Introduction
Breast cancer is currently the most common cancer
among Chinese women (1,2). For a number of women with breast
cancer, breast lumpectomy with intraoperative pathological
assessment is the preferred treatment to make an intraoperative
decision (3). Breast cancer surgery
performed under general anesthesia is associated with a potential
risk of complications, which may make the patient feel unpleasant
and delay patient recovery after surgery (4,5). Minor
breast surgery requires fast and effective local anesthetic
techniques with minimal side effects to allow the patient to
recover quickly (6,7). Local anesthesia alone can make patients
feel uncomfortable and distressed during surgery (8,9). Thus,
the application of conscious sedation techniques may reduce the
need for local anesthesia.
Dexmedetomidine is a highly selective
α2-adrenoreceptor agonist that induces sedation
providing improved hemodynamic stability without eliciting
respiratory depression (10–13). Previous studies have reported that
dexmedetomidine is rapidly and efficiently absorbed after
intranasal administration, and is better tolerated compared with
intravenous administration (14–17).
Additionally, intranasal dexmedetomidine has been successfully used
for conscious sedation under local anesthesia in numerous minor
surgeries, including neurotologic procedures and dental surgery
(18–20). In addition, intranasal administration
of dexmedetomidine has been investigated in studies involving
children, which demonstrated that intranasal administration may be
a feasible alternative in patients requiring light sedation
(21,22); however, more attention should be paid
to the differences in dexmedetomidine dosage between children and
adults (15–17,20–22). A
number of studies have reported the application of intravenous
dexmedetomidine in breast lumpectomy (23–25).
Based on the similar efficacy between intravenous and intranasal
administration, the present study hypothesized that intranasal
dexmedetomidine may be effective in breast lumpectomy procedures.
In order to evaluate the efficacy and identify the optimal dose of
intranasal dexmedetomidine for conscious sedation, and to make
patients more comfortable and cooperative during surgery, the
modified Observer's Assessment of Alertness/Sedation (OAA/S) score
(22), bispectral index (BIS) and
pain were monitored. Additionally, systolic blood pressure (SBP),
heart rate (HR), oxygen saturation (SpO2) and
respiratory rate (RR) were monitored to evaluate the side effects
during breast lumpectomy under local anesthesia. The optimal
dexmedetomidine dose that yielded the best sedation and the least
adverse effects was also investigated.
Materials and methods
Study design
The present prospective randomized, double-blinded,
placebo-controlled, single-center study was approved by the Ethics
Committee of Tianjin Medical University Cancer Institute &
Hospital (approval no. bc201512; Tianjin, China), and was
registered at ClinicalTrials.gov (trial registration no.
NCT02675049). Participants were enrolled during February and March
2016 from Tianjin Medical University Cancer Institute &
Hospital, and written informed consent was obtained. All procedures
were performed in accordance with the ethical standards of the
responsible committee on human experimentation (institutional and
national) and with the Helsinki Declaration of 1975, as revised in
2000.
Inclusion criteria
Patients included in the present study were women
aged between 20 and 60 years. Patients with ASA (American Society
of Anesthesiologists) physical status I and II (26) who were scheduled for breast
lumpectomy at the Tianjin Medical University Cancer Institute and
Hospital were enrolled in the study. A total of 100 patients were
recruited.
Exclusion criteria
The exclusion criteria were a history of heart
block, upper respiratory tract infection, asthma, allergy to
dexmedetomidine or local anesthetics, memory or cognitive
dysfunction, pregnancy, lack of understanding of the consent
process, impaired liver or renal function, hypertension, concurrent
application of β-receptor blockers, and a history of drug or
alcohol abuse. Baseline demographic (age and body weight) and
clinical ASA status characteristics were recorded.
Intervention
The patients were instructed to fast at least 6 h
prior to surgery. No premedication was administered and patients
were sent to the induction room 1 h prior to surgery. The patients
were monitored routinely for electrocardiography, SpO2,
non-invasive blood pressure and BIS. Prior to dexmedetomidine
administration, the operative, sedation and pain assessment
procedures were explained. The patients were assigned randomly
before surgery on the basis of a computer-generated random number
table (complete randomization) at a 1:1:1:1 allocation ratio to
receive 1, 1.5 or 2 µg.kg−1 dexmedetomidine or 0.9%
saline (placebo) intranasally 45 min before surgery. Both the
patients and the investigators were blinded to the randomized
intervention. An independent investigator and an anesthesiologist
who were unaware of patient allocation prepared and administered
the drug or placebo. A parenteral preparation of 100
µg.ml−1 dexmedetomidine (Ai Bei Ning; Jiangsu Heng-rui
Medicine Co., Ltd.) was used without further dilution. An
equivalent volume of the placebo (0.9% saline) or undiluted
dexmedetomidine was administered evenly by bilateral nasal dripping
while the patients were in a recumbent position ~45 min prior to
surgery. All surgical procedures were performed by the same
surgical team comprising of three surgeons. Local anesthetic was
administered by local infiltration with 1% lidocaine ~5 min before
surgery and the volume was recorded. Inadequate analgesia was
managed by local anesthetic infiltration into the surgical
site.
Measurements and outcomes
The primary outcome was OAA/S, which was measured
after drug administration, during surgery and during recovery in
the post-anesthesia care unit (PACU). A score of 4 or 5 was
considered the optimal OAA/S score in our study group. The
secondary outcomes were BIS, pain [scored using a numerical rating
scale (NRS)] (27), vital signs
(SBP, HR, SpO2 and RR), adverse effects and satisfaction
with sedation. Adverse effects included hypotension (defined as SBP
<90 mmHg), bradycardia (defined as HR <50 bpm), oxygen
desaturation (defined as SpO2 <92%), respiratory
depression (defined as a ventilatory frequency <10/min), nausea
and vomiting. The surgical condition, graded by the surgeon and
patient, has been described in a previous study (28).
Baseline data were recorded before the commencement
of surgery. The observation indices were measured 15, 30 and 45 min
after drug administration, at which point resection commenced. The
measurements were then taken every 5 min during surgery, and every
10 min after surgery in the PACU.
