Introduction
Patients with bone metastases commonly present with
spinal metastases, and 60–70% of patients with advanced cancer
develop spinal metastases during the progression of the disease
(1,2). Spinal metastases are asymptomatic when
their size and number are small; however, they gradually progress
and frequently lead to skeletal-related events, including radiation
therapy (RT) due to painful vertebral metastases, vertebral body
fracture and malignant spinal cord compression (MSCC) (1). In patients without a neurological
deficit, the pain from vertebral bone metastases can be controlled
by RT in most cases (3). Our
previous study reported that disappearance of pain and
re-ossification were achieved at 3 months following RT in patients
with painful vertebral bone metastases without paralysis (4). In addition, no patients presented with
pain that was difficult to control with conventional radiotherapy
or required surgery. Therefore, it was concluded that early
diagnosis and treatment of bone metastases can lead to improved
results (3,4).
MSCC is one of the most serious complications of
cancer, and 2.5–5% of patients with advanced cancer develop MSCC
(1,2). MSCC can occur with all types of cancer;
however, MSCC is most frequently observed in patients with breast,
prostate or lung cancer (5,6).
In MSCC, the spinal cord is damaged by compression
or vascular compromise due to tumor growth, which leads to
neurological dysfunction (5,6). MSCC is an oncological emergency since
it causes motor weakness, sensory deficits, walk disturbance, and
urinary and bowel dysfunction that markedly impair the quality of
life (7–9).
The importance of local control of bone metastases
is increasing since patients with advanced cancer live longer due
to recent advances in systemic therapies. Therefore, early
diagnosis and treatment of MSCC, and a prophylactic approach for
MSCC are crucial. However, several days may pass between the
diagnosis and treatment of the neurological deficit (‘Doctor's
Delay’), and the severity of the neurological deficit is often
advanced at the time of referral to orthopedists and it is
frequently difficult to improve the neurological deficit (8,9).
In most cases, back pain in the metastatic vertebral
bone is the primary symptom preceding neurological dysfunction
(5,10). Therefore, guidelines for bone
metastases recommend that clinicians watch for early suspicious
signs of MSCC and make a prompt diagnosis by spine magnetic
resonance imaging (MRI) (5,10). According to the MSCC guidelines of
the National Institute for Health and Clinical Excellence in the
United Kingdom, spine MRI is recommended as soon as possible when
symptoms of MSCC, including back pain and neurological deficit,
appear in patients with a history of cancer (5).
Doctors should communicate and share information
regarding bone metastases in order to facilitate their treatment,
since bone metastases occur in various types of cancer. It is
crucial to establish a multidisciplinary treatment system for the
management of bone metastases in hospitals (11–18).
Recently, numerous institutions in Japan have organized cancer
boards, including doctors, nurses and other paramedical staff, in
order to focus on the management of bone metastases (19). However, the actual benefit of these
cancer boards has not been clearly identified.
To the best of our knowledge, no study has reported
a decreased incidence of neurological deficits due to spinal
metastases when a multidisciplinary treatment system was
implemented. Therefore, the present study investigated the change
in the number of patients with neurological deficits before and
after the establishment of a multidisciplinary treatment system for
bone metastases. Furthermore, the differences in the time intervals
to MRI and RT before and after the establishment of the
multidisciplinary treatment system were examined.
Materials and methods
Patients and methods. The file records of 45
patients (24 male and 21 female; age, 30–82 years) who were
followed-up in Shikoku Cancer Center (Matsuyama, Japan) for
advanced cancer, presented with bone metastases and developed
neurological deficits due to MSCC before (between April 2007 and
March 2012; group A) and after (between April 2012 and March 2017;
group B) the establishment of the multidisciplinary treatment
system at the Shikoku Cancer Center were retrospectively evaluated.
Patients were excluded if they had paralysis at their first
presentation or the origin of the neurological deficit was the
brain.
In 2012, a multidisciplinary treatment system for
bone metastases was established at the Shikoku Cancer Center to
prevent neurological deficits caused by vertebral bone metastases.
Patients with bone metastases were identified by radiologists via
regular computed tomography (CT). Contrast-enhanced CT was
performed routinely.
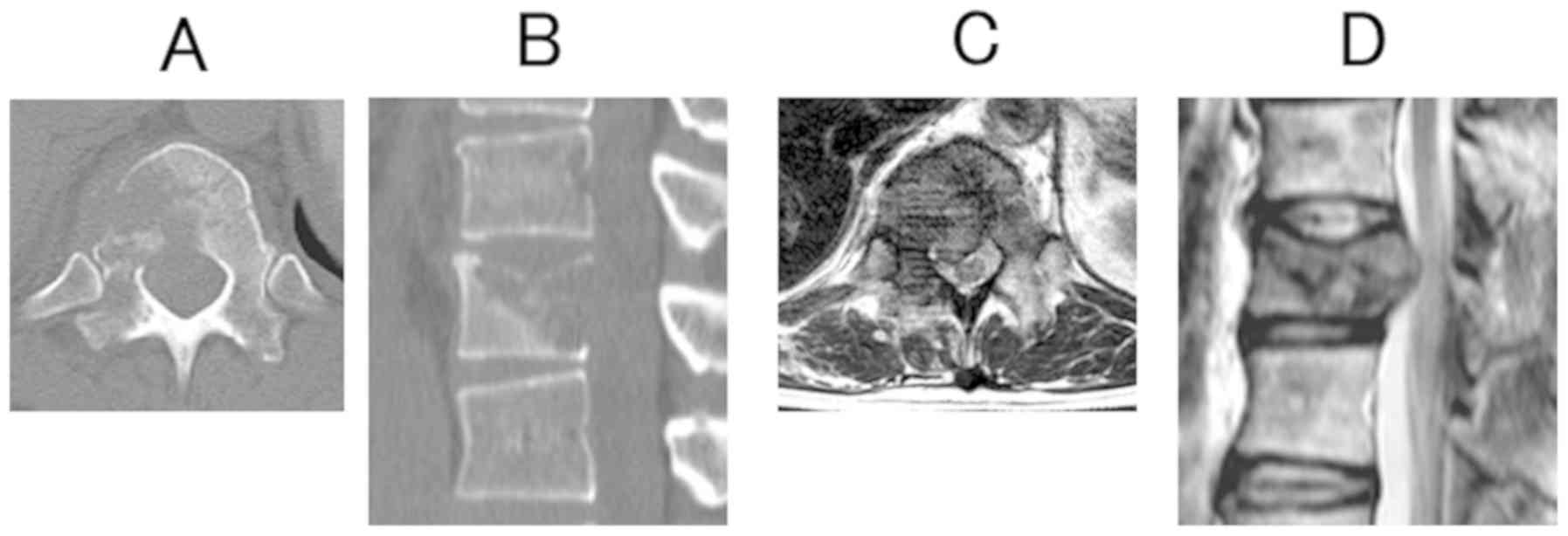
The radiologists paid attention to destruction of
the vertebral column cortex since this indicates the existence of
MSCC (Fig. 1A-D). When radiologists
confirmed MSCC or severe vertebral body destruction (destruction of
>1/3 of a vertebral body) during image interpretation, they
described these findings in their reports and wrote comments
recommending referral of the patients to orthopedists for a
consultation or to receive RT. Physicians could refer patients to
orthopedists or radiotherapists after reading these comments.
