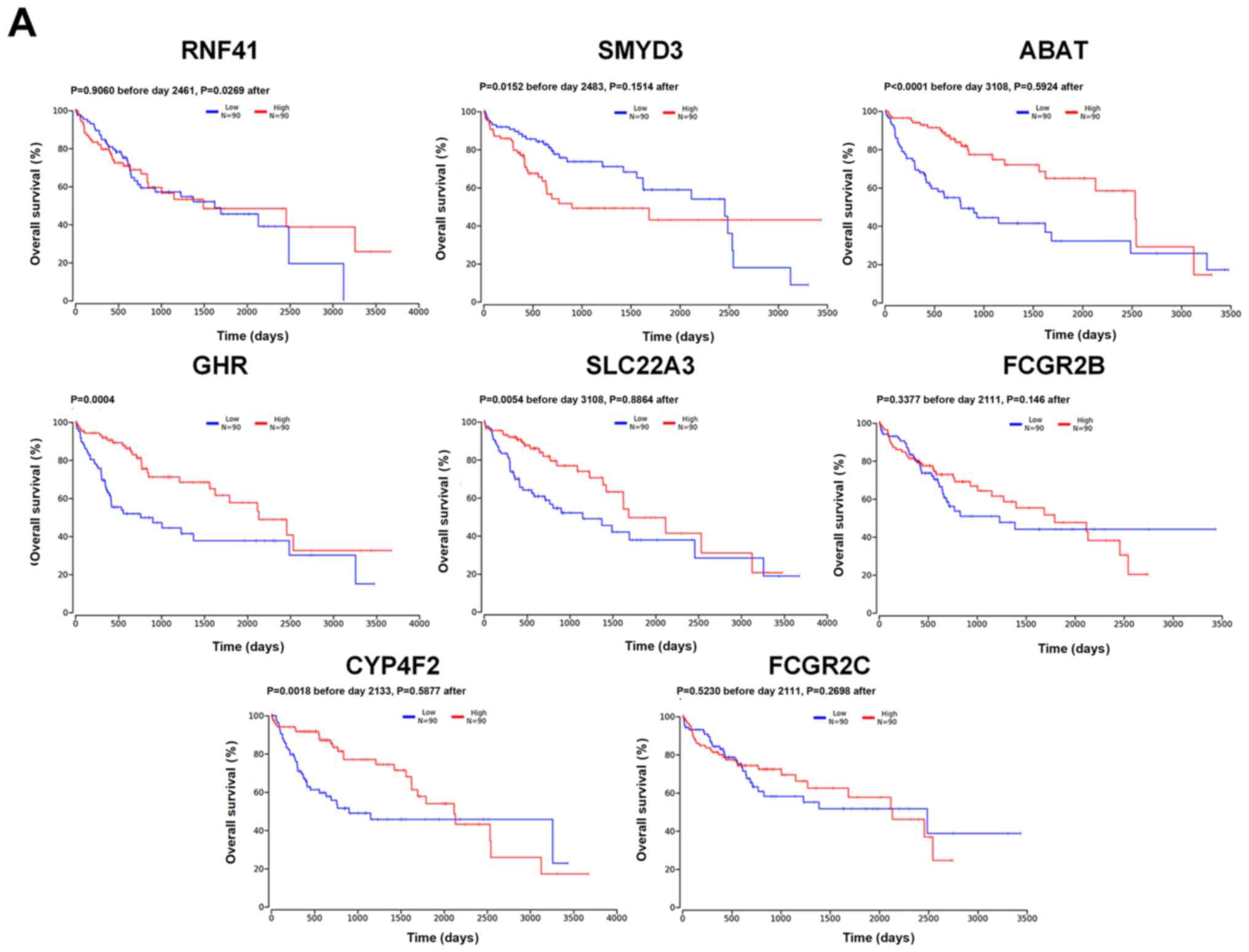
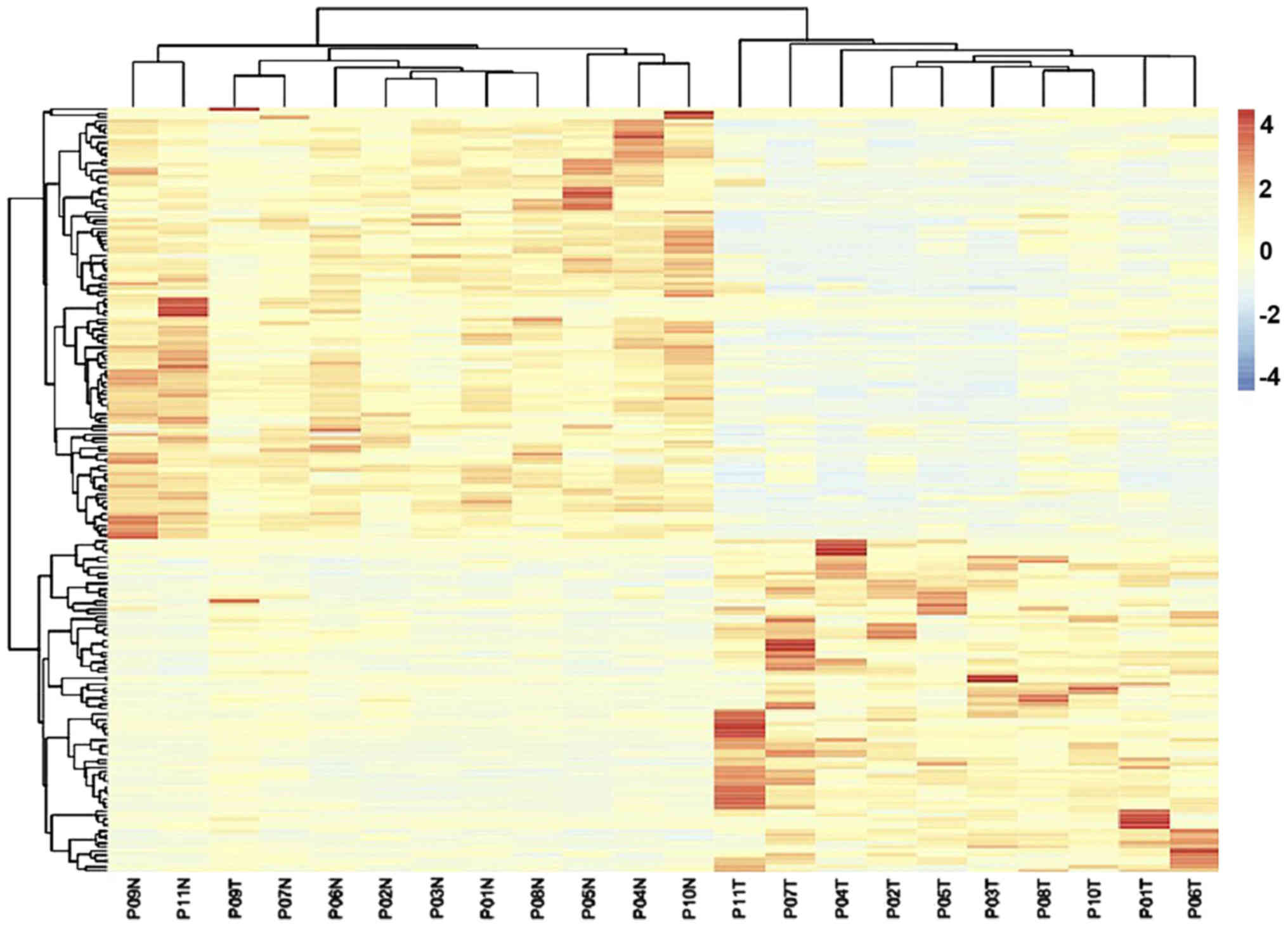
