Introduction
According to the GLOBOCAN 2018 statistics,
colorectal cancer (CRC) is the fourth (6.1%) most common malignancy
after lung (11.6%), breast (11.6%) and prostate cancer (7.1%),
whereas the mortality rate of CRC is the second highest (9.2%) in
the world, following lung cancer (18.4%) (1). A total of 1.8 million CRC cases were
diagnosed, and nearly 880,000 cases of CRC-associated mortality
occurred in 2018 worldwide (1). An
improvement in the treatment options of CRC is a significant
challenge that requires attention. Additionally, the prioritization
of primary prevention and early detection is also required.
MicroRNAs (miRNAs/miRs) belong to a class of short
non-coding RNAs that are involved in cancer progression by binding
to the complementary site of mRNAs, which often leads to
translational repression or mRNA degradation (2). Various miRNAs, including let-7, miR-34a
and the miR-200 family, have been identified as potential
biomarkers or therapeutic molecules (3). Previous studies have reported that
miR-101 is downregulated in various types of cancer and suppresses
cancer cell progression, such as bladder cancer (4), breast cancer (5), hepatocellular carcinoma (6) and CRC (7). However, the comprehensive mechanisms
underlying the effects of miR-101 on CRC progression have not been
fully elucidated.
Ras-associated protein-1 (Rap1) serves a vital role
in the regulation of cancer progression (8). This gene is a member of the Ras-like
small GTPase family and contains two highly homologous isoforms,
Rap1a and Ras-related protein Rap1b (Rap1b) (9). Several studies have proposed potential
roles of Rap1b in tumorigenesis and demonstrated the upregulation
of Rap1b in several types of cancer (10,11). A
previous study reported that the loss of Rap1b can attenuate
thyroid cancer (12). Tang et
al (13) indicated that Rap1b
promotes the invasion and migration of hepatocellular carcinoma
cells by regulating Twist 1. Jia et al (14) discovered that Rap1b can promote the
progression of esophageal squamous cell carcinoma. Furthermore, Li
et al (15) reported that
metastasis associated lung adenocarcinoma transcript 1 can promote
the proliferation of glioma cells by acting as a sponge for
miR-101. In hepatocellular carcinoma, Sheng et al (16) reported that miR-101 regulates the
progression of hepatocellular carcinoma by interacting with Rap1b.
Based on these findings and assumptions, it was hypothesized that
miR-101 may interact with Rap1b to suppress the progression of
CRC.
The present study demonstrated that miR-101
inhibited CRC progression via regulation of Rap1b. The present
study provided insights into the underlying mechanism of miR-101 in
CRC and facilitates development of therapeutic strategies for
CRC.
Materials and methods
Clinical specimens
The tissues were collected from 50 patients (31
males and 19 females) with a median age of 48 years (range, 29–78
years) between April 2015 and June 2018 from Shanghai Ninth
People's Hospital (Shanghai, China). Written informed consent was
obtained from all patients prior to the study start. CRC tumor
tissues were obtained via surgical resections of 50 patients, and
adjacent normal tissues were obtained from the distal edge of each
resection <10 cm away from the tumor. The inclusion criteria for
the present study were as follows, the patients were newly
diagnosed and agreed to a 5-year follow up period. The exclusion
criteria were as follows, patients diagnosed with other diseases
and patients that had received chemotherapy or radiotherapy prior
to surgery. All tissue samples were obtained and immediately stored
at −80°C prior to further experiments. The present study was
approved by the Ethics Committee of Shanghai Ninth People's
Hospital (Shanghai, China).
Cell lines
Human CRC cells (SW620), human colon mucosal cells
(NCM460) and 293T cells were obtained from American Type Culture
Collection. The cell lines were cultured in RPMI-1640 medium and
supplemented with 10% fetal bovine serum. The 293T cells were
cultured in Dulbecco's modified Eagle's medium (Gibco; Thermo
Fisher Scientific, Inc.) with 10% fetal bovine serum (FBS; Thermo
Fisher Scientific, Inc.) and 1% penicillin-streptomycin (Thermo
Fisher Scientific, Inc). All cell lines were maintained at 37°C in
a humidified atmosphere with 5% CO2.
