Introduction
Despite notable progress in the ability to
preoperatively diagnose hepatic tumors due to recent technological
advances in radiological imaging techniques, it may be difficult to
obtain a precise preoperative diagnosis for liver tumors (1,2).
Intrahepatic cholangiocarcinoma (ICC) has been reported to exhibit
various imaging features such as irregular margins, thin peripheral
arterial enhancement with progressive centripetal filling and
delayed enhancement of the central portions, depending on the
tumor's location, size and intratumoral components (3). Additionally, computed topography (CT)
findings in cholangiocellular carcinoma (CoCC) have demonstrated
the histological features of the tumor (4). Furthermore, enhanced CT indicated the
early enhancement and delayed washout of CoCC, similar to
hepatocellular carcinoma (HCC) (5),
or ringed enhancement at the periphery during the early phase and
persistent enhancement during the delayed phase, similar to ICC on
enhanced CT (6). Therefore,
percutaneous needle biopsy examinations may be performed to
diagnose liver tumors preoperatively. However, needle biopsy may
yield different results depending on the region of the tumor that
is biopsied due to the heterogeneous distribution of cells in liver
tumors (7). The present study reports
a case of mass-forming ICC, which mimicked CoCC during imaging and
needle biopsy examination.
Case report
A liver tumor was incidentally located in a
51-year-old female during a medical check-up in September 2016. The
patient had no significant medical, family, including with regard
to genetic information, or psychosocial history of liver tumors. A
routine physical examination produced normal results. No peripheral
blood-, blood chemistry- or hemostasis-associated abnormalities
were determined, except for a slightly elevated serum γ-glutamyl
transpeptidase level of 63 U/l (normal range, <50 U/l). The
patient's tumor marker levels, including those of carcinoembryonic
antigen [0.7 ng/ml (normal range, <5.0 ng/ml)], carbohydrate
antigen 19-9 [7.5 U/ml (normal range, <37 U/ml)], α-fetoprotein
[AFP; 4.3 U/ml (normal range, <20 U/ml)], and protein induced by
vitamin K absence or antagonists-II [14 U/ml (normal range, <40
U/ml)], were within the normal limits. Abdominal ultrasonography
revealed a 50-mm tumor in the lateral segment of the liver.
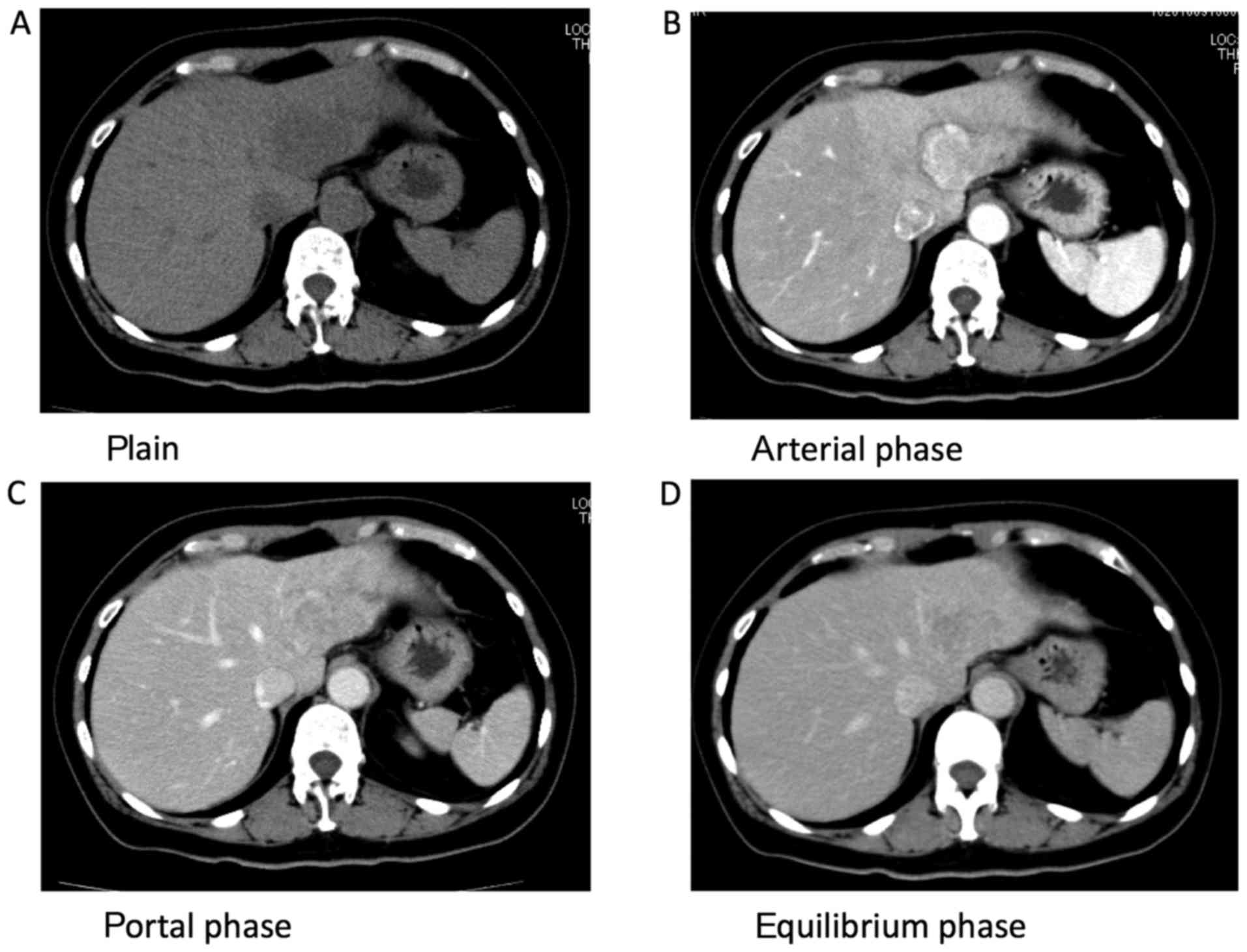
Computed tomography (CT) demonstrated non-homogeneous enhancement
in the arterial phase, persistent enhancement in the portal and
equilibrium phases (Fig. 1A-D), and
swelling and enhancement of the para-aortic lymph nodes.
