Introduction
Hepatocellular carcinoma (HCC) is a common solid
tumor displaying inferior prognosis and a high recurrence rate,
which results in extensive worldwide mortality (1). Due to the development of innovative
treatments like hepatic transplantation, hepatic resection,
chemotherapy, and radiofrequency ablation, the survival rates of
patients have improved (2); however,
an effective treatment for the complete cure of HCC has yet to be
developed. Although significant efforts have been made to elucidate
the disease progression and to develop valuable therapies, the
molecular mechanisms in HCC are still mostly unknown. Hence, there
is an imperative need to enhance the prognosis of the disease.
Oncolytic viruses (OVs), which can be engineered to selectively
replicate intracellularly and destroy tumor tissues, have been
applied as an efficacious solution against tumors. Numerous OVs
have been designed to exploit their antitumor effects; for
instance, pexastimogene devacirepvec (Pexa-Vec) is currently in a
phase III trial for the treatment of HCC (3). In this context, the safety and efficacy
of adenovirus vectors for gene delivery should also be demonstrated
in the future.
The coxsackie and adenovirus receptor (CAR), a
transmembrane constituent of the tight junctions in epithelial
tissue, has been originally discovered as a viral attachment site
that is essential for virus uptake (4). Walters et al (5) established the role of CAR in gene
transfer and as a principal receptor for the coxsackie B virus and
adenovirus. Pandha et al (6)
demonstrated that CAR expression was strongly correlated with
adenovirus infection, adhesion and transgene expression. Attenuated
adenoviruses, which may be replication-incompetent to transmitted
therapeutic genes or viruses, can be employed in cancer therapy
(7). Therefore, the expression of CAR
is considered a crucial factor for the effectiveness of
adenovirus-based therapeutics.
Analysis of CAR expression in several classes of
tumors produced diverse results. It has been reported that CAR
expression was low in colon, lung and bladder (8–11) tumors,
predominantly in poorly differentiated and advanced-stage cancers
(9,12,13).
Additionally, downregulated CAR expression predicted an inferior
clinical result for gastric and bladder cancer patients (9,14).
Contrastingly, CAR upregulation has been detected in endometrial,
ovarian, cervical and breast cancers, along with neuroblastomas and
medulloblastomas (15–20). Moreover, elevated CAR expression was
correlated with inferior prognosis in breast and lung cancers
(12,21). Therefore, there is a need to confirm
if these outcomes indicate a disparity in CAR expression or a
result from racial and procedural variations.
In the present investigation, immunohistochemistry
was employed to evaluate CAR expression in HCC and neighboring
healthy tissue specimens in tissue microarrays (TMAs). A greater
sample size was chosen to acquire data for an improved
understanding of the function of CAR in the progression of HCC.
Additionally, probable targets for adenovirus-mediated therapeutic
strategies related to CAR expression were established.
Materials and methods
HCC patients in tissue microarray
HCC protein expression levels were assessed with
immunohistochemical staining of tissue microarrays, which were
purchased from Shanghai Biochip Co., Ltd. (Shanghai, China). The
patient samples were collected from Zhejiang Provincial People's
Hospital (Hangzhou, China) and all patients provided written
informed consent. The study was approved by the Ethics Committee of
Zhejiang Provincial People's Hospital. The TMAs contained a total
of 396 formalin-fixed, paraffin-embedded archived samples from a
total of 198 HCC patients from the Chinese Han population, in
addition to 198 corresponding controls derived from adjacent normal
tissue samples.
The patient cohort consisted of 159 males and 39
females, with a median age of 55 years (range, 27–91 years) at the
time of surgery. All patients had follow-up records for >5
years. The survival time was calculated from the date of surgery to
the follow-up deadline or mortality.