Reverse transcription-quantitative
(RT-q)PCR
Total RNA was extracted from CRC tissues, adjacent
normal tissues, SW620 cells and NCM460 cells using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.), and was reverse transcribed into cDNA by using a PrimeScript
RT reagent kit (Takara Bio, Inc.) at 37°C for 15 min, according to
the manufacturer's protocol. The expression levels of miR-101 and
Rap1b were determined using a ViiATM 7 Real-Time PCR system (Thermo
Fisher Scientific, Inc.). qPCR was subsequently performed using the
SYBR®-Green Real-Time PCR Master mix (Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol. The
following thermocycling conditions were used to detect miR-101: An
initial predenaturation step at 50°C for 2 min, followed by 40
cycles of denaturation at 95°C for 10 min and annealing at 60°C for
1 min. For other factor detection, the thermocycling conditions
were as follows: An initial predenaturation step at 94°C for 5 min,
followed by 40 cycles of denaturation at 95°C for 30 sec, annealing
at 60°C for 30 sec and extension at 72°C for 20 sec. The following
primer sequences were used for the qPCR: Rap1b; forward,
5′-TTTATTCCATCACAGCACAGTCC-3′ and reverse,
5′-TTTCTGTTAATTTGCCGCACTAGG-3′; miR-101; forward,
5′-CGGCGGTACAGTACTGTGATAA-3′ and reverse,
5′-CTGGTGTCGTGGAGTCGGCAATTC-3′; GAPDH; forward,
5′-GATGATCTTGAGGCTGTTGTC-3′ and reverse, 5′-CAGGGCTGCTTTTAACTCTG-3′
and U6; forward, 5′-CTCGCTTCGGCAGCACATATACTA-3′ and reverse,
5′-ACGAATTTGCGTGTCATCCTTGCG-3′. Relative expression levels were
quantified using the 2−ΔΔCq method (17) and normalized to the internal
reference genes GAPDH and U6.
Western blot analysis
Western blot analysis was performed in order to
detect Rap1b expression as previously described by Joseph et
al (18). Total protein was
extracted from transfected SW620 cells using
radioimmunoprecipitation assay buffer (Beyotime Institute of
Biotechnology). Total protein was quantified using a bicinchoninic
acid assay and 10 µg protein/lane was separated via SDS-PAGE on a
10% gel. Proteins were subsequently transferred onto polyvinylidene
difluoride membrane and blocked in 5% non-fat milk for 2 h at room
temperature. The membranes were incubated with the following
primary antibodies, anti-Rap1b (1:1,000, cat. no. 2326; Cell
Signaling Technology, Inc.) and anti-GAPDH (1:1,000, cat. no. 5174;
Cell Signaling Technology, Inc.) overnight at 4°C. Membranes were
washed three times with TBS with 0.1% Tween-20. Following the
primary incubation, membranes were incubated with horseradish
peroxidase-labeled secondary antibodies (1:1,000, cat. no.
ab205718; Abcam) for 2 h at 37°C. Protein bands were visualized
using SuperSignal™ West Femto Maximum Sensitivity Substrate (Thermo
Fisher Scientific, Inc.), according to the manufacturer's protocol.
GAPDH was used as an internal control.
Transfection
miR-101 mimics, anti-miR-101 and their respective
negative controls (miRNA mimic/inhibitor) were synthesized by
Shanghai GenePharma Co., Ltd. The full length of Rap1b was
subcloned into pcDNA3.1 to overexpress Rap1b, with empty pcDNA3.1
vector serving as the control. Transfection of the cells with the
miR-101 mimics (10 nM), anti-miR-101 (10 nM), miRNA mimic/inhibitor
(10 nM) and vectors (10 nM) were performed using
Lipofectamine® 2000 (Thermo Fisher Scientific, Inc.).
Co-transfection of the cells with miR-101 mimic and Rap1b was
performed using Lipofectamine® 2000 (Thermo Fisher
Scientific, Inc.), the subsequent experiments were performed at 48
h post-transfection. The sequences of oligonucleotides used were as
follows: miR-101 mimics, 5′-UACAGUACUGUGAUAACUGAA-3′; miRNA mimics,
5′-UUUGUACUACACAAAAGUACUG-3′; anti-miR-101,
5′-UUCAGUUAUCACAGUACUGUA-3′ and miRNA inhibitor control,
5′-UCACAACCUCCUAGAAAGAGUAGA-3′.
Cell proliferation, migration and
invasion assays
An MTT assay was used to determine cell
proliferation, whereas wound healing and Transwell assays were
conducted to measure cell migratory and invasive activities,
respectively. All of these assays were performed as previously
described (16).
Flow cytometry
Cell apoptosis was detected in three independent
experiments with the Annexin V-FITC/Propidium Iodide Apoptosis
Detection kit (40302ES20), according to the manufacturer's
protocol. Cells were subsequently detected using the FACSCalibur
flow cytometer (BD Biosciences) and analyzed using FlowJo software
(version 7.6.1; FlowJo LLC). The apoptotic rate was presented as
the percentage of Annexin V-positive cells among the total
cells.
Luciferase reporter assay
293T cells were transfected with the Firefly
luciferase plasmid (Promega Corporation) containing wild-type or
mutant 3′-untranslated region (UTR) of Rap1b, using
Lipofectamine® 2000 (Thermo Fisher Scientific, Inc.).