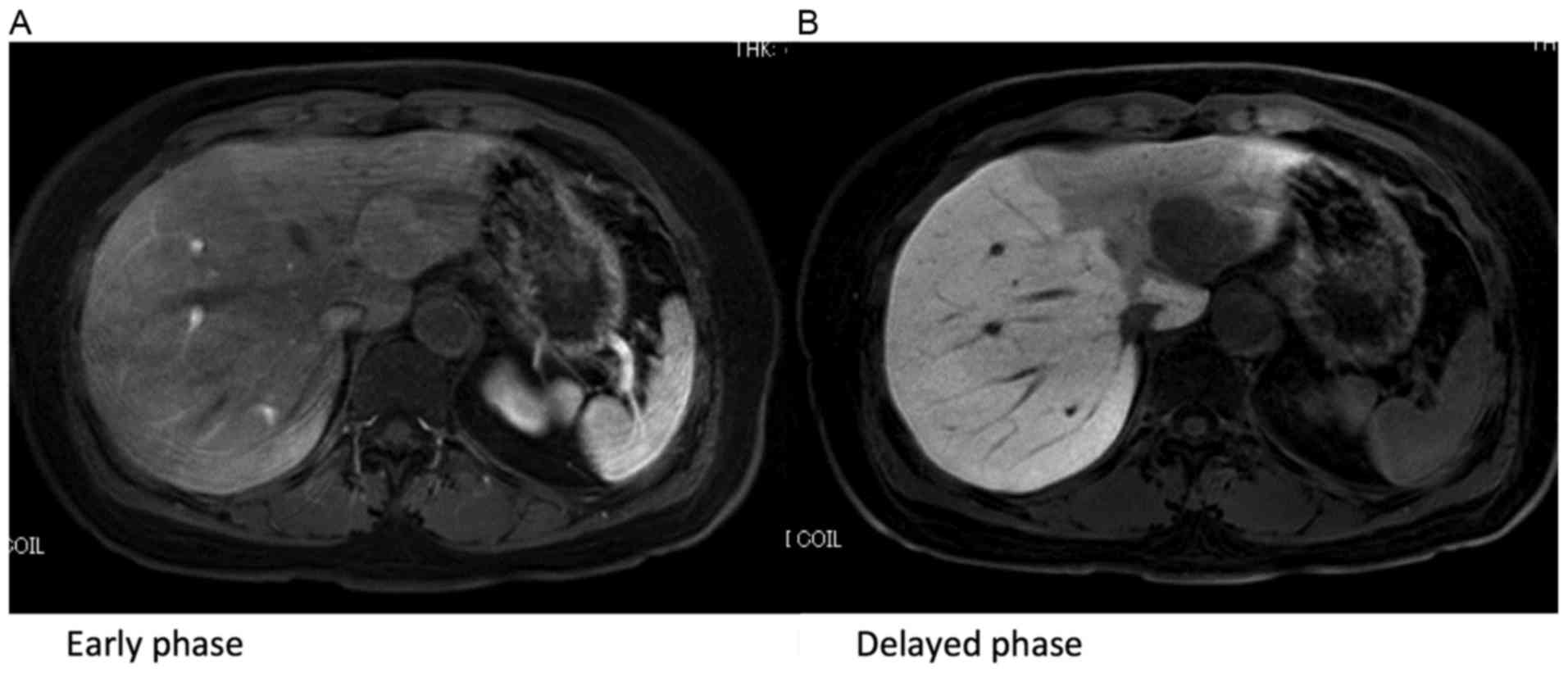
Gadolinium-ethoxybenzyl diethylenetriamine-pentaacetic
acid-enhanced magnetic resonance imaging revealed low signal
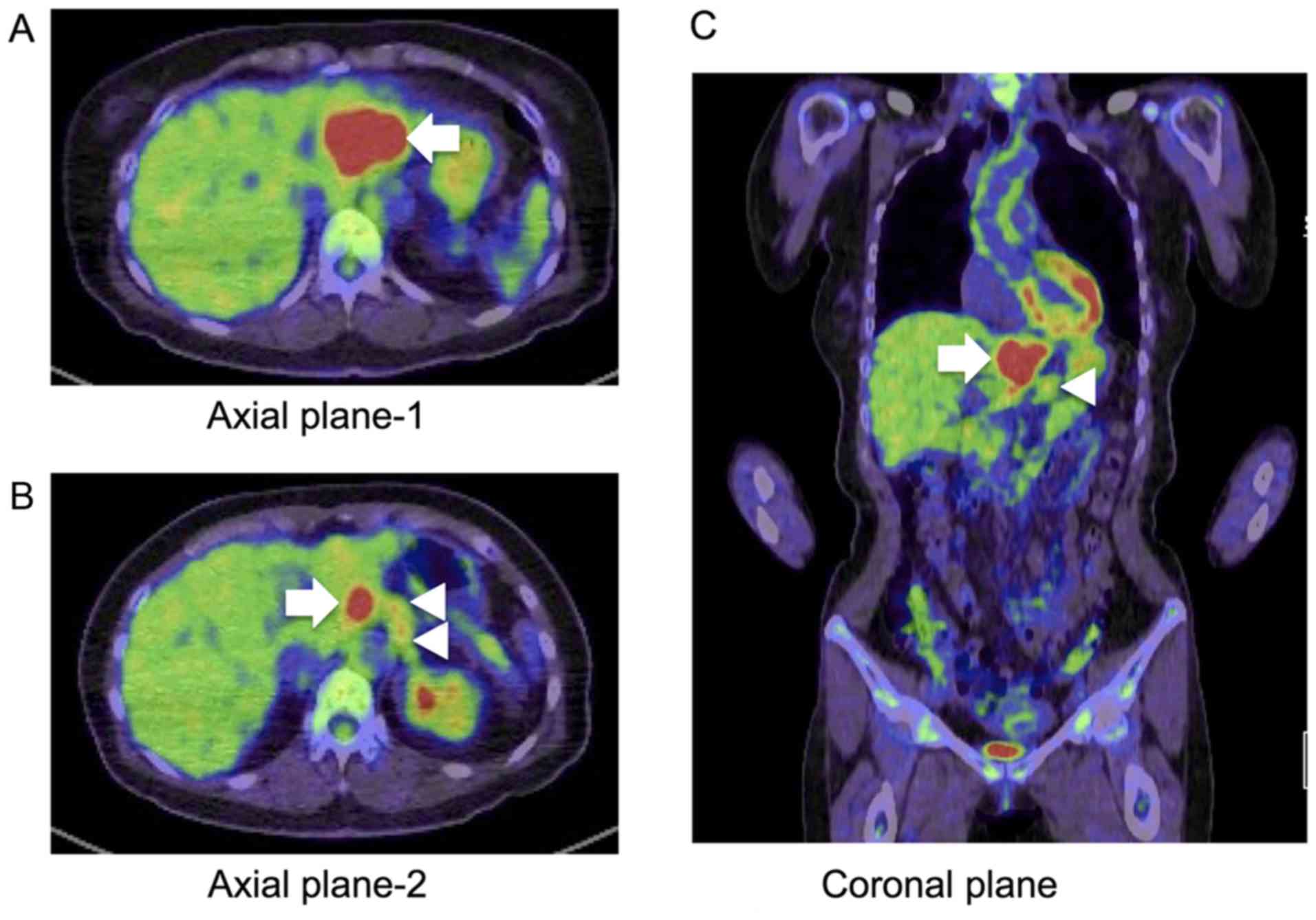
intensity in the hepatobiliary phase (Fig. 2A and B). Positron emission
tomography/CT indicated fludeoxyglucose uptake by the liver tumor
and lymph nodes (Fig. 3A-C). Due to
the absence of underlying liver disease, elevated tumor marker
levels and specific imaging findings, ultrasonography-guided
percutaneous needle biopsy of the liver tumor was performed.