Immunohistochemical staining
Immunohistochemical staining was performed according
to the standard method. Briefly, 5-µm sections from the TMAs were
baked at 70°C for 2 h. Then, the sections were de-paraffinized in
xylene, rehydrated using a gradient of ethanol concentrations,
boiled in 1 mM TE buffer with a high-pressure cooker for 3 min for
antigen retrieval, blocked with 3% hydrogen peroxide for 15 min to
inhibit endogenous peroxidase activity and incubated with 10% goat
non-immune serum (Invitrogen; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) for 20 min to reduce background non-specific
staining. Subsequently, TMA sections were incubated with rabbit
anti-human primary polyclonal antibody against CAR (dilution,
1:400; cat. no. Sc-15405; Santa Cruz Biotechnology, Inc., Dallas,
TX, USA) overnight at 4°C, and then incubated with biotin-labeled
secondary antibody (1:200; cat. no. AB-2548649) at room temperature
for 15 min, followed by incubation with HRP-conjugated streptavidin
(1:100; cat. no. AB-11155398; both Invitrogen; Thermo Fisher
Scientific, Inc) at room temperature for 15 min. Then, color
development was performed with a DAB Substrate kit (Dako, Glostrup,
Denmark). Finally, the sections were counterstained with
hematoxylin, dehydrated, cleared and mounted.
Evaluation of the immunohistochemical
stainings
Immunohistochemical stainings of CAR were scored by
two pathologists independently, based on the intensity and the
proportion of positively stained cells. Staining intensity was
evaluated with a four-tiered grading system: 0, negative; 1, weak;
2, moderate; and 3, strong. The percentage of positive cells were
scored as follows: 0, no cells stained, 1, 1–25% of cells stained;
2, 26–50% of cells stained; 3, 51–75% of cells stained; and 4,
>75% of cells stained. Scores for intensity and percentage of
positive cells were multiplied. Scores ≤6 were used to define
tumors with low CAR expression and scores >6 with high CAR
expression.
Statistical analysis
Statistical analysis was performed using Statistical