Sample size
The sample size was calculated according to the
previous study by Yuen et al (29). The mean modified OAA/S scores for the
1 and 1.5 µg.kg−1 dexmedetomidine groups in the present
study were 5.2 and 4.6, respectively, and the standard deviation
(SD) was 0.5, which led to a standardized difference of 1.2 SDs
between the 1 and 1.5 µg.kg−1 groups. Based on this
result, the present study needed to have ~90% power to detect a
1.2-SD difference between any two of the three dexmedetomidine
groups using two-sided Student's t-tests conducted using a
Bonferroni-adjusted P<0.0167 significance level. Calculations
using PASS2011 software (version 11.0.10; NCSS, LLC) demonstrated
that 20 subjects per group provide the two-sided Student's t-test
with 89.6% power at a P<0.0167 significance level, thereby
satisfying power requirements. To maintain ~90% power in the event
of a 20% dropout rate, the sample size was increased to 25 subjects
per group for a total of 100 study subjects.
Statistical analysis
All statistical analyses were performed using SPSS
software version 18.0 (SPSS Inc). Continuous variables following a
normal distribution were expressed as mean ± SD and analyzed using
Student's t-test. Categorical variables and continuous variables
following an abnormal distribution were expressed as median and
interquartile range, and were assessed using Mann-Whitney U test.
To detect differences between the groups in terms of primary and
secondary outcomes, a two-step procedure was employed. First, the
placebo and 1 µg.kg−1 dexmedetomidine groups were
compared in terms of OAA/S, BIS, NRS, SBP, HR, RR or frequency of
‘good’ surgical conditions. If the difference was not statistically
significant (P>0.05), no further analysis was performed.
However, if P<0.05, the three dexmedetomidine groups were
compared with each other. P<0.0167 (Bonferroni-adjusted P-value)
was considered to indicate a statistically significant
difference.
Results
Baseline characteristics
A total of 100 hundred patients were separated into
four groups, with each group consisting of 25 patients. The mean
(SD) age of the patients was 37.5 (11.5), 41.1 (10.6), 41.9 (7.0)
and 42.3 (11.1) years, and the mean weight was 62.5 (11.5), 59.7
(8.7), 58.5 (7.3) and 60.2 (12.7) kg in the placebo group and 1,
1.5, and 2 µg.kg−1 dexmedetomidine groups, respectively
(Table I). No significant difference
in demographic data and clinical features was observed among the
groups (Table I). All patients met
