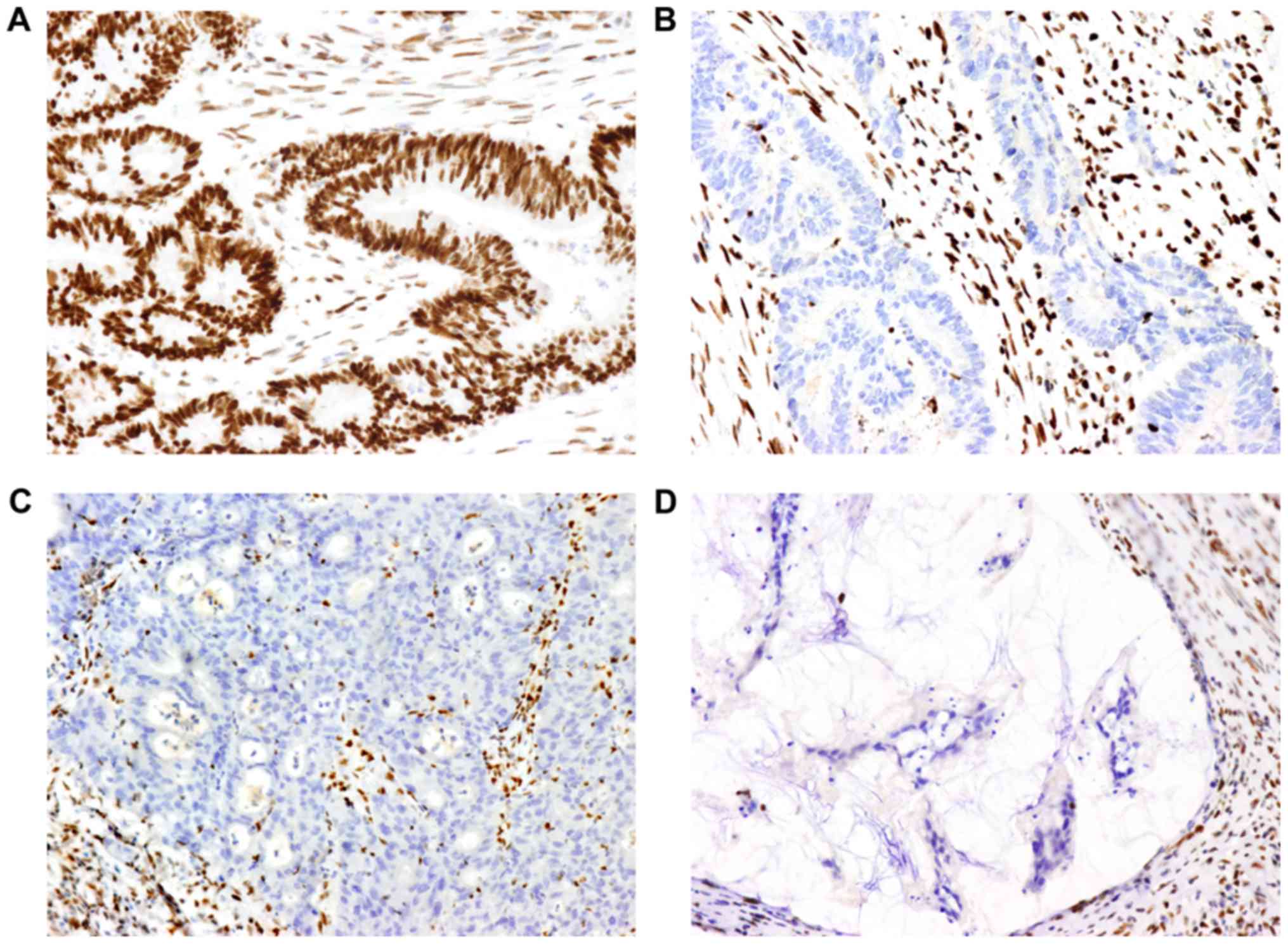
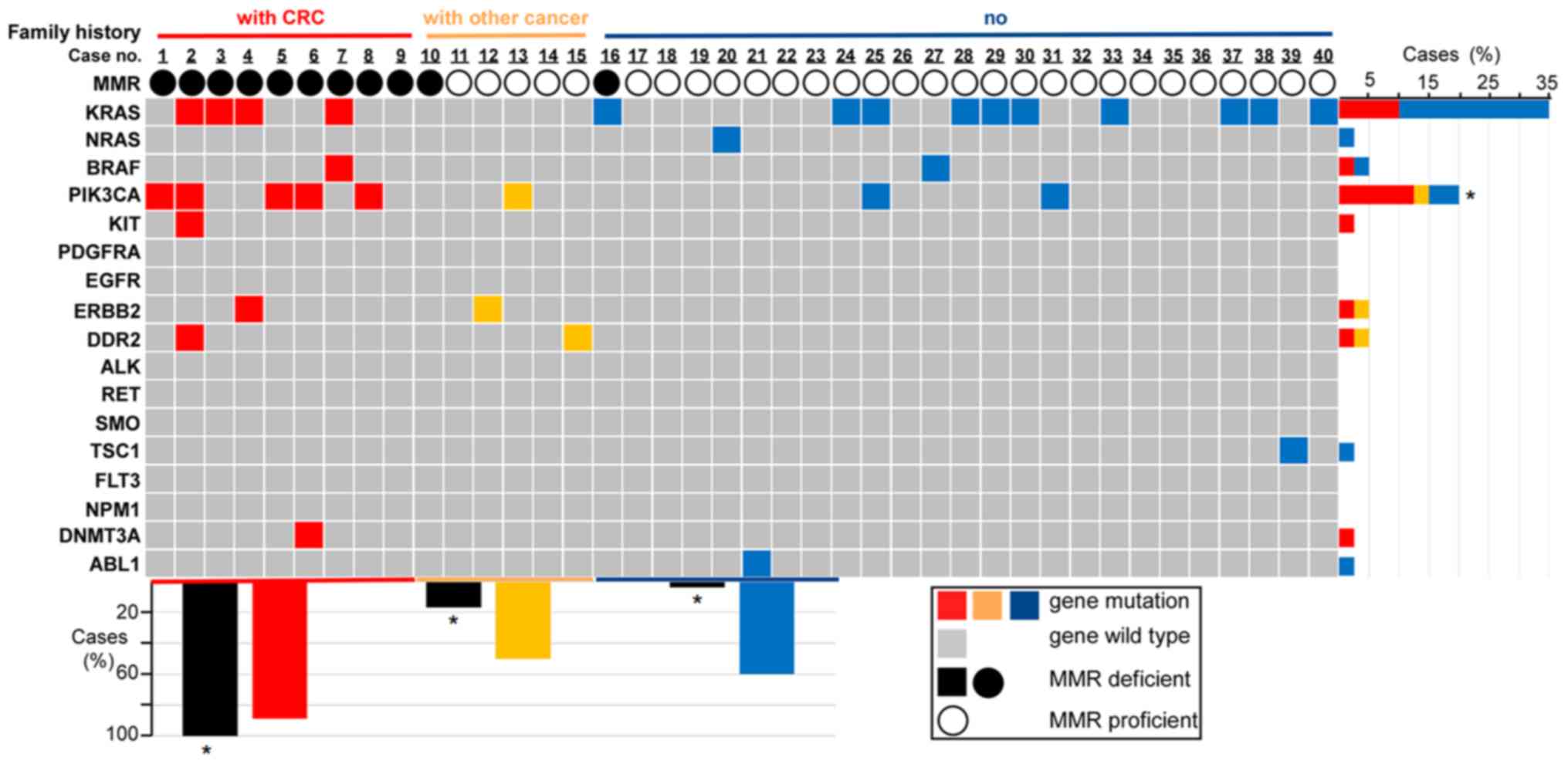
|
1
|
Stigliano V, Sanchez-Mete L, Martayan A
and Anti M: Early-onset colorectal cancer: A sporadic or inherited
disease? World J Gastroenterol. 20:12420–12430. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Singh KE, Taylor TH, Pan CG, Stamos MJ and
Zell JA: Colorectal cancer incidence among young adults in
California. J Adolesc Young Adult Oncol. 3:176–184. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bailey CE, Hu CY, You YN, Bednarski BK,
Rodriguez-Bigas MA, Skibber JM, Cantor SB and Chang GJ: Increasing
disparities in the age-related incidences of colon and rectal
cancers in the United States, 1975–2010. JAMA Surg. 150:17–22.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ,
Meester RGS, Barzi A and Jemal A: Colorectal cancer statistics,
2017. CA Cancer J Clin. 67:177–193. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vuik FE, Nieuwenburg SA, Bardou M,
Lansdorp-Vogelaar I, Dinis-Ribeiro M, Bento MJ, Zadnik V, Pellisé
M, Esteban L, Kaminski MF, et al: Increasing incidence of
colorectal cancer in young adults in Europe over the last 25 years.