Program for Social Sciences (SPSS) software 13.0 (SPSS Inc.,
Chicago, IL, USA). The χ2-tests were applied to assess
the statistical significance of the associations between CAR
expression and clinicopathological parameters, respectively. Cox
proportional hazards regression model was used to perform
multivariate survival analysis to assess predictors associated with
prognosis. P<0.05 was considered to indicate a statistically
significant difference.
Results
CAR expression in HCC is inferior to
that in adjacent healthy tissue samples
Immunohistochemistry was employed to evaluate the
distribution of HCC, where immunostaining of the tissues
predominantly affected the cell membrane and cytoplasm (Fig. 1). Positive CAR expression was observed
in 114 out of 198 (57%) normal liver tissue samples, which was
slightly superior to that of HCC (56%, 111/198,
χ2=174.7, P>0.05).
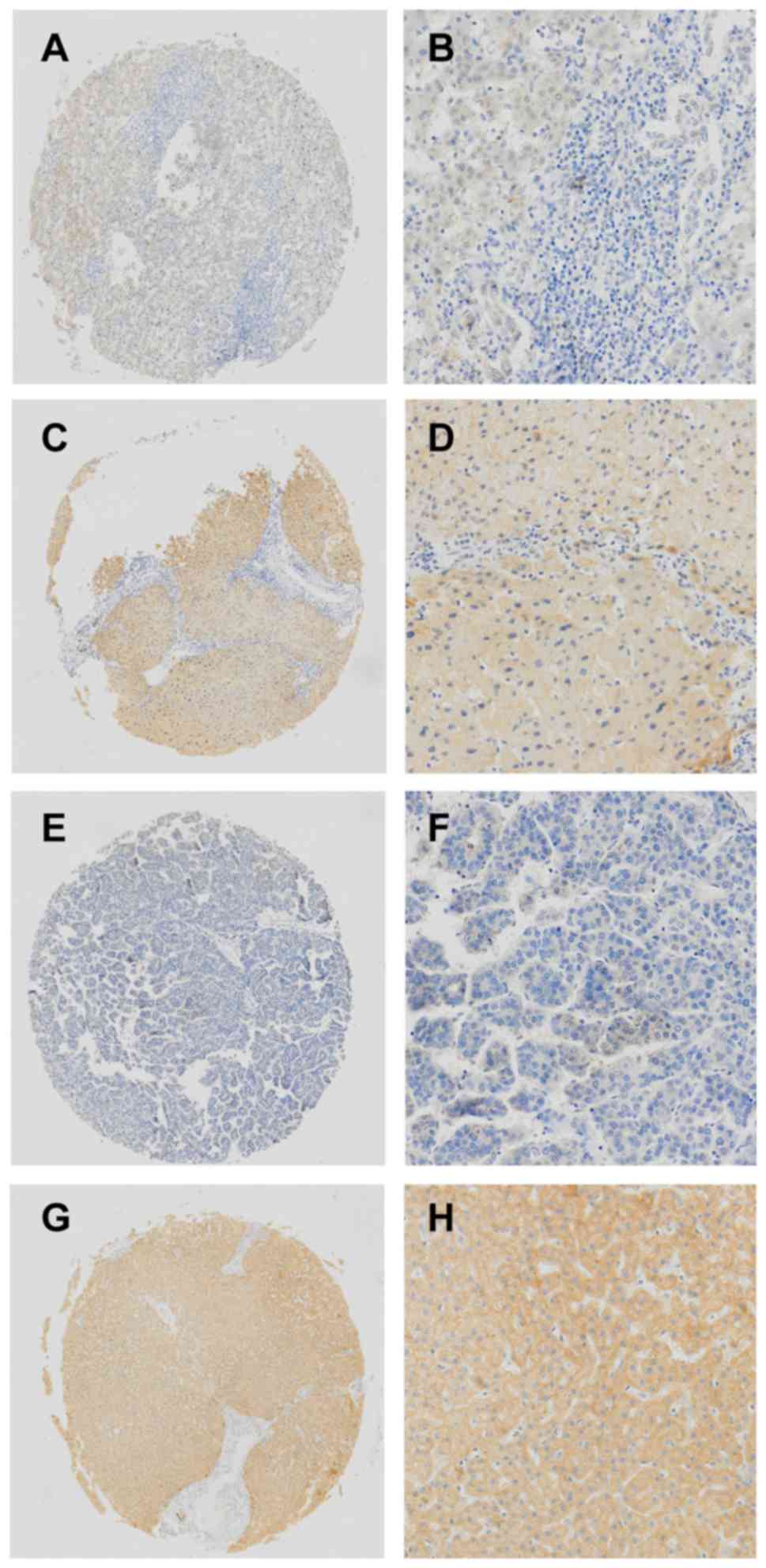 | Figure 1.Immunohistochemical analysis of CAR in
liver cancer and normal tissues. (A and B) Low expression of CAR in
normal liver tissues adjacent to cancerous tissues. (C and D) High
expression of CAR in normal liver tissues adjacent to cancerous
tissues and positive staining, primarily in the membrane and
cytoplasm. (E and F) Low expression of CAR in the tumor sample. (G
and H) High expression of CAR in the tumor sample and positive
staining, mainly in the membrane and cytoplasm. (A, C, E, and G)
Original magnification, ×200; (B, D, F, and H) original
magnification, ×800. CAR, Coxsackie and adenovirus receptor. |
Patient characteristics and the
association between CAR expression and clinicopathological
parameters of HCC
With the aim of exploring whether CAR is related to
HCC progression, the expression of CAR was studied with respect to
clinicopathological features in HCC. The outcomes established that
the occurrence of CAR immunopositivity was not significantly
dependent upon sex, age, metastasis, microvascular invasion, HBs
antigen, cirrhosis and AFP (Table
I).
 | Table I.Expression of CAR in HCC tissues. |
Table I.
Expression of CAR in HCC tissues.
