Case report
In October 2016, a 58-year-old male patient was
referred to the Hepatobiliary Department of Qilu Hospital (Jinan,
China) for review. The patient had previously been diagnosed with
sigmoid cancer (first primary) and left lobe thyroid cancer (second
primary) in May 2015. A left hemicolectomy and radical surgery were
undertaken for the thyroid cancer. The final pathology results for
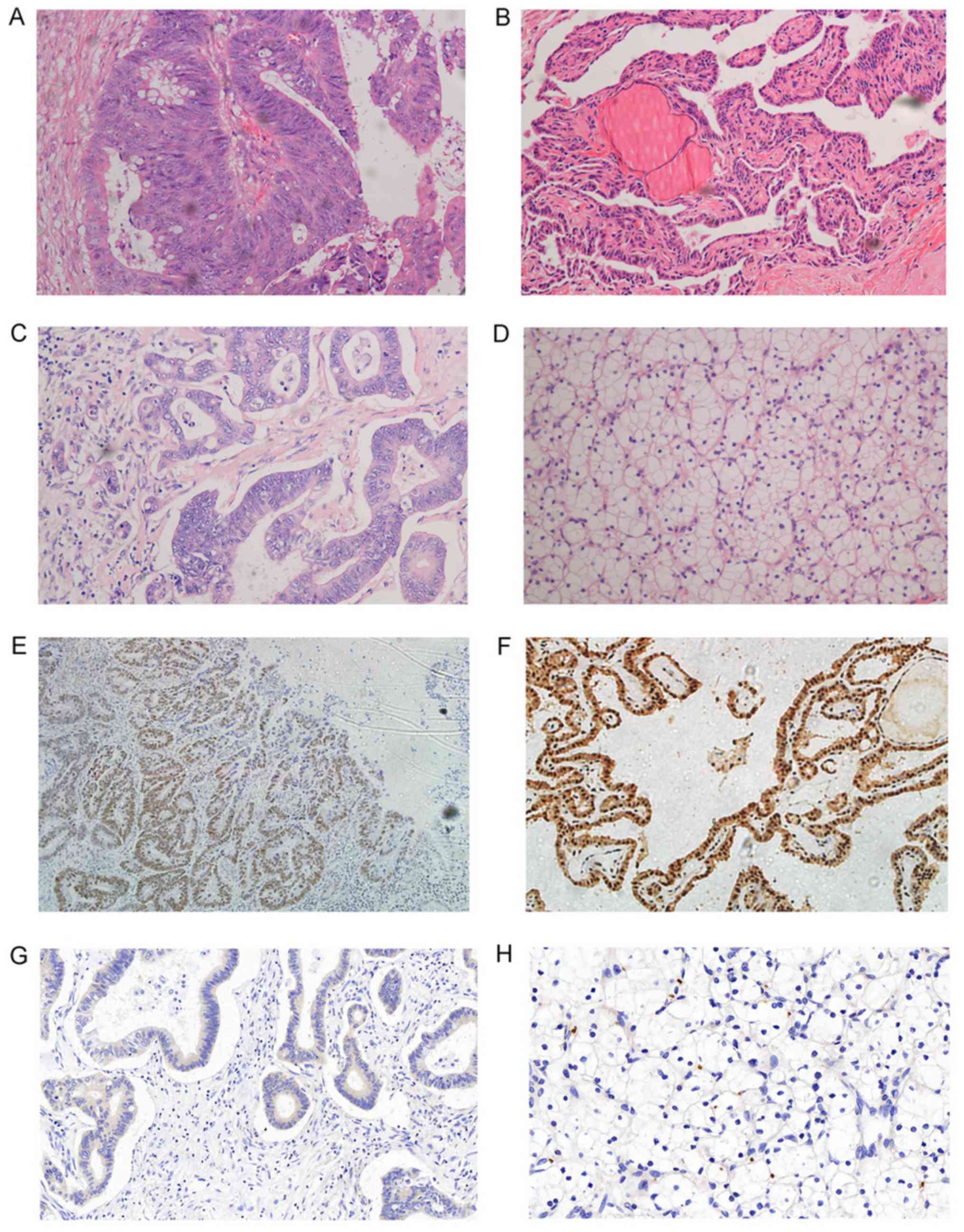
sigmoid colon cancer revealed well-differentiated adenocarcinoma,
polypus type, incisal surface square 2.8×0.3 cm, and invasion of
the stratum sub-mucosa. There were no carcinoma cells at the anodic
and bottom incisal margin, and no metastasis to the lymph nodes
(Fig. 1A). The final pathology
results for thyroid cancer revealed left lobe micro-papillary
carcinoma, and right lobe lymphatic nodule thyroiditis (Fig. 1B). As the sigmoid colon cancer was in
the early stages (pT1, pN0, M0; Stage I), adjuvant chemotherapy was
