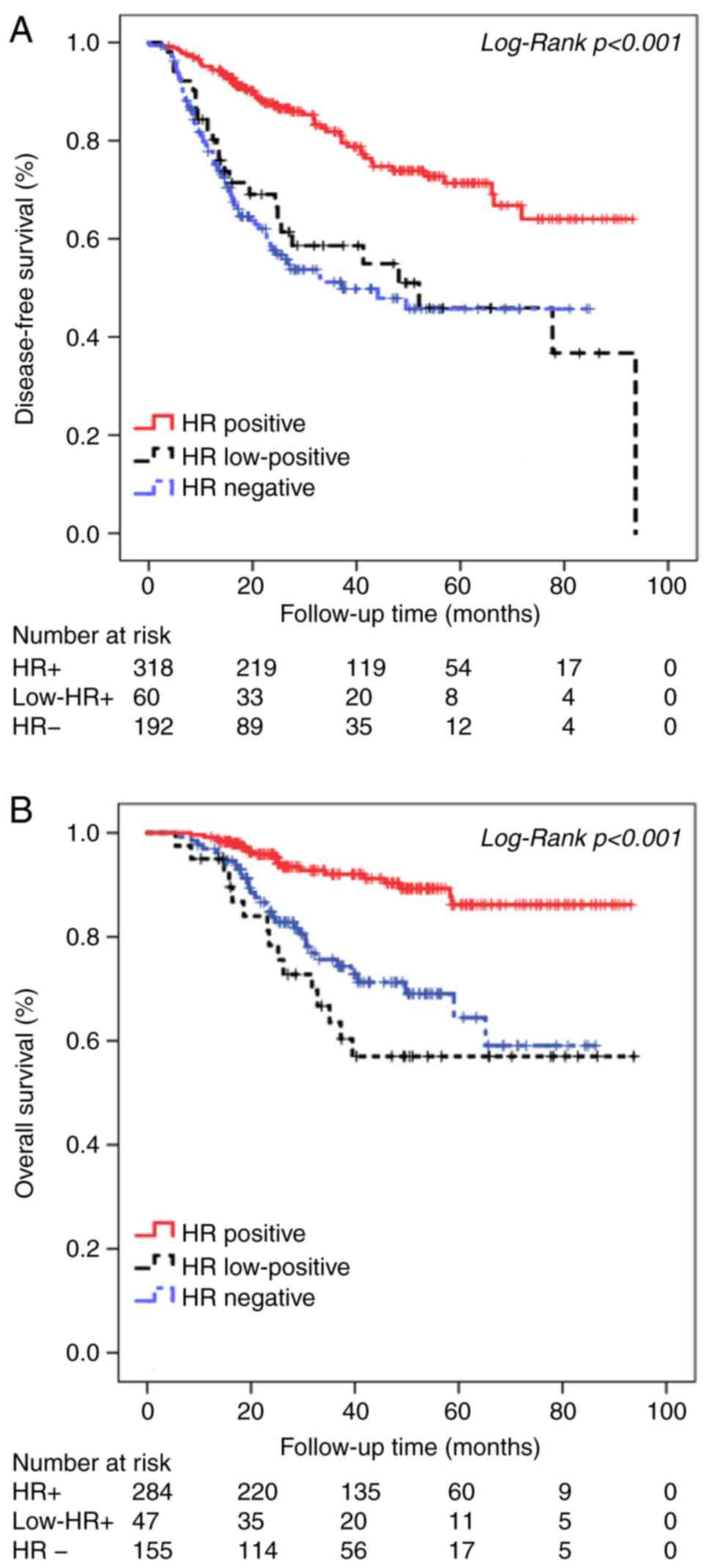
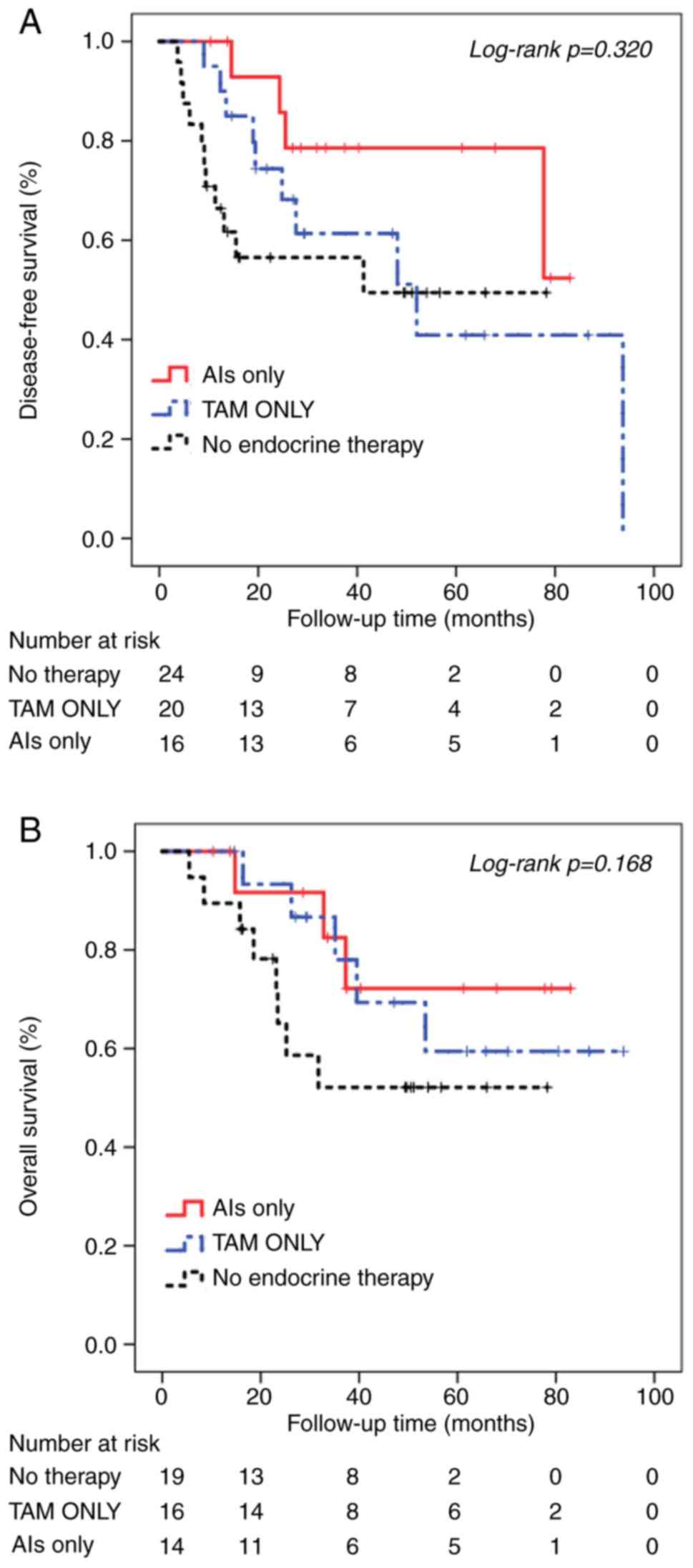
|
1
|
Early Breast Cancer Trialists'
Collaborative Group (EBCTCG), : Effects of chemotherapy and
hormonal therapy for early breast cancer on recurrence and 15-year
survival: An overview of the randomised trials. Lancet.
365:1687–1717. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Waks AG and Winer EP: Breast cancer
treatment: A review. JAMA. 321:288–300. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Goldhirsch A, Glick JH, Gelber RD, Coates
AS, Thürlimann B and Senn HJ; Panel members, : Meeting highlights:
International expert consensus on the primary therapy of early
breast cancer 2005. Ann Oncol. 16:1569–1583. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hammond ME, Hayes DF, Dowsett M, Allred
DC, Hagerty KL, Badve S, Fitzgibbons PL, Francis G, Goldstein NS,
Hayes M, et al: American Society of Clinical Oncology/College Of
American Pathologists guideline recommendations for
immunohistochemical testing of estrogen and progesterone receptors
in breast cancer. J Clin Oncol. 28:2784–2795. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gloyeske NC, Dabbs DJ and Bhargava R: Low
ER+ breast cancer: Is this a distinct group? Am J Clin Pathol.