Histological examination of 10% formalin-fixed (24
h, room temperature), 3-µm paraffin-embedded sections was performed
using hematoxylin-eosin (HE) and mucin staining. For HE staining,
sections were stained with 0.12 g/v% hematoxylin solution produced
by Hematoxylin (C.I. 75290) cryst. (cat. no. 104302; Merck KGaA,
Darmstadt, Germany) three times (3 min/stain) at room temperature,
and with eosin Y (cat. no. 058-00062; Wako Pure Chemical
Industries, Ltd., Osaka, Japan) two times (1.5 min/stain) at room
temperature. For mucin staining, sections were stained with
Schiff's Reagent (cat. no. 40921; Muto Pure Chemicals Co. Ltd.,
Tokyo, Japan) for 15 min at room temperature, and with Alcian Blue
Stain Solution pH 2.5 for Histology (cat. no. 40852; Muto Pure
Chemicals Co. Ltd.) for 20 min at room temperature. Staining was
detected under a light microscope.
For immunohistochemistry, 10% formalin-fixed (24 h,
room temperature), 3-µm paraffin-embedded sections were analyzed.
The following commercially available primary antibodies were used:
Anti-cytokeratin (CK)7 (cat. no. M7018; 1:200; Dako; Agilent
Technologies, Inc., Santa Clara, CA, USA), CK19 (cat. no. M0888;
1:100; Dako; Agilent Technologies, Inc.), CK20 (cat. no. M7019;
1:100; Dako; Agilent Technologies, Inc.), glypican 3 (cat. no.
718021; Nichirei Bioscience, Inc., Tokyo, Japan), CEA (cat. no.
A115; 1:2,000; Dako; Agilent Technologies, Inc.), cluster of
differentiation 56 (CD56; cat. no. 713331; Nichirei Bioscience,
Inc.), AFP (cat. no. A0008; 1:500; Dako; Agilent Technologies,
Inc.), hepatocyte-specific antigen (cat. no. NCL-HSA; 1:50;
Novocastra; Leica Microsystems GmbH, Wetzler, Germany), epithelial
membrane antigen (EMA; cat. no. M0613; 1:200; Dako; Agilent
Technologies, Inc.) and KIT (cat. no. 713391; Nichirei Bioscience,
Inc.). Briefly, sections were prepared as follows: For glypican 3
detection, samples underwent heat-induced epitope-retrieval [boiled
at 100°C for 40 min in Heat Processor Solution pH 9 (cat. no.
715291; 1:10; Nichirei Bioscience, Inc.)]; for CD56 detection,
samples underwent heat-induced epitope retrieval [boiled at 100°C
for 40 min in Heat Processor Solution pH 6 (cat. no. 715281; 1:10;
Nichirei Bioscience, Inc.)]; for CK7, 19 and 20 detection, samples
underwent, proteinase-induced epitope retrieval [protease K (cat.
no. 715231; Nichirei Bioscience, Inc.)] at room temperature for 10
min). After blocking with 3v/v% hydrogen peroxidase solution (cat.
no. 715142; Nichirei Bioscience, Inc.) for 5 min at room
temperature, the sections were incubated with primary antibodies
for 30 min at room temperature, followed by incubation with
Histofine® Simple Stain MAX PO (MULTI) (cat. no. 724152;
Nichirei Bioscience, Inc.) for 30 min, and finally with
Histofine® Simple Stain DAB substance kit (cat. no.
725191; Nichirei Bioscience, Inc.; 5 min ×2 incubations). The steps
following incubation with primary antibodies were performed under a
HISTOSTAINER 48A autostaining machine (Nichirei Bioscience, Inc.)
at room temperature.
A histopathological examination of the biopsy
specimen demonstrated spindle-shaped tumor cells with slightly
eosinophilic cytoplasm and enlarged nuclei, arranged in a cord-like
pattern. An immunohistochemical examination was performed,
according to the manufacturers' protocols; the results indicated
that the tumor cells were positive for CK7 and glypican 3,
partially positive for CD56 and negative for CK20, AFP, and
hepatocyte-specific antigen (Fig.
4A-E). EMA staining revealed positivity in the luminal
membranes of the tumor cells (Fig.
4F) (8,9). No mucus production was detected with
Periodic acid Schiff-alcian blue (pH 2.5) staining. According to
these findings, the patient was preoperatively diagnosed with CoCC
(cT2N1M0 stage II, according to the eighth edition of the
Tumor-Node-Metastasis Classification of Malignant Tumors developed
by the Union for International Cancer Control) (10). The patient underwent left hepatectomy
and lymphadenectomy 72 days after her first visit to the National
Defense Medical College Hospital. The regional lymph nodes, which
were identified based on preoperative imaging and an intraoperative
assessment, were removed. The regional lymph nodes were located in
the hepatic hilum and hepatoduodenal ligament, posterior to the
upper portion of the pancreatic head, and at the common hepatic
artery and para-aortic stations. The operative time was 510 min,
and the total amount of intraoperative blood loss was 721 g. The
patient's postoperative recovery was uneventful, and she was
discharged from the hospital on postoperative day 15.