The cells were subsequently transfected with NC, miR-101 mimic or
anti-miR-101 (all from Ambion; Thermo Fisher Scientific, Inc.).
After 48 h of cell culture, the cells were harvested and lysed.
Luciferase activity was measured using the the Dual-Luciferase
Reporter Analysis kit (Promega Corporation). Normalized relative
light units represent Firefly luciferase activity/Renilla
luciferase activity.
The Cancer Genome Atlas (TCGA)
analysis
The clinical information and miR-101 expression
profile of the CRC cases were obtained from TCGA database
(https://cancergenome.nih.gov), within
the colorecteral cancer dataset (TCGA-COAD) (19), using the R software package (version
3.1.3) (20). The expression levels
of each hub gene were defined as either high (expression value ≥
median value) or low (expression value < median value). The
survival curves were analyzed via the Kaplan-Meier method, and
univariate survival analysis was performed using a log-rank
test.
Statistical analysis
The software package SPSS (version 22.0; IBM Corp.)
was used for statistical analysis. The data derived from each
experiment were presented as the mean ± standard deviation. A
paired Student's t-test was used to compare parameters between two
groups, and one-way ANOVA and Tukey's test were used to evaluate
the differences among multiple groups. P<0.05 was considered to
indicate a statistically significant difference. The experiments
were performed >3 times.
Results
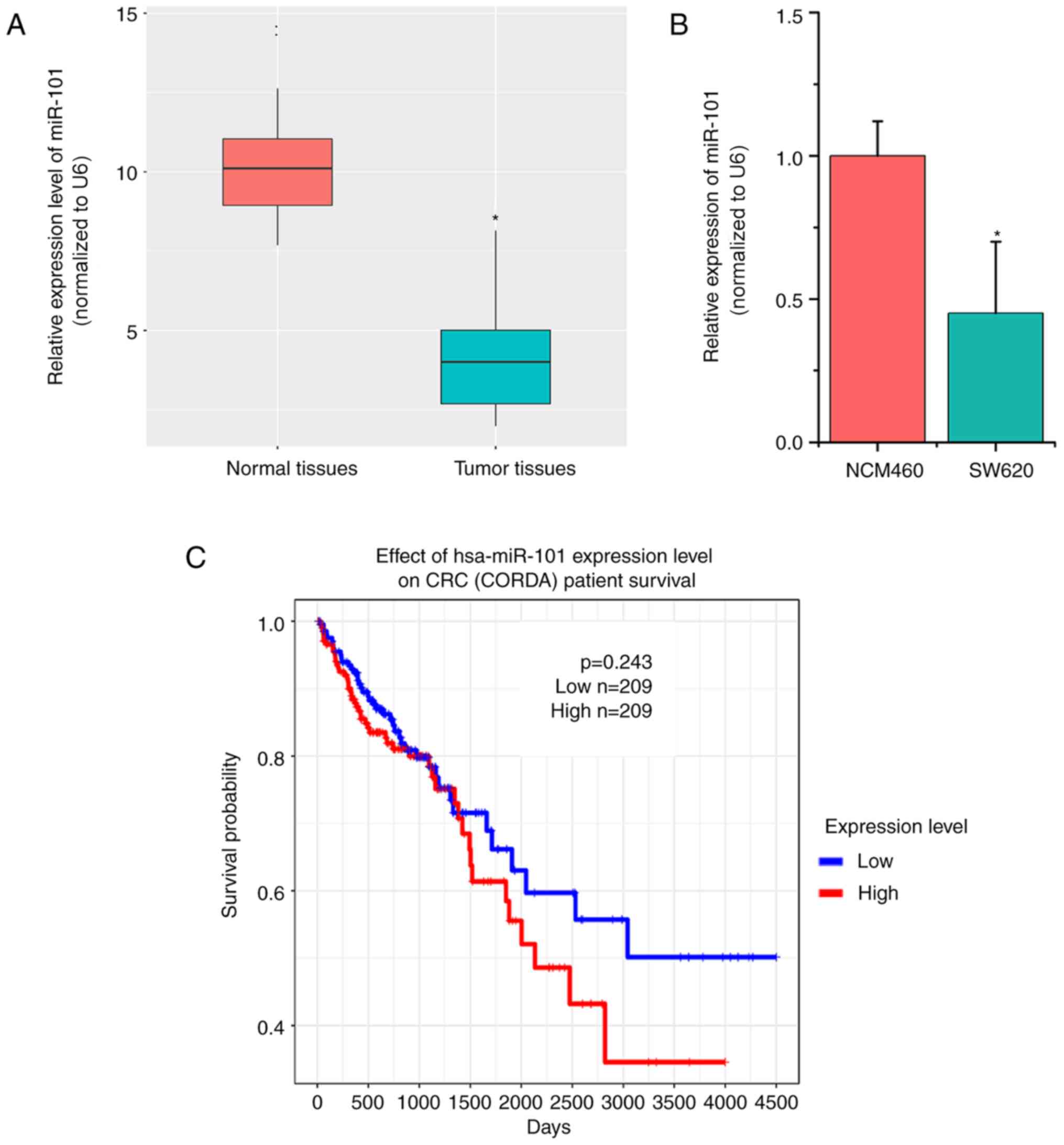
miR-101 expression is downregulated in
CRC tissues and cells
In order to explore the detailed role of miR-101 in
CRC, 50 paired tumor tissues and their adjacent normal tissues were
collected. Subsequently, the expression levels of miR-101 were
detected. RT-qPCR results indicated that miR-101 expression was
significantly lower in CRC tumor tissues compared with in normal
tissues (P<0.05; Fig. 1A;). The
present study further examined miR-101 expression in CRC cells
(SW620) and human colon mucosal cells (NCM460), which indicated
similar results (P<0.05; Fig.