|
|
| CAR expression |
|
|---|
|
|
|
|
|
|---|
| Clinical
parameters | No. | Low | High | P-value |
|---|
| Age, years |
|
|
| 0.989 |
|
<55 | 75 | 33 | 42 |
|
| ≥55 | 123 | 54 | 69 |
|
| Sex |
|
|
| 0.303 |
| Male | 159 | 67 | 92 |
|
|
Female | 39 | 20 | 19 |
|
| Size, cm |
|
|
| 0.019 |
|
<5 | 100 | 36 | 64 |
|
| ≥5 | 95 | 50 | 45 |
|
| Tumor number |
|
|
| 0.407 |
|
Single | 161 | 73 | 88 |
|
|
Multiple | 37 | 14 | 23 |
|
| Edmondson
grade |
|
|
| 0.009 |
|
I+II | 127 | 47 | 80 |
|
|
III | 71 | 40 | 31 |
|
| Metastasis |
|
|
| 0.987 |
| M0 | 182 | 80 | 102 |
|
| M1 | 16 | 7 | 9 |
|
| Microvascular
invasion |
|
|
| 0.880 |
|
Absence | 65 | 33 | 32 |
|
|
Presence | 73 | 38 | 35 |
|
| HBs antigen |
|
|
| 0.744 |
|
Negative | 43 | 18 | 25 |
|
|
Positive | 150 | 67 | 83 |
|
| Cirrhosis |
|
|
| 0.701 |
|
Negative | 62 | 26 | 36 |
|
|
Positive | 136 | 61 | 75 |
|
| AFP |
|
|
| 0.274 |
|
<50 | 101 | 44 | 57 |
|
|
≥50 | 97 | 49 | 48 |
|
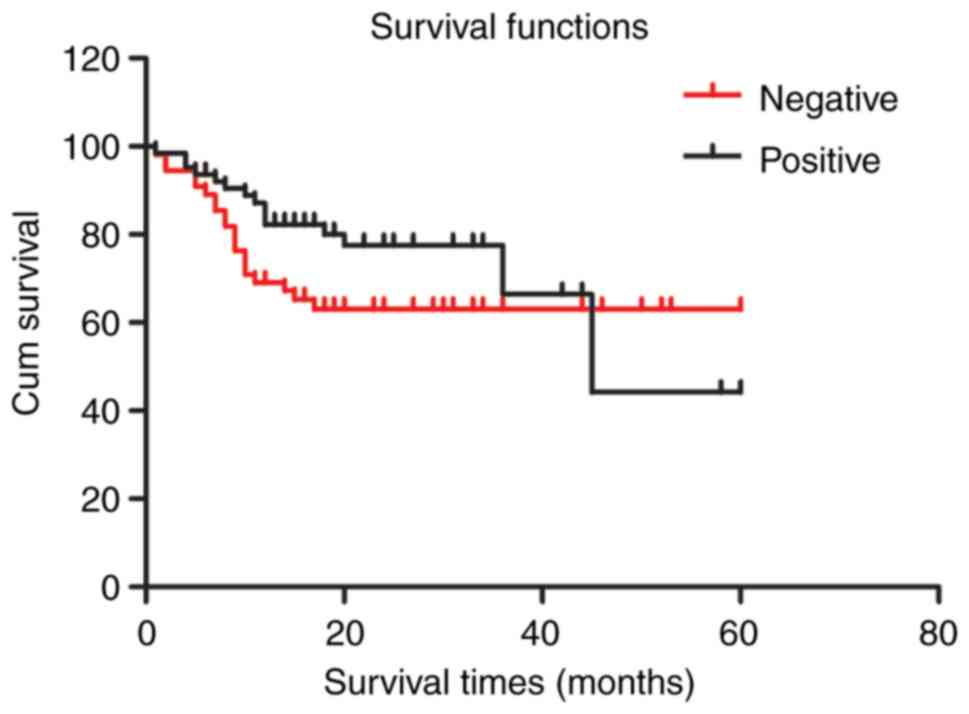
Survival analysis reveals that CAR
immunopositivity does not significantly diminish survival
times
The average survival time of CAR-positive liver
cancer patients (42.201±4.056 months) was not significantly
different than that of CAR-negative patients (40.934±3.409 months,
P=0.240). The Kaplan-Meier survival curve indicated that CAR
expression was not significantly associated to overall survival in
HCC patients (Fig. 2). Additionally,
the prognosis factors of HCC were analyzed by Cox-regression
analysis. Univariate analysis indicated that tumor size (P=0.005),
metastasis (P<0.001), microvascular invasion (P=0.016),
Edmondson grade (P<0.001) and AFP (P=0.002) were independent
prognostic factors in patients with HCC. Multivariate analysis
revealed that distant metastases (P<0.001), tumor number
(P=0.001), Edmondson grade (P=0.028) and AFP (P=0.008) were
independent prognostic factors for patients with HCC (Table II).
 | Table II.Univariate and multivariate
Cox-regression analyses of the clinicopathological features in HCC
patients. |
Table II.