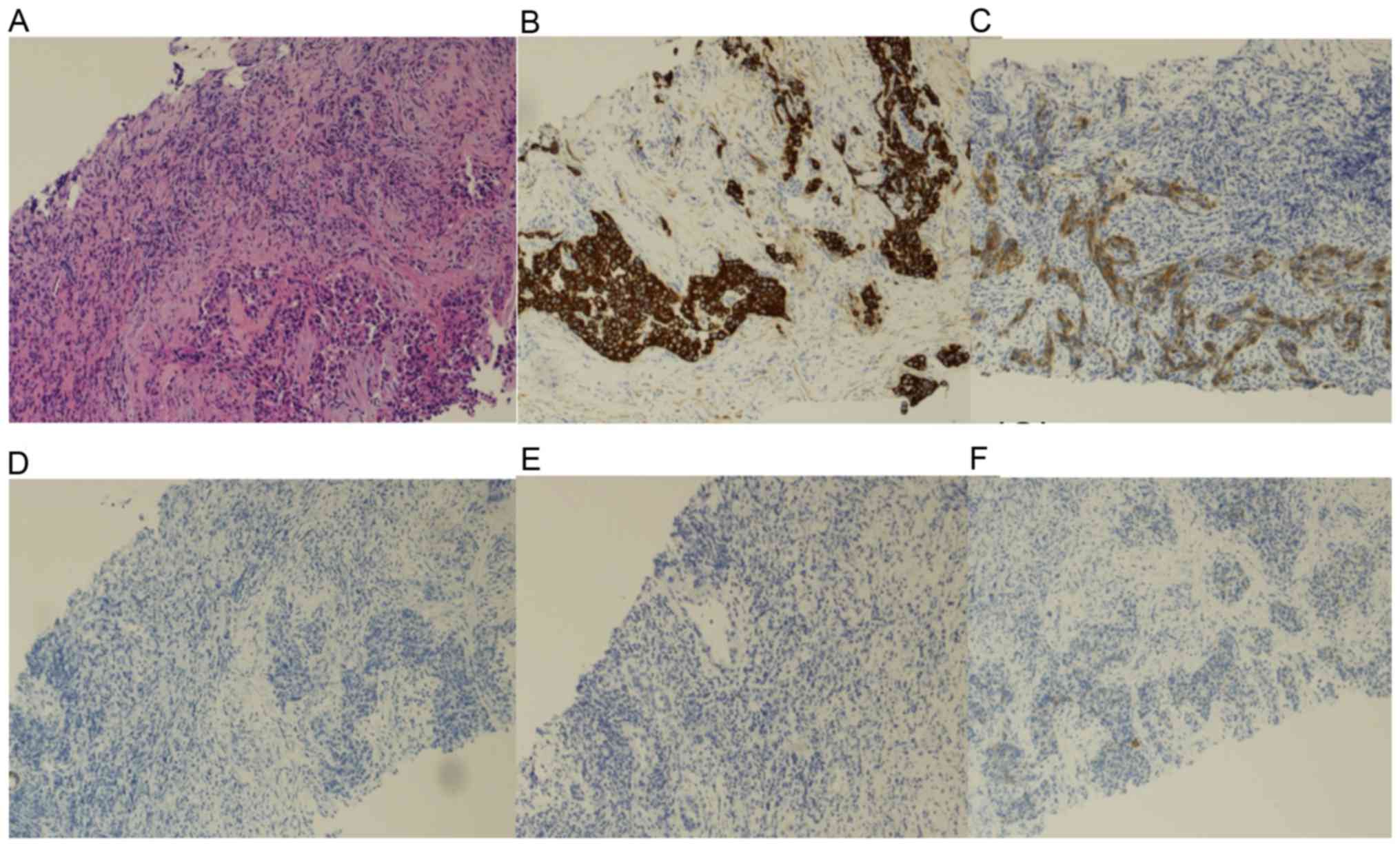 | Figure 4.Histopathological analysis of the
liver tumor obtained during the needle biopsy. (A)
Hematoxylin-eosin staining of the microscopic specimen demonstrated
that the tumor cells had slightly eosinophilic cytoplasm and round
nuclei with clear nucleoli. Immunohistochemical staining of (B)
CK7, (C) EMA, (D) CD56, (E) AFP (F) and glypican 3 demonstrated
that the tumor cells were diffusely positive for CK7, positive for
EMA (in the luminal membranes), and negative for CD56, AFP and
glypican 3 (magnification, ×20). CD56, cluster of differentiation;
CK, cytokeratin; AFP, α-fetoprotein; EMA, epithelial membrane
antigen. |
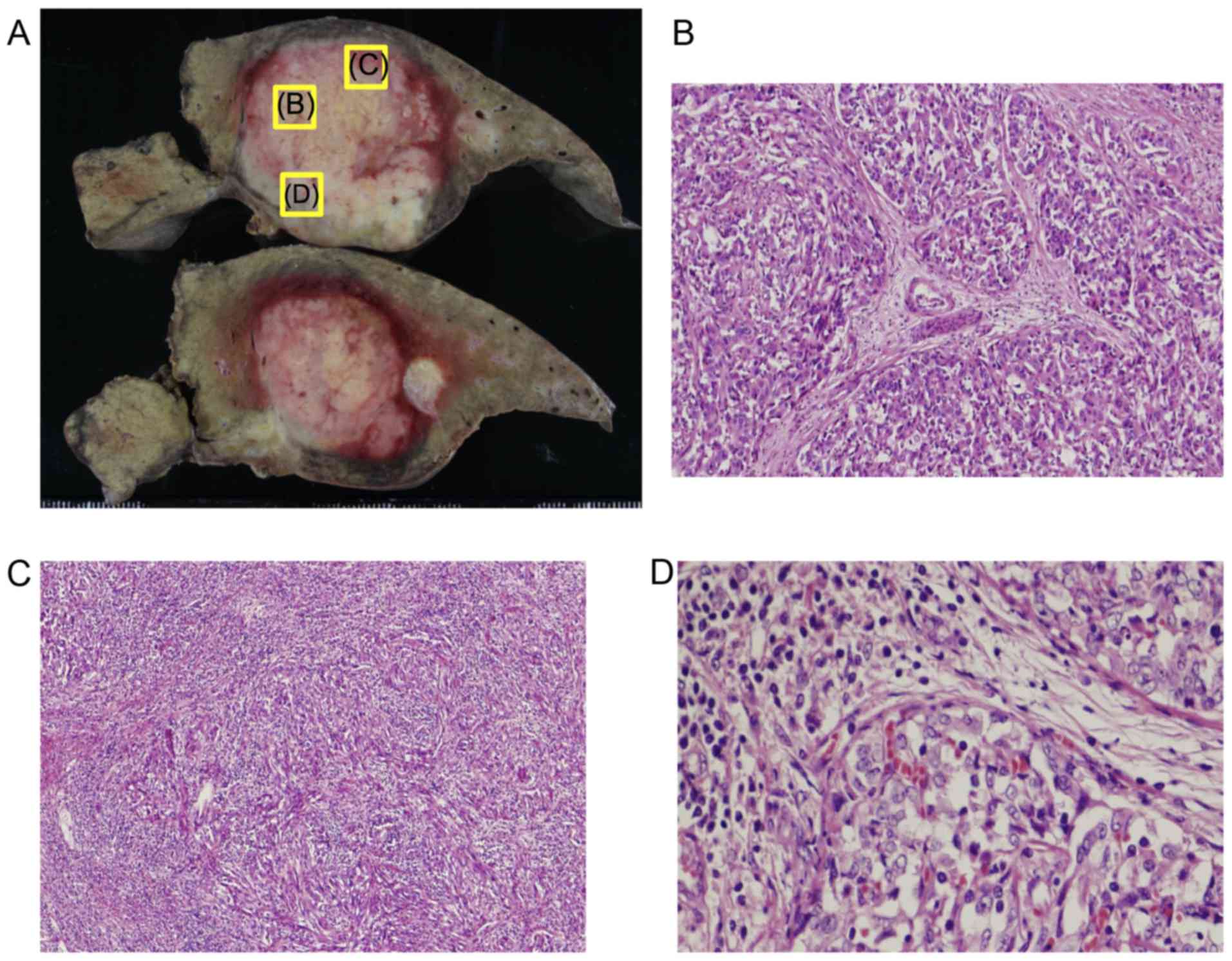
The resected liver tumor measured 46×45×35 mm
(Fig. 5A). The tumor was
histologically composed of three parts: i) Polygonal eosinophilic
carcinoma cells arranged in a cord- or nest-like pattern; ii)
spindle-shaped carcinoma cells with an irregular growth pattern;
and iii) a number of cancer nests with irregular tubular structures
(Fig. 5B-D). Additionally, mucin
production was present in a portion of this tumor. This tumor did