The orthopedists met with the patients following
referral by physicians. They evaluated the severity of the
neurological deficit (motor function) according to the Frankel
classification (20). Furthermore,
they determined whether the origin of the neurological deficit was
the brain, the spinal cord or other peripheral nerves. They also
evaluated the spinal instability in order to decide whether
conservative treatment (with or without orthosis) or surgery was
required (3).
Additionally, physicians were advised to consider
the existence of back pain in patients with vertebral bone
metastases due to the associated-risk of developing paralysis. A
rapid referral system was established to provide urgent access to
MRI scanning, referral to orthopedists and administration of RT for
the urgent management of neurological deficits caused by MSCC.
It was difficult to determine the precise number of
patients with bone metastases. Therefore, the rate of patients with
neurological deficits among those who received RT for vertebral
metastases was investigated. The numbers of patients who received
RT before and after the establishment of the system were 286 and
206, respectively (Table SI). The
primary tumor sites of patients before the establishment of the
system were lung (94 patients), breast (67), prostate (27), stomach
(17) colorectum (12) and other (69). The primary tumor sites
of patients after the establishment of the system were lung (65
patients), breast (46), prostate (34), colorectum (13), stomach (8) and other (40). The severity of
neurological deficits at the time of their recognition by
physicians and at the time of treatment in groups A and B were
evaluated using the Frankel classification (20).
The time intervals between the recognition of
neurological deficits by physicians and MRI scanning and the start
of treatment were also investigated. Furthermore, the rate of
referral to orthopedists for neurological deficits before treatment
and the use of bone-modifying agents (BMAs), such as denosumab or
zoledronic acid were evaluated in groups A and B.
The present study was approved by the Ethical Review
Board of Shikoku Cancer Center (Matsuyama, Japan) and conducted in
accordance with the World Medical Association Declaration of
Helsinki.
Statistical analysis
Categorical variables were analyzed using
χ2 and Fisher's exact tests. Continuous variables were
analyzed using a Mann-Whitney test. P<0.05 was considered to
indicate a statistically significant difference. The Kaplan-Meier
method was used to estimate the overall survival rate in the two
groups and the log-rank test was used to compare differences in the
overall survival. All statistical analyses were performed with the
statistical computing software R (R version 3.5.0; R Core
Team).
Results
MSCC prior to the establishment of the
multidisciplinary treatment system. MSCC occurred in 38 patients in
group A (Tables I and SII). The number of patients with
neurological deficits in each of the 5 years was 8, 7, 6, 10 and 7,
respectively. The primary tumor sites were breast (12 patients),
lung (8 patients), prostate (8 patients), colorectum (4 patients),
stomach (4 patients), bile duct (1 patient) and multiple myeloma (1
patient). The affected locations in the spine were the cervical
spine (1 patient), thoracic spine (30 patients) and lumbar spine (7
patients). The severity of the neurological deficit was Frankel A,
B, C and D for 1, 1, 13 and 23 patients, respectively (Table SII). Furthermore, one patient
underwent surgery following RT, whereas the other patients received
RT only.
 | Table I.Characteristics of patients with bone
metastases included in this study. |
Table I.
Characteristics of patients with bone
metastases included in this study.
| Characteristic | Group A, n
(n=38) | Group B, n (n=7) |
|---|
| Primary cancer
site |
|
Breast | 12 | 3 |
| Lung | 8 | 4 |
|
Prostate | 8 | 0 |
|
Colorectal | 4 | 0 |
|
Stomach | 4 | 0 |
|
Others | 2 | 0 |
| Radiation site |
| Cervical
spine | 1 | 1 |
| Thoracic
spine | 30 | 6 |
| Lumbar
spine | 7 | 0 |
| Severity of
neurological deficit (Frankel classification) |
|
|
| A | 1 | 0 |
| B | 1 | 0 |
| C | 13 | 3 |
| D | 23 | 4 |
MSCC after the establishment of the
multidisciplinary treatment system
MSCC occurred in 7 patients in group B (Tables I and SII). The number of patients with
neurological deficits in each of the five years was 0, 2, 1, 2 and
2, respectively. The primary tumor sites were lung (4 patients) and
breast (3 patients). The affected locations in the spine were the
cervical spine (1 patient) and thoracic spine (6 patients). The
severity of neurological deficit was Frankel C and D for 3 and 4
patients, respectively (Table SII).
All patients from group B received RT.
Comparison between the number and
severity of neurological deficits before and after the
establishment of the multidisciplinary treatment system
Among the patients who received RT, the number of
patients with neurological deficits was 38/286 (13.2%) and 7/206
(3.4%) before and after the system was introduced, respectively
(P<0.01). In patients with breast cancer, the rate of
neurological deficits was higher in group A (12/67; 18%) compared
with in group B (3/46; 6.5%); however, the difference was not
identified to be significant (P=0.08). In patients with prostate
cancer, the rate of neurological deficits was significantly higher
in group A (8/27; 30%) compared with group B (0/34; 0%; P<0.01).
In patients with cancer of the digestive organs, the rate of
neurological deficits was significantly higher in group A (9/63;
14%) compared with group B (0/34; 0%; P=0.02). In patients with
lung cancer, the rate of neurological deficits was higher in group
A (8/97; 8.2%) compared with group B (4/65; 6.2%), although the
difference was not identified to be significant (P=0.62) (Table SI).