not administered; 3× cantharidis capsules were taken twice daily,
for 11 months. Routine, post-surgical, examination, including blood
tests, abdominal and pelvic CT scans, and a colonoscopy were
performed. Tumor marker analysis indicted that carcinoembryonic
antigen (CEA) (230.10 ng/ml), cancer antigen (CA)-199 (89.23 U/ml)
and CA-724 (8.80 U/ml), and neuron-specific enolase (NSE) (21.68
ng/ml), among other markers, including CYFr21-1, squamous cell
carcinoma antigen, pro-gastrin releasing peptide, α-fetoprotien,
CA-125, Ferrari, total prostate-specific antigen and serum sialic
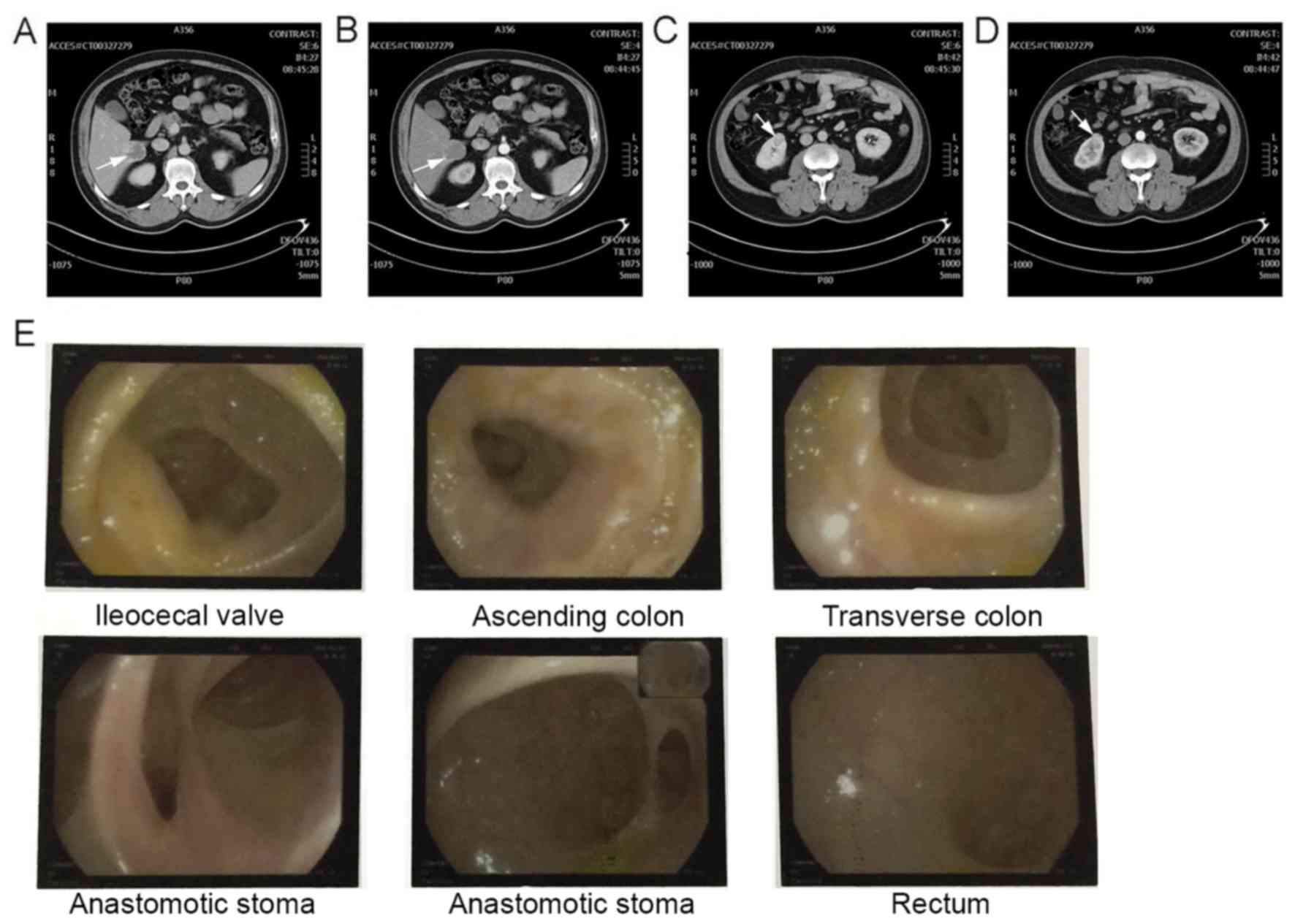
acid were within the normal range. CT scan suggested that,
according to the patient history, the space-occupying lesion
located in the V and VIII liver segments may have been the
metastatic tumor; the VII segment tumor may have been an hemangioma
(Fig. 2A and B); and the
space-occupying lesion of the right kidney anus perineum may have
been primary kidney cancer (third primary) (Fig. 2C and D).