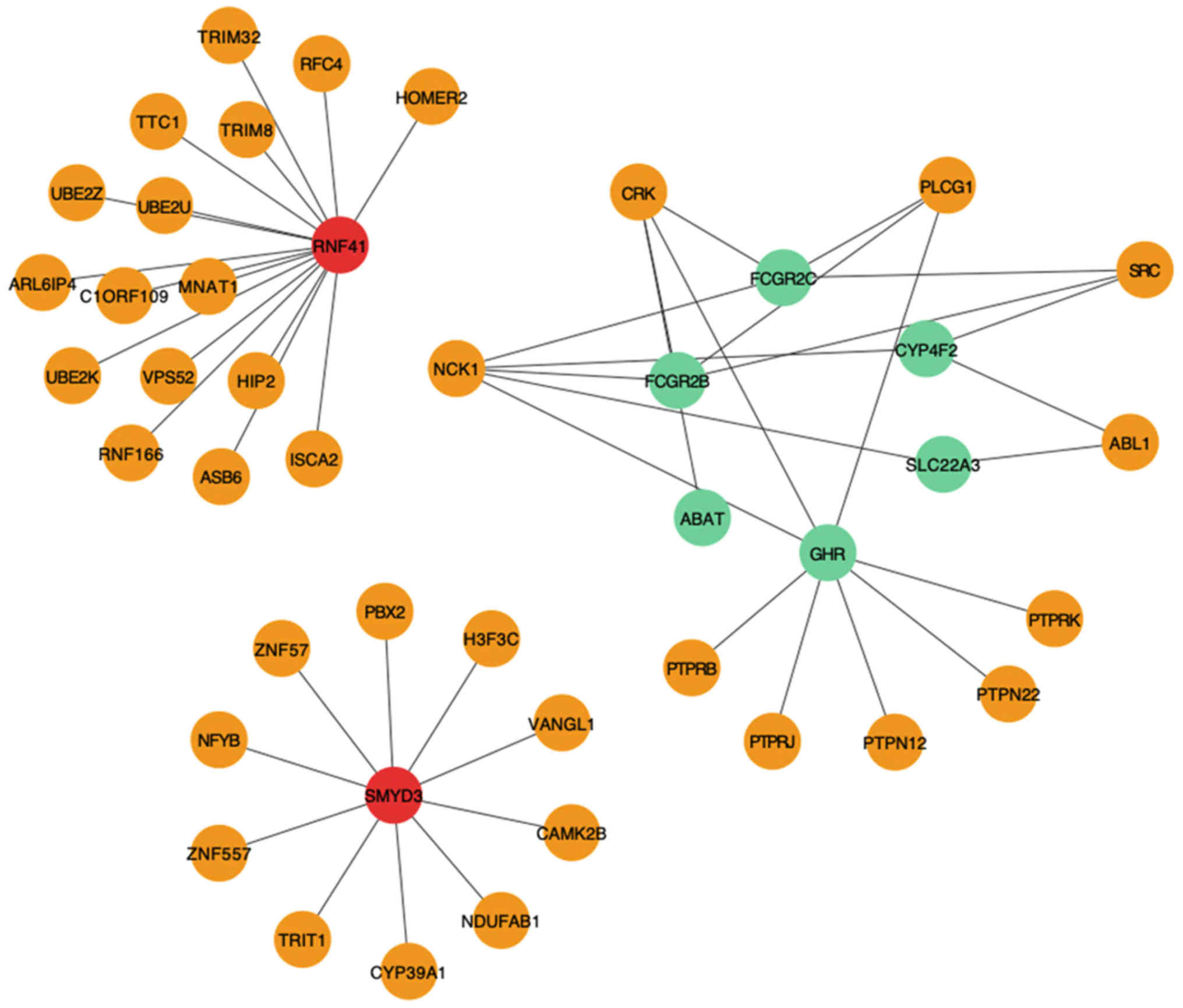
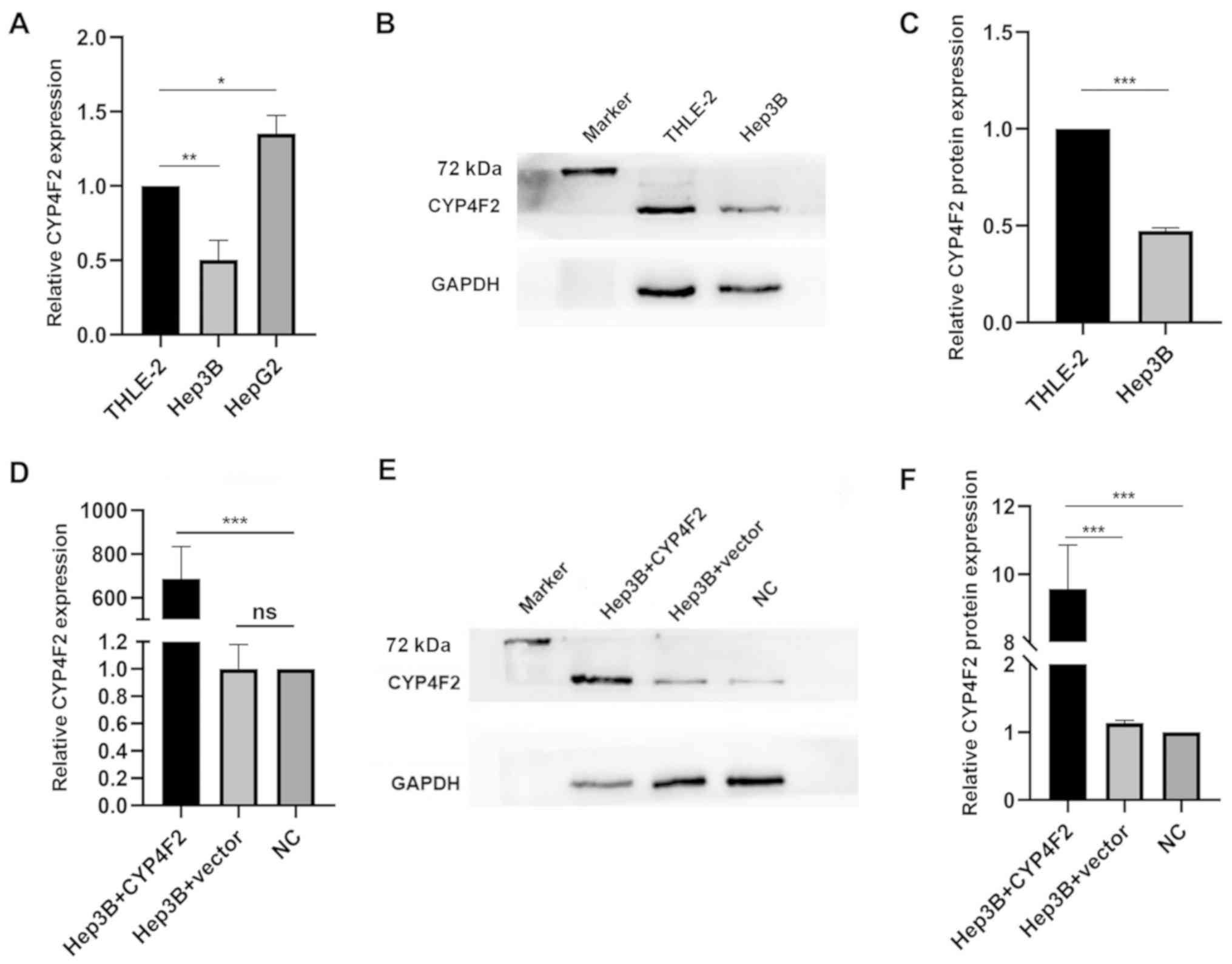