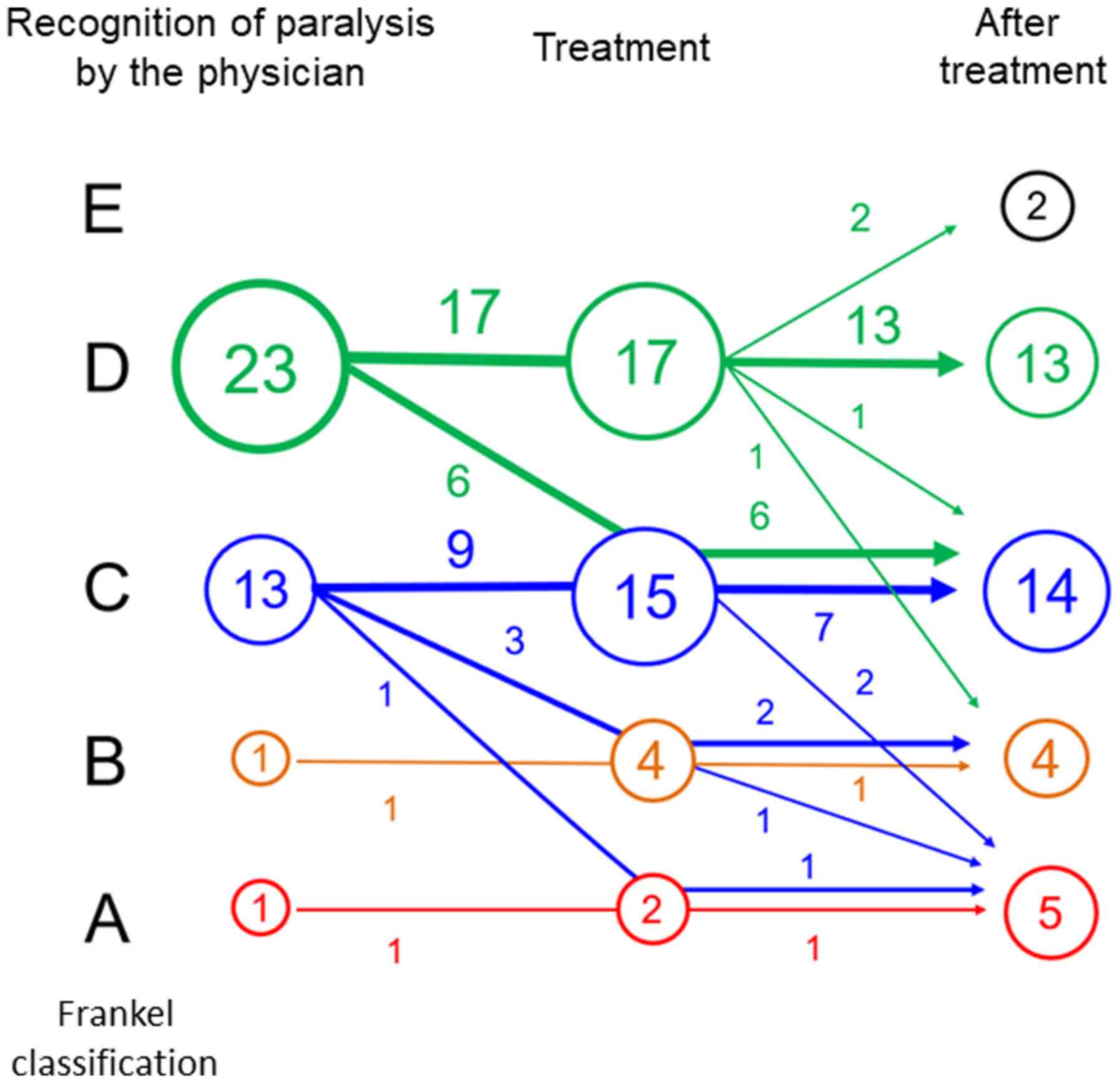
In group A, the severity of neurological deficits
was Frankel A, B, C and D for 1, 1, 13 and 23 patients,
respectively, at the time of their recognition by physicians
(Fig. 2; Table SII). In 10/38 patients (26%), the
severity of neurological deficit was worse at the time of
treatment. Following treatment, improvement in the severity of
neurological deficit was achieved for 2 patients (5.3%) with
Frankel D, but not in patients with Frankel A, B or C. In patients
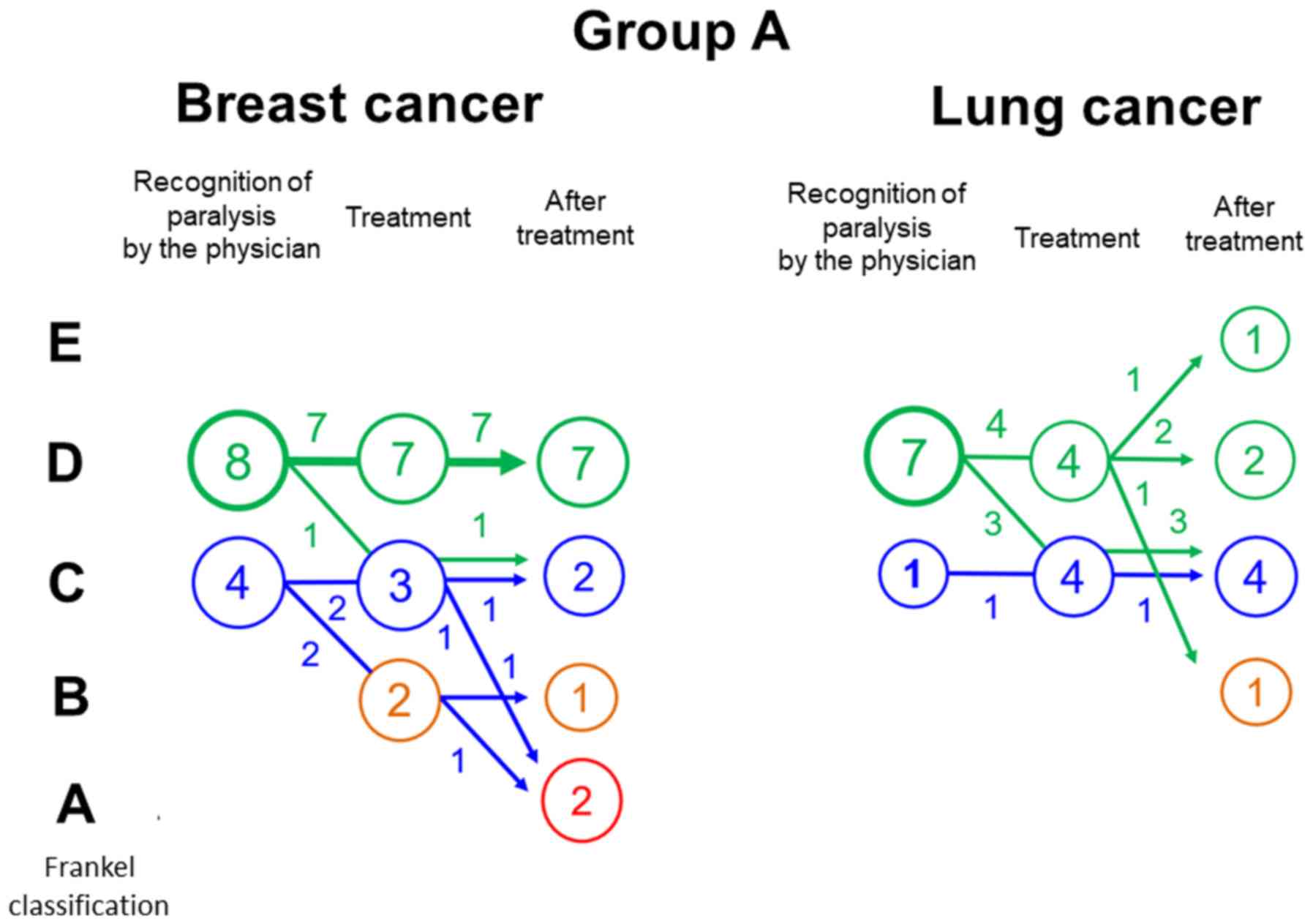
with breast cancer, the severity of neurological deficit was
Frankel C and D for 4 and 8 patients, respectively, at the time of
recognition by physicians. In 3/12 patients (25%), the severity of
neurological deficit was worse at the time of treatment. After
treatment, no patient attained improvement in the severity of
neurological deficit (Fig. 3). In
patients with lung cancer, the severity of neurological deficit was
Frankel C and D in 1 and 7 patients, respectively, at the time of
recognition by physicians. In 3/8 patients (38%), the severity of
neurological deficit was worse at the time of treatment.