The colonoscopy results were satisfactory, with no
recurrence noted (Fig. 2E). To
further investigate the characteristics of the liver and kidney
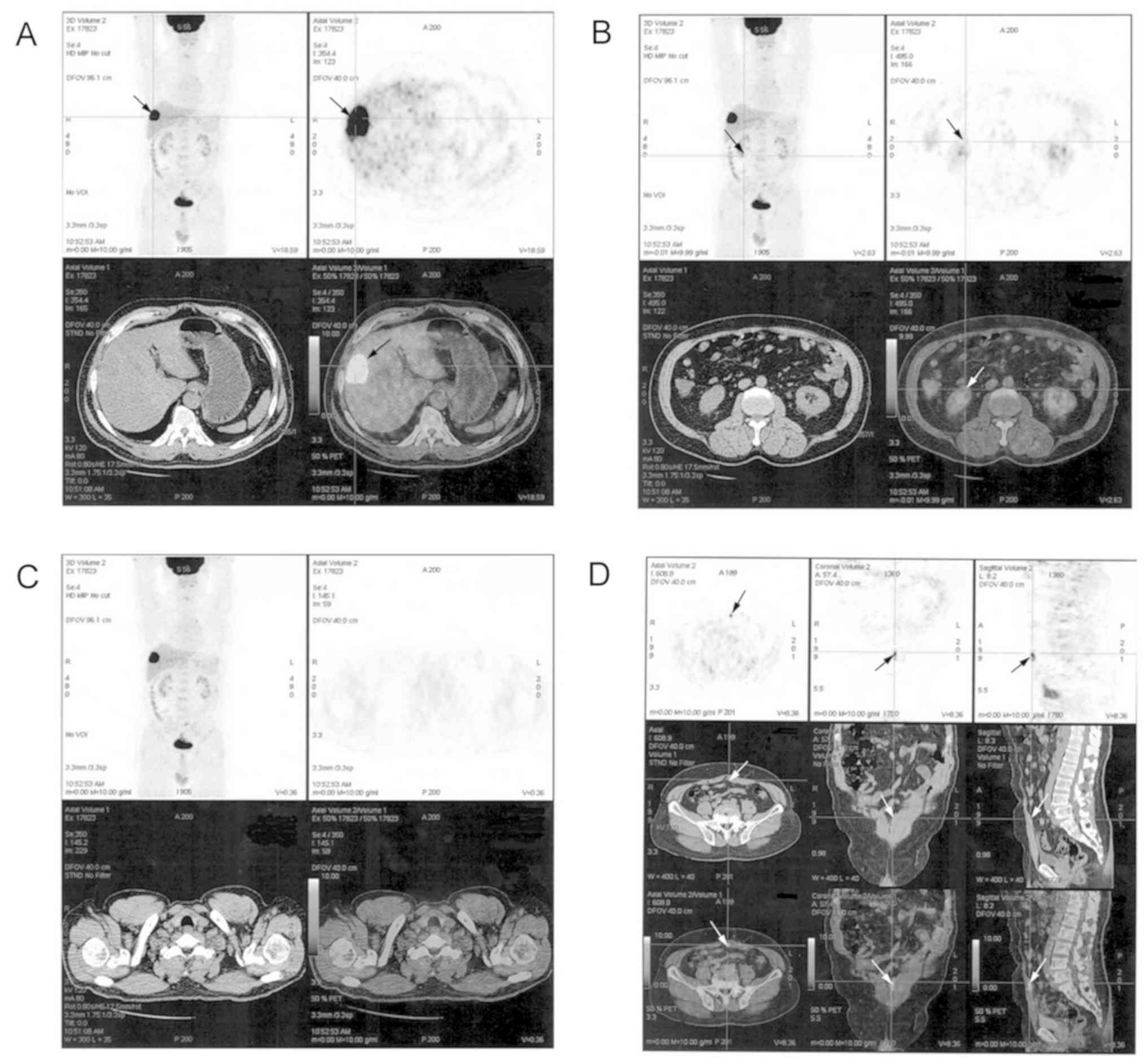
tumors, PET-CT examination was conducted, the results of which
indicated that: i) The tumor in the right anterior lobe of the
liver may have been a metastatic lesion (Fig. 3A); ii) combined with the CT scan
results, it was supposed that the tumor of the right kidney anus
perineum may have been malignant (hypo-metabolism type) (Fig. 3B); iii) 1.5 years after radical
surgery for left lobe thyroid cancer, no recurrence was observed
(Fig 3C); iv) a small node in the
left anterior-inferior abdominal exhibited high absorbance of FDG,
possibly indicating metastasis or a responsive modification
(Fig. 3D).