Univariate and multivariate
Cox-regression analyses of the clinicopathological features in HCC
patients.
|
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|
|---|
| Parameters | No. | HR | 95.0% CI for
HR | P-value | HR | 95.0% CI for
HR | P-value |
|---|
| Age (<55
years/≥55 years) | 75/123 | 0.651 | 0.409–1.038 | 0.071 | 0.335 | 0.119–0.943 | 0.058 |
| Sex
(male/female) | 159/39 | 1.575 | 0.922–1.575 | 0.096 | 0.623 | 0.202–1.922 | 0.410 |
| Tumor size (<50
mm/≥50 mm) | 100/95 | 1.954 | 1.219–3.132 | 0.005 | 1.425 | 0.423–4.799 | 0.567 |
| Tumor number
(single/multiple) | 161/37 | 1.239 | 0.678–2.264 | 0.487 | 11.26 | 2.573–49.279 | 0.001 |
|
Metastasis(M0/M1) | 182/16 | 4.821 | 2.551–9.112 |
<0.001 | 26.004 | 5.762–7.352 |
<0.001 |
| Microvascular
invasion (−/+) | 65/73 | 1.89 | 1.127–3.171 | 0.016 | 0.688 | 0.216–2.194 | 0.527 |
| Edmondson grade
(I+II/III) | 127/71 | 2.783 | 1.735–4.464 |
<0.001 | 2.984 | 1.124–7.923 | 0.028 |
| Cirrhosis
(−/+) | 62/136 | 1.153 | 0.698–1.905 | 0.577 | 3.52 | 0.949–10.052 | 0.06 |
| AFP (<50
µg/l/≥50 µg/l) | 101/97 | 2.51 | 1.395–4.517 | 0.002 | 4.245 | 1.455–12.386 | 0.008 |
| CAR expressions
(−/+) | 88/110 | 0.667 | 0.345–1.289 | 0.228 | 0.677 | 0.206–2.225 | 0.520 |
Discussion
HCC is a common lethal cancer which leads to
large-scale mortality worldwide (22). At present, there are numerous
limitations to the treatment of HCC; hence, there is a necessity
for the development of innovative and efficient therapeutics
(23–25). It has been reported that low
expression of CAR was detected in colon, lung and bladder (8–11) tumors
and downregulated CAR expression predicted an inferior clinical
result for gastric and bladder cancer patients (9,14).
Conversely, CAR upregulation has been detected in endometrial,
ovarian, cervical, breast cancers, along with neuroblastomas and
medulloblastomas (15–20). Moreover, increased CAR expression was
correlated with inferior prognosis in breast and lung cancers
(12,21). However, in the present study, relative
to normal tissues, slightly decreased CAR expression was detected
in HCC samples and the Kaplan-Meier survival curve indicated that
CAR expression was not significantly associated to overall survival
in HCC patients. CAR immunopositivity in tumors was not dependent
upon sex, age, differentiation or metastasis in HCC patients, which
was in agreement with earlier observations by Stecker et al
(26). Also, positive expression was
observed in 56% of the samples from patients with hepatic
metastasis, which was the same as those devoid of metastasis.
Although, the relevant molecular mechanism is important, a
limitation of this study is that it lacks some functional
experiments to study the relevant mechanisms. In subsequent
studies, with the purpose of exploring the relevant molecular
mechanisms of CAR, CAR expression will be altered (downregulation
and upregulation), followed by the detection of the expression of
related genes through fluorogenic quantitative PCR and western
blotting.
Recently, cancer virotherapy, principally mediated
by oncolytic viruses (OVs), has gained popularity as a new and
potent strategy in the field of cancer therapy (27–29).