Improvement in the severity of neurological deficit after treatment
was attained in 1 patient (12.5%) with Frankel D (Fig. 3).
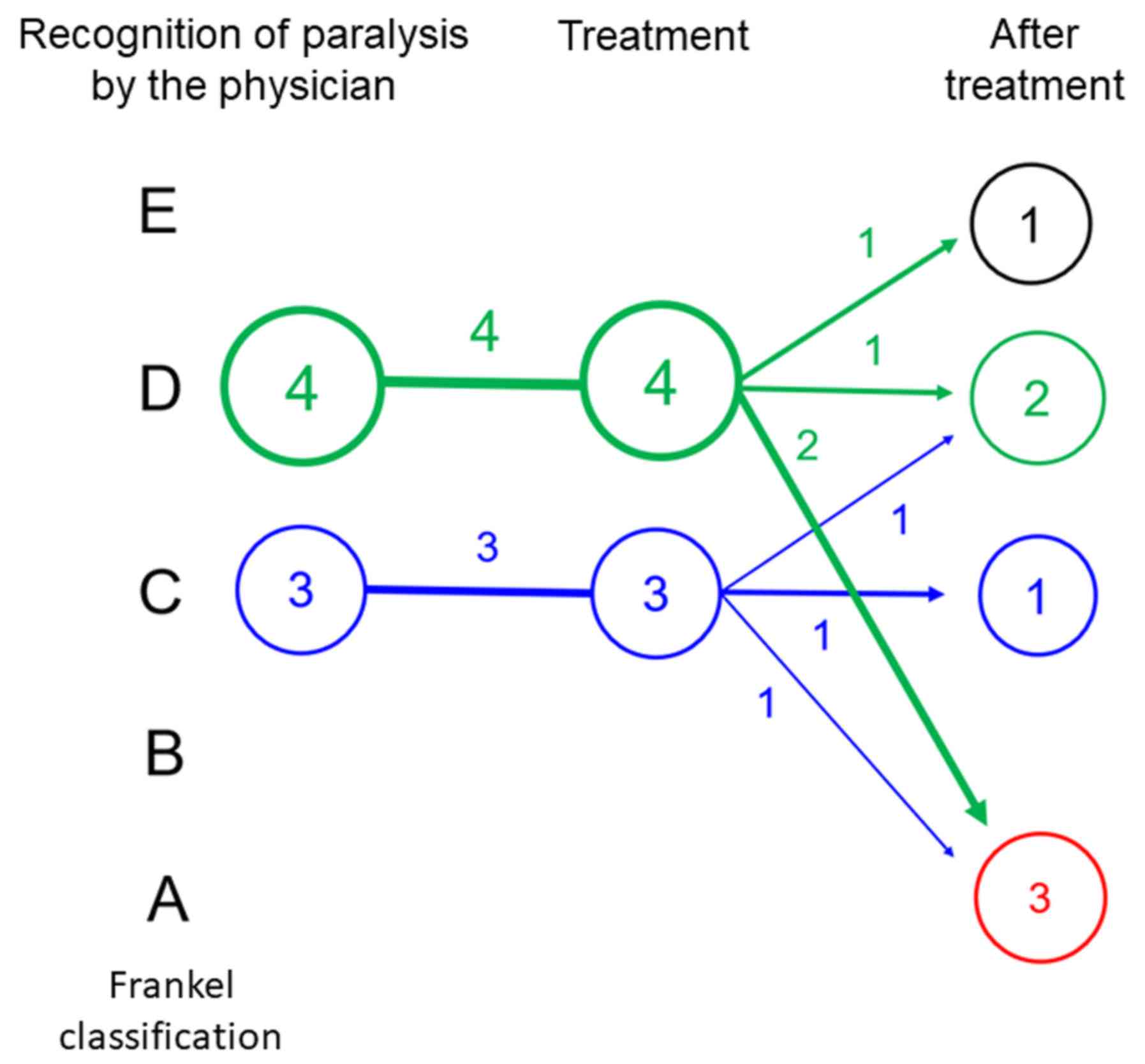
In group B, the severity of neurological deficit was
Frankel C and D in 3 and 4 patients, respectively, at the time of
recognition by physicians. No severe neurological deficits (Frankel
A and B) and no deterioration of neurological deficits were
observed at the time of treatment. After treatment, improvement in
the severity of neurological deficit was achieved by 2 patients
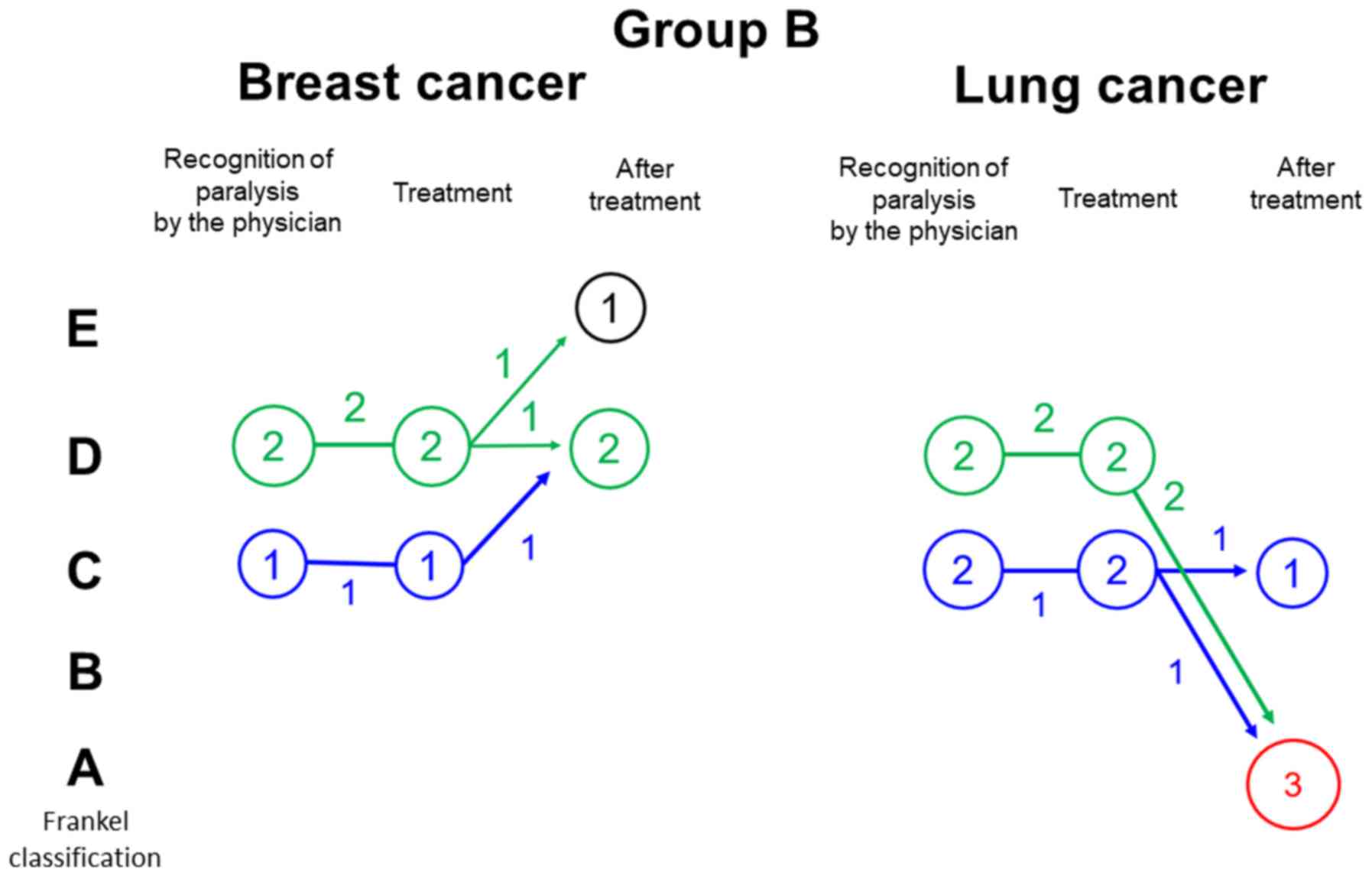
(28.6%; Fig. 4). In patients with
breast cancer, the severity of neurological deficit was Frankel C
and D in 1 and 2 patients, respectively, at the time of recognition
by physicians. Improvement of neurological deficits was attained in
2 patients after treatment (66.7%; Fig.
5). In patients with lung cancer, the severity of neurological
deficit was Frankel C and D in 2 patients each at the time of
recognition by physicians. No patient achieved improvement in the
severity of neurological deficits after treatment (Fig. 5).
The rate of patients with improvement in the