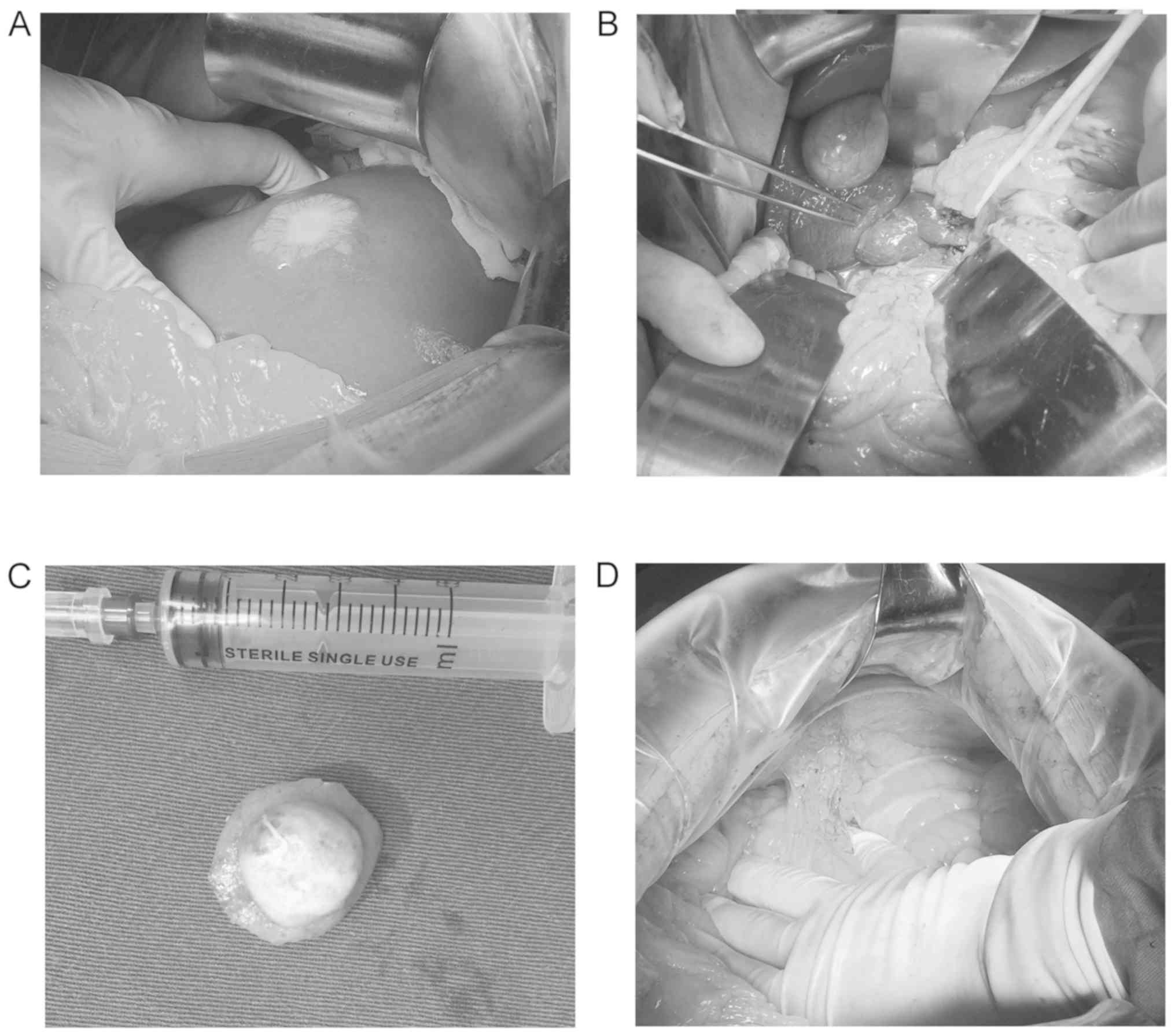
As a result of these findings, surgical excision of
the metastatic liver tumor and hepatic hemangioma, in addition to a
partial nephrectomy, were conducted on October 11th, 2016 (Fig. 4). The final pathology examination
results revealed: i) V and VIII segment liver, well-differentiated
adenocarcinoma in the liver tissue (incision square, 4.0×2.7 cm),
which, according to the patient history, is the metastatic tumor
from the primary colon cancer (Fig.