Consequently, the safety and efficacy of adenovirus vectors for
gene delivery should also be illustrated in the future. The first
OV therapy for cancer was approved in 2015 after more than a
century of extensive research. However, this is considered as a
modest victory when compared to the numerous small molecule
anticancer agents and antibody therapies that have gained approval
for clinical use over the previous thirty years. Although OVs are
markedly effective as anticancer agents with low toxicity in
vitro and in vivo, their efficacy as a single agent
therapy is limited. In this context, a combination of gene therapy
and therapeutically valuable OVs, such as oncolytic adenoviruses
(OAds), is one of the most potent therapeutic approaches.
Furthermore, OAds have been genetically improved to take advantage
of the altered tumor environment.
In the last twenty years, viral and non-viral
vector-mediated gene therapy has been established as a potential
therapeutic strategy for a range of cancers and other serious
diseases which would otherwise be deemed incurable with traditional
drugs (28). Previously, several
trials focusing on the combination of OAds with immunostimulatory-,
proapoptotic- or tumor suppressor genes were carried out and
revealed enhanced antitumor potency (28,30–33).
Clinically detected cancers may have escaped antitumor immune
mechanisms during their growth; hence, the probability of potent
immunotherapies for their cure is currently becoming a clinical
reality (34). Furthermore, a
circadian crosstalk exists among OAds, TRAIL and IL-12 in the
stimulation of antitumor immunity (35). Angiogenesis is vital for
tumorigenesis; accordingly, several studies have ascertained that
OAds can impair tumor-mediated angiogenesis (36). Apoptosis is a programmed cell death
process which eliminates cancer cells and other detrimental cells
to sustain homoeostasis; thus, it presents a potential target for
innovative cancer therapeutics. In this context, a combination
therapy of OAd-expressing TRAIL with an additional type of
immunostimulant cytokine (IL-24) was demonstrated to be related to
the activation of an apoptotic caspase cascade (primarily
caspases-3 and −8) in HCC (37).
A future direction of cancer research is to find a
new combination therapy with OAds to treat tumors. Conversely, CAR
is vital for ensuring virus uptake, gene transfer and acts as a
principal receptor for the coxsackie B virus and adenovirus. In
summary, CAR expression could serve as a biomarker for studying and
estimating the results of gene therapy, and increasing its
expression may enhance cellular sensitivity to adenovirus
infection. Therefore, there is a potential need to research new
therapies and to detect the expression of the CAR,
concurrently.
Acknowledgements
Not applicable.
Funding
The present article was supported by the National
Science Foundation of China (nos. 81602706 and 81570198), funds
from the Department of Science Technology of Zhejiang Province (no.
2017C33116), the Zhejiang Medical Technology Plan Project (no.
2016KYA018), and the State Administration of Traditional Chinese
Medicine of Zhejiang (no. 2017ZB006).
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
SW and XM conceived and designed the study. XY, SL,
HW and WC performed the experiments. XY wrote the paper. HW, WC, XM
and SW reviewed and edited the manuscript. All authors read and
approved the manuscript and agree to be accountable for all aspects
of the research in ensuring that the accuracy or integrity of any
part of the study are appropriately investigated and resolved.
Ethics approval and consent to
participate
The patient samples were collected from Zhejiang
Provincial People's Hospital and all patients provided written
informed consent. The study was approved by the Ethics Committee of
Zhejiang Provincial People's Hospital.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Altekruse SF, McGlynn KA, Dickie LA and
Kleiner DE: Hepatocellular carcinoma confirmation, treatment, and
survival in surveillance, epidemiology, and end results registries,
1992–2008. Hepatology. 55:476–482. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
US National Library of Medicine.
ClinicalTrials.gov. http://www.Clinicaltrials.gov/ct2/Show/NCT025627552015
|
|
4
|
Cohen CJ, Shieh JT, Pickles RJ, Okegawa T,
Hsieh JT and Bergelson JM: The coxsackievirus and adenovirus
receptor is a transmembrane component of the tight junction. Proc
Natl Acad Sci USA. 98:15191–15196. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Walters RW, Freimuth P, Moninger TO,
Ganske I, Zabner J and Welsh MJ: Adenovirus fiber disrupts
CAR-mediated intercellular adhesion allowing virus escape. Cell.