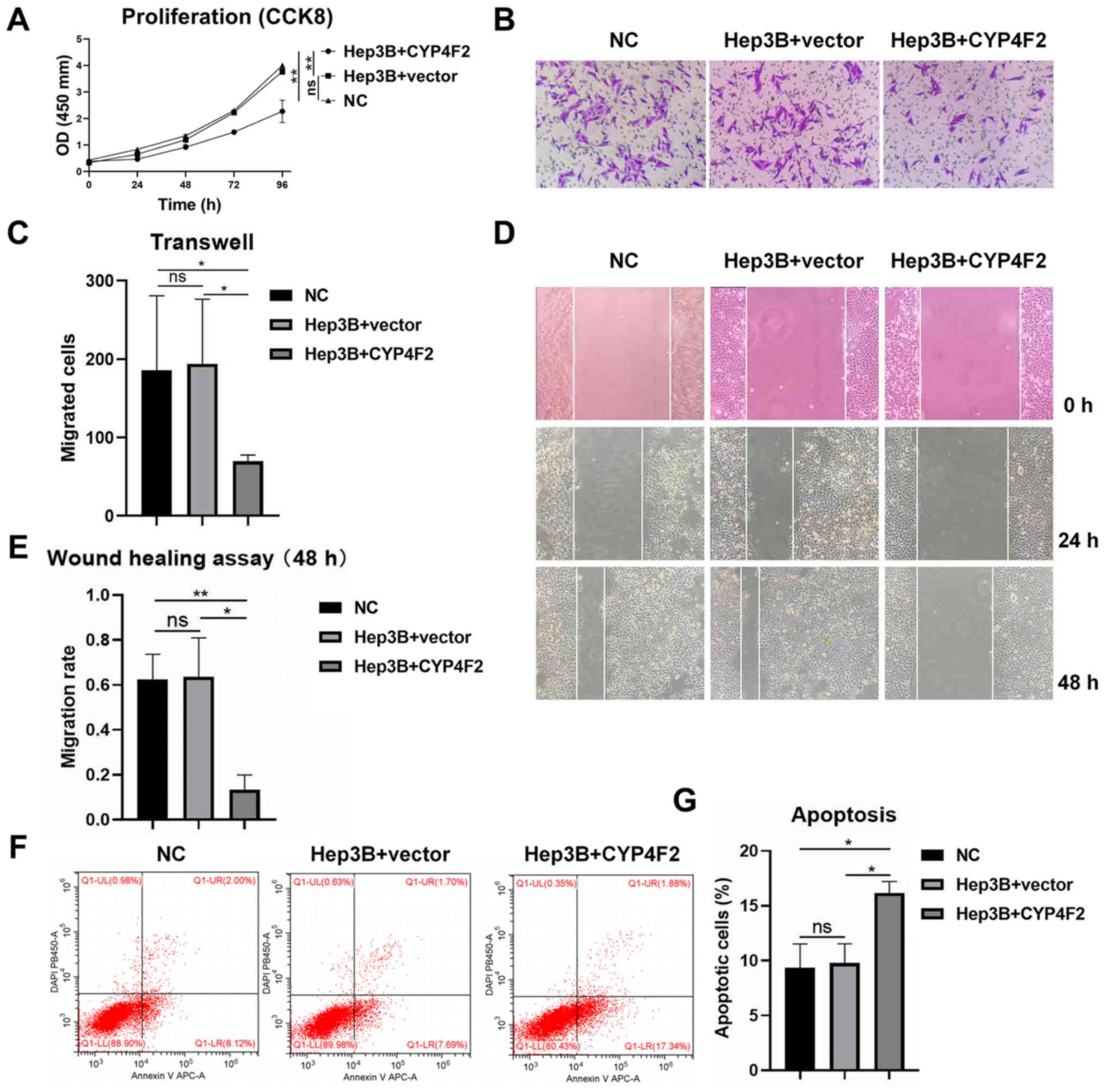
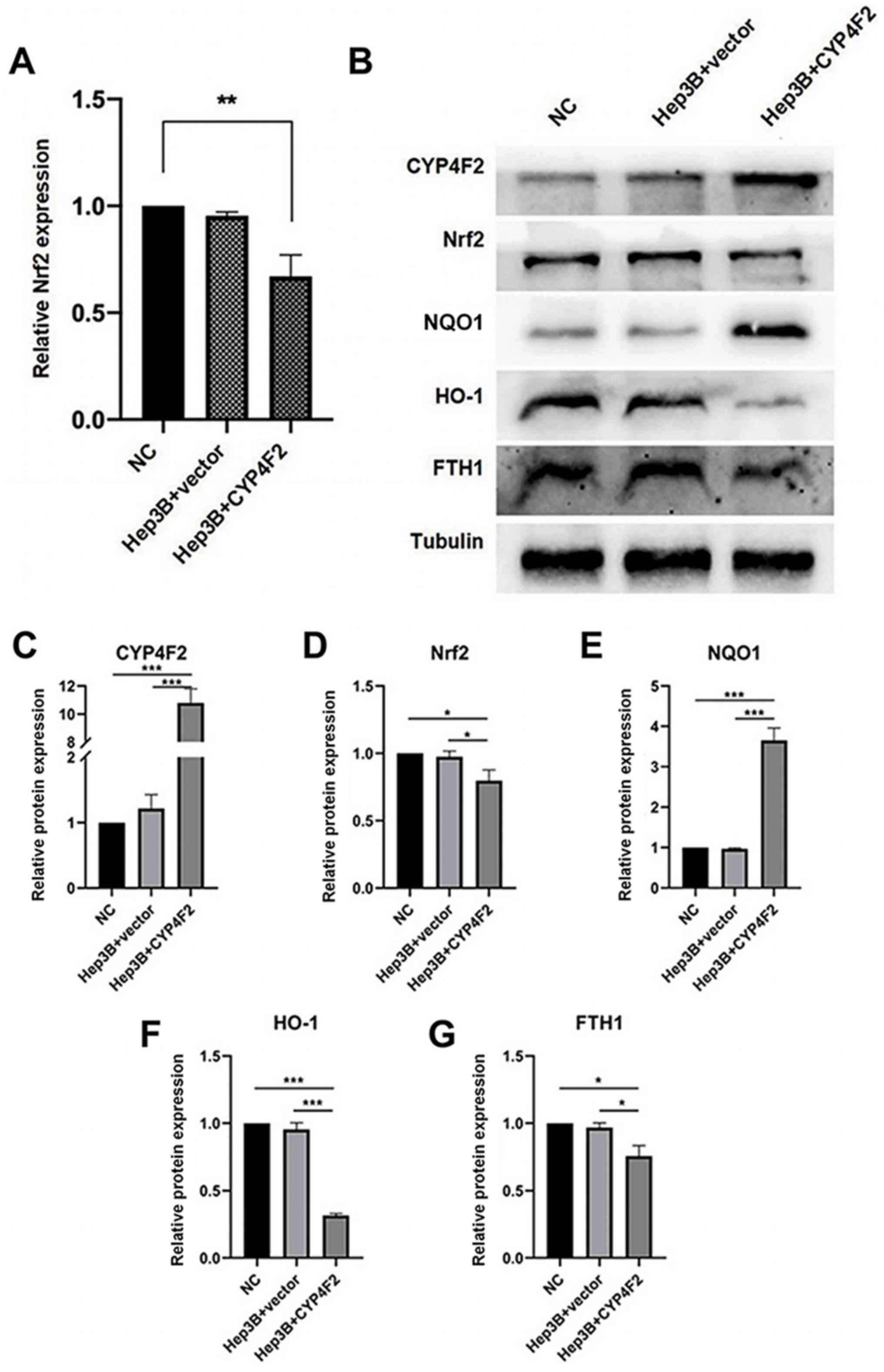
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
El-Serag HB: Epidemiology of viral
hepatitis and hepatocellular carcinoma. Gastroenterology.
