Introduction
Glomus tumor is a mesenchymal neoplastic
proliferation composed of modified smooth muscle cells arising from
the perivascular structure called a glomus body (1). This tumor occurs most commonly in the
peripheral soft tissues and extremities, and is seldom found in
internal organs (2). Gastric glomus
tumors are rare, with an estimated frequency of approximately 1%
that of gastrointestinal stromal tumors (GISTs) (3,4).
In this report, we present a case of gastric glomus
tumor in a 39-year-old male who was treated by a laparoscopic
distal gastrectomy. The clinical characteristics of previously
reported cases are also discussed.
Case report
A 39-year-old Japanese man was referred to our
hospital for further examination of gastric submucosal tumor (SMT)
diagnosed during a medical check-up. His past medical history and
family history were unremarkable. The laboratory findings were
almost within normal limits, as were serum levels of
carcinoembryonic antigen and cancer antigen 19-9.
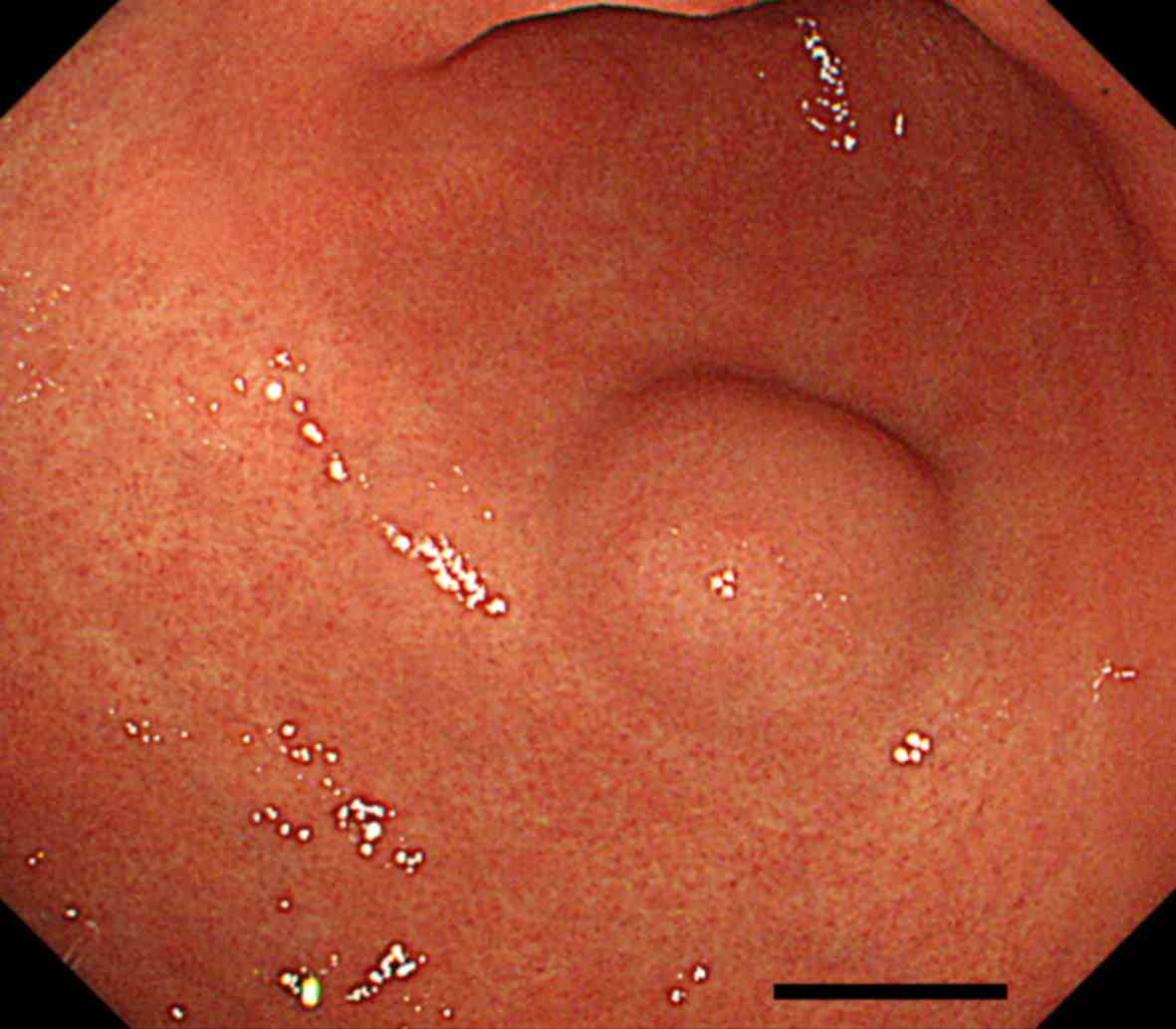
Esophagogastroduodenoscopy (EGD) revealed a SMT of approximately 2
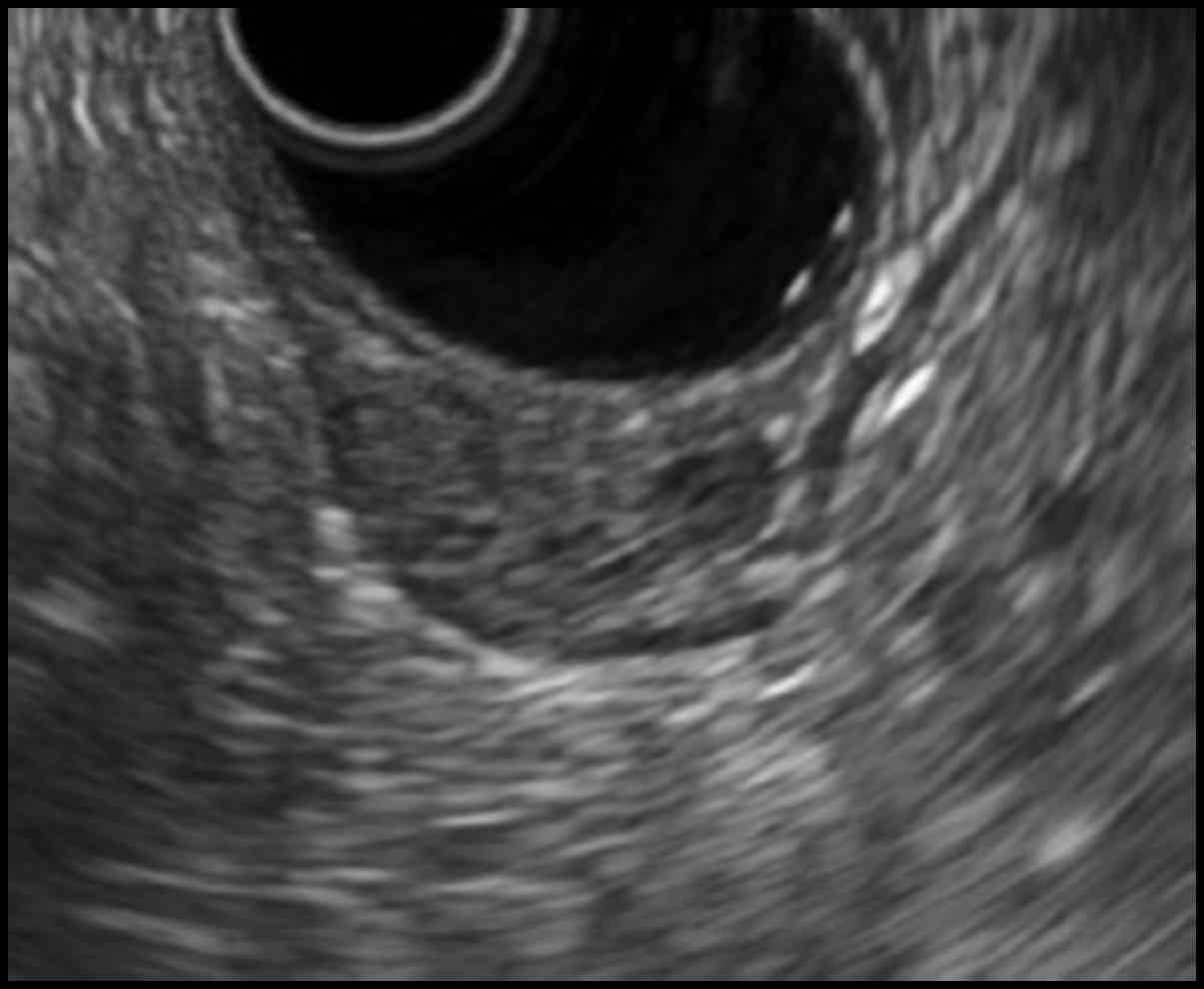
cm in size in the greater curvature side of the antrum (Fig. 1). Endoscopic ultrasonography (EUS)
demonstrated a hypoechoic solid mass lesion with a small anechoic
component in the third or fourth tissues layer, suggesting a lesion