the eligibility criteria and completed the study (Fig. 1). The mean (SD) time between
dexmedetomidine administration and the start of surgery was 47.4
(10.8), 52.4 (24.3), 47.2 (20.0) and 43.7 (13.6) min in the placebo
group and 1, 1.5, and 2 µg.kg−1 dexmedetomidine groups,
respectively (P=0.397; data not shown).
 | Table I.Baseline demographic and clinical
characteristics and intraoperative aspects. |
Table I.
Baseline demographic and clinical
characteristics and intraoperative aspects.
|
Characteristics | Placebo (n=25) | 1
µg.kg−1 dexmedetomidine (n=25) | 1.5
µg.kg−1 dexmedetomidine (n=25) | 2
µg.kg−1 dexmedetomidine (n=25) |
|---|
| Age, years | 37.5±11.5 | 41.1±10.6 | 41.9±7.0 | 42.3±11.1 |
| Body weight,
kg | 62.5±11.5 | 59.7±8.7 | 58.5±7.3 | 60.2±12.7 |
| ASA |
|
|
|
|
| I | 10 | 12 | 13 | 14 |
| II | 15 | 13 | 12 | 11 |
| Preoperative
BIS | 97.1±2.1 | 92.3±18.1 | 96.4±2.6 | 96.4±1.8 |
| Preoperative
OAA/S | 6.0 | 6.0 | 6.0 | 6.0 |
| Preoperative
NRS | 0 | 0 | 0 | 0 |
| Duration from DEX
to surgery, min | 47.4±11.8 | 52.4±24.3 | 47.1±20.0 | 43.7±13.6 |
| Duration of
surgery, min | 25.6±9.6 | 24.3±8.9 | 28.6±13.0 | 30.3±16.4 |
| Total volume-local
anesthetic used, ml | 31.3±15.1 | 27.7±15.6 | 29.6±19.4 | 29.3±18.7 |
| Surgical conditions
graded ‘good’a | 18 (72.0%) | 23 (92.0%) | 25 (100.0%) | 24 (96.0%) |
Outcomes
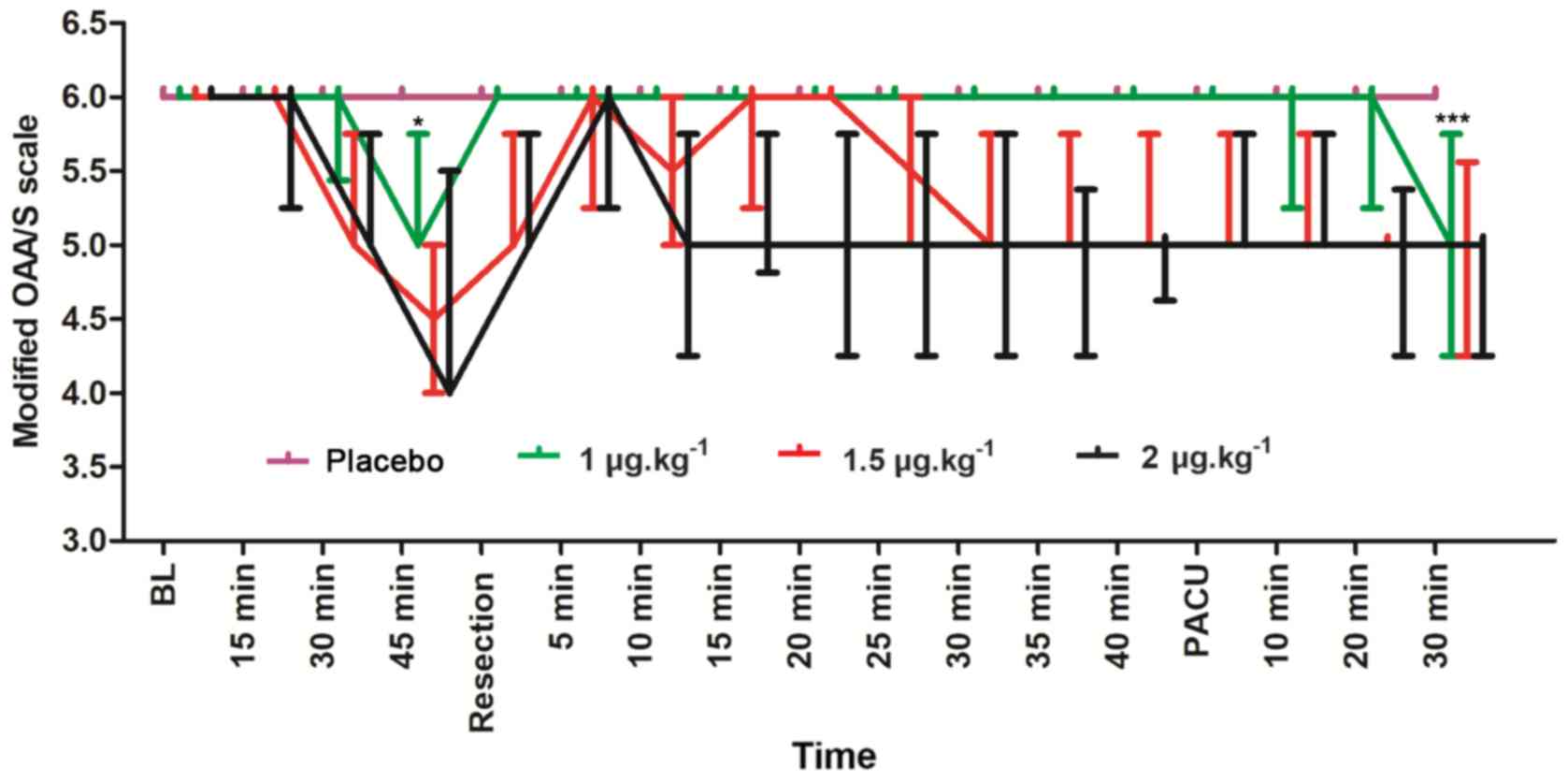
The modified OAA/S scores of the four groups at
different time intervals before and after administration of the
drug were investigated (Fig. 2). The
modified OAA/S scores were significantly lower in the 1
µg.kg−1 group compared with the placebo group 45 min
after intranasal (U=204.0; P=0.0003) and after 30 min in the PACU
(U=516.5; P<0.001). The 2 µg.kg−1 group exhibited
greater sedation compared with the 1.5 µg.kg−1 group but
these differences did not have any statistical significance
(U=68.0; P=0.300) at 45 min after intranasal administration; also,
no significant difference was observed in the level of sedation
between the 1.5 and 1 µg.kg−1 groups (U=59.5; P=0.026)
at 45 min after intranasal administration.
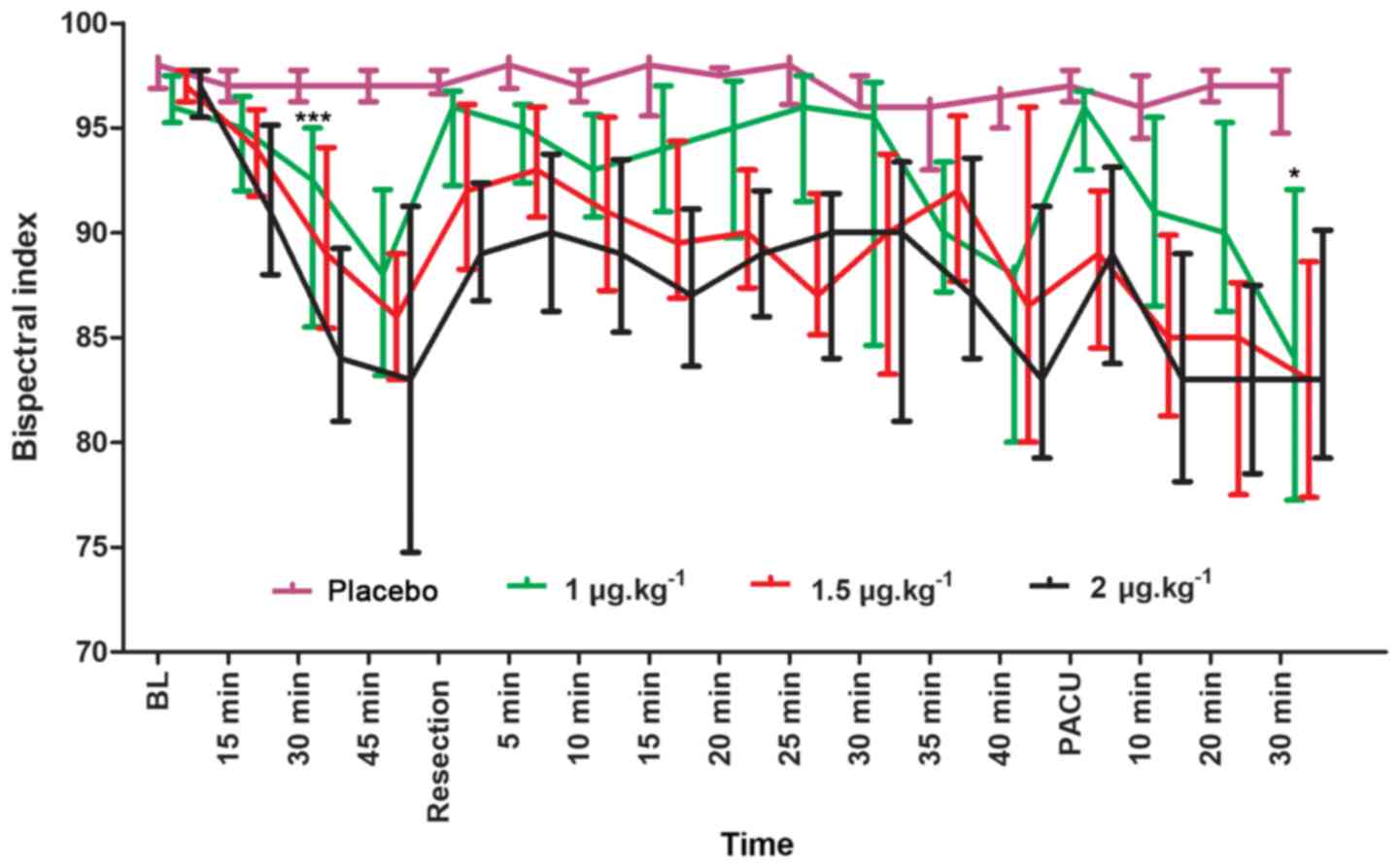
The BIS scores between the placebo group and other
groups were compared using Mann-Whitney U test. Compared with that
of the placebo group, the 1 µg.kg−1 group had
significantly lower BIS scores at 30 min after intranasal
administration (BIS=91; U=383.0; P<0.001) and after 30 min in
the PACU (BIS=84; U=327.0; P=0.002; Fig.