severity of neurological deficit after treatment was significantly
increased in all patients (group A, 5.3%; group B, 28.5%;
P<0.001) and in patients with breast cancer (group A, 0%; group
B, 66.7%; P<0.01), but not in patients with lung cancer (group
A, 12.5%; group B, 0%; P=0.46) following the establishment of the
system.
MRI scanning
The interval between the recognition of neurological
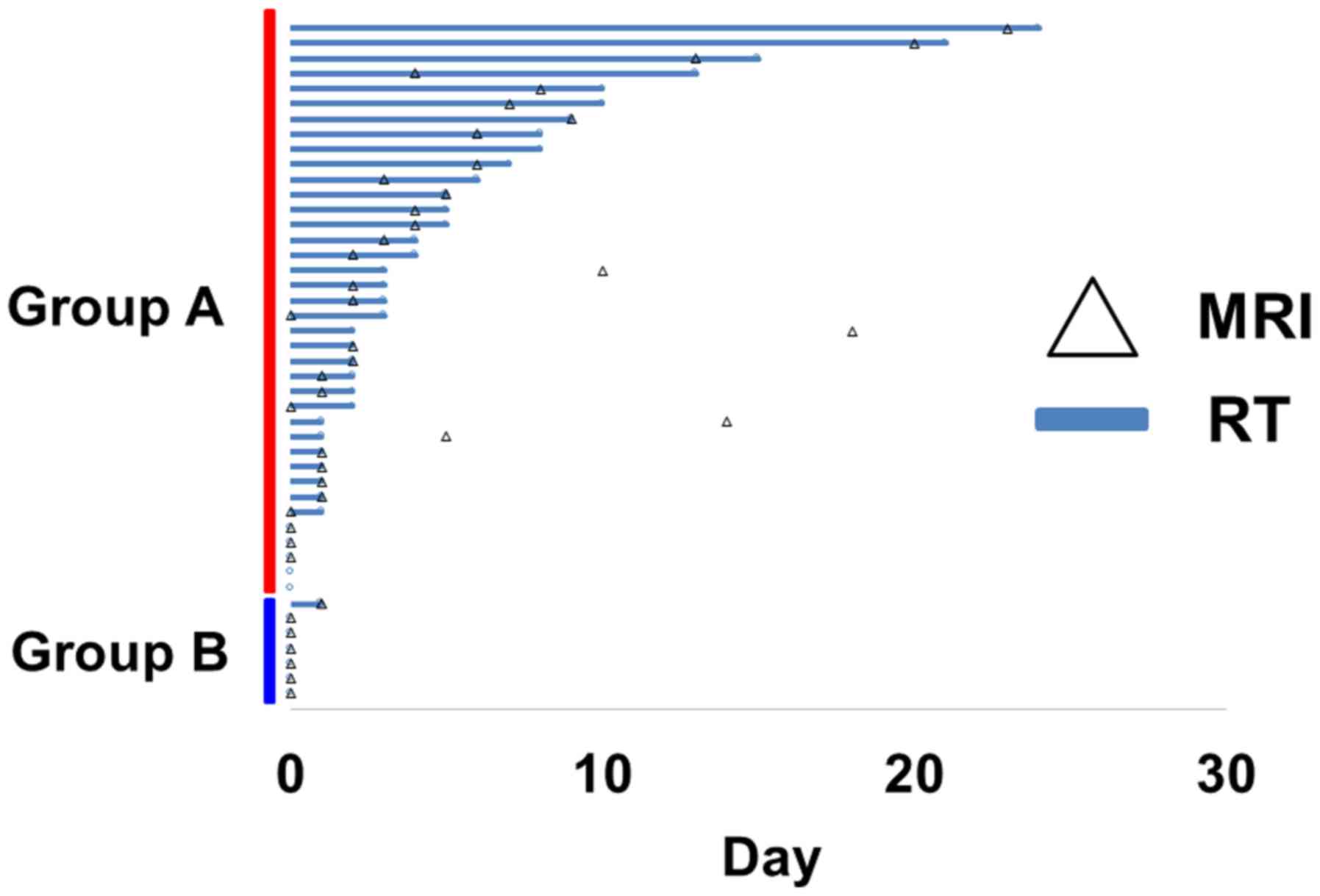
deficits by physicians and MRI scanning was 0–23 days (median, 3
days) in group A (Fig. 6, Table SII). MRI was not performed in 2
patients. The interval between the recognition of neurological
deficits by physicians and MRI scanning was 0 days in 7 patients,
1–3 days in 13 patients, 4–7 days in 8 patients and >7 days in 8
patients. The interval between the recognition of neurological
deficits by physicians and MRI scanning was 0–1 days (median, 0
days) in group B. MRI was performed on the same day as the
recognition of neurological deficits by physicians in 5 patients
(71%) in group B. The interval between the recognition of
neurological deficits by physicians and MRI scanning was
significantly decreased after the establishment of the system
(P<0.01; Table SI).