142:1264–1273.e1. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Plummer M, de Martel C, Vignat J, Ferlay
J, Bray F and Franceschi S: Global burden of cancers attributable
to infections in 2012: A synthetic analysis. Lancet Glob Health.
4:e609–616. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Thorgeirsson SS and Grisham JW: Molecular
pathogenesis of human hepatocellular carcinoma. Nat Genet.
31:339–346. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Iizuka N, Oka M, Yamada-Okabe H, Mori N,
Tamesa T, Okada T, Takemoto N, Hashimoto K, Tangoku A, Hamada K, et
al: Differential gene expression in distinct virologic types of
hepatocellular carcinoma: Association with liver cirrhosis.
Oncogene. 22:3007–3014. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ye QH, Qin LX, Forgues M, He P, Kim JW,
Peng AC, Simon R, Li Y, Robles AI, Chen Y, et al: Predicting
hepatitis B virus-positive metastatic hepatocellular carcinomas
using gene expression profiling and supervised machine learning.
Nat Med. 9:416–423. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tateishi R and Omata M; Nature Publishing
Group, : Hepatocellular carcinoma in 2011: Genomics in
hepatocellular carcinoma - a big step forward. Nat Rev
Gastroenterol Hepatol. 9:69–70. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shibata T and Aburatani H: Exploration of
liver cancer genomes. Nat Rev Gastroenterol Hepatol. 11:340–349.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nakagawa H and Shibata T: Comprehensive
genome sequencing of the liver cancer genome. Cancer Lett.
340:234–240. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fujimoto A, Totoki Y, Abe T, Boroevich KA,
Hosoda F, Nguyen HH, Aoki M, Hosono N, Kubo M, Miya F, et al:
Whole-genome sequencing of liver cancers identifies etiological
influences on mutation patterns and recurrent mutations in
chromatin regulators. Nat Genet. 44:760–764. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kan Z, Zheng H, Liu X, Li S, Barber TD,
Gong Z, Gao H, Hao K, Willard MD, Xu J, et al: Whole-genome
sequencing identifies recurrent mutations in hepatocellular
carcinoma. Genome Res. 23:1422–1433. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Guichard C, Amaddeo G, Imbeaud S, Ladeiro
Y, Pelletier L, Maad IB, Calderaro J, Bioulac-Sage P, Letexier M,
Degos F, et al: Integrated analysis of somatic mutations and focal
copy-number changes identifies key genes and pathways in
hepatocellular carcinoma. Nat Genet. 44:694–698. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Huang J, Zhao L, Yang P, Chen Z, Tang NZ,
Ruan X and Chen Y: Genome-Wide Transcriptome Analysis of CD36
Overexpression in HepG2.2.15 Cells to Explore Its Regulatory Role
in Metabolism and the Hepatitis B Virus Life Cycle. PLoS ONE.
11:e01647872016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pan Q, Long X, Song L, Zhao D, Li X, Li D,
Li M, Zhou J, Tang X, Ren H, et al: Transcriptome sequencing
identified hub genes for hepatocellular carcinoma by weighted-gene
co-expression analysis. Oncotarget. 7:38487–38499. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang H, Weng X, Ye J, He L, Zhou D and
Liu Y: Promoter hypermethylation of TERT is associated with
hepatocellular carcinoma in the Han Chinese population. Clin Res
Hepatol Gastroenterol. 39:600–609. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Trapnell C, Pachter L and Salzberg SL:
TopHat: Discovering splice junctions with RNA-Seq. Bioinformatics.