1B). In addition, the association between miR-101 expression
and the prognosis of patients with CRC was investigated using TCGA
data. The overall survival curves indicated no apparent association
between miR-101 expression levels and the prognosis of patients
with CRC (P=0.243; Fig. 1C).
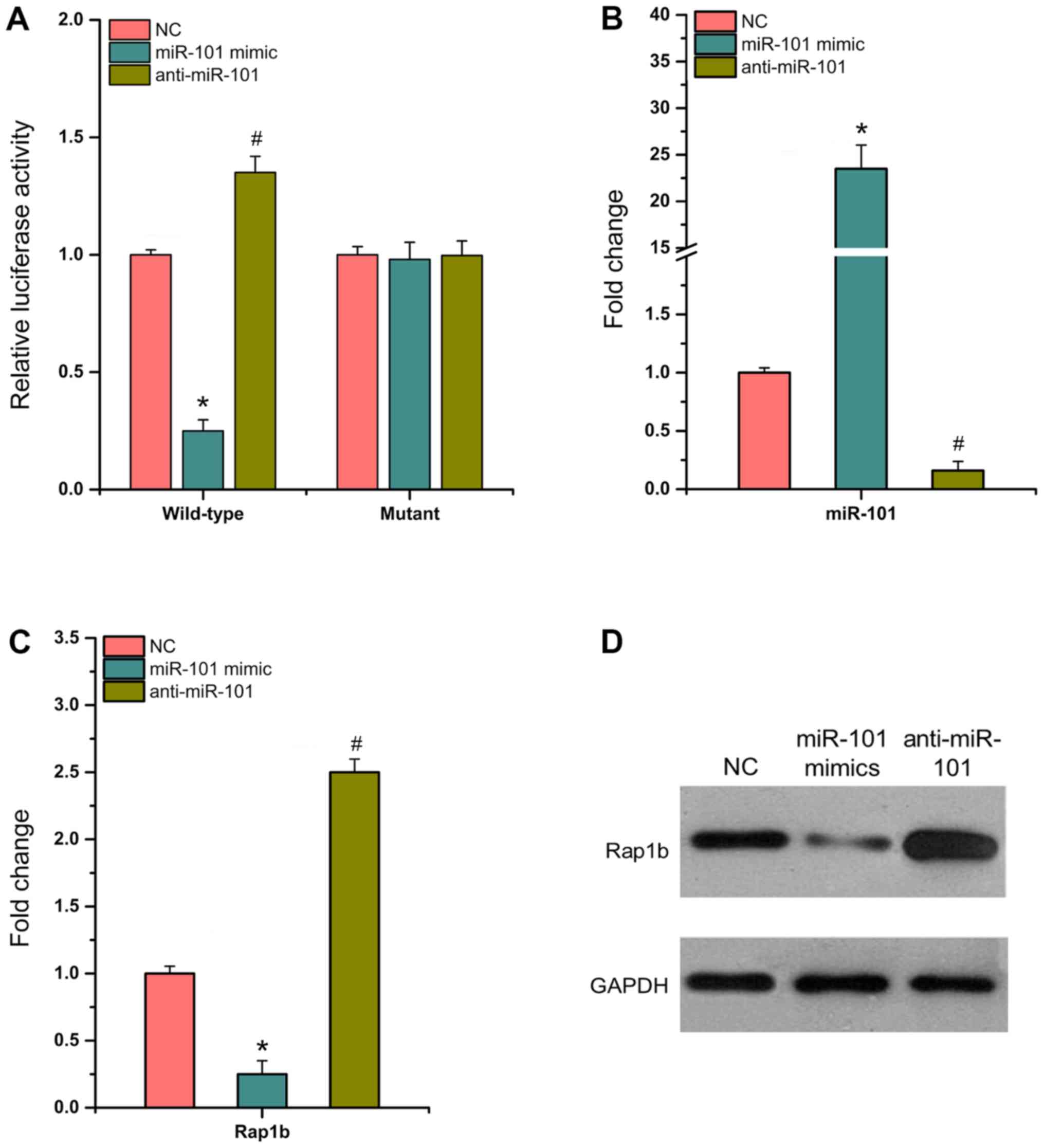
miR-101 directly targets Rap1b and
inhibits its expression
The present study used dual luciferase reporter
assays to detect luciferase activity in 293T cells containing
wild-type or mutant 3′-UTR, which were transfected with
anti-miR-101, miR-101 mimic and NC. The results indicated that in
wild-type SW620 cells, miR-101 mimic significantly decreased
luciferase activity by ~75% compared with that noted in NC cells,
whereas anti-miR-101 significantly enhanced luciferase activity by
~40% (P<0.05; Fig. 2A). The
transfection efficiency of miR-101 mimic and anti-miR-101 in SW620
cells was confirmed (P<0.05; Fig.