3). Additionally, the BIS scores of the 1 µg.kg−1
group were significantly greater compared with those of the 1.5 and
2 µg.kg−1 groups 30 min after resection (U=34.5;
P=0.004; and U=36.5; P=0.002, respectively); however, there was no
significant difference at 30 min in the PACU (U=203.5; P=0.596; and
U=208.5; P=0.684, respectively). In addition, no significant
difference was observed between the BIS scores of the 1.5 and 2
µg.kg−1 groups.
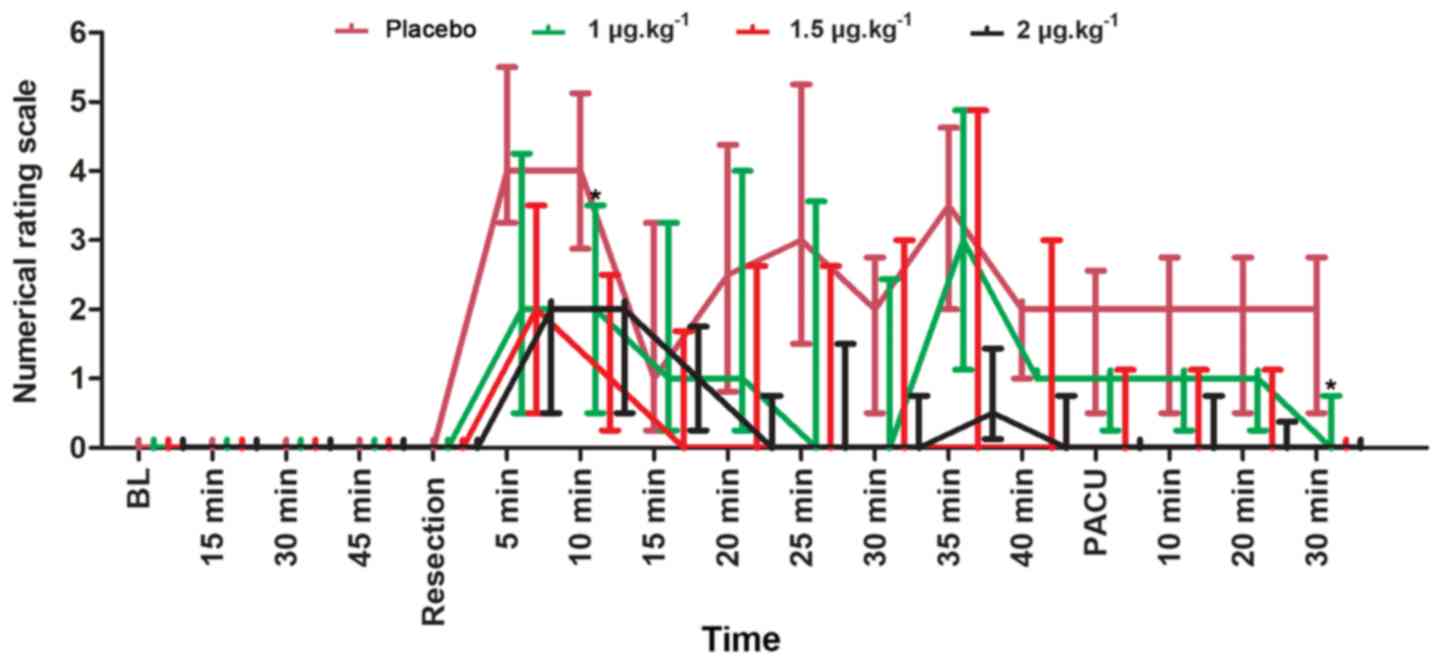
The median pain NRS scores at each sample time were
collected and presented in Fig. 4.
The patients in the placebo group had a significantly higher pain
score compared with patients in the 1 µg.kg−1 group 10
min into surgery (U=436.5; P=0.015) and after 30 min in the PACU
(U=405.0; P=0.012; Fig. 4). The NRS
scores in the 1 µg.kg−1 group were comparable to those
in the 1.5 and 2 µg.kg−1 groups 10 min into surgery
(U=262.5; P=0.318; and U=254.0; P=0.245, respectively) and after 30
min in the PACU (U=194.5; P=0.079; and U=223.0; P=0.037,
respectively; Fig. 4). In addition,
no significant differences were observed in the volume of local
anesthetic administered between the four groups (P=0.280, Table I).