in the muscular layer of the stomach (Fig. 2).
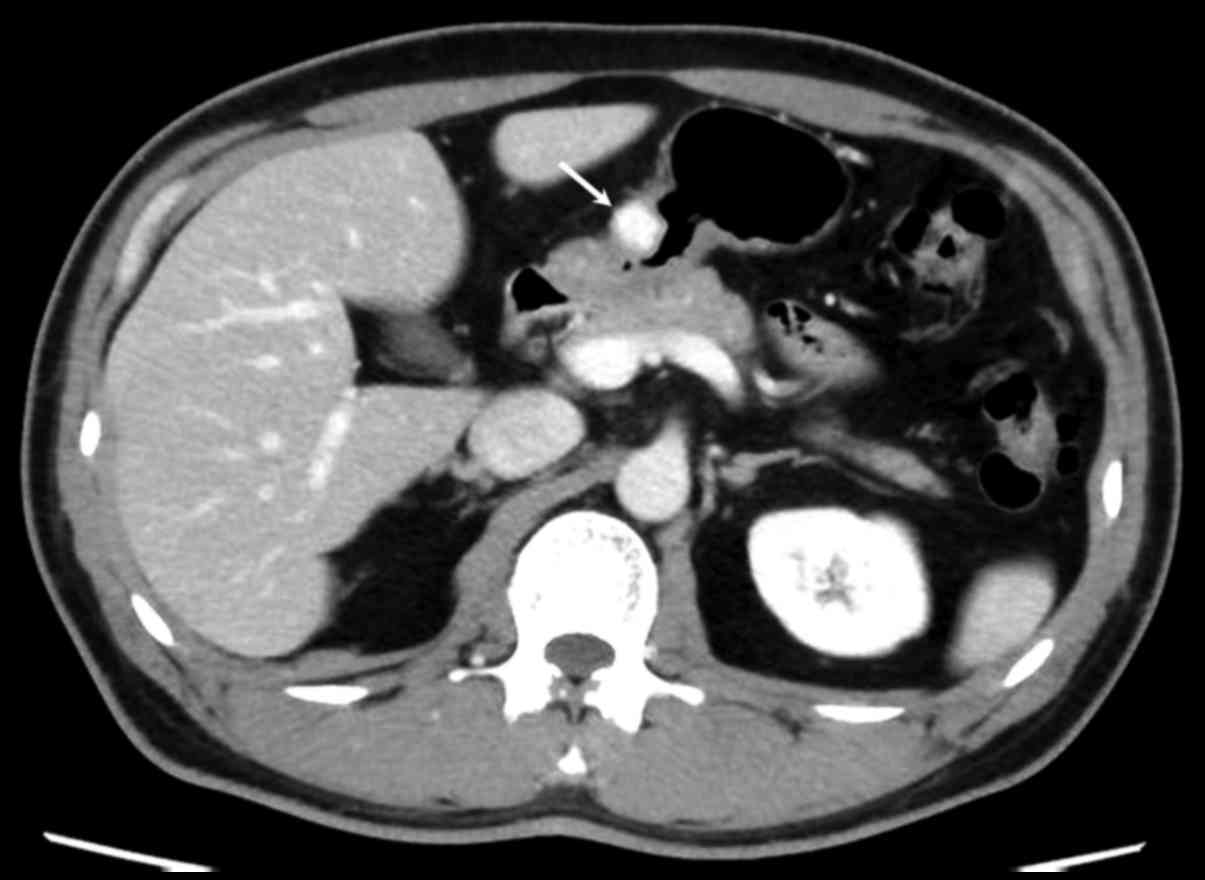
Abdominal contrast-enhanced computed tomography
revealed a 1.5-cm, well-defined mass lesion in the gastric antrum
with strong, homogeneous enhancement in the early (Fig. 3). To obtain a definite diagnosis,
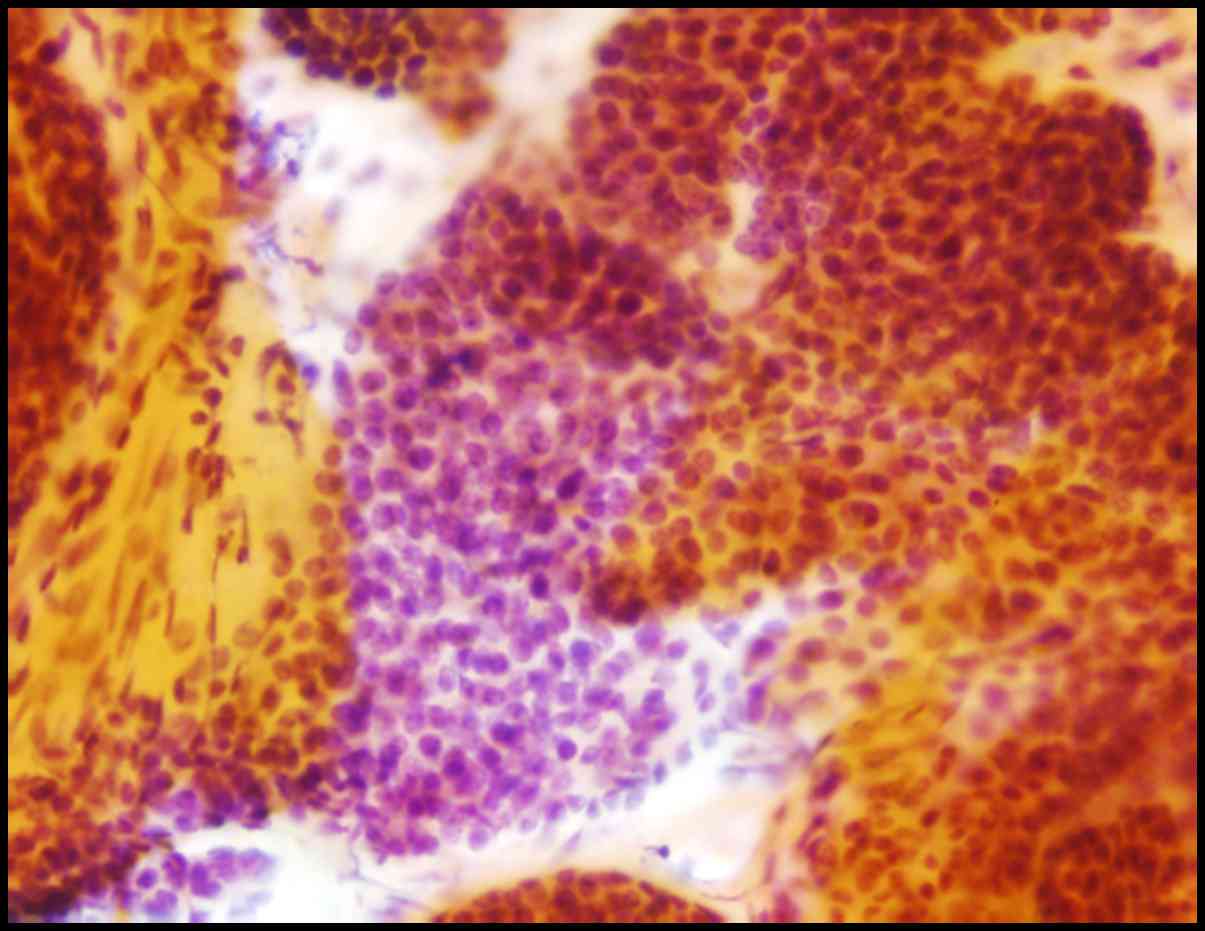
EUS-guided fine-needle aspiration (EUS-FNA) was performed using a
22-gauge needle. The cytopathology smear of the lesion obtained by