2B). RT-qPCR and western blotting assays demonstrated that the
expression levels of Rap1b were significantly decreased by miR-101
mimic (Fig. 2C and D; P<0.05).
Overall, the data indicated that miR-101 could directly bind to
Rap1b and downregulate its expression in CRC cells.
miR-101 inhibits the development and
progression of CRC cells
MTT and flow cytometry assays indicated that miR-101
overexpression could significantly inhibit cell proliferation and
promote apoptosis (Fig. 3A and B).
Wound healing assays further demonstrated that miR-101 mimic
decreased SW620 cell migration (Fig.
3C). Transwell assays indicated that miR-101 mimic suppressed
SW620 cell invasion (Fig. 3D).
Overall, the results indicate that miR-101 acts as a tumor
suppressor for CRC.
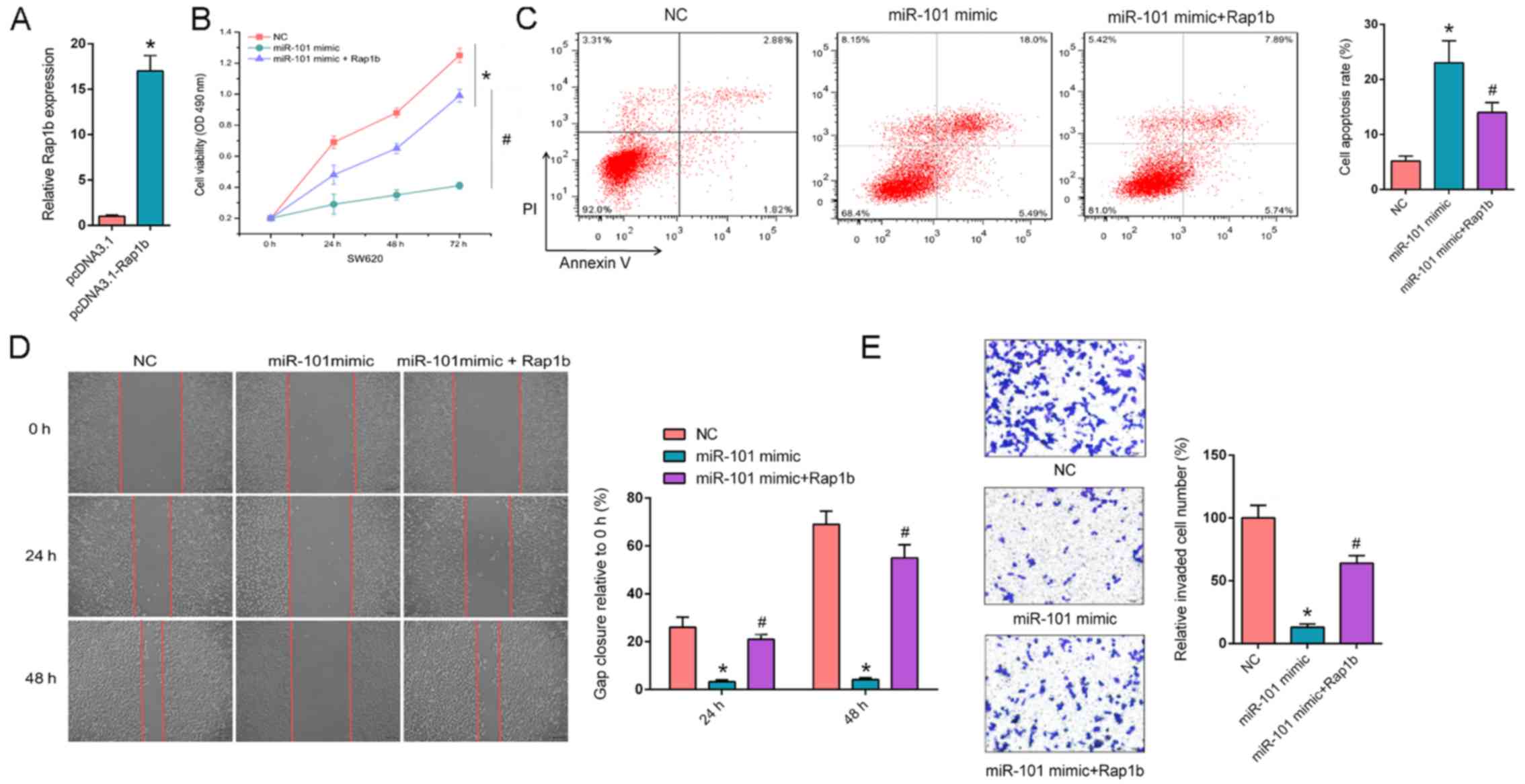 | Figure 3.Overexpression of Rap1b rescues
miR-101 mimic-attenuated CRC progression. (A) RT-qPCR revealed the
relative Rap1b expression in SW620 cells transfected with pcDNA3.1
and pcDNA3.1- Rap1b. (B) MTT assays revealed the cell proliferation
rate of SW620 cells transfected with NC, miR-101 mimic or miR-101
mimic + Rap1b at different time points (0, 24, 48 and 72 h). (C)
Flow cytometry revealed cell apoptosis of SW620 cells transfected
with NC, miR-101 mimic or miR-101 mimic + Rap1b. (D) Wound healing
assays revealed migration abilities of SW620 cells transfected with
NC, miR-101 mimic or miR-101 mimic + Rap1b at 0, 24 and 48 h. (E)
Transwell invasion assays demonstrated invasion abilities of SW620
cells transfected with NC, miR-101 mimic or miR-101 mimic + Rap1b.
*P<0.05 vs. NC; #P<0.05 vs. miR-101 mimic. Rap1b,
Ras-related protein Rap1b; NC, negative control; miR-101,
microRNA-101; PI, propidium iodide; RT-qPCR, reverse
transcription-quantitative PCR. |
Overexpression of Rap1b rescues
miR-101 mimic-attenuated CRC progression
In order to further determine whether the
interaction between miR-101 and Rap1b could affect the progression
of CRC, Rap1b was transfected into miR-101 mimic-expressing SW620