Treatment initiation
The interval between the recognition of neurological
deficits by physicians and the start of treatment was 0–24 days
(median, 3 days) in group A (Table
SII). The interval between the recognition of neurological
deficits by physicians and the start of treatment was 0 days in 5
patients, 1–3 days in 17 patients, 4–7 days in 7 patients, and
>7 days in 9 patients (Table
SII). The interval between the recognition of neurological
deficits by physicians and the start of treatment was 0–1 days
(median, 0 days) in group B (Table
SII). Treatment was provided on the same day in all but 2
patients in group B. The interval between the recognition of
neurological deficits by physicians and treatment was significantly
decreased after the establishment of the system (P<0.01)
(Table SII).
Referral to orthopedists
The rates of referral to orthopedists before
treatment in groups A and B were 17/38 (45%) and 7/7 (100%),
respectively (P<0.01) (Table
SII). The use of BMAs before the recognition of neurological
deficits by physicians was 15/38 (39%) and 7/7 (100%) in groups A
and B, respectively (P<0.01) (Table
SII). BMAs were administered at the time of diagnosis of bone
metastases to all patients in group B.
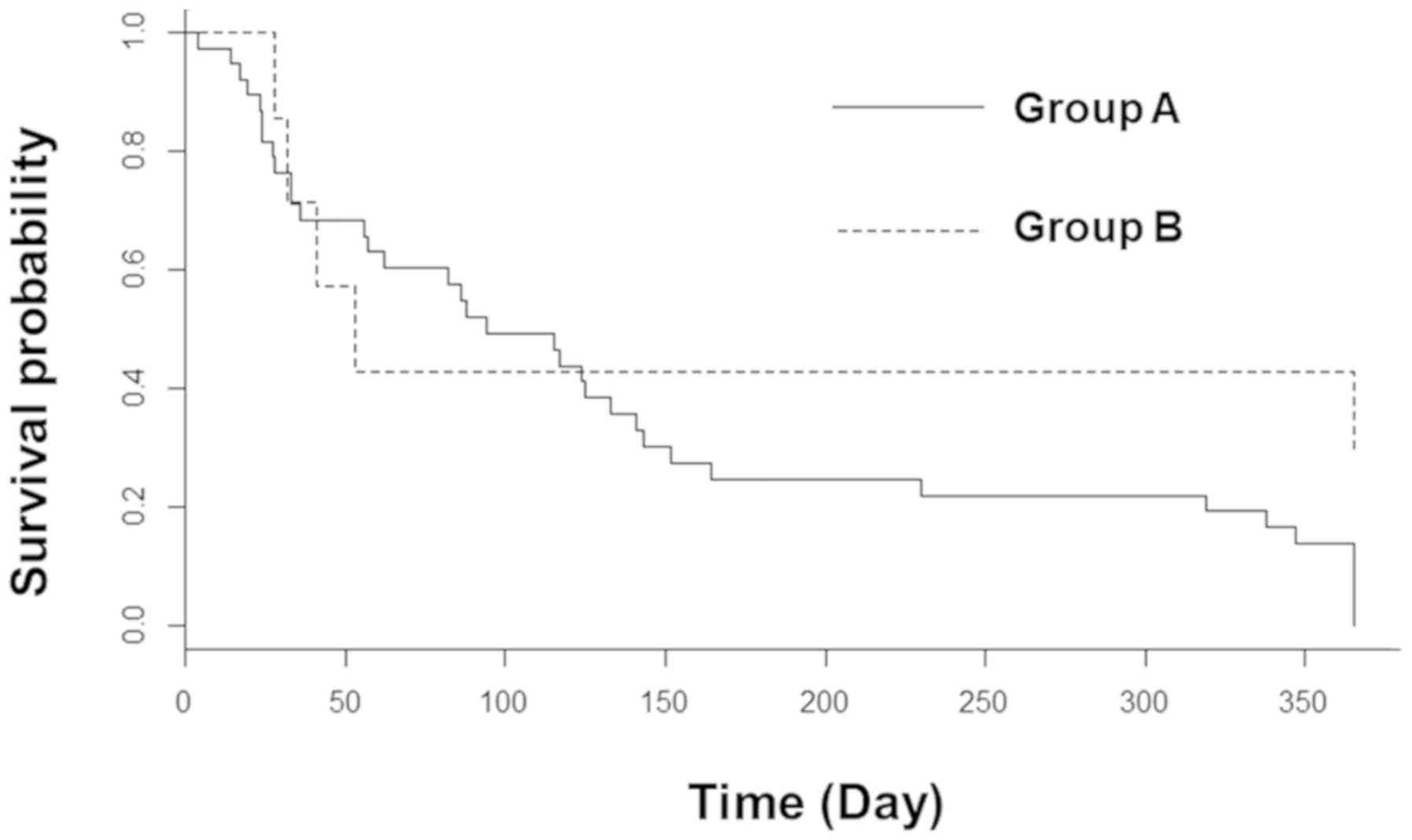
Overall survival rates
At the last follow-up, 37/38 patients and 5/7
patients had died in groups A and B, respectively. The overall
survival rates were 24 and 43% in groups A and B, respectively, at
6 months following RT with no significant difference identified
(P=0.36; Fig. 7).
Discussion
The present study reported a significant improvement
in the rate of neurological deficit cases when using a
multidisciplinary treatment system for patients with bone
metastases. Furthermore, the rate of patients with improvement in
the severity of neurological deficit after treatment was improved
after the establishment of the system.
An innovative treatment approach, including
intensity-modulated RT, vertebroplasty and minimally invasive
surgery, has been developed (21).