EUS-FNA revealed the proliferation of small, round to oval tumor
cells with round nuclei and scant cytoplasm (Fig. 4). The patient underwent laparoscopic
distal gastrectomy with reginal lymph node dissection under a
clinical diagnosis of neuroendocrine neoplasm (NEN).
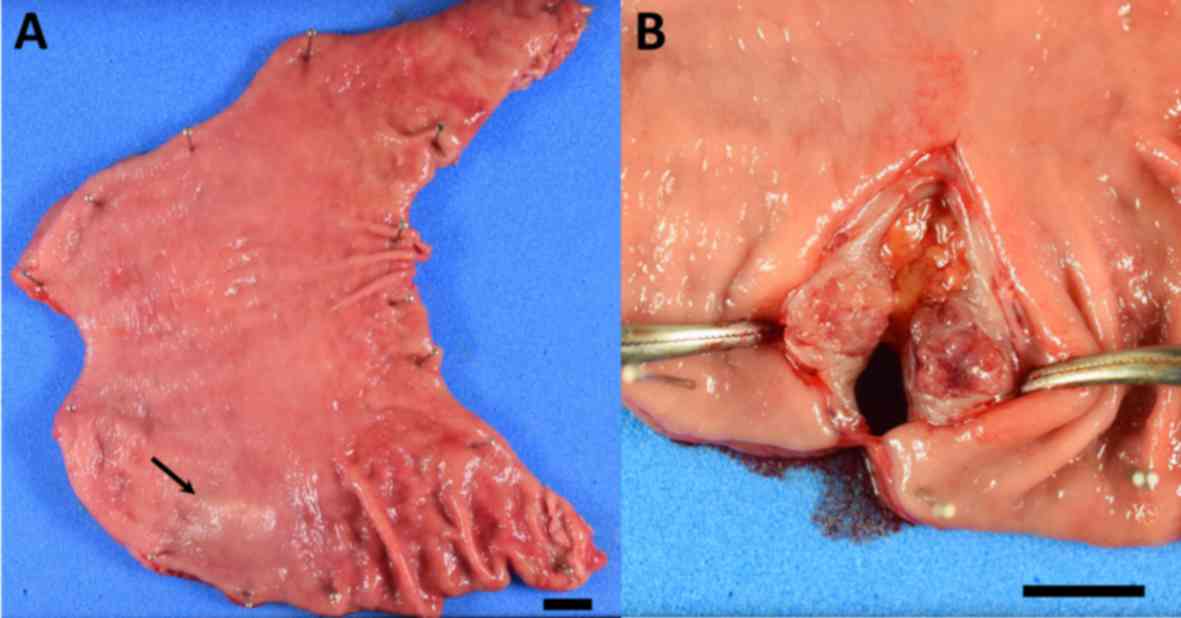
Gross examination of the surgically resected
specimen showed a SMT (Fig. 5A,
arrow) that appeared in cross-section as a well-circumscribed,
solid tumor measuring 1.5×1.0 cm (Fig.