1C); ii) VII segment liver, cavernous hemangioma; iii) kidney,
clear-cell carcinoma of the kidney (II level nuclear) with a
diameter of 2.3 cm (Fig. 1D); iv)
abdominal wall, hyperplastic collagen fiber and vascular tissue.
Post-surgery, the patient recovered well, and was administered one
course of FOLFORX6 chemotherapy prior to discharge (November 3rd,
2016). In subsequent follow-up, the patient had recovered
markedly.
For resected specimens of colon, thyroid, liver
metastatic tumor and renal cell carcinoma, immunohistochemical
staining of integrin αvβ6 was performed, the results of which are
detailed below. Experimental methods were as follows: Mouse
anti-human monoclonal antibody (clone 6.2A1; IgG1) against integrin
αvβ6 was obtained from Biogen (Abcam, Cambridge, MA, USA). Patient
tissue sections were deparaffinized and hydrated, and heat-induced
epitope retrieval was performed using Borg decloaking high pH
buffer in the Biocare decloaking chamber (Biocare Medical, LLC,
Pacheco, CA, USA). Endogenous peroxidase activity was blocked with
3% hydrogen peroxide for 5 min at room temperature. The slides were
incubated with an Endogenous Avidin-Biotin Blocking kit (cat. no.
ab64212; Abcam) for 10 min, followed by further incubations with
the integrin αvβ6 primary antibody 6.2A1 (1:500) overnight at 4°C.
The following day, biotinylated anti-mouse IgG (1:200; cat. no.
ab207996; Abcam) was applied to the slides for 20 min at 37°C,
which were subsequently treated with 50 µl horseradish
peroxidase-labeled streptoantibiotin (Dako; Agilent Technologies,
Inc., Santa Clara, CA, USA) for 15 min at 37°C and Betazoid
Diaminobenzidine (Biocare Medical, LLC, Pacheco, CA, USA) for color
development for 2 min at room temperature. The slides were
counterstained with hematoxylin for 1 min (Dako; Agilent
Technologies, Inc.), rinsed with water for 2 min, and dehydrated
with alcohol and xylene for several seconds, before a coverslip was
affixed. Normal mouse IgG (1:200; cat. no. ab188776; Abcam) was
substituted for primary antibody as the negative control and
incubated with the slides overnight at 4°C. Unless otherwise
indicated, incubations were performed at room temperature. On all
slides, >5% of the cells stained positively for integrin
αvβ6.
Discussion
MPC refers to the presence of ≥2 pathologically
diagnosed primary cancers, in ≥1 organ of the same patient. The
diagnosis of MPC is based on the Warren standards (1): i) All tumors identified after the
pathological examination are malignant; ii) each tumor is
independent of the others, and the possibility of transfer is
excluded; and iii) all tumors originate from different organs.
According to differences in primary tumor occurrence times,
simultaneous MPC refers to cases wherein all tumors appear within 6
months; otherwise, it is known as non-simultaneous or metachronous
MPC.
MPC is relatively rare, with a reported incidence
range of 0.8–6.6% among the general population (2–4); however,
the Surveillance Epidemiology and End Results report issued by the
National Cancer Institute states that between 1975 and 2000, the
incidence increased to ~16% (5). This
difference may be associated with location, environment, living
habits, quality and availability of medical care, the type of
cancer and statistical research methods. The incidence of triple or
quadruple malignant cancer in one patient remains rare, with the
incidence of quadruple cancer reported to be <0.1% (6–8). Thus,
cases of MPC may be overlooked or misdiagnosed. MPC typically
originates in pairs of organs or same system organs, such as the
gastrointestinal, respiratory or urological systems. Male patients
have an increased tendency to develop gastrointestinal cancer, and
female patients are more prone to breast and gastrointestinal
cancer (9,10). Therefore, when a patient presents with
these particular cancer types, clinicians should investigate
whether an accompanying malignancy has occurred elsewhere. Baigrie
(11) reported that colon cancer is
frequently present in cases of MPC, particularly in patients
between 50 and 60 years of age.