However, improvement of the neurological deficit is difficult once
it occurs. Therefore, early diagnosis, treatment and prevention of
MSCC are crucial in order to minimize treatment delay. In numerous
cases, a neurological deficit does not occur suddenly, and back
pain appears as a prodromal symptom for several weeks prior.
Therefore, several guidelines for bone metastases recommend that
clinicians watch for this early sign of MSCC and provide an
emergency diagnosis by using spine MRI (5,10).
Furthermore, the occurrence of MSCC should be considered when back
pain appears in a patient with spinal metastases (so-called ‘Red
Flag’), and emergency imaging, including CT or MRI, should be
performed. When imaging cannot be performed immediately, the most
recent imaging should be reviewed, and the presence of vertebral
body collapse or asymptomatic MSCC should be confirmed. If
treatment is initiated before a neurological deficit fully
develops, it can be prevented in certain patients (preventable
paralysis).
In the present study, the incidence of paralysis
decreased following the establishment of the system. The rate of
patients with neurological deficit and the rate of patients with
improvement in the severity of neurological deficit after treatment
were significantly improved. In addition, among patients who
received RT, the rate of patients with neurological deficit was
38/286 (13.2%) and 7/206 (3.4%) before and after the establishment
of the system, respectively, and this difference was identified to
be significant. Although the rate of patients with neurological
deficit was lower in all types of cancer, there was no significant
difference observed between patients with breast and lung
cancer.
Numerous patients presented with severe levels of
neurological deficit (Frankel A and B), and the severity of
neurological deficit was worse at the time of treatment in 26% of
patients before the establishment of the system. However, no severe
level of neurological deficit and no deterioration of the
neurological deficit were seen at the time of treatment after the
establishment of the system.
The rate of patients with improvement in the
severity of neurological deficit was 5.3 and 28.5% before and after
the establishment of the system, respectively, which was
significantly different. Urgent treatment with the system could
lead to the prevention of the deterioration of the neurological
deficit at the time of treatment and improvement of the
neurological deficit.
The rate of patients with improvement in the
severity of neurological deficit after treatment was significantly
improved in patients with breast cancer, but not in patients with
lung cancer, after the establishment of the system. This can be
explained by the difference in the sensitivity to RT between
patients with breast and lung cancer. Since breast cancer is more
sensitive to RT (4), early treatment
may lead to an improvement in the severity of neurological
deficits.
It has been hypothesized that the increased
awareness of back pain as a sign of MSCC and subsequent earlier
referral to orthopedists could lead to the early diagnosis and
treatment of MSCC. Additionally, the system depends on the capacity
of radiologists to identify patients with vertebral body collapse
who may require a potential prophylactic approach to prevent
neurological deficit.
Previous studies have demonstrated that specific
systems developed for earlier diagnosis and treatment can decrease
treatment delays, which can lead to improved neurological outcomes
of patients (11,18). Savage et al (11) reported the establishment of a rapid
referral system designed to provide urgent access to MRI scanning
and urgent referral to oncology specialists for the management of
MSCC. Key steps in the management of patients with suspicion of
MSCC would include the capacity of physicians to directly connect
with the oncology specialist in a tertiary center in order to
provide access to urgent MRI scanning for patients. Furthermore,
physicians can provide an alert card to the patients with high risk
of MSCC in order to encourage self-referral to the hospital when
back pain, which is a potential symptom of MSCC, occurs. By using
this approach, it has been reported that the rate of patients with
major impairment decreased from 80 to 62% (11). Allan et al (18) reported a similar system for improving
the time to diagnosis via a hotline by which physicians can
directly connect with the oncology specialist in a tertiary center
for patients with suspicion of MSCC and in urgent need for MRI
scanning. Using this approach, it has been reported that the time
to MRI is shortened, and that the proportion of patients who are
unable to walk at MRI decreases from 46 to 23% (18). These results suggest that the
establishment of such a system is effective in daily clinical
practice and that it can improve the time to referral.
Furthermore, the present study reported a
significant decrease in the time from the recognition of a
neurological deficit by physicians to MRI scanning and treatment
after the establishment of the treatment system for bone
metastases. Approximately 40% of patients underwent MRI at >3
days following recognition of a neurological deficit by physicians,
and in 26% of the patients, the severity of the neurological
deficit was worse at the time of the treatment before the system
was implemented. The rate of referral to orthopedists before
treatment was only 45%. Conversely, all patients were referred to
orthopedists, 71% of patients underwent MRI and RT on the same day,
and the remaining 29% of patients underwent MRI and RT the day
after the recognition of neurological deficit by physicians after
the system was implemented.
The use of BMAs was also increased following the
establishment of the system. Although the use of BMAs before the
recognition of a neurological deficit by physicians was only 39% in
patients before the establishment of the system, they were
administered at the time of diagnosis of bone metastases in all
patients after the establishment of the system. This could be due
to the increased efforts of physicians to treat patients with bone
metastases.
A novel multidisciplinary treatment system is
required to establish an effective and intensive diagnosis and
treatment system for patients with bone metastases (19). Hirai et al (17) demonstrated that early diagnosis
through surveillance of bone metastases in patients with
hepatocellular carcinoma can prevent fractures and paralysis, which
can lead to an improved quality of life. Recently, cancer boards,
including teams of doctors specialized in orthopedy, oncology,
palliative care, radiotherapy and radiology, nurses, physical
therapists, occupational therapists and medical social workers,
focusing on the management of bone metastases were organized in a
number of institutions in Japan (19). In certain institutions, radiologists
review all imaging studies of patients with bone metastases and
can, thus, identify patients that may require a prophylactic
approach to prevent paralysis (18).
With this system, a common understanding of the importance of
managing bone metastases is established among all members of the
cancer board.
Although the multidisciplinary treatment system is
effective for the management of bone metastases, this system
presents some limitations. With this system, it can be difficult to
identify patients with imminent development of MSCC in a timely
manner. For example, in certain patients with osteoblastic bone
metastases and extravertebral metastases, it can be hard for the
radiologist to identify MSCC by routine CT alone. Furthermore,
physicians can occasionally overlook signs of MSCC, including back
pain, motor dysfunction and sensory disturbance if they are not
severe. In addition, patients do not always mention the back pain
to physicians or do not come to the hospital when lower leg
weakness is not severe. There is currently no system in The Shikoku
Cancer Center (Matsuyama, Japan) that could be used to inform or
educate patients about back pain as an early warning sign of MSCC,
and alert them to go to hospital earlier. Therefore, neurological
deficits cannot be prevented in some patients. The rate of early
diagnosis and prevention of potential neurological deficits could
be improved if patients with bone metastases had been informed
about the importance of contacting physicians when complaining of
back pain, especially when the pain is accompanied by neurological
signs. Since MSCC occurs in only a small percentage of patients
with bone metastases, it may be reasonable to select patients at
high risk for education.