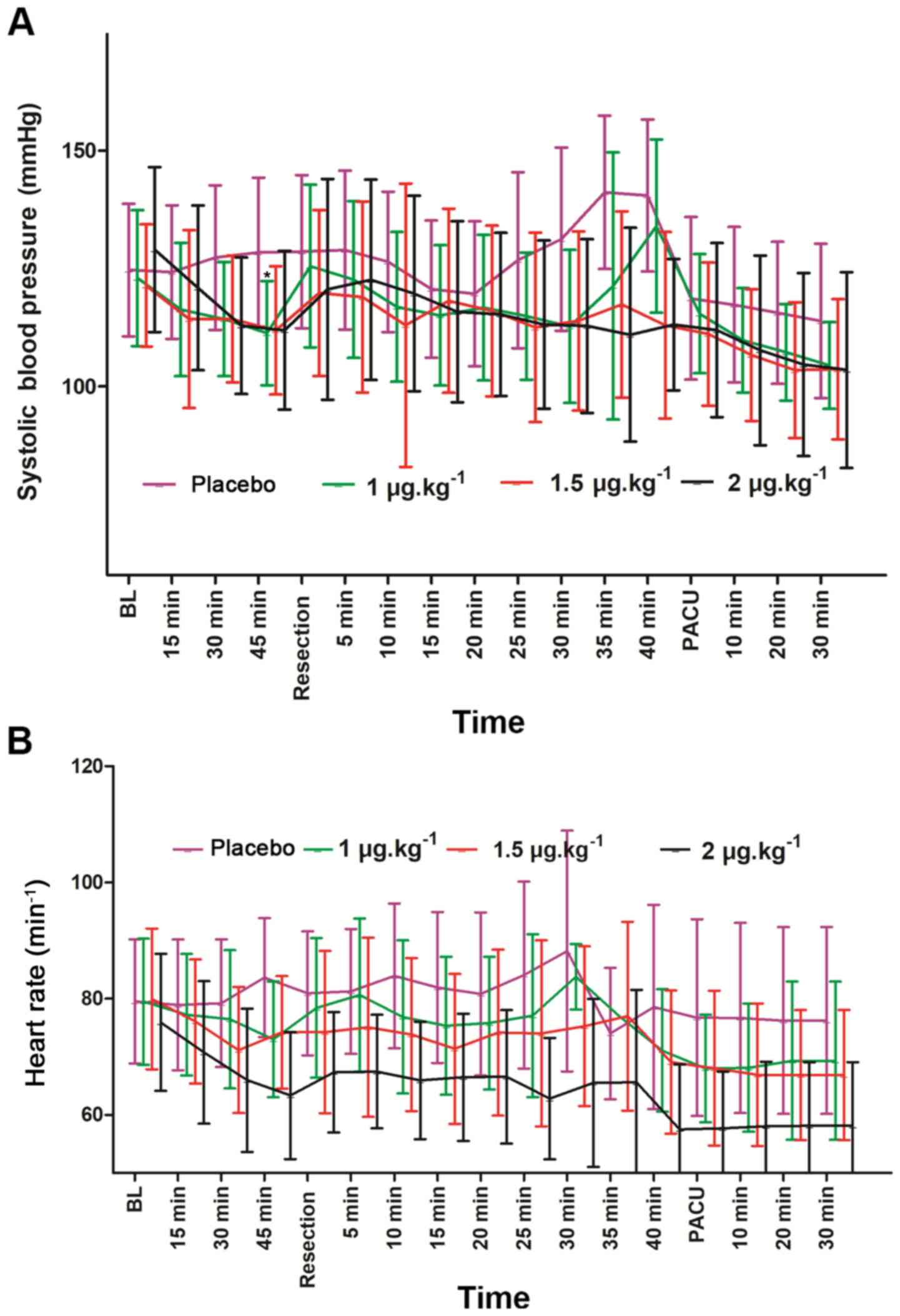
The three dexmedetomidine groups exhibited
significant decreases in mean value of SBP during surgery and
recovery compared with the baseline values (Fig. 5A). In addition, compared with the
placebo group, the SBP of the 1 µg.kg−1 group was
significantly lower 45 min after intranasal dexmedetomidine
administration (P=0.001). Additionally, all three dexmedetomidine
groups exhibited significantly lower HR 45 min after drug
administration and after 30 min in the PACU compared with the
control group (Fig. 5B). In
addition, compared with the placebo group, the 1 µg.kg−1
group had similar HR reduction during surgery and recovery
(Fig. 5B).
Dexmedetomidine administration had no significant
effect on SpO2 levels or RR compared with the baseline
values (P>0.05) and no significant differences were observed in
the SpO2 and RR between the four groups (data not
shown). In addition, the intranasal administration was well
tolerated by all patients; none of them developed local irritation
or pain due to the administration, or complained of any unpleasant
smell or taste associated with intranasal drug or placebo
administration.
Safety outcomes
Only the 2 µg.kg−1 group developed
bradycardia (4% of total patients) and significant hypotension (1%
of total patients) after dexmedetomidine administration. Most
patients were asymptomatic and remained untreated. Only one patient
with bradycardia required treatment with 0.5 mg atropine. The other
two dexmedetomidine groups did not develop bradycardia or
hypotension. No significant differences were observed in the
incidence of nausea and vomiting between the four groups (P=0.286;
data not shown).
Patient satisfaction
In the dexmedetomidine groups, the patients
expressed more comfortable experiences during surgery, which were
indicative of satisfactory sedation (P=0.157). The surgeons graded
the surgical conditions as ‘good’ (indicating adequate sedation)
more frequently in the dexmedetomidine groups compared with the
placebo group (72%; P=0.066), but no significant differences were
observed between the dexmedetomidine groups (P=0.768, Table I).
Discussion
The present study evaluated the efficacy and dose of
combined intranasal dexmedetomidine and local anesthesia for
conscious sedation during breast lumpectomy. Patients who received
dexmedetomidine were significantly more sedated, experienced less
pain, and were more satisfied with the sedation compared with
patients in the placebo group. In addition, the results of the
present study suggested that 1.5 µg.kg−1 was the optimal
dose of intranasal dexmedetomidine, which yielded satisfactory
sedation with good surgical condition, patient sedation
satisfaction, and no serious adverse events.