The definite cause of MPC is not clear. Though there
are a number of factors that may influence its occurrence
(including smoking, and exposure to carcinogens or carcinogenic
environments) there are numerous defining characteristics of MPC.
With improvements in living standards and medical technology, life
expectancy is increasing, in accordance with the probability of
acquiring a malignancy. It was reported by Curtis et al
(12) that patients with cancer were
14% more likely to develop an additional tumor compared with the
general population. Additionally, it was deduced by Jiao et
al (6) that this phenomenon was
associated with increased genetic instability, and the reduction of
tumor immunity in cancer patients.
In addition, the wide application of diagnostic
imaging has led to increased radiation exposure; this is
particularly prominent in developed countries (13), where during routine follow-up
examinations, patients are exposed through imaging procedures
including X-rays and CT scans (14).
Frequent doses of radiation may increase the occurrence of further
malignancies (15). Studies have
confirmed that the occurrence of head and neck cancer, particularly
thyroid cancer, is associated with long-term radiological
examination (16); specifically, the
over-diagnosis and treatment of small adenocarcinoma was reported
to influence the occurrence of thyroid cancer (17,18). This
is consistent with the case reported in the current study.
Furthermore, the carcinogenic effects of chemotherapy and endocrine
therapy may contribute to the development of MPC. For example,
studies have demonstrated that in patients with breast cancer who
received long-term treatment with tamoxifen or raloxifene, the
incidence of endometrial cancer was increased 2–3-fold compared
with healthy volunteers (19,20).
Integrin αvβ6 is an integrin subtype expressed only
in epithelial cells; its primary ligand is fibronectin (FN). In
healthy epithelial cells the expression of integrin αvβ6 is rare
(21), although it is increased
substantially in response to injury and/or inflammation, and in
epithelial tumors (including gastric carcinoma and colon cancer)
(22–24). The de novo expression of
integrin αvβ6 has been reported to modulate a number of
characteristics of colon carcinoma cells, including adhesion and
spreading on fibronectin, proliferation in collagen gels, tumor
growth, invasion and metastasis, and apoptosis (25–28). In
the present case report, immunohistochemical staining of integrin
αvβ6 was conducted, which revealed positive expression of integrin
αvβ6 in numerous tissues, including those from the colon cancer
primary site (Fig. 1E), the left lobe
of the thyroid (Fig. 1F), and the
hepatic metastatic tumor (Fig. 1G).
Expression of integrin αvβ6 was negative in clear cell carcinoma of
the kidney (Fig. 1H). In a previous
study, it was identified that the rate of positive integrin αvβ6
expression in hepatic metastatic foci was 71.4% (29). Our previous study regarding the
associations between integrin αvβ6 and thyroid cancer revealed the
positive expression rate of integrin αvβ6 in thyroid papillary
carcinoma to be 79.03% (49/62), 78.57% (22/26) in thyroid
follicular carcinoma, and 100% in metastatic lymph nodes (10/10)
(30). Combined with additional
previous data, it was proposed that integrin αvβ6 may have an
important role in MPC.
Renal clear cell carcinoma (RCC) originates from the
malignant transformation of renal tubular epithelial cells. Due to
its expression in malignant epithelial tumors, integrin αvβ6 may
also be expressed in RCC. However, in this patient study, RCC
specimens were negative for integrin αvβ6 expression, supported by
the lack of literature reporting integrin αvβ6 expression in RCC
tissue. Negative expression is predicted to be due to the loss of
cytoplasm from RCC carcinoma cells during the preparation of the
tissue sections. The loss of lipid components and vacuolization of
the cytoplasm forms the so-called ‘transparent’ component; as
integrin αvβ6 is predominantly expressed in the epithelial cell
membrane and cytoplasm, cytoplasmic depletion in RCC may
potentially negate the expression of integrin αvβ6.
The incidence of MPC is increasing, thus there is an
urgent need to develop the understanding of MPC pathogenesis,
diagnosis and treatment. Current treatment regimens for MPC differ
from those indicated for traditional recurrent or metastatic
malignant tumors. For MPC, radical excision of the tumors is
preferred. When faced with that for recurrent or metastatic
malignant tumors, the prognosis for MPC is comparatively more
favorable (31,32).
With improvements in anti-tumor treatment
technology, the prognosis for tumor patients has improved, though
there has been a corresponding rise in the incidence of MPC. The
aim of the clinician should be to improve understanding of MPC,
reduce misdiagnosis and missed diagnosis, make early diagnoses to
facilitate earlier treatment, and to administer radical surgery,
radiotherapy and chemotherapy, and targeted therapy to improve
clearance rates and prolong patient survival. The current case
demonstrates that integrin αvβ6 is positively expressed in MPC.