Another limitation of the system is the variable
background of patients with bone metastases. Since patients
presented with various types of primary tumor, metastatic sites,
systemic therapies and general status at the time of the
neurological deficit caused by MSCC, the individual prognoses were
different. These factors could be responsible for the absence of a
significant difference in the overall survival rates before and
after the establishment of the system presented in the present
study. However, these limitations are common in the study of bone
metastases.
In conclusion, the present study demonstrated that
the times between the recognition of a neurological deficit by
physicians to MRI scanning and the treatment were significantly
decreased after the establishment of the multidisciplinary
treatment system for bone metastases. In addition, no deterioration
of neurological deficits was observed at the time of treatment
after the establishment of the multidisciplinary treatment system.
Furthermore, the rate of patients with improvement in the severity
of neurological deficits after treatment was significantly
increased. The establishment of a multidisciplinary treatment
system for patients with bone metastases may therefore be useful
for the early diagnosis and treatment and prevention of MSCC in
patients with bone metastases.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
EN, KN, TF, TT and YA designed the study, and
collected and analyzed data. RN, TK and TO analyzed data. YS and SS
treated the patients presented in this manuscript and collected and
analyzed data. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Shikoku Cancer Center (Matsuyama, Japan).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Harel R and Angelov L: Spine metastases:
Current treatments and future directions. Eur J Cancer.
46:2696–2707. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sciubba DM, Petteys RJ, Dekutoski MB,
Fisher CG, Fehlings MG, Ondra SL, Rhines LD and Gokaslan ZL:
Diagnosis and management of metastatic spine disease. A review. J
Neurosurg Spine. 13:94–108. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nakata E, Sugihara S, Kataoka M, Yamashita
N, Furumatsu T, Takigawa T, Tetsunaga T and Ozaki T: Early response
assessment of palliative conventional radiotherapy for painful
uncomplicated vertebral bone metastases. J Orthop Sci. 23:912–917.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nakata E, Sugihara S, Kataoka M, Yamashita
N, Furumatsu T, Takigawa T, Tetsunaga T and Ozaki T: Early response
assessment of re-ossification after palliative conventional
radiotherapy for vertebral bone metastases. J Orthop Sci.
24:332–336. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
White BD, Stirling AJ, Paterson E,
Asquith-Coe K and Melder A; Guideline Development Group, :
Diagnosis and management of patients at risk of or with metastatic
spinal cord compression: Summary of NICE guidance. BMJ.
337:a25382008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Solberg A and Bremnes RM: Metastatic
spinal cord compression: Diagnostic delay, treatment, and outcome.
Anticancer Res. 19:677–684. 1999.PubMed/NCBI
|
|
7
|
Shibata H, Kato S, Sekine I, Abe K, Araki
N, Iguchi H, Izumi T, Inaba Y, Osaka I, Kato S, et al: Diagnosis
and treatment of bone metastasis: Comprehensive guideline of the
Japanese Society of Medical Oncology, Japanese Orthopedic
Association, Japanese Urological Association, and Japanese Society
for Radiation Oncology. ESMO Open. 16:e0000372016. View Article : Google Scholar
|
|
8
|
Levack P, Graham J, Collie D, Grant R,
Kidd J, Kunkler I, Gibson A, Hurman D, McMillan N, Rampling R, et
al: Don't wait for a sensory level-listen to the symptoms: A
prospective audit of the delays in diagnosis of malignant cord
compression. Clin Oncol (R Coll Radiol). 14:472–480. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tsukada Y, Nakamura N, Ohde S, Akahane K,
Sekiguchi K and Terahara A: Factors that delay treatment of
symptomatic metastatic extradural spinal cord compression. J
Palliat Med. 18:107–113. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Loblaw DA, Mitera G, Ford M and Laperriere
NJ: A 2011 updated systematic review and clinical practice
guideline for the management of malignant extradural spinal cord
compression. Int J Radiat Oncol Biol Phys. 84:312–317. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Savage P, Sharkey R, Kua T, Schofield L,
Richardson D, Panchmatia N, Papanastasopoulos P, Williams M,
Falconer A, Power D, et al: Malignant spinal cord compression: NICE
guidance, improvements and challenges. QJM. 107:277–282. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vieillard MH and Thureau S:
Multidisciplinary meetings dedicated to bone metastases: A
historical perspective and rationale. Bull Cancer. 100:1135–1139.
2013.(In French). View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Blum RH, Novetsky D, Shasha D and
Fleishman S: The multidisciplinary approach to bone metastases.
Oncology (Williston Park). 17:845–857; discussion 862–863, 867.
2003.PubMed/NCBI
|
|
14
|
Ibrahim T, Flamini E, Fabbri L, Serra P,
Mercatali L, Ricci R, Sacanna E, Falasconi MC, Casadei R, Galassi
R, et al: Multidisciplinary approach to the treatment of bone
metastases: Osteo-Oncology Center, a new organizational model.
Tumori. 95:291–297. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Thavarajah N, Wong K, Zhang L, Bedard G,
Wong E, Tsao M, Danjoux C, Barnes E, Sahgal A, Dennis K, et al:
Continued success in providing timely palliative radiation therapy
at the Rapid Response Radiotherapy Program: A review of 2008–2012.
Curr Oncol. 20:e206–e211. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bongiovanni A, Recine F, Fausti V, Foca F,
Casadei R, Falasconi MC, Oboldi D, Sansoni E, Fabbri L, Micheletti
S, et al: Ten-year experience of the multidisciplinary Osteoncology
Center. Support Care Cancer. 27:3395–3402. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hirai T, Shinoda Y, Tateishi R, Asaoka Y,
Uchino K, Wake T, Kobayashi H, Ikegami M, Sawada R, Haga N, et al:
Early detection of bone metastases of hepatocellular carcinoma
reduces bone fracture and paralysis. Jpn J Clin Oncol. 49:529–536.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Allan L, Baker L, Dewar J, Eljamel S,
Grant RM, Houston JG, McLeay T, Munro AJ and Levack P: Suspected
malignant cord compression-improving time to diagnosis via a
‘hotline’: A prospective audit. Br J Cancer. 100:1867–1872. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kimura T: Multidisciplinary approach for
bone metastasis: A review. Cancers (Basel). 10(pii): E1562018.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Frankel HL, Hancock DO, Hyslop G, Melzak
J, Michaelis LS, Ungar GH, Vernon JD and Walsh JJ: The value of
postural reduction in the initial management of closed injuries of
the spine with paraplegia and tetraplegia. I. Paraplegia.
7:179–192. 1969.PubMed/NCBI
|
|
21
|
Ejima Y, Matsuo Y and Sasaki R: The
current status and future of radiotherapy for spinal bone
metastases. J Orthop Sci. 20:585–592. 2015. View Article : Google Scholar : PubMed/NCBI
|