141:697–701. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Raghav KP, Hernandez-Aya LF, Lei X,
Chavez-Macgregor M, Meric-Bernstam F, Buchholz TA, Sahin A, Do KA,
Hortobagyi GN and Gonzalez-Angulo AM: Impact of low
estrogen/progesterone receptor expression on survival outcomes in
breast cancers previously classified as triple negative breast
cancers. Cancer. 118:1498–1506. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Masuda N, Lee SJ, Ohtani S, Im YH, Lee ES,
Yokota I, Kuroi K, Im SA, Park BW, Kim SB, et al: Adjuvant
capecitabine for breast cancer after preoperative chemotherapy. N
Engl J Med. 376:2147–2159. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Reddy SM, Barcenas CH, Sinha AK, Hsu L,
Moulder SL, Tripathy D, Hortobagyi GN and Valero V: Long-term
survival outcomes of triple-receptor negative breast cancer
survivors who are disease free at 5 years and relationship with low
hormone receptor positivity. Br J Cancer. 118:17–23. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Prabhu JS, Korlimarla A, Desai K,
Alexander A, Raghavan R, Anupama C, Dendukuri N, Manjunath S,
Correa M, Raman N, et al: A majority of low (1–10%) ER positive
breast cancers behave like hormone receptor negative tumors. J
Cancer. 5:156–165. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Landmann A, Farrugia DJ, Zhu L, Diego EJ,
Johnson RR, Soran A, Dabbs DJ, Clark BZ, Puhalla SL, Jankowitz RC,
et al: Low estrogen receptor (ER)-positive breast cancer and
neoadjuvant systemic chemotherapy: Is response similar to typical
ER-positive or ER-negative disease? Am J Clin Pathol. 150:34–42.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yi M, Huo L, Koenig KB, Mittendorf EA,
Meric-Bernstam F, Kuerer HM, Bedrosian I, Buzdar AU, 0Symmans WF,
Crow JR, et al: Which threshold for ER positivity? a retrospective
study based on 9639 patients. Ann Oncol. 25:1004–1011. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Amin MB, Edge S, Greene F, Byrd DR,
Brookland RK, Washington MK, Gershenwald JE, Compton CC, Hess KR,
Sullivan DC, et al: AJCC cancer staging manual. New York: 2017,
View Article : Google Scholar
|
|
13
|
von Minckwitz G, Raab G, Caputo A, Schütte
M, Hilfrich J, Blohmer JU, Gerber B, Costa SD, Merkle E, Eidtmann
H, et al: Doxorubicin with cyclophosphamide followed by docetaxel
every 21 days compared with doxorubicin and docetaxel every 14 days
as preoperative treatment in operable breast cancer: The GEPARDUO
study of the german breast group. J Clin Oncol. 23:2676–2685. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bines J, Earl H, Buzaid AC and Saad ED:
Anthracyclines and taxanes in the neo/adjuvant treatment of breast
cancer: Does the sequence matter? Ann Oncol. 25:1079–1085. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gradishar WJ, Anderson BO, Balassanian R,
Blair SL, Burstein HJ, Cyr A, Elias AD, Farrar WB, Forero A,
Giordano SH, et al: NCCN guidelines insights: Breast cancer,
Version 1.2017. J Natl Compr Canc Netw. 15:433–451. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Prat A, Cheang MC, Martin M, Parker JS,
Carrasco E, Caballero R, Tyldesley S, Gelmon K, Bernard PS, Nielsen
TO and Perou CM: Prognostic significance of progesterone
receptor-positive tumor cells within immunohistochemically defined
luminal A breast cancer. J Clin Oncol. 31:203–209. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bae SY, Kim S, Lee JH, Lee HC, Lee SK, Kil
WH, Kim SW, Lee JE and Nam SJ: Poor prognosis of single hormone
receptor- positive breast cancer: Similar outcome as
triple-negative breast cancer. BMC Cancer. 15:1382015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cancello G, Maisonneuve P, Rotmensz N,
Viale G, Mastropasqua MG, Pruneri G, Montagna E, Iorfida M, Mazza
M, Balduzzi A, et al: Progesterone receptor loss identifies Luminal
B breast cancer subgroups at higher risk of relapse. Ann Oncol.