Gut. 68:1820–1826. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Al-Barrak J and Gill S: Presentation and
outcomes of patients aged 30 years and younger with colorectal
cancer: A 20-year retrospective review. Med Oncol. 28:1058–1061.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mork ME, You YN, Ying J, Bannon SA, Lynch
PM, Rodriguez-Bigas MA and Vilar E: High prevalence of hereditary
cancer syndromes in adolescents and young adults with colorectal
cancer. J Clin Oncol. 33:3544–3549. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Siegel RL, Jakubowski CD, Fedewa SA, Davis
A and Azad NS: Colorectal Cancer in the Young: Epidemiology,
prevention, management. Am Soc Clin Oncol Educ Book. 40:1–14.
2020.PubMed/NCBI
|
|
9
|
Yantiss RK, Goodarzi M, Zhou XK, Rennert
H, Pirog EC, Banner BF and Chen YT: Clinical, pathologic, and
molecular features of early-onset colorectal carcinoma. Am J Surg
Pathol. 33:572–582. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Smith G, Carey FA, Beattie J, Wilkie MJ,
Lightfoot TJ, Coxhead J, Garner RC, Steele RJ and Wolf CR:
Mutations in APC, Kirsten-ras, and p53-alternative genetic pathways
to colorectal cancer. Proc Natl Acad Sci USA. 99:9433–9438. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vogelstein B, Fearon ER, Hamilton SR, Kern
SE, Preisinger AC, Leppert M, Nakamura Y, White R, Smits AM and Bos
JL: Genetic alterations during colorectal-tumor development. N Engl
J Med. 319:525–532. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Okugawa Y, Grady WM and Goel A: Epigenetic
alterations in colorectal cancer: Emerging biomarkers.
Gastroenterology. 149:1204–1225 e12. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Peltomaki P and de la Chapelle A:
Mutations predisposing to hereditary nonpolyposis colorectal
cancer. Adv Cancer Res. 71:93–119. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Haraldsdottir S, Hampel H, Tomsic J,
Frankel WL, Pearlman R, de la Chapelle A and Pritchard CC: Colon
and endometrial cancers with mismatch repair deficiency can arise
from somatic, rather than germline, mutations. Gastroenterology.
147:1308–1316 e1. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
National Comprehensive Cancer Network, .
Colon cancer treatment guidelines. October
19–2018
|
|
16
|
National Comprehensive Cancer Network, .
Rectal cancer treatment guidelines. August
7–2018
|
|
17
|
Shaw AT and Engelman JA: ALK in lung
cancer: Past, present, and future. J Clin Oncol. 31:1105–1111.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Perez-Carbonell L, Ruiz-Ponte C, Guarinos
C, Alenda C, Paya A, Brea A, Egoavil CM, Castillejo A, Barbera VM,
Bessa X, et al: Comparison between universal molecular screening
for Lynch syndrome and revised Bethesda guidelines in a large
population-based cohort of patients with colorectal cancer. Gut.
61:865–872. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xu Z, Huo X, Ye H, Tang C, Nandakumar V,
Lou F, Zhang D, Dong H, Sun H, Jiang S, et al: Genetic mutation
analysis of human gastric adenocarcinomas using ion torrent
sequencing platform. PLoS One. 9:e1004422014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cancer Genome Atlas Network: Comprehensive
molecular characterization of human colon and rectal cancer.
Nature. 487:330–337. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chang DT, Pai RK, Rybicki LA, Dimaio MA,
Limaye M, Jayachandran P, Koong AC, Kunz PA, Fisher GA, Ford JM, et
al: Clinicopathologic and molecular features of sporadic
early-onset colorectal adenocarcinoma: An adenocarcinoma with
frequent signet ring cell differentiation, rectal and sigmoid
involvement, and adverse morphologic features. Mod Pathol.
25:1128–1139. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
O'Connell JB, Maggard MA, Liu JH, Etzioni
DA, Livingston EH and Ko CY: Do young colon cancer patients have
worse outcomes? World J Surg. 28:558–562. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ballester V, Rashtak S and Boardman L:
Clinical and molecular features of young-onset colorectal cancer.
World J Gastroenterol. 22:1736–1744. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li SKH and Martin A: Mismatch repair and
colon cancer: Mechanisms and therapies explored. Trends Mol Med.