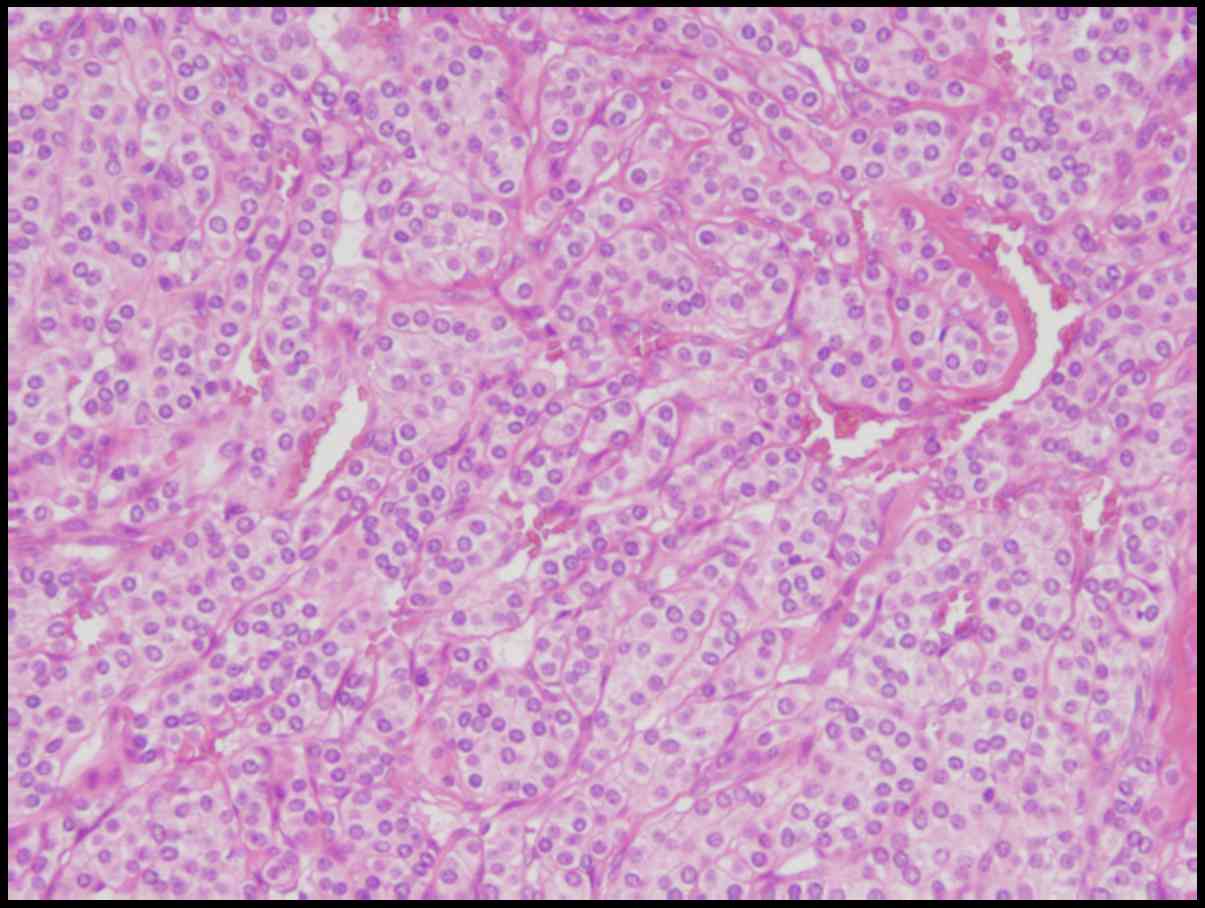
5B). Microscopical examination reveals solid proliferation of
tumor cells with oval-shaped nuclei and scanty cytoplasm around the
disorganized vessels in the submucosa and the muscular layer
(Fig. 6). Immunohistochemical
staining showed a positive signal for α-smooth muscle actin, but no
staining for desmin, c-kit, CD34, S-100, chromogranin A,
synaptophysin, and CD56. Approximately 1% of the cells were
positive for the proliferation marker Ki-67. Therefore, we
diagnosed a glomus tumor of the stomach. The postoperative course
was uneventful, and he was discharged on postoperative day 10.