One of the main causes of postoperative
complications is general anesthesia, which is frequently used for
sedation, and causes nausea and vomiting in 50% of cases (7). Local anesthesia may be an alternative
to general anesthesia for this type of surgery (5). Dexmedetomidine is an effective drug for
conscious sedation in patients who undergo minor surgery (14,17,30,31). A
previous study demonstrated that intranasal dexmedetomidine was
noninvasive and more tolerable compared with intravenous
administration (14). In addition,
the intranasal route is safe, effective, comfortable and convenient
(14,22,32–34).
Iirola et al (28)
demonstrated that administration of intranasal dexmedetomidine had
a high bioavailability of 65% (35-93%) and could potentially have
useful sedative effects in surgical procedures. To the best of our
knowledge, to date there is no study that has reported the efficacy
of intranasal dexmedetomidine for conscious sedation during breast
lumpectomy under local anesthesia. The present study demonstrated
that patients who received intranasal dexmedetomidine exhibited
improved clinical sedation and analgesia compared with patients in
the placebo group. In addition, the surgeons considered that the
surgical conditions in the dexmedetomidine groups were superior
compared with those in the placebo group. It was also demonstrated
that intranasal dexmedetomidine could provide good clinical
analgesia and sedation for a long duration of time of up to at
least 70 min after surgery had started.
To identify the optimal intranasal dose providing
the best sedation while inducing minimal side effects, three
different intranasal doses were tested in the present study. These
doses (1, 1.5 and 2 µg.kg−1) were chosen on the basis of
previous studies (29,31,35).
However, only a few studies have assessed intranasal
dexmedetomidine in adults (14,16,17).
Because of the different requirements of anesthesia and surgery
between children and adults, and as the selected doses were based
on previous studies in adults, the dose of dexmedetomidine for
sedation may be different (15–17,20–22,31,32). The
results of the present study demonstrated that there was a
dose-dependent increase in sedation levels when dexmedetomidine was
given intranasally, which was consistent with the findings of
previous studies (25,36). In the current study, the level of
sedation was significantly greater in the dexmedetomidine groups
compared with that in the placebo group. Notably, Yuen et al
(29) also demonstrated that 1 and
1.5 µg.kg−1 intranasal dexmedetomidine produced
clinically significant sedation in healthy volunteers. In addition,
patients receiving 1 µg.kg−1 intranasal dexmedetomidine
for unilateral third molar surgery with local anesthesia were more
sedated perioperatively with greater postoperative pain relief
compared with intranasal water (17). Additionally, Zhang et al
(37) reported that 1
µg.kg−1 intranasal dexmedetomidine appeared to be safe
and efficacious for patients undergoing elective
electrochemotherapy for facial vascular malformations. The present
study demonstrated that the 1.5 µg.kg−1 group achieved
greater sedation compared with the 1 µg.kg−1 group,
although it did not reach a statistically significant difference.
In addition, the 2 µg.kg−1 group also did not achieve a
significant difference compared with the 1.5 µg.kg−1
group. However, 1.5 µg.kg−1 may be the optimal dose even
if there were no significant differences.
The NRS pain scores also indicated that the
dexmedetomidine groups experienced greater analgesia compared with
the placebo group, as they felt less pain and were more tolerant.
Mohta et al (38) reported
that paravertebral block using intravenous dexmedetomidine in
patients undergoing breast cancer surgery provided greater
analgesia. Dexmedetomidine administration has been reported to
result in significant bradycardia and hypotension (39). When dexmedetomidine was administered
in adults before stimulation or intervention (i.e., surgery), it
attenuated the hemodynamic response to stimulation and reduced
arterial blood pressure and HR (40). The present study observed that,
regardless of dose, dexmedetomidine significantly decreased SBP
during surgery and recovery compared with that of the placebo
group, but this hemodynamic change did not cause patient
discomfort. There was also a significant drop in HR compared with
the baseline values at the same time point in the dexmedetomidine
groups. Although the 1 µg.kg−1 group had a similar HR to
the placebo group during surgery and recovery, the two higher dose
groups had a lower HR during surgery and/or recovery. These changes
may account for the relatively high rates of profound hypotension
and bradycardia in the 2 µg.kg−1 group. Notably, the
most frequently reported adverse events associated with
dexmedetomidine treatment are hypotension and bradycardia (41). Although patients in the 2
µg.kg−1 group achieved a relatively greater level of
sedation and analgesia, the cardiovascular side effects rendered
this dose suboptimal. Since the optimal dose should yield
sufficient sedation and analgesia without such adverse effects, the
results of the present study suggested that the optimal dose of
dexmedetomidine for intranasal administration may be 1.5
µg.kg−1. Notably, patients who received 1.5
µg.kg−1 dexmedetomidine remained aware during surgery
and tolerated the procedure well, which allowed patients to
cooperate with the surgeon to perform various required
behaviors.
The present study had some limitations. First, the
postoperative pain relief was not monitored after 30 min in the
PACU. Whether intranasal dexmedetomidine can enhance postoperative
pain relief under local anesthesia in breast lumpectomy requires
further investigation. Second, intranasal dexmedetomidine was
administered by dripping the solution into both nostrils with a 1
ml syringe. An optimal delivery system may have more consistent
results. One such system would be the mucosal atomization device
nasal spray that was used in a previous study to deliver intranasal
dexmedetomidine during third molar extraction (16). Third, although the results of the
present study together with previous study (42) indicated the efficacy and safety of
intranasal dexmedetomidine administration during breast lumpectomy,
future studies need to be conducted to focus on the safety in a
larger sample size. Fourth, individual differences in drug
tolerance may also provide bias since the time point for surgery
was 45 min after intranasal dexmedetomidine was selected according
to previous studies (14,28–29)
instead of plasma concentration.
In conclusion, patients undergoing breast lumpectomy
surgery who received intranasal 1.5 µg.kg−1
dexmedetomidine attained significant and satisfactory sedation
without experiencing any adverse effects compared with patients who
received 1 and 2 µg.kg−1 intranasal dexmedetomidine.
Future studies should be conducted to focus on the efficacy and
safety in a larger sample size.