22:274–289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Douillard JY, Oliner KS, Siena S,
Tabernero J, Burkes R, Barugel M, Humblet Y, Bodoky G, Cunningham
D, Jassem J, et al: Panitumumab-FOLFOX4 treatment and RAS mutations
in colorectal cancer. N Engl J Med. 369:1023–1034. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shen Y, Wang J, Han X, Yang H, Wang S, Lin
D and Shi Y: Effectors of epidermal growth factor receptor pathway:
The genetic profiling of KRAS, BRAF, PIK3CA, NRAS mutations in
colorectal cancer characteristics and personalized medicine. PLoS
One. 8:e816282013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Samuels Y, Wang Z, Bardelli A, Silliman N,
Ptak J, Szabo S, Yan H, Gazdar A, Powell SM, Riggins GJ, et al:
High frequency of mutations of the PIK3CA gene in human cancers.
Science. 304:5542004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Grady WM and Carethers JM: Genomic and
epigenetic instability in colorectal cancer pathogenesis.
Gastroenterology. 135:1079–1099. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ley TJ, Ding L, Walter MJ, McLellan MD,
Lamprecht T, Larson DE, Kandoth C, Payton JE, Baty J, Welch J, et
al: DNMT3A mutations in acute myeloid leukemia. N Engl J Med.
363:2424–2433. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yan XJ, Xu J, Gu ZH, Pan CM, Lu G, Shen Y,
Shi JY, Zhu YM, Tang L, Zhang XW, et al: Exome sequencing
identifies somatic mutations of DNA methyltransferase gene DNMT3A
in acute monocytic leukemia. Nat Genet. 43:309–315. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kim MS, Kim YR, Yoo NJ and Lee SH:
Mutational analysis of DNMT3A gene in acute leukemias and common
solid cancers. APMIS. 121:85–94. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li WL, Xiao MS, Zhang DF, Yu D, Yang RX,
Li XY and Yao YG: Mutation and expression analysis of the IDH1,
IDH2, DNMT3A, and MYD88 genes in colorectal cancer. Gene.
546:263–270. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Van Cutsem E, Cervantes A, Adam R, Sobrero
A, Van Krieken JH, Aderka D, Aranda Aguilar E, Bardelli A, Benson
A, Bodoky G, et al: ESMO consensus guidelines for the management of
patients with metastatic colorectal cancer. Ann Oncol.
27:1386–1422. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Al-Shamsi HO, Alhazzani W and Wolff RA:
Extended RAS testing in metastatic colorectal cancer-Refining the
predictive molecular biomarkers. J Gastrointest Oncol. 6:314–321.
2015.PubMed/NCBI
|
|
35
|
Chou CL, Chang SC, Lin TC, Chen WS, Jiang
JK, Wang HS, Yang SH, Liang WY and Lin JK: Differences in
clinicopathological characteristics of colorectal cancer between
younger and elderly patients: An analysis of 322 patients from a
single institution. Am J Surg. 202:574–582. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bai Y, Kim JY, Watters JM, Fang B, Kinose
F, Song L, Koomen JM, Teer JK, Fisher K, Chen YA, et al: Adaptive
responses to dasatinib-treated lung squamous cell cancer cells
harboring DDR2 mutations. Cancer Res. 74:7217–7228. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Heinrich MC, Corless CL, Demetri GD,
Blanke CD, von Mehren M, Joensuu H, McGreevey LS, Chen CJ, Van den
Abbeele AD, Druker BJ, et al: Kinase mutations and imatinib
response in patients with metastatic gastrointestinal stromal
tumor. J Clin Oncol. 21:4342–4349. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Soverini S, Hochhaus A, Nicolini FE,
Gruber F, Lange T, Saglio G, Pane F, Muller MC, Ernst T, Rosti G,
et al: BCR-ABL kinase domain mutation analysis in chronic myeloid
leukemia patients treated with tyrosine kinase inhibitors:
Recommendations from an expert panel on behalf of European Leukemia
Net. Blood. 118:1208–1215. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
McConechy MK, Talhouk A, Li-Chang HH,
Leung S, Huntsman DG, Gilks CB and McAlpine JN: Detection of DNA
mismatch repair (MMR) deficiencies by immunohistochemistry can
effectively diagnose the microsatellite instability (MSI) phenotype
in endometrial carcinomas. Gynecol Oncol. 137:306–310. 2015.
View Article : Google Scholar : PubMed/NCBI
|