not exhibit histological features, and had admixtures of small
monotonous glands, reflecting so-called antler-like anastomosing
patterns as characteristics of CoCC (Fig.
6A). Immunohistochemical staining demonstrated that the tumor
was positive for CKs 7, 19 (Fig. 6B and
C) and EMA (Fig. 6D), and
negative for AFP (Fig. 6E),
hepatocyte-specific antigen, glypican 3, CD56 and KIT (Fig. 6F-H). EMA staining indicated that all
of the tumor components in the resected specimen were cytoplasmic
positive, but the normal liver was negative. This tumor did not
exhibit characteristics of hepatic progenitor cells or stem cells.
Based on these final histopathological and immunohistochemical
findings, the tumor was diagnosed as a poorly differentiated ICC
with anaplastic changes, not CoCC (Table
I). The patient is still alive and has not suffered any
recurrence at 15 months after surgery.
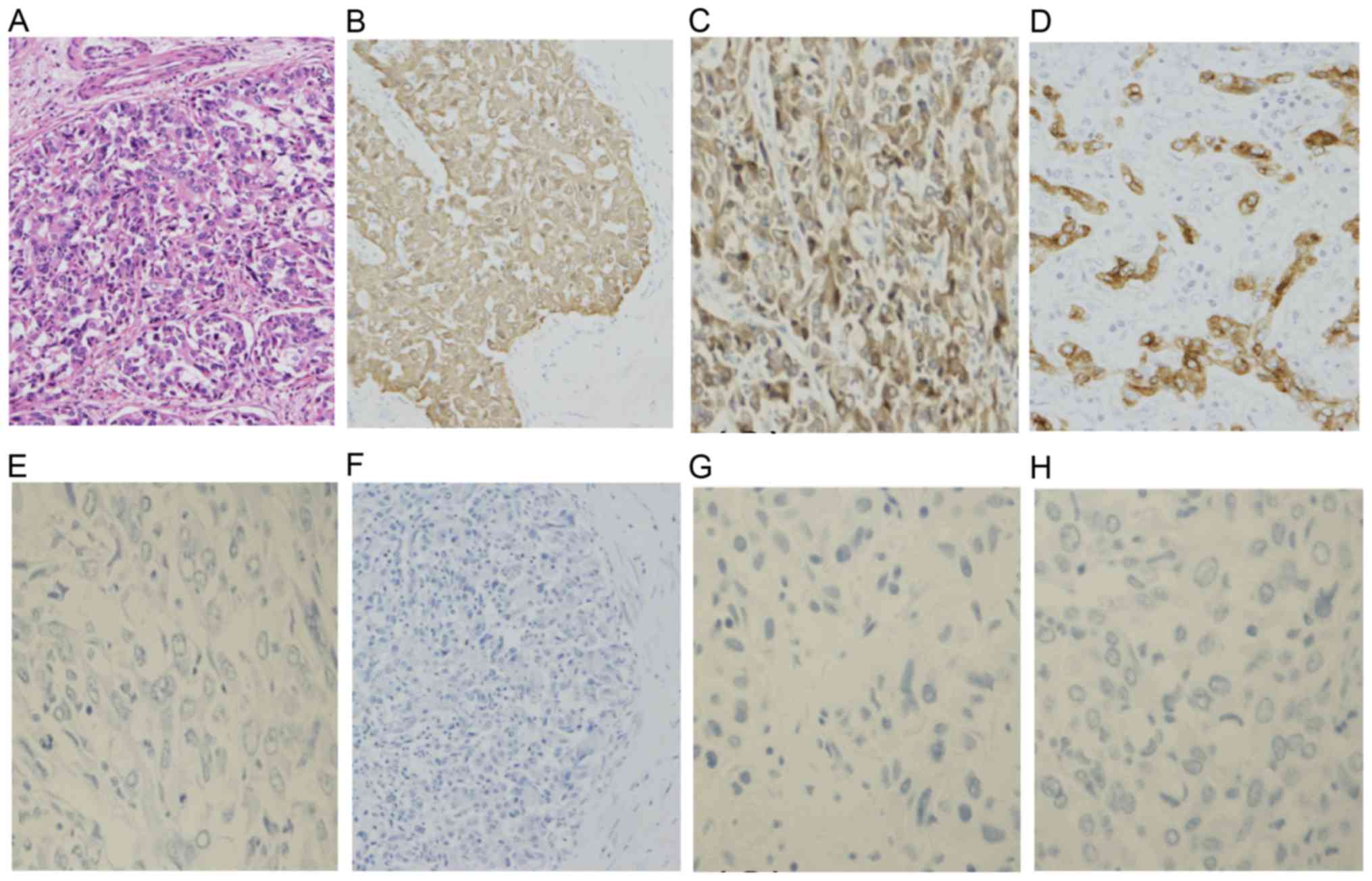 | Figure 6.Histopathological analysis of
resected liver tumor. (A) Hematoxylin-eosin staining demonstrated
tumor cells with enlarged round nuclei and eosinophilic cytoplasm.
