cohort. Genes Nutr. 8:549–560. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yuan JM, Ross RK, Gao YT, Qu YH, Chu XD
and Yu MC: Prediagnostic levels of serum micronutrients in relation
to risk of gastric cancer in Shanghai, China. Cancer Epidemiol
Biomarkers Prev. 13:1772–1780. 2004.PubMed/NCBI
|
|
19
|
Luo HZ, Mi DH, Jing TZ, Xu Q, Yang WS,
Yang GQ, Wang SZ, Liu GH and Su CY: Risk factors of gastric cancer
in Wuwei City-an endemic region of gastric cancer. Ai Zheng.
24:563–566. 2005.(In Chinese). PubMed/NCBI
|
|
20
|
Cai M, Chen H and Shi K: Effects of
selenium, beta-carotene, vitamin C, vitamin B_(12) and Ge-132 on
human gastric cancer cell proliferation. Acta Univ Med Secondae
Shanghai. 14:225–228. 1994.
|
|
21
|
Koo J, Dong K and Park K: The inhibitory
effect of Resveratrol on the growth of human gastric cancer cells
(AGS) in vitro. Cancer Res. 68:5664. 2008.
|
|
22
|
Choi YH, Yong JK, Kim SH, Seon HM, Bokyung
S, Kim DH, Hwang SY, Kim M, Yoon JH, Moon HY, et al: Abstract 1773:
MHY-449 induces apoptotic cell death through ROS- and
caspase-dependent pathways in AGS human gastric cancer cells.
Cancer Res. 75:1773. 2015. View Article : Google Scholar
|
|
23
|
González-Reyes S, Martínez L and Tovar JA:
Vitamin C rescues in part the effects of nitrofen on cultured human
pneumocytes. Pediatr Surg Int. 20:258–262. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sun X, Wu S and Xing D: The reactive
oxygen species-Src-Stat3 pathway provokes negative feedback
inhibition of apoptosis induced by high-fluence low-power laser
irradiation. FEBS J. 277:4789–4802. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ding L, Xu Y, Zhang W, Deng Y, Si M, Du Y,
Yao H, Liu X, Ke Y, Si J and Zhou T: MiR-375 frequently
downregulated in gastric cancer inhibits cell proliferation by
targeting JAK2. Cell Res. 20:784–793. 2010. View Article : Google Scholar : PubMed/NCBI
|