24:661–668. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Braun L, Mietzsch F, Seibold P,
Schneeweiss A, Schirmacher P, Chang-Claude J, Peter Sinn H and
Aulmann S: Intrinsic breast cancer subtypes defined by estrogen
receptor signalling-prognostic relevance of progesterone receptor
loss. Mod Pathol. 26:1161–1171. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Guarneri V, Broglio K, Kau SW,
Cristofanilli M, Buzdar AU, Valero V, Buchholz T, Meric F,
Middleton L, Hortobagyi GN and Gonzalez-Angulo AM: Prognostic value
of pathologic complete response after primary chemotherapy in
relation to hormone receptor status and other factors. J Clin
Oncol. 24:1037–1044. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Carey LA, Dees EC, Sawyer L, Gatti L,
Moore DT, Collichio F, Ollila DW, Sartor CI, Graham ML and Perou
CM: The triple negative paradox: Primary tumor chemosensitivity of
breast cancer subtypes. Clin Cancer Res. 13:2329–2334. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cui X, Schiff R, Arpino G, Osborne CK and
Lee AV: Biology of progesterone receptor loss in breast cancer and
its implications for endocrine therapy. J Clin Oncol. 23:7721–7735.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Pan H, Gray R, Braybrooke J, Davies C,
Taylor C, McGale P, Peto R, Pritchard KI, Bergh J, Dowsett M, et
al: 20-Year risks of breast-cancer recurrence after stopping
endocrine therapy at 5 Years. N Engl J Med. 377:1836–1846. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Breast International Group (BIG) 1–98
Collaborative Group and, ; Thurlimann B, Keshaviah A, Coates AS,
Mouridsen H, Mauriac L, Forbes JF, Paridaens R, Castiglione-Gertsch
M, Gelber RD, et al: A comparison of letrozole and tamoxifen in
postmenopausal women with early breast cancer. N Engl J Med.
353:2747–2757. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cuzick J, Sestak I, Baum M, Buzdar A,
Howell A, Dowsett M and Forbes JF; ATAC/LATTE investigators, :
Effect of anastrozole and tamoxifen as adjuvant treatment for
early-stage breast cancer: 10-year analysis of the ATAC trial.
Lancet Oncol. 11:1135–1141. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bevers TB, Helvie M, Bonaccio E, Calhoun
KE, Daly MB, Farrar WB, Garber JE, Gray R, Greenberg CC, Greenup R,
et al: Breast cancer screening and diagnosis, Version 3.2018, NCCN
clinical practice guidelines in oncology. J Natl Compr Canc Netw.
16:1362–1389. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Paik S, Shak S, Tang G, Kim C, Baker J,
Cronin M, Baehner FL, Walker MG, Watson D, Park T, et al: A
multigene assay to predict recurrence of tamoxifen-treated,
node-negative breast cancer. N Engl J Med. 351:2817–2826. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
van't Veer LJ, Dai H, van de Vijver MJ, He
YD, Hart AA, Mao M, Peterse HL, van der Kooy K, Marton MJ,
Witteveen AT, et al: Gene expression profiling predicts clinical
outcome of breast cancer. Nature. 415:530–536. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sestak I, Dowsett M, Zabaglo L,
Lopez-Knowles E, Ferree S, Cowens JW and Cuzick J: Factors
predicting late recurrence for estrogen receptor-positive breast
cancer. J Natl Cancer Inst. 105:1504–1511. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Filipits M, Rudas M, Jakesz R, Dubsky P,
Fitzal F, Singer CF, Dietze O, Greil R, Jelen A, Sevelda P, et al:
A new molecular predictor of distant recurrence in ER-positive,
HER2-negative breast cancer adds independent information to
conventional clinical risk factors. Clin Cancer Res. 17:6012–6020.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Buus R, Sestak I, Kronenwett R, Denkert C,
Dubsky P, Krappmann K, Scheer M, Petry C, Cuzick J and Dowsett M:
Comparison of endopredict and EPclin with oncotype DX recurrence
score for prediction of risk of distant recurrence after endocrine
therapy. J Natl Cancer Inst. 108:2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jerevall PL, Ma XJ, Li H, Salunga R, Kesty
NC, Erlander MG, Sgroi DC, Holmlund B, Skoog L, Fornander T, et al:
Prognostic utility of HOXB13:IL17BR and molecular grade index in
early-stage breast cancer patients from the Stockholm trial. Br J
Cancer. 104:1762–1769. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dowsett M, Sestak I, Regan MM, Dodson A,
Viale G, Thürlimann B, Colleoni M and Cuzick J: Integration of
clinical variables for the prediction of late distant recurrence in
patients with estrogen receptor-positive breast cancer treated with
5 years of endocrine therapy: CTS5. J Clin Oncol. 36:1941–1948.
2018. View Article : Google Scholar : PubMed/NCBI
|