25:1105–1111. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Langmead B, Trapnell C, Pop M and Salzberg
SL: Ultrafast and memory-efficient alignment of short DNA sequences
to the human genome. Genome Biol. 10:R252009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Trapnell C, Williams BA, Pertea G,
Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ and Pachter
L: Transcript assembly and quantification by RNA-Seq reveals
unannotated transcripts and isoform switching during cell
differentiation. Nat Biotechnol. 28:511–515. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Dennis G Jr, Sherman BT, Hosack DA, Yang
J, Gao W, Lane HC and Lempicki RA: DAVID: Database for Annotation,
Visualization, and Integrated Discovery. Genome Biol. 4:32003.
View Article : Google Scholar
|
|
20
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al The Gene Ontology Consortium, : Gene ontology: Tool for the
unification of biology. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ogata H, Goto S, Sato K, Fujibuchi W, Bono
H and Kanehisa M: KEGG: Kyoto Encyclopedia of Genes and Genomes.
Nucleic Acids. 27:29–34. 1999. View Article : Google Scholar
|
|
22
|
Nepusz T, Yu H and Paccanaro A: Detecting
overlapping protein complexes in protein-protein interaction
networks. Nat Methods. 9:471–472. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Anaya J: OncoLnc: Linking TCGA survival
data to mRNAs, miRNAs, and lncRNAs. PeerJ Computer Science. PeerJ
Inc. 2:e672016.
|
|
24
|
Qiu P and Sheng J: A two-stage procedure
for comparing hazard rate functions. J R Stat Soc B. 70:191–208.
2008.
|
|
25
|
Uhlen M, Zhang C, Lee S, Sjöstedt E,
Fagerberg L, Bidkhori G, Benfeitas R, Arif M, Liu Z, Edfors F, et
al: A pathology atlas of the human cancer transcriptome. Science.
357:eaan25072017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Das J and Yu H: HINT: High-quality protein
interactomes and their applications in understanding human disease.
BMC Syst Biol. 6:922012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Johnson AL, Edson KZ, Totah RA and Rettie
AE: Cytochrome P450 ω-Hydroxylases in Inflammation and Cancer. Adv
Pharmacol. 74:223–262. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Raghunath A, Sundarraj K, Arfuso F, Sethi
G and Perumal E: Dysregulation of Nrf2 in Hepatocellular Carcinoma:
Role in Cancer Progression and Chemoresistance. Cancers (Basel).
10:4812018. View Article : Google Scholar
|
|
30
|
Alves AC, Ribeiro D, Horta M, Lima JLFC,
Nunes C and Reis S: The daunorubicin interplay with mimetic model
membranes of cancer cells: A biophysical interpretation. Biochim
Biophys Acta Biomembr. 1859:941–948. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Herath NI, Devun F, Herbette A, Lienafa
M-C, Chouteau P, Sun J-S, Dutreix M and Denys A: Potentiation of
doxorubicin efficacy in hepatocellular carcinoma by the DNA repair
inhibitor DT01 in preclinical models. Eur Radiol. 27:4435–4444.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Fearon KC, McMillan DC, Preston T,
Winstanley FP, Cruickshank AM and Shenkin A: Elevated circulating
interleukin-6 is associated with an acute-phase response but
reduced fixed hepatic protein synthesis in patients with cancer.