Discussion
Herein we describe a rare case of glomus tumor of
the stomach treated by laparoscopic distal gastrectomy. In
preparing this report, we searched for English-language articles on
similar cases published from 2000 to 2016 in the Medline and PubMed
databases, using the keywords ‘glomus tumor’, ‘stomach’, and
‘laparoscopy’ to obtain data on age, gender, tumor location, tumor
size, depth of invasion, histological type, treatment, and outcome
for each patient. To the best of our knowledge, this is only the
seventh reported case of a gastric glomus tumor treated
laparoscopically in the English literature.
Table I lists
clinicopathological features of the six previously reported cases
(1,3,5–8) and the present case. The median patient
age was 50 (range 33–70 years), and the male-to-female ratio was
2:5. Gastric tumor in the lower third of the stomach was reported
in six cases, with the remaining patient showing a lesion in the
middle-third of the stomach. Treatment consisted of laparoscopic
wedge resection in three patients, laparoscopic distal gastrectomy
in two patients, laparoscopy and endoscopy cooperative surgery
(LECS) in one patient, and non-exposed endoscopic wall inversion
surgery in the remaining patient. The median tumor size was 1.6 cm
(range 0.8–3.5 cm).
 | Table I.Clinicopathological data for reported
cases of gastric glomus tumor treated laparoscopically. |
Table I.
Clinicopathological data for reported
cases of gastric glomus tumor treated laparoscopically.
| Author | Age (years) | Sex | Tumor location | Tumor diameter
(cm) | Treatment | (Refs.) |
|---|
| Minoda et
al | 50 | Female | L, Ant | 1.2 | LWR | (3) |
| Halawani et
al | 33 | Female | L, Gre | 1.6 | LDG | (5) |
| Zhao et
al | 51 | Female | M, Less | 2 | LWR | (6) |
| Kato et
al | 52 | Male | L, Gre | 3.5 | LECS | (7) |
| Castro et
al | 70 | Female | L, Gre | 2.5 | LWR | (8) |
| Ebi et al | 45 | Female | L, Less | 1.5 | NEWS | (1) |
| Present case | 39 | Male | L, Gre | 0.8 | LDG |
|
As gastric glomus tumor lacks specific clinical and
endoscopic characteristics, it is difficult to distinguish them
from the more common GIST or NEN including carcinoid tumor by
conventional imaging (9,10). EUS-FNA is an effective method for
obtaining pathological specimens of gastric SMT with a high rate of
diagnostic accuracy (7). In the
present case, the Giemsa staining of cytological smear obtained by
EUS-FNA showed solid clusters of tumor cells with round nuclei and
scant cytoplasm. On the basis of these histological findings, a
preoperative diagnosis of a gastric NEN was made. However, because
NEN comprises oval or spindle tumor cells arranged in cords or
nests with thin-walled blood vessels, they can be extremely
difficult to distinguish from glomus tumor. Consequently, a
definitive diagnosis of gastric glomus tumors cannot be confirmed
by standard endoscopy until histological results including
immunohistochemical examination are considered (11,12). If
the immunohistochemical staining of the specimens were performed,
taking into the possibility of glomus tumor consideration, an
accurate preoperative diagnosis might have been possible.
Complete resection of the lesion is necessary for
accurate characterization and to treat the patient appropriately
due to the possibility of malignant transformation, even though
most glomus tumors are benign (11).
Such efforts to treat SMT of the stomach have been improved by
recent advances in endoscopic technique and devices that allow not
only laparoscopic resection including LECS and non-exposed
endoscopic wall-inversion surgery (NEWS), but also endoscopic
resection (11,13).