110:789–799. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pandha HS, Stockwin LH, Eaton J, Clarke
IA, Dalgleish AG, Todryk SM and Blair GE: Coxsackie B and
adenovirus receptor, integrin and major histocompatibility complex
class I expression in human prostate cancer cell lines:
Implications for gene therapy strategies. Prostate Cancer Prostatic
Dis. 6:6–11. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kasuya H, Takeda S, Shimoyama S, Shikano
T, Nomura N, Kanazumi N, Nomoto S, Sugimoto H and Nakao A:
Oncolytic virus therapy-foreword. Curr Cancer Drug Targets.
7:123–125. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Abdolazimi Y, Mojarrad M, Pedram M and
Modarressi MH: Analysis of the expression of coxsackievirus and
adenovirus receptor in five colon cancer cell lines. World J
Gastroenterol. 13:6365–6369. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Anders M, Vieth M, Rocken C, Röcken C,
Ebert M, Pross M, Gretschel S, Schlag PM, Wiedenmann B, Kemmner W
and Höcker M: Loss of the coxsackie and adenovirus receptor
contributes to gastric cancer progression. Br J Cancer.
100:352–359. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang Y, Thorne S, Hannock J, Francis J, Au
T, Reid T, Lemoine N, Kirn D and Halldén G: A novel assay to assess
primary human cancer infectibility by replication-selective
oncolytic adenoviruses. Clin Cancer Res. 11:351–360.
2005.PubMed/NCBI
|
|
11
|
Yamashita M, Ino A, Kawabata K, Sakurai F
and Mizuguchi H: Expression of coxsackie and adenovirus receptor
reduces the lung metastatic potential of murine tumor cells. Int J
Cancer. 121:1690–1696. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wunder T, Schmid K, Wicklein D, Groitl P,
Dobner T, Lange T, Anders M and Schumacher U: Expression of the
coxsackie adenovirus receptor in neuroendocrine lung cancers and
its implications for oncolytic adenoviral infection. Cancer Gene
Ther. 20:25–32. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wunder T, Schumacher U and Friedrich RE:
Coxsackie adenovirus receptor expression in carcinomas of the head
and neck. Anticancer Res. 32:1057–1062. 2012.PubMed/NCBI
|
|
14
|
Matsumoto K, Shariat SF, Ayala GE, Rauen
KA and Lerner SP: Loss of coxsackie and adenovirus receptor
expression is associated with features of aggressive bladder
cancer. Urology. 66:441–446. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dietel M, Häfner N, Jansen L, Dürst M and
Runnebaum IB: Novel splice variant CAR 4/6 of the coxsackie
adenovirus receptor is differentially expressed in cervical
carcinogenesis. J Mol Med (Berl). 89:621–630. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Giaginis CT, Zarros AC, Papaefthymiou MA,
Papadopouli AE, Sfiniadakis IK and Theocharis SE: Coxsackievirus
and adenovirus receptor expression in human endometrial
adenocarcinoma: Possible clinical implications. World J Surg Oncol.
6:592008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Martin TA, Watkins G and Jiang WG: The
Coxsackie-adenovirus receptor has elevated expression in human
breast cancer. Clin Exp Med. 5:122–128. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Martino TA, Petric M, Weingartl H,
Bergelson JM, Opavsky MA, Richardson CD, Modlin JF, Finberg RW,
Kain KC, Willis N, et al: The coxsackie-adenovirus receptor (CAR)
is used by reference strains and clinical isolates representing all
six serotypes of coxsackievirus group B and by swine vesicular
disease virus. Virology. 271:99–108. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Persson A, Fan X, Widegren B and Englund
E: Cell type- and region-dependent coxsackie adenovirus receptor
expression in the central nervous system. J Neurooncol. 78:1–6.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Reimer D, Steppan I, Wiedemair A, Concin
N, Hofstetter G, Marth C, Müller-Holzner E and Zeimet AG: Soluble
isoforms but not the transmembrane form of coxsackie-adenovirus
receptor are of clinical relevance in epithelial ovarian cancer.