Acknowledgements
The authors would like to thank Professor Jing-Bo
Zhai (Institute of Traditional Chinese Medicine, Tianjin University
of Traditional Chinese Medicine) for their assistance with the
statistical analysis.
Funding
This study was supported by the National Natural
Science Foundation of China (grant no. 81501140).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HWZ and YJY conceptualized the study and analyzed
data. YJY, PZ, FX, XBZ, SSH, DYG and YHX acquired and analyzed the
data. YJY, PZ, FX, XBZ, SSH, DYG, YHX and HWZ wrote the manuscript.
HWZ and YJY revised the manuscript from a critical perspective for
important intellectual content. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
This study was approved by the Ethics Committee of
Tianjin Medical University Cancer Institute & Hospital
(approval no. bc201512) and was registered at ClinicalTrials.gov (registration no. NCT02675049).
Written informed consent to participate was obtained from the
patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
PACU
|
post-anesthesia care unit
|
|
CONSORT
|
Consolidated Standards of Reporting
Trials
|
|
OAA/S
|
Observer's Assessment of
Alertness/Sedation
|
|
ASA
|
American Society of
Anesthesiologists
|
|
BIS
|
bispectral index
|
|
NRS
|
numerical rating scale
|
|
SBP
|
systolic blood pressure
|
|
HR
|
heart rate
|
|
SpO2
|
oxygen saturation
|
|
RR
|
respiratory rate
|
|
ANOVA
|
analysis of variance
|
References
|
1
|
Hong W and Dong E: The past, present and
future of breast cancer research in China. Cancer Lett. 35:1–5.
2014. View Article : Google Scholar
|
|
2
|
Li H, Zheng RS, Zhang SW, Zeng HM, Sun KX,
Xia CF, Yang ZX, Chen WQ and He J: Incidence and mortality of
female breast cancer in China, 2014. Zhonghua Zhong Liu Za Zhi.
40:166–171. 2018.(In Chinese). PubMed/NCBI
|
|
3
|
Tevis SE, Neuman HB, Mittendorf EA, Kuerer
HM, Bedrosian I, DeSnyder SM, Thompson AM, Black DM, Scoggins ME,
Sahin AA, et al: Multidisciplinary intraoperative assessment of
breast specimens reduces number of positive margins. Ann Surg
Oncol. 25:2932–2938. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sessler DI, Pei L, Huang Y, Fleischmann E,
Marhofer P, Kurz A, Mayers DB, Meyer-Treschan TA, Grady M, Tan EY,
et al: Recurrence of breast cancer after regional or general
anaesthesia: A randomised controlled trial. Lancet. 394:1807–1815.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pusch F, Freitag H, Weinstabl C, Obwegeser
R, Huber E and Wildling E: Single-injection paravertebral block
compared to general anaesthesia in breast surgery. Acta
Anaesthesiol Scand. 43:770–774. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Manfè AZ, Marchesini M, Bortolato A,
Feltracco P and Lumachi F: Ropivacaine versus levobupivacaine for
minor breast surgery in outpatients: Inversion of postoperative
pain relief efficacy. In Vivo. 26:1075–1077. 2012.PubMed/NCBI
|
|
7
|
Calì Cassi L, Biffoli F, Francesconi D,
Petrella G and Buonomo O: Anesthesia and analgesia in breast
surgery: The benefits of peripheral nerve block. Eur Rev Med
Pharmacol Sci. 21:1341–1345. 2017.PubMed/NCBI
|
|
8
|
FitzGerald S, Odor PM, Barron A and Pawa
A: Breast surgery and regional anaesthesia. Best Pract Res Clin
Anaesthesiol. 33:95–110. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Burgess C, Cornelius V, Love S, Graham J,
Richards M and Ramirez A: Depression and anxiety in women with
early breast cancer: Five year observational cohort study. BMJ.
330:7022005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sezen G, Demiraran Y, Seker IS, Karagoz I,
Iskender A, Ankarali H, Ersoy O and Ozlu O: Does premedication with
dexmedetomidine provide perioperative hemodynamic stability in
hypertensive patients? BMC Anesthesiol. 14:1132014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tang C, Hu Y, Zhang Z, Wei Z, Wang H, Geng
Q, Shi S, Wang S, Wang J and Chai X: Dexmedetomidine with
sufentanil in intravenous patient-controlled analgesia for relief
from postoperative pain, inflammation, and delirium after
esophageal cancer surgery. Biosci Rep. 40:BSR201934102020.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kang MH, Lee HJ, Lim YJ, Jeon YT, Hwang JW
and Park HP: Preoperative dexmedetomidine attenuates hemodynamic
responses to hydrodissection in patients undergoing robotic
thyroidectomy. J Anesth. 29:191–197. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kang R, Jeong JS, Ko JS, Lee SY, Lee JH,
Choi SJ, Cha S and Lee JJ: Intraoperative dexmedetomidine
attenuates norepinephrine levels in patients undergoing
transsphenoidal surgery: A randomized, placebo-controlled trial.
BMC Anesthesiol. 20:1002020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Poonai N, Spohn J, Vandermeer B, Ali S,
Bhatt M, Hendrikx S, Trottier ED, Sabhaney V, Shah A, Joubert G and
Hartling L: Intranasal dexmedetomidine for procedural distress in
children: A systematic review. Pediatrics. 145:e201916232020.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yang F, Liu Y, Yu Q, Li S, Zhang J, Sun M,
Liu L, Lei Y, Tian Q, Liu H and Tu S: The role of parents in
reporting sedation outcomes in our analysis of 17 948 pediatric
patients undergoing procedural sedation with a combination of
intranasal dexmedetomidine and ketamine. Paediatr Anaesth.
29:12082019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cheung CW, Ng KF, Liu J, Yuen MY, Ho MH
and Irwin MG: Analgesic and sedative effects of intranasal
dexmedetomidine in third molar surgery under local anaesthesia. Br
J Anaesth. 107:430–437. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nooh N, Sheta SA, Abdullah WA and
Abdelhalim AA: Intranasal atomized dexmedetomidine for sedation
during third molar extraction. Int J Oral Maxillofac Surg.