Combined with previous research, this may implicate integrin αvβ6
in the pathogenesis of MPC, and aid future research into the
etiology of MPC.
Acknowledgements
Not applicable.
Funding
This study was supported by the Natural Sciences
Foundation of Shandong Province (grant no. ZR2015HM042).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CP led patient diagnosis and treatment, and
manuscript authorship. ZL performed the immunohistochemical
staining. HG, was the resident carer, responsible for the daily
care of the patient, recorded the clinical laboratory examination
results, and retrieved the related literature. XZ was responsible
for data collation, statistical analysis and figure creation. XW
was responsible for pathological procedures and diagnoses. CZ
performed the radical nephrectomy. JN as the chief surgeon,
performed the radical resection of sigmoid colon cancer and
excision of liver metastases from sigmoid colon cancer. He was
additionally involved the immunohistochemical staining experiment
and statistical analysis of data. As the Dean of the Institute of
Laparoscopic Minimally Invasive Surgery of Shandong University, he
gave the final approval of the article.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient provided informed consent for the use of
the data in this study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ueno M, Muto T, Oya M, Ota H, Azekura K
and Yamaguchi T: Multiple primary cancer: An experience at the
Cancer Institute Hospital with special reference to colorectal
cancer. Int J Clin Oncol. 8:162–167. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Demandante CG, Troyer DA and Miles TP:
Multiple primary malignant neoplasms: Case report and a
comprehensive review of the literature. Am J Clin Oncol. 26:79–83.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jones AS, Morar P, Phillips DE, Field JK,
Husband D and Helliwell TR: Second primary tumours in patients with
head and neck squamous cell carcinoma. Cancer. 75:1343–1353. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Aydiner A, Karadeniz A, Uygun K, Tas S,
Tas F, Disci R and Topuz E: Multiple primary neoplasms at a single
institution: Differences between synchronous and metachronous
neoplasms. Am J Clin Oncol. 23:364–370. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ries LA, Eisner MP, Kosary CL, Hankey BF,
Miller BA and Clegg L: SEER Cancer Statistics Review. 1975–2000.
National Cancer Institute; Bethesda, MD: 2003
|
|
6
|
Jiao F, Hu H and Wang LW: Quadruple
primary malignancy patient with survival time more than 20 years.
World J Gastroenterol. 19:1498–1501. 2003. View Article : Google Scholar
|
|
7
|
Jones P: Five separate malignancies in one
patient. Br Med J. 1:15331976. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shankar PS: Case report. Five primary
cancers in one patient Postgrad Med. 61:281–282. 1977.PubMed/NCBI
|
|
9
|
Liu LY, Sheng SH, Zhang ZY and Xu JH: A
case of matrix-producing carcinoma of the breast with micoglandular
adenosis and review of literature. Int J Clin Exp Pathol.
8:8568–8572. 2015.PubMed/NCBI
|
|
10
|
Ye Y, Neil AL, Wills KE and Venn AJ:
Temporal trends in the risk of developing multiple primary cancers:
A systematic review. BMC Cancer. 16:8492016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Baigrie RJ: Seven different primary
cancers in a single patient. A case report and review of multiple
primary malignant neoplasia. Eur J Surg Oncol. 17:81–83.
1991.PubMed/NCBI
|
|
12
|
Curtis RE, Freedman DM, Ron E, Ries LAG,
Hacker DG, Edwards BK, Tucker MA and Fraumeni JF Jr: New
malignancies among cancer survivors: SEER cancer registries,
1973–2000. National Cancer Institute; Bethesda, MD, NIH Publ.: No.