cells and the transfection efficiency of Rap1b was measured. The
expression of Rap1b significantly increased in SW620 cells
transfected with pcDNA3.1-Rap1b compared with SW620 cells
transfected with pcDNA3.1 (P<0.05; Fig. 3A). Furthermore, MTT assays
demonstrated that Rap1b overexpression significantly enhanced
miR-101-suppressed cell proliferation (P<0.05; Fig. 3B). Flow cytometry assays indicated
that aberrantly expressed Rap1b significantly decreased the
apoptosis rate induced by miR-101 mimic (P<0.05; Fig. 3C). In addition, Rap1b reversed the
suppressive effect of miR-101 on cell migration and invasion in
SW620 cells (P<0.05; Fig. 3D and
E, respectively). In summary, the results of the present study
suggest that Rap1b is a key downstream effector of
miR-101-regulated CRC phenotypes.
Discussion
Abnormal expression of miRNAs is a feature of human
malignancies and frequently leads to either oncogenesis or tumor
suppression (21,22). Increasing evidence has demonstrated
that miR-101 is downregulated in various types of cancer and
inhibits the development and progression of cancer (23,24). Liu
et al (25) reported that
miR-101 can interact with cancer susceptibility 2c to promote
astrocytoma tumorigenesis. Furthermore, Jiang et al
(26) demonstrated that miR-101 is
involved in CRC metastasis via the miR-101-O-linked
β-N-acetylglucosamine/enhancer of zeste 2 polycomb repressive
complex 2 subunit regulatory feedback circuit. Xiong et al
(27) revealed that CRC cell
proliferation and migration are regulated by the zinc finger E-box
binding homeobox 1 (ZEB1)-transcription factor AS1/miR-101/ZEB1
axis. The present study determined that miR-101 was significantly
downregulated in CRC tissues and cells according to RT-qPCR assays.
Additionally, SW620 cells transfected with miR-101 mimic exhibited
decreased growth rates compared with NC cells. In addition, it was
demonstrated that the upregulation of miR-101 promoted cell
apoptosis. Wound healing and Transwell assays demonstrated that
miR-101 could suppress CRC cell migration and invasion.