Immunohistochemical staining of (B) CK7, (C) CK19, (D) EMA, (E)
HSA, (F) glypican 3, (G) CD56 and (H) KIT demonstrated that the
tumor cells were diffusely positive for CK7 and CK19, positive for
EMA (in the luminal membranes), and negative for HSA, CD56 and KIT.
HSA, hepatocyte-specific antigen; CD56, cluster of differentiation
56; CK, cytokeratin; EMA, epithelial membrane antigen
(magnification, ×20). |
 | Table I.Comparison of histologic finding
findings between needle biopsy specimen and surgical specimen. |
Table I.
Comparison of histologic finding
findings between needle biopsy specimen and surgical specimen.
| Special or
immunohistochemical staining | Needle biopsy
specimen | Surgical
specimen |
Characteristics |
|---|
| PAS and/or
alcian-blue | Mucin production
(−) | Mucin production
(+) | ICC |
| EMA | Membranous
positive | Membranous
positive |
|
| CK7 | Positive | Positive |
|
| CK19 | Not determined | Positive |
|
| CK20 | Negative | Negative |
|
| CEA | Not determined | Negative |
|
| AFP | Negative | Negative | HCC |
| HSA | Negative | Negative |
|
| Glypican 3 | Positive |
|
|
| CD56 | Partially
positive | Negative | Stem/progenitor
cell |
| KIT | Negative | Negative |
|
Discussion
ICC is the second most common type of primary liver
cancer behind HCC in Japan in 2010 (1,11–16). A number of previous studies have
reported rapidly rising rates of ICC over the recent decades
globally (13,17). ICC has similar risk factors to HCC,
including cirrhosis of the liver, chronic viral hepatitis, alcohol
abuse, diabetes mellitus and obesity (18). As ICC exhibits various radiological
features, including capsular retraction, satellite nodules, and
peripheral biliary dilatation, depending on the tumor's location,
size, and intratumoral components (3,19), a
definitive pathological diagnosis is required for appropriate
treatment planning. However, CoCC is a rare type of primary liver
cancer (20). There are a number of
reports regarding the definition of CoCC. Recently, CoCC was
classified as a stem-cell subtype of combined hepatocellular and
cholangiocarcinoma (CHC) according to the 2010 World Health
Organization classification (18,21).
While, in Japan, CoCC was reclassified as a type of primary liver
cancer according to he General Rules for the Clinical and
Pathological Study of Primary Liver Cancer, which were developed by
the Liver Cancer Study Group of Japan (22). Yano et al (23) reported that when the HCC or ICC area
accounts for ≥10% of the tumor, the tumor should be diagnosed as
CHC. CoCC frequently exhibits HCC-like and ICC areas within the
tumor, but these areas compose of <10% of the tumor (24). Therefore, CoCC should be different
from CHC. Histologically, ICC resembles small intrahepatic bile
ducts, including interlobular bile ducts (13). Conversely, CoCC is considered to
originate from the peripheral biliary duct system, including
cholangioles/ductules and the canals of Hering, where hepatic
progenitor cells are located (25).
In a previous study, CoCC did not exhibit mucin production,
although ICC did (25).
Immunohistochemically, luminal EMA expression and positive CD56 are
observed in CoCC, whereas cytoplasmic EMA expression and negative
CD56 expression are usually observed in ICC (26). The histopathological features of these
tumors are described in Table II.
CoCC is rare primary malignant liver tumor, and therefore there are
a limited number of reports regarding its prognosis (20,27). The
tumor doubling times of CoCC and ICC were reported to be 285 and 70
days, respectively (28). These data
indicate that slow growth may be a characteristic of CoCC (28). Previous reports demonstrated CoCC
primarily exhibited improved prognosis following resection,
compared with ICC (20,29).
 | Table II.Histopathological features of CoCC
and ICC. |
Table II.
Histopathological features of CoCC
and ICC.
| Histopathological
features | CoCC | ICC |
|---|
| Origin of
tumor |
Cholangioles/ductules and the canals of
Hering | Small intrahepatic
bile ducts, including interlobular bile ducts |
| EMA
immunohistochemical staining results | Membranous
positive | Cytoplasmic
positive |
| Mucus
production | Negative | Positive |
| Classification
according to the General Rules for the Clinical and Pathological
Study of Primary Liver Cancer (9) | An independent
disease entity | ICC |
| World Health
Organization classification 4th edition (21) | Subtype of CHC | ICC |
In the present case, a preoperative diagnosis based
on a combination of imaging and pathological information was
subsequently determined to be incorrect. The characteristic imaging
findings of CoCC include a combination of early peripheral
enhancement, the presence of intratumoral portal tracts, the
absence of bile duct dilation and delayed contrast enhancement
(5). However, the characteristic
imaging findings of ICC include peripheral rim-like enhancement,
central delayed enhancement, irregular tumor margins and capsular
retraction (28,30). Therefore, the imaging findings of ICC
are similar to those of CoCC. However, a number of hepatic tumors
do not exhibit specific imaging findings. For example, the tumor in
the present case exhibited HCC-like early enhancement in the
arterial phase and ICC-like delayed enhancement in the portal and
equilibrium phases; therefore, a percutaneous needle biopsy was
performed. The biopsy specimen only included the part of the tumor
containing spindle-shaped tumor cells with slightly eosinophilic
cytoplasm and enlarged nuclei, which were arranged in a cord-like
pattern. An immunohistochemical examination of the whole biopsy
specimen demonstrated positive for CK7 and luminal EMA. However,
the resected tumor revealed three distinct parts with different
histological features, and an immunohistochemical examination
indicated that the cytoplasm of the tumor cells was positive for
CK7 and EMA. As was demonstrated in the present case,
histopathological examinations of percutaneous needle biopsy
samples do not always result in a correct diagnosis due to only
small tumor samples being obtained (21). Intratumoral heterogeneity is
increasingly recognized as a factor that has a major impact on
diagnosis and the personalized treatment of a number of cancer
types (3,19). A previous study indicated that ICC
exhibits substantial intratumoral heterogeneity, which should be
considered when producing therapeutic decisions based on single
biopsy examination (3,19). Recently, numerous studies involving
advanced technology have demonstrated that ICC can originate from
multiple types of cells and exhibits heterogeneity in its genetic
background and microenvironment (3,19). The
existence of two different stem cell compartments and the
associated cell lineages may explain why ICC can develop from
various types of cells (9). It has
been indicated that ICC should be reclassified into ICC that
originate from hepatic stem cell-derived lineages and ICC that
originate from biliary tree stem/progenitor cell-derived lineages
in the peribiliary glands or the epithelia of large bile ducts
(13,31). The genetic heterogeneity of ICC was
examined in previous studies (7,19), which
indicated that the genetic variability in ICC could be caused by
the complex interplay among a number of factors. A total of two
genetically distinct stem cell niches exist along the biliary tree,
and they exhibit differing susceptibility to various risk factors
(19). Additionally, genetically
distinct tumor cell sub-clones can co-exist with founder cells,
which harbor the majority of the tumor-borne genetic mutations
(19).