42:857–862. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Svrakic M, Pollack A, Huncke TK and Roland
JT Jr: Conscious sedation and local anesthesia for patients
undergoing neurotologic and complex otologic procedures. Otol
Neurotol. 35:e277–e285. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu S, Wang Y, Zhu Y, Yu T and Zhao H:
Safety and sedative effect of intranasal dexmedetomidine in
mandibular third molar surgery: A systematic review and
meta-analysis. Drug Des Devel Ther. 13:1301–1310. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mondardini MC, Amigoni A, Cortellazzi P,
Di Palma A, Navarra C, Picardo SG, Puzzutiello R, Rinaldi L, Vitale
F, Marinosci GZ and Conti G: Intranasal dexmedetomidine in
pediatrics: Update of current knowledge. Minerva Anestesiol.
85:1334–1345. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Messeha MM and El-Morsy GZ: Comparison of
intranasal dexmedetomidine compared to midazolam as a premedication
in pediatrics with congenital heart disease undergoing cardiac
catheterization. Anesth Essays Res. 12:170–175. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gupta A, Dalvi NP and Tendolkar BA:
Comparison between intranasal dexmedetomidine and intranasal
midazolam as premedication for brain magnetic resonance imaging in
pediatric patients: A prospective randomized double blind trial. J
Anaesthesiol Clin Pharmacol. 33:236–340. 2017.PubMed/NCBI
|
|
23
|
Goyal S, Gupta KK and Mahajan V: A
comparative evaluation of intravenous dexmedetomidine and fentanyl
in breast cancer surgery: A prospective, randomized, and controlled
trial. Anesth Essays Res. 11:611–616. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nakanishi T, Yoshimura M and Toriumi T:
Pectoral nerve II block, transversus thoracic muscle plane block,
and dexmedetomidine for breast surgery in a patient with
achondroplasia: A case report. JA Clin Rep. 5:472019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mohamed SA, Fares KM, Mohamed AA and
Alieldin NH: Dexmedetomidine as an adjunctive analgesic with
bupivacaine in paravertebral analgesia for breast cancer surgery.
Pain Physician. 17:E589–E598. 2014.PubMed/NCBI
|
|
26
|
Mak PH, Campbell RC and Irwin MG; American
Society of Anesthesiologists, : The ASA physical status
classification: Inter-observer consistency. American society of
anesthesiologists. Anaesth Intensive Care. 30:633–640. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wikström L, Nilsson M, Broström A and
Eriksson K: Patients' self-reported nausea: Validation of the
numerical rating scale and of a daily summary of repeated numerical
rating scale scores. J Clin Nurs. 28:959–968. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Iirola T, Vilo S, Manner T, Aantaa R,
Lahtinen M, Scheinin M and Olkkola KT: Bioavailability of
dexmedetomidine after intranasal administration. Eur J Clin
Pharmacol. 67:825–831. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yuen VM, Irwin MG, Hui TW, Yuen MK and Lee
LH: A double-blind, crossover assessment of the sedative and
analgesic effects of intranasal dexmedetomidine. Anesth Analg.
105:374–380. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang HM, Shi XY, Zhou JL and Xia YF:
Comparison of dexmedetomidine and propofol for conscious sedation
in inguinal hernia repair: A prospective, randomized, controlled
trial. J Int Med Res. 45:533–539. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Miller JW, Divanovic AA, Hossain MM,
Mahmoud MA and Loepke AW: Dosing and efficacy of intranasal
dexmedetomidine sedation for pediatric transthoracic
echocardiography: A retrospective study. Can J Anaesth. 63:834–841.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sheta SA, Al-Sarheed MA and Abdelhalim AA:
Intranasal dexmedetomidine vs. midazolam for premedication in
children undergoing complete dental rehabilitation: A
double-blinded randomized controlled trial. Paediatr Anaesth.
24:181–189. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Plambech MZ and Afshari A: Dexmedetomidine
in the pediatric population: A review. Minerva Anestesiol.
81:320–332. 2015.PubMed/NCBI
|
|
34
|
Jia JE, Chen JY, Hu X and Li WX: A
randomised study of intranasal dexmedetomidine and oral ketamine
for premedication in children. Anaesthesia. 68:944–949. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ryu DS, Lee DW, Choi SC and Oh IH:
Sedation protocol using dexmedetomidine for third molar extraction.
J Oral Maxillofac Surg. 74:926.e1–7. 2016. View Article : Google Scholar
|
|
36
|
Ebert TJ, Hall JE, Barney JA, Uhrich TD
and Colinco MD: The effects of increasing plasma concentrations of
dexmedetomidine in humans. Anesthesiology. 93:382–394. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhang X, Bai X, Zhang Q, Wang X and Lu L:
The safety and efficacy of intranasal dexmedetomidine during
electrochemotherapy for facial vascular malformation: A
double-blind, randomized clinical trial. J Oral Maxillofac Surg.
71:1835–1842. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Mohta M, Kalra B, Sethi AK and Kaur N:
Efficacy of dexmedetomidine as an adjuvant in paravertebral block
in breast cancer surgery. J Anesth. 30:252–260. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Su F and Hammer GB: Dexmedetomidine:
Pediatric pharmacology, clinical uses and safety. Expert Opin Drug
Saf. 10:55–66. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Scholz J and Tonner PH:
Alpha2-adrenoceptor agonists in anaesthesia: A new paradigm. Curr
Opin Anaesthesiol. 13:437–442. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Piao G and Wu J: Systematic assessment of
dexmedetomidine as an anesthetic agent: A meta-analysis of
randomized controlled trials. Arch Med Sci. 10:19–24. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Das R, Das RK, Sahoo S and Nanda S: Role
of dexmedetomidine as an anaesthetic adjuvant in breast cancer
surgery as a day-care procedure: A randomised controlled study.
Indian J Anaesth. 62:182–187. 2018. View Article : Google Scholar : PubMed/NCBI
|