In conclusion, although gastric glomus tumor is
extremely rare, physicians should take this disease into
consideration especially when SMT is observed in the lower third of
the stomach. To avoid overtreatment of the glomus tumor, every
effort including EUS-FNA and immunohistochemical examination must
be carried out to make accurate diagnosis. Further studies and
assessments by the accumulation of additional cases are needed to
establish precise diagnostic criteria and to distinguish other
diseases presenting as SMT of the stomach.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets during and/or analysed during the
current study available from the corresponding author on reasonable
request.
Authors' contributions
TN and MK designed the study. TN, ST, KF, JI, SU,
ST, HM, HK, MK and KH acquired, analyzed and interpreted the data
within the study. TN and KH finalized the manuscript and submitted
the paper for publication. All authors edited the manuscript for
intellectual content. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient for publication of this case report and any accompanying
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ebi M, Sugiyama T, Yamamoto K, Saito T,
Inoue T, Yamaguchi Y, Tamura Y, Izawa S, Hijikata Y, Funaki Y, et
al: A gastric glomus tumor resected using non-exposed endoscopic
wall-inversion surgery. Clin J Gastroenterol. 10:508–513. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yamagata S, Shoji T, Kawashima S, Narasaka
T, Sato S, Kawamura M, Matoba N and Wakasa H: Glomus tumor of the
stomach. Tohoku J Exp Med. 78:202–208. 1962. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Minoda Y, Akahoshi K, Oya M, Kubokawa M,
Motomura Y and Nakamura K: Gastric glomus tumor diagnosed by
endoscopic ultrasound-guided fine-needle aspiration biopsy: Report
of a case. Fukuoka Igaku Zasshi. 105:105–109. 2014.PubMed/NCBI
|
|
4
|
Miettinen M, Paal E, Lasota J and Sobin
LH: Gastrointestinal glomus tumors: A clinicopathologic,
immunohistochemical, and molecular genetic study of 32 cases. Am J
Surg Pathol. 26:301–311. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Halawani HM, Khalife M, Safadi B, Rida K,
Boulos F and Khalifeh F: Laparoscopic antral resection with
Billroth I reconstruction for a gastric glomus tumor. Int J Surg
Case Rep. 5:1128–1131. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhao XJ, Wang HH, Sheng JQ and Li N:
Laparoscopic resection of a gastric glomangioma. Endoscopy. 46
Suppl 1 UCTN:E73–E74. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kato S, Kikuchi K, Chinen K, Murakami T
and Kunishima F: Diagnostic utility of endoscopic ultrasound-guided
fine-needle aspiration biopsy for glomus tumor of the stomach.
World J Gastroenterol. 21:7052–7058. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Castro Ruiz C, Carlinfante G, Zizzo M,
Giunta A, Ronzoni R, Azzolini F and Pedrazzoli C: Glomus tumor of
the stomach: GI image. J Gastrointest Surg. 21:1099–1101. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lin YM, Chiu NC, Li AF, Liu CA, Chou YH
and Chiou YY: Unusual gastric tumors and tumor-like lesions:
Radiological with pathological correlation and literature review.
World J Gastroenterol. 23:2493–2504. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Papadelis A, Brooks CJ and Albaran RG:
Gastric glomus tumor. J Surg Case Rep. 2016(pii): rjw1832016.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang Y, Zhou P, Xu M, Chen W, Li Q, Ji Y
and Yao L: Endoscopic diagnosis and treatment of gastric glomus
tumors. Gastrointest Endosc. 73:371–375. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Baek YH, Choi SR, Lee BE and Kim GH:
Gastric glomus tumor: Analysis of endosonographic characteristics
and computed tomographic findings. Dig Endosc. 25:80–83. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Namikawa T and Hanazaki K: Laparoscopic
endoscopic cooperative surgery as a minimally invasive treatment
for gastric submucosal tumor. World J Gastrointest Endosc.
7:1150–1156. 2015. View Article : Google Scholar : PubMed/NCBI
|