The microenvironmental factors associated with ICC
include cancer-associated fibroblast and macrophage components
(32). Crosstalk among different
microenvironmental components is involved in the activation of
ICC-associated oncogenic signaling (33). Additionally, an association between
the imaging phenotypes and hypoxia-associated molecular profiles of
ICC was demonstrated in a previous study (34).
The standard treatment strategy for cases of ICC
involving a single intrahepatic nodule and no evidence of
metastasis is surgical resection (18); however, patients that demonstrate
extensive intrahepatic metastases/vascular invasion and/or notable
regional lymph node metastasis should not undergo resection
(35). Although the removal of the
regional lymph nodes is recommended for its prognostic value
(36), the present case involved
notably swollen para-aortic lymph nodes. However, no appropriate
treatment for CoCC has been established due to the rarity of the
condition. Although Ariizumi et al (20) reported that patients with CoCC
exhibited favorable long-term survival time following curative
surgery, the effect of resection with curative intent in cases of
CoCC involving possible metastasis to the regional lymph nodes is
unknown. In the present case, if the ICC had been correctly
diagnosed based on the preoperative biopsy examination, surgical
resection would not have been performed. Considering the difficulty
of differentiating between ICC and CoCC, adopting a policy of
resective surgery for these types of hepatic tumors is
reasonable.
In conclusion, ICC is difficult to distinguish from
other intrahepatic tumor types, including CoCC. Considering the
difficulty of obtaining a definitive preoperative diagnosis,
performing surgery as a diagnostic treatment may be reasonable in
cases involving tumors that exhibit characteristics of ICC and
other liver lesions without distant metastasis.
Acknowledgements
The authors would like to thank Professor Takehiro
Otsubo (St. Marianna University School of Medicine), Professor
Tomoaki Ichikawa (Saitama Medical University International Medical
Center) and Professor Makoto Mochizuki (Teikyo University), for the
fruitful discussion regarding this patient at the 56th Liver Cancer
Cases Conference.
Funding
No funding was received.
Availability of data and materials
The data used and/or analyzed during this published
article are available from the corresponding author on reasonable
request.
Authors' contributions
KN, TE and JY collaborated in the conception and
design of the study. KN, AK, MM, MN, MH, TN and SA acquired the
data. KN, TE, HTa, SO, YK, ES, YY, SH, HTs, KH, HU and JY performed
data analysis and interpretation. All authors were involved in
writing the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient provided written informed consent for
the publication of their data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kitajima K, Shiba H, Nojiri T, Uwagawa T,
Ishida Y, Ichiba N and Yanaga K: Intrahepatic cholangiocarcinoma
mimicking hepatic inflammatory pseudotumor. J Gastrointest Surg.
11:398–402. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Adam SZ, Parthasarathy S and Miller FH:
Intrahepatic cholangiocarcinomas mimicking other lesions. Abdom
Imaging. 40:2345–2354. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Walter D, Döring C, Feldhahn M, Battke F,
Hartmann S, Winkelmann R, Schneider M, Bankov K, Schnitzbauer A,
Zeuzem S, et al: Intratumoral heterogeneity of intrahepatic
cholangiocarcinoma. Oncotarget. 8:14957–14968. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kozaka K, Matsui O, Kobayashi S, Koda W,
Minami T, Kitao A, Inoue D, Yoneda N, Yoshida K, Toshima F, et al:
Dynamic CT findings of cholangiolocellular carcinoma: Correlation
with angiography-assisted CT and histopathology. Abdom Radiol (NY).
42:861–869. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tasch JJ and Dube N: An unusual
presentation of advanced intrahepatic cholangiocarcinoma: When
biopsy results fail. Am J Case Rep. 19:35–40. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ishii N, Suzuki H, Tsukagoshi M, Watanabe
A, Kubo N, Araki K, Wada S and Kuwano H: Giant cholangiolocellular
carcinoma with early recurrence that was difficult to distinguish
from cholangiocellular carcinoma: Report of a case. Int Surg.
100:1111–1116. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bragazzi MC, Ridola L, Safarikia S, Matteo
SD, Costantini D, Nevi L and Cardinale V: New insights into
cholangiocarcinoma: Multiple stems and related cell lineages of
origin. Ann Gastroenterol. 31:42–55. 2018.PubMed/NCBI
|
|
8
|
Schlageter M, Terracciano LM, D'Angelo S
and Sorrentino P: Histopathology of hepatocellular carcinoma. World
J Gastroenterol. 20:15955–15964. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vijgen S, Terris B and Rubbia-Brandt L:
Pathology of intrahepatic cholangiocarcinoma. Hepatobiliary Surg
Nutr. 6:22–34. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Brierley JD, Gospodarowicz MK and
Wittekind C: TNM Classification of Malignant Tumours. 8th edition.
Wiley-Blackwell; Oxford: pp. 2722017
|
|
11
|
Nakanuma Y, Harada K, Ishikawa A, Zen Y
and Sasaki M: Anatomic and molecular pathology of intrahepatic
cholangiocarcinoma. J Hepatobiliary Pancreat Surg. 10:265–281.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Khan SA, Thomas HC, Davidson BR and
Taylor-Robinson SD: Cholangiocarcinoma. Lancet. 366:1303–1314.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Nakanuma Y, Sato Y, Harada K, Sasaki M, Xu
J and Ikeda H: Pathological classification of intrahepatic
cholangiocarcinoma based on a new concept. World J Hepatol.