Ann Surg. 213:26–31. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Perlmutter DH, Dinarello CA, Punsal PI and
Colten HR: Cachectin/tumor necrosis factor regulates hepatic
acute-phase gene expression. J Clin Invest. 78:1349–1354. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Brodie MJ, Boobis AR, Bulpitt CJ and
Davies DS: Influence of liver disease and environmental factors on
hepatic monooxygenase activity in vitro. Eur J Clin
Pharmacol. 20:39–46. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Toyokuni S: Iron-induced carcinogenesis:
The role of redox regulation. Free Radic Biol Med. 20:553–566.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Likhitrattanapisal S, Tipanee J and
Janvilisri T: Meta-analysis of gene expression profiles identifies
differential biomarkers for hepatocellular carcinoma and
cholangiocarcinoma. Tumour Biol. 37:12755–12766. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shirakami Y, Lee S-A, Clugston RD and
Blaner WS: Hepatic metabolism of retinoids and disease
associations. Biochim Biophys Acta. 1821:124–136. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Bert F, Bellier C, Lassel L, Lefranc V,
Durand F, Belghiti J, Mentre F and Fantin B: Risk factors for
Staphylococcus aureus infection in liver transplant
recipients. Liver Transplant. 11:1093–1099. 2005. View Article : Google Scholar
|
|
39
|
Sarris ME, Moulos P, Haroniti A,
Giakountis A and Talianidis I: Smyd3 Is a Transcriptional
Potentiator of Multiple Cancer-Promoting Genes and Required for
Liver and Colon Cancer Development. Cancer Cell. 29:354–366. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jansen MPHM, Sas L, Sieuwerts AM, Van
Cauwenberghe C, Ramirez-Ardila D, Look M, Ruigrok-Ritstier K,
Finetti P, Bertucci F, Timmermans MM, et al: Decreased expression
of ABAT and STC2 hallmarks ER-positive inflammatory breast cancer
and endocrine therapy resistance in advanced disease. Mol Oncol.
9:1218–1233. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kaulsay KK, Zhu T, Bennett W, Lee KO and
Lobie PE: The effects of autocrine human growth hormone (hGH) on
human mammary carcinoma cell behavior are mediated via the hGH
receptor. Endocrinology. 142:767–777. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gründemann D, Schechinger B, Rappold GA
and Schömig E: Molecular identification of the
corticosterone-sensitive extraneuronal catecholamine transporter.
Nat Neurosci. 1:349–351. 1998. View
Article : Google Scholar : PubMed/NCBI
|
|
43
|
Vollmar J, Lautem A, Closs E, Schuppan D,
Kim YO, Grimm D, Marquardt JU, Fuchs P, Straub BK, Schad A, et al:
Loss of organic cation transporter 3 (Oct3) leads to enhanced
proliferation and hepatocarcinogenesis. Oncotarget.
8:115667–115680. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Alvarellos ML, Sangkuhl K, Daneshjou R,
Whirl-Carrillo M, Altman RB and Klein TE: PharmGKB summary: Very
important pharmacogene information for CYP4F2. Pharmacogenet
Genomics. 25:41–47. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Gandhi AV, Saxena S, Relles D, Sarosiek K,
Kang CY, Chipitsyna G, Sendecki JA, Yeo CJ and Arafat HA:
Differential expression of cytochrome P450 omega-hydroxylase
isoforms and their association with clinicopathological features in
pancreatic ductal adenocarcinoma. Ann Surg Oncol. 20 (Suppl
3):S636–S643. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Eun HS, Cho SY, Lee BS, Seong I-O and Kim
K-H: Profiling cytochrome P450 family 4 gene expression in human
hepatocellular carcinoma. Mol Med Rep. 18:4865–4876.
2018.PubMed/NCBI
|
|
47
|
Tsunedomi R, Iizuka N, Hamamoto Y,
Uchimura S, Miyamoto T, Tamesa T, Okada T, Takemoto N, Takashima M,
Sakamoto K, et al: Patterns of expression of cytochrome P450 genes
in progression of hepatitis C virus-associated hepatocellular
carcinoma. Int J Oncol. 27:661–667. 2005.PubMed/NCBI
|
|
48
|
López-Terrada D, Cheung SW, Finegold MJ
and Knowles BB: Hep G2 is a hepatoblastoma-derived cell line. Hum
Pathol. 40:1512–1515. 2009. View Article : Google Scholar
|
|
49
|
Ma Q: Role of nrf2 in oxidative stress and
toxicity. Annu Rev Pharmacol Toxicol. 53:401–426. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Li R, Jia Z and Zhu H: Regulation of Nrf2
Signaling. React Oxyg Species (Apex). NIH Public Access. 8:312–322.
2019.
|
|
51
|
Zhang M, Zhang C, Zhang L, Yang Q, Zhou S,
Wen Q and Wang J: Nrf2 is a potential prognostic marker and
promotes proliferation and invasion in human hepatocellular
carcinoma. BMC Cancer. 15:531–12. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Ryoo I-G, Choi B-H, Ku S-K and Kwak M-K:
High CD44 expression mediates p62-associated NFE2L2/NRF2 activation
in breast cancer stem cell-like cells: Implications for cancer stem
cell resistance. Redox Biol. 17:246–258. 2018. View Article : Google Scholar : PubMed/NCBI
|