05-5302. 2006
|
|
13
|
Linet MS, Slovis TL, Miller DL, Kleinerman
R, Lee C, Rajaraman P and Berrington de Gonzalez A: Cancer risks
associated with external radiation from diagnostic imaging
procedures. CA Cancer J Clin. 62:75–100. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Meyerhardt JA, Mangu PB, Flynn PJ, Korde
L, Loprinzi CL, Minsky BD, Petrelli NJ, Ryan K, Schrag DH, Wong SL,
et al: Follow-up care, surveillance protocol, and secondary
prevention measures for survivors of colorectal cancer: American
society of clinical oncology clinical practice guideline
endorsement. J Clin Oncol. 31:4465–4470. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mathews JD, Forsythe AV, Brady Z, Butler
MW, Goergen SK, Byrnes GB, Giles GG, Wallace AB, Anderson PR,
Guiver TA, et al: Cancer risk in 680000 people exposed to computed
tomography scans in childhood or adolescence: Data linkage study of
11 million Australians. BMJ. 346:f23602013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kim C, Bi X, Pan D, Chen Y, Carling T, Ma
S, Udelsman R and Zhang Y: The risk of second cancers after
diagnosis of primary thyroid cancer is elevated in thyroid
microcarcinomas. Thyroid. 23:575–582. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Morgan DJ, Dhruva SS, Wright SM and
Korenstein D: Update on medical practices that should be questioned
in 2015. JAMA Intern Med. 175:1960–1964. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Davies L and Welch HG: Increasing
incidence of thyroid cancer in the United States, 1973–2002. JAMA.
295:2164–2167. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Matesich SM and Shapiro CL: Second cancer
after breast cancer treatment. Semin Oncol. 30:740–748. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Williams-Brown MY, Salih SM, Xu X,
Veenstra TD, Saeed M, Theiler SK, Diaz-Arrastila CR and Salama SA:
The effect of tamoxifen and raloxifene on estrogen metabolism and
endometrial cancer risk. J Steroid Biochem Mol Biol. 126:78–86.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Breuss JM, Gillett N, Lu L, Sheppard D and
Pytela R: Restricted distribution of integrin beta 6 mRNA in
primate epithelial tissue. J Histochem Cytochem. 41:1521–1527.
1999. View Article : Google Scholar
|
|
22
|
Breuss JM, Gallo J, DeLisser HM,
Klimanskaya IV, Folkesson HG, Pittet JF, Nishimura SL, Aldape K,
Landers DV, Carpenter W, et al: Expression of the beta 6 integrin
subunit in development, neoplasia and tissue repair suggests a role
in epithelial remodeling. J Cell Sci. 108:2241–2251.
1995.PubMed/NCBI
|
|
23
|
Bates RC, Bellovin DI, Brown C, Maynard E,
Wu B, Kawakatsu H, Sheppard D, Oettgen P and Mercurio AM:
Transcriptional activation of integrin beta 6 during the
epithelial-mesenchymal transition defines a novel prognostic
indicator of aggressive colon carcinoma. J Clin Invest.
115:339–347. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang ZY, Xu KS, Wang JS, Yang GY, Wang W,
Wang JY, Niu WB, Liu EY, Mi YT and Niu J: Integrin alpha v beta 6
acts as a prognostic indicator in gastric carcinoma. Clin Ongol.
20:61–66. 2008. View Article : Google Scholar
|
|
25
|
Niu J, Gu X, Turton J, Meldrum C, Howard
EW and Agrez M: Integrin-mediated signalling of gelatinase B
secretion in colon cancer cells. Biochem Biophys Res Commun.
249:287–291. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Agrez M, Gu XH, Turton J, Meldrum C, Niu
J, Antalis T and Howard EW: The alpha v beta 6 integrin induces
gelatinase B secretion in colon cancer cells. Int J Cancer.
81:90–97. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang GY, Xu KS, Pan ZQ, Zhang ZY, Mi YT,
Wang JS, Chen R and Niu J: Integrin alpha v beta 6 mediates the
potential for colon cancer cells to colonize in and metastasize to
the liver. Cancer Sci. 99:879–887. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu S, Wang J, Niu W, Liu E, Wang J, Peng
C, Lin P, Wang B, Khan AQ, Gao H, et al: The β6-integrin-ERK/MAP
kinase pathway contributes to chemo resistance in colon cancer.
Cancer Lett. 328:325–334. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang GY, Xu KS, Pan ZQ, Zhang ZY, Mi YT,
Wang JS, Chen R and Niu J: Integrin alpha v beta 6 mediates the
potential for colon cancer cells to colonize in and metastasize to
the liver. Cancer Sci. 99:879–887. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liu S, Liang B, Gao H, Zhang F, Wang B,
Dong X and Niu J: Integrin αvβ6 as a novel marker for diagnosis and
metastatic potential of thyroid carcinoma. Head Neck Oncol.
5:72013.
|
|
31
|
Koide N, Adachi W, Koike S, Watanabe H,
Yazawa K and Amano J: Synchronous gastric tumors associated with
esophageal cancer: A retrospective study of twenty-four patients.
Am J Gastroenterol. 93:758–762. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chai W, Gong FY, Zhang WL, Wen Y and Cui
LF: Multiple primary cancer in the female genital system. Medicine.
96:472017. View Article : Google Scholar
|