Previous studies have demonstrated that Rap1b is
involved in the progression of various types of cancer and that it
often acts as an oncogene to promote cancer cell proliferation
(28–30). Peng et al (31) highlighted that miR-100 can regulate
CRC cell proliferation and invasion by targeting Rap1b, and Guo
et al (32) demonstrated that
miR-139 acts as a sponge of Rap1b to affect the progression of CRC,
which indicated that Rap1b may regulate CRC progression through
multiple mechanisms. Therefore, it was inferred that miR-101 could
bind to the complementary sequence of Rap1b and may affect the
biological functions of CRC cells. Initially, a dual luciferase
reporter assay was applied to confirm the interaction between
miR-101 and Rap1b. miR-101 mimic significantly downregulated Rap1b
expression in wild-type SW620 cells. Furthermore, overexpression of
Rap1b could rescue miR-101 mimic-attenuated cell growth, migration
and invasion.
Collectively, the present study identified a
molecular axis of miR-101/Rap1b underlying CRC progression.
However, in vivo experiments are required to further
validate these findings in follow-up studies. It remains necessary
to identify downstream signaling pathways that are regulated by
Rap1b.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
ZZ and BL designed the study. HX and YD analyzed the
data and prepared the figures. ZZ and BL drafted the initial
manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the Shanghai Ninth People's Hospital (Shanghai,
China). Written informed consent was obtained from all patients
prior to the study start.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rupaimoole R and Slack FJ: MicroRNA
therapeutics: Towards a new era for the management of cancer and
other diseases. Nat Rev Drug Discov. 16:203–222. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liu D, Li Y, Luo G, Xiao X, Tao D, Wu X,
Wang M, Huang C, Wang L, Zeng F and Jiang G: LncRNA SPRY4-IT1
sponges miR-101-3p to promote proliferation and metastasis of
bladder cancer cells through up-regulating EZH2. Cancer Lett.
388:281–291. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Qian K, Liu G, Tang Z, Hu Y, Fang Y, Chen
Z and Xu X: The long non-coding RNA NEAT1 interacted with miR-101
modulates breast cancer growth by targeting EZH2. Arch Biochem
Biophys. 615:1–9. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu Y, Tan J, Ou S, Chen J and Chen L:
MicroRNA-101-3p suppresses proliferation and migration in
hepatocellular carcinoma by targeting the HGF/c-Met pathway. Invest
New Drugs. Mar 30–2019.(Epub ahead of print). doi:
10.1007/s10637-019-00766-8.
|
|
7
|
Chen LG, Xia YJ and Cui Y: Upregulation of
miR-101 enhances the cytotoxic effect of anticancer drugs through
inhibition of colon cancer cell proliferation. Oncol Rep.
38:100–108. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hattori M and Minato N: Rap1 GTPase:
Functions, regulation, and malignancy. J Biochem. 134:479–484.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pei W, Tao L, Zhang LW, Zhang S, Cao J,
Jiao Y, Tong J and Nie J: Circular RNA profiles in mouse lung
tissue induced by radon. Environ Health Prev Med. 22:362017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang Y, Li M, Yan Y, Zhang J, Sun K, Qu
JK, Wang JS and Duan XY: Expression of RAP1B is associated with
poor prognosis and promotes an aggressive phenotype in gastric
cancer. Oncol Rep. 34:2385–2394. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin KT, Yeh YM, Chuang CM, Yang SY, Chang
JW, Sun SP, Wang YS, Chao KC and Wang LH: Glucocorticoids mediate
induction of microRNA-708 to suppress ovarian cancer metastasis
through targeting Rap1B. Nat Commun. 6:59172015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Huk DJ, Ashtekar A, Magner A, La Perle K
and Kirschner LS: Deletion of Rap1b, but not Rap1a or Epac1,
reduces protein kinase A-mediated thyroid cancer. Thyroid.
28:1153–1161. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tang Z, Peng H, Chen J, Liu Y, Yan S, Yu
G, Chen Q, Tang H and Liu S: Rap1b enhances the invasion and
migration of hepatocellular carcinoma cells by up-regulating Twist
1. Exp Cell Res. 367:56–64. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Jia Z, Yang Y, Dengyan Z, Chunyang Z,
Donglei L, Kai W and Song Z: RAP1B, a DVL2 binding protein,
activates Wnt/beta-catenin signaling in esophageal squamous cell
carcinoma. Gene. 611:15–20. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li Z, Xu C, Ding B, Gao M, Wei X and Ji N:
Long non-coding RNA MALAT1 promotes proliferation and suppresses
apoptosis of glioma cells through derepressing Rap1B by sponging
miR-101. J Neurooncol. 134:19–28. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sheng Y, Ding S, Chen K, Chen J, Wang S,
Zou C, Zhang J, Cao Y, Huang A and Tang H: Functional analysis of
miR-101-3p and Rap1b involved in hepatitis B virus-related
hepatocellular carcinoma pathogenesis. Biochem Cell Biol.