2:419–427. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sakamoto Y, Kokudo N, Matsuyama Y,
Sakamoto M, Izumi N, Kadoya M, Kaneko S, Ku Y, Kudo M, Takayama T,
et al: Proposal of a new staging system for intrahepatic
cholangiocarcinoma: Analysis of surgical patients from a nationwide
survey of the Liver Cancer Study Group of Japan. Cancer. 122:61–70.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Konstantinidis IT, Koerkamp Groot B, Do
RK, Gönen M, Fong Y, Allen PJ, D'Angelica MI, Kingham TP, DeMatteo
RP, Klimstra DS, et al: Unresectable intrahepatic
cholangiocarcinoma: Systemic plus hepatic arterial infusion
chemotherapy is associated with longer survival in comparison with
systemic chemotherapy alone. Cancer. 122:758–765. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Buettner S, van Vugt JL, IJzermans JN and
Koerkamp Groot B: Intrahepatic cholangiocarcinoma: Current
perspectives. Onco Targets Ther. 10:1131–1142. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Malhi H and Gores GJ: Cholangiocarcinoma:
Modern advances in understanding a deadly old disease. J Hepatol.
45:856–867. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Doherty B, Nambudiri VE and Palmer WC:
Update on the diagnosis and treatment of cholangiocarcinoma. Curr
Gastroenterol Rep. 19:22017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Brandi G, Farioli A, Astolfi A, Biasco G
and Tavolari S: Genetic heterogeneity in cholangiocarcinoma: A
major challenge for targeted therapies. Oncotarget. 6:14744–14753.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ariizumi S, Kotera Y, Katagiri S, Nakano
M, Nakanuma Y, Saito A and Yamamoto M: Long-term survival of
patients with cholangiolocellular carcinoma after curative
hepatectomy. Ann Surg Oncol. 21 Suppl 3:S451–S458. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Liu L, Wang W, Chen B, Xie W, Zhang Y,
Zhang Z, Li Q, Li S, Hua Y, Shen S and Peng B: Combined
hepatocellular and cholangiocarcinoma-WHO classi cation-based
analysis of long-term prognosis after surgery. Int J Clin Exp
Pathol. 10:6601–6612. 2017.
|
|
22
|
Kudo M, Kitano M, Sakurai T and Nishida N:
General rules for the clinical and pathological study of primary
liver cancer, nationwide follow-up survey and clinical practice
guidelines: The outstanding achievements of the Liver Cancer Study
Group of Japan. Dig Dis. 33:765–770. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yano Y, Yamamoto J, Kosuge T, Sakamoto Y,
Yamasaki S, Shimada K, Ojima H, Sakamoto M, Takayama T and Makuuchi
M: Combined hepatocellular and cholangiocarcinoma-a
clinicopathologic study of 26 resected cases. Jpn J Clin Oncol.
33:283–287. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Komuta M, Spee B, Vander Borght S, De Vos
R, Verslype C, Aerts R, Yano H, Suzuki T, Matsuda M, Fujii H, et
al: Clinicopathological study on cholangiolocellular carcinoma
suggesting hepatic progenitor cell origin. Hepatology.
47:1544–1556. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yoh T, Kato T, Hirohata Y, Nakamura Y,
Nakayama H and Okamura R: Cholangiolocellular carcinoma with rapid
progression initially showing abnormally elevated serum
alfa-fetoprotein. Clin J Gastroenterol. 9:257–260. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hoshino H, Ohta M, Ito M, Uchimura K,
Sakai Y, Uehara T, Low S, Fukushima M and Kobayashi M: Apical
membrane expression of distinct sulfated glycans represents a novel
marker of cholangiolocellular carcinoma. Lab Invest. 96:1246–1255.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Akabane S, Ban T, Kouriki S, Tanemura H,
Nakazaki H, Nakano M and Shinozaki N: Successful surgical resection
of ruptured cholangiolocellular carcinoma: A rare case of a primary
hepatic tumor. World J Hepatol. 9:752–756. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ishii N, Araki K, Yamanaka T, Handa T,
Tsukagoshi M, Igarashi T, Watanabe A, Kubo N, Aishima S, Kuwano H
and Shirabe K: Small cholangiolocellular carcinoma that was
difficult to distinguish from cholangiocellular carcinoma: A case
report. Surg Case Rep. 3:1032017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Komuta M, Govaere O, Vandecaveye V, Akiba
J, Van Steenbergen W, Verslype C, Laleman W, Pirenne J, Aerts R,
Yano H, et al: Histological diversity in cholangiocellular
carcinoma reflects the different cholangiocyte phenotypes.
Hepatology. 55:1876–1888. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Motosugi U, Ichikawa T, Nakajima H, Araki
T, Matsuda M, Suzuki T, Fujii H, Nakazawa T and Yamaguchi H:
Cholangiolocellular carcinoma of the liver-imaging findings. J
Comput Assist Tomogr. 33:682–688. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Cardinale V, Carpino G, Reid L, Gaudio E
and Alvaro D: Multiple cells of origin in cholangiocarcinoma
underlie biological, epidemiological and clinical heterogeneity.
World J Gastrointest Oncol. 4:94–102. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Patel T: New insights into the molecular
pathogenesis of intrahepatic cholangiocarcinoma. J Gastroenterol.
49:165–172. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Raggi C, Invernizzi P and Andersen JB:
Impact of microenvironment and stem-like plasticity in
cholangiocarcinoma: Molecular networks and biological concepts. J
Hepatol. 62:198–207. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sadot E, Simpson AL, Do RK, Gonen M, Shia
J, Allen PJ, D'Angelica MI, DeMatteo RP, Kingham TP and Jarnagin
WR: Cholangiocarcinoma: Correlation between molecular profiling and
imaging phenotypes. PLoS One. 10:e01329532015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shimada K, Sano T, Nara S, Esaki M,
Sakamoto Y, Kosuge T and Ojima H: Therapeutic value of lymph node
dissection during hepatectomy in patients with intrahepatic
cholangiocellular carcinoma with negative lymph node involvement.
Surgery. 145:411–416. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Adachi T and Eguchi S: Lymph node
dissection for intrahepatic cholangiocarcinoma: A critical review
of the literature to date. J Hepatobiliary Pancreat Sci.
21:162–168. 2014. View Article : Google Scholar : PubMed/NCBI
|