Int J Cancer. 120:2568–2575. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang Y, Wang S, Bao Y, Ni C, Guan N, Zhao
J, Salford LG, Widegren B and Fan X: Coxsackievirus and adenovirus
receptor expression in non-malignant lung tissues and clinical lung
cancers. J Mol Histol. 37:153–160. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jiang G, Zhang L, Zhu Q, Bai D, Zhang C
and Wang X: CD146 promotes metastasis and predicts poor prognosis
of hepatocellular carcinoma. J Exp Clin Cancer Res. 35:382016.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Stotz M, Gerger A, Haybaeck J, Kiesslich
T, Bullock MD and Pichler M: Molecular targeted therapies in
hepatocellular carcinoma: Past, present and future. Anticancer Res.
35:5737–5744. 2015.PubMed/NCBI
|
|
24
|
Wada Y, Takami Y, Tateishi M, Ryu T,
Mikagi K and Saitsu H: The efficacy of continued sorafenib
treatment after radiologic confirmation of progressive disease in
patients with advanced hepatocellular carcinoma. PLoS One.
11:e01464562016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bhayani NH, Jiang Y, Hamed O, Kimchi ET,
Staveley-O'Carroll KF and Gusani NJ: Advances in the pharmacologic
treatment of hepatocellular carcinoma. Curr Clin Pharmacol.
10:299–304. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Stecker K, Vieth M, Koschel A, Wiedenmann
B, Röcken C and Anders M: Impact of the coxsackievirus and
adenovirus receptor on the adenoma-carcinoma sequence of colon
cancer. Br J Cancer. 104:1426–1433. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jebar AH, Errington-Mais F, Vile RG, Selby
PJ, Melcher AA and Griffin S: Progress in clinical oncolytic
virus-based therapy for hepatocellular carcinoma. J Gen Virol.
96:1533–1550. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang YG, Huang PP, Zhang R, Ma BY, Zhou XM
and Sun YF: Targeting adeno-associated virus and adenoviral gene
therapy for hepatocellular carcinoma. World J Gastroenterol.
22:326–337. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bartlett DL, Liu Z, Sathaiah M,
Ravindranathan R, Guo Z, He Y and Guo ZS: Oncolytic viruses as
therapeutic cancer vaccines. Mol Cancer. 12:1032013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Larson C, Oronsky B, Scicinski J, Fanger
GR, Stirn M, Oronsky A and Reid TR: Going viral: A review of
replication-selective oncolytic adenoviruses. Oncotarget.
6:19976–19989. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pesonen S, Kangasniemi L and Hemminki A:
Oncolytic adenoviruses for the treatment of human cancer: Focus on
translational and clinical data. Mol Pharm. 8:12–28. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Choi JW, Lee JS, Kim SW and Yun CO:
Evolution of oncolytic adenovirus for cancer treatment. Adv Drug
Deliv Rev. 64:720–729. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wold WS and Toth K: Adenovirus vectors for
gene therapy, vaccination and cancer gene therapy. Curr Gene Ther.
13:421–433. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ma Y, Shurin GV, Peiyuan Z and Shurin MR:
Dendritic cells in the cancer microenvironment. J Cancer. 4:36–44.
2013. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gomez-Gutierrez JG, Nitz J, Sharma R,
Wechman SL, Riedinger E, Martinez-Jaramillo E, Sam Zhou H and
McMasters KM: Combined therapy of oncolytic adenovirus and
temozolomide enhances lung cancer virotherapy in vitro and in vivo.
Virology. 487:249–259. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Saito Y, Sunamura M, Motoi F, Abe H, Egawa
S, Duda DG, Hoshida T, Fukuyama S, Hamada H and Matsuno S:
Oncolytic replication-competent adenovirus suppresses tumor
angiogenesis through preserved E1A region. Cancer Gene Ther.
13:242–252. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Cai Y, Liu X, Huang W, Zhang K and Liu XY:
Synergistic antitumor effect of TRAIL and IL-24 with complete
eradication of hepatoma in the CTGVT-DG strategy. Acta Biochim
Biophys Sin (Shanghai). 44:535–543. 2012. View Article : Google Scholar : PubMed/NCBI
|