92:152–162. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Joseph JV, Conroy S, Pavlov K, Sontakke P,
Tomar T, Eggens-Meijer E, Balasubramaniyan V, Wagemakers M, den
Dunnen WF and Kruyt FA: Hypoxia enhances migration and invasion in
glioblastoma by promoting a mesenchymal shift mediated by the
HIF1α-ZEB1 axis. Cancer Lett. 359:107–116. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Collins FS and Barker AD: Mapping the
cancer genome. Pinpointing the genes involved in cancer will help
chart a new course across the complex landscape of human
malignancies. Sci Am. 296:50–57. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li L, Zhu Z, Zhao Y, Zhang Q, Wu X, Miao
B, Cao J and Fei S: FN1, SPARC, and SERPINE1 are highly expressed
and significantly related to a poor prognosis of gastric
adenocarcinoma revealed by microarray and bioinformatics. Sci Rep.
9:78272019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hassanlou M, Soltani BM, Medlej A, Kay M
and Mowla SJ: Hsa-miR-6165 downregulates insulin-like growth
factor-1 receptor (IGF-1R) expression and enhances apoptosis in
SW480 cells. Biol Chem. Nov 8–2019.(Epub ahead of print). doi:
10.1515/hsz-2018-0421.
|
|
22
|
Zhu QL, Zhan DM, Chong YK, Ding L and Yang
YG: MiR-652-3p promotes bladder cancer migration and invasion by
targeting KCNN3. Eur Rev Med Pharmacol Sci. 23:8806–8812.
2019.PubMed/NCBI
|
|
23
|
Cao C, Xu Y, Du K, Mi C, Yang C, Xiang L,
Xie Y and Liu W: LINC01303 functions as a competing endogenous RNA
to regulate EZH2 expression by sponging miR-101-3p in gastric
cancer. J Cell Mol Med. 23:7342–7348. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang H, Wang X, Hu B, Zhang F, Wei H and
Li L: Circular RNA ZFR accelerates non-small cell lung cancer
progression by acting as a miR-101-3p sponge to enhance CUL4B
expression. Artif Cells Nanomed Biotechnol. 47:3410–3416. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liu C, Sun Y, She X, Tu C, Cheng X, Wang
L, Yu Z, Li P, Liu Q, Yang H, et al: CASC2c as an unfavorable
prognosis factor interacts with miR-101 to mediate astrocytoma
tumorigenesis. Cell Death Dis. 8:e26392017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Jiang M, Xu B, Li X, Shang Y, Chu Y, Wang
W, Chen D, Wu N, Hu S, Zhang S, et al: O-GlcNAcylation promotes
colorectal cancer metastasis via the miR-101-O-GlcNAc/EZH2
regulatory feedback circuit. Oncogene. 38:301–316. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xiong WC, Han N, Wu N, Zhao KL, Han C,
Wang HX, Ping GF, Zheng PF, Feng H, Qin L and He P: Interplay
between long noncoding RNA ZEB1-AS1 and miR-101/ZEB1 axis regulates
proliferation and migration of colorectal cancer cells. Am J Transl
Res. 10:605–617. 2018.PubMed/NCBI
|
|
28
|
Wilson JM, Lorimer E, Tyburski MD and
Williams CL: β-Adrenergic receptors suppress Rap1B prenylation and
promote the metastatic phenotype in breast cancer cells. Cancer
Biol Ther. 16:1364–1374. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhang M, Zhou S, Zhang L, Zhang J, Cai H,
Zhu J, Huang C and Wang J: miR-518b is down-regulated, and involved
in cell proliferation and invasion by targeting Rap1b in esophageal
squamous cell carcinoma. FEBS Lett. 586:3508–3521. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li Y, Liu Y, Shi F, Cheng L and She J:
Knockdown of Rap1b enhances apoptosis and autophagy in gastric
cancer cells via the PI3K/Akt/mTOR pathway. Oncol Res. 24:287–293.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Peng H, Luo J, Hao H, Hu J, Xie SK, Ren D
and Rao B: MicroRNA-100 regulates SW620 colorectal cancer cell
proliferation and invasion by targeting RAP1B. Oncol Rep.
31:2055–2062. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Guo H, Hu X, Ge S, Qian G and Zhang J:
Regulation of RAP1B by miR-139 suppresses human colorectal
carcinoma cell proliferation. Int J Biochem Cell Biol.
44:1465–1472. 2012. View Article : Google Scholar : PubMed/NCBI
|