Introduction
The highest incidence of colorectal cancer (CRC) is
observed in certain European countries, Australia, New Zealand,
Northern America and East Asia (1).
The global burden of CRC is expected to persist at least until the
year 2035 (2,3). The highest number of cancer-associated
deaths are caused by cancers of the lungs, prostate gland, and
colon/rectum in males and cancers of the lungs, breasts and
colon/rectum in females (4). CRC is
the most commonly diagnosed cancer type in Taiwan (5). Despite significant improvements
achieved with multidisciplinary treatment, CRC remains one of the
major causes of cancer-associated mortality. Of note, the death
rate for CRC decreased by 53% from 1970 to 2016 (4).
A growing body of evidence supports the concept of
perioperative treatment for CRC. Concurrent chemoradiation (CCRT)
followed by surgery and adjuvant chemotherapy has become the
standard treatment for locally advanced rectal cancer (6). An analysis of the National Cancer
Database (NCDb) revealed that neoadjuvant radiotherapy (RT) for
clinical T4 stage (cT4) disease may be associated with superior R0
resection rates and improved overall survival (OS) (7). The clinical T4b classification refers
to advanced tumors with direct invasion of adjacent structures and
those tumors are unresectable (8).
Excision repair cross-complementing (ERCC) genes
encode proteins involved in a complex DNA repair mechanism that is
responsible for the removal of DNA lesions and the maintenance of
chromosome stability. ERCC1 gene polymorphisms have been
investigated as a potential predictive biomarkers of the efficacy
of oxaliplatin and platinum treatment in various cancer types
(9). Increased ERCC1 expression is
associated with clinical resistance to platinum-based chemotherapy.
In a previous study by our group, ERCC1 overexpression was
indicated to be an independent predictor of poor disease-free
survival (DFS) and OS in patients with stage III CRC who received
adjuvant 5-fluorouracil/leucovorin/oxaliplatin (FOLFOX) (10). Despite advancements in aggressive
surgery and combination chemotherapy, the prognosis of cT4b CRC
remains poor, with a 5-year survival rate of 15.7–38.5% (11–14).
Therefore, identifying potential prognostic biomarkers and
optimizing the therapeutic sequence administered to patients with
cT4b CRC to improve clinical outcomes are crucial. Whether
neoadjuvant treatment for cT4b CRC improves the resectability of
tumors and patient survival has remained to be fully determined. A
previous study by our group demonstrated that neoadjuvant
chemoradiotherapy is feasible and safe with a prominent pathologic
complete response in locally advanced colon cancer (15). In the present study, its capacity to
convert the tumor into a resectable tumor was comprehensively
studied for patients with cT4b CRC after neoadjuvant CCRT.
Materials and methods
Patients
The present study assessed patients with
unresectable cT4b and clinical nodal stage N1-2 (cN1-2) CRC between
February 2015 and March 2019 at Kaohsiung medical university
hospital. A total of 20 consecutive patients were treated with
preoperative CCRT by using the FOLFOX regimen (15). The resectability of their tumors was
assessed through imaging techniques such as CT, after 6 to 12
cycles of FOLFOX with duration of 12 to 24 weeks. Only patients who
did not respond to this first-line CCRT regimen were included in
the present study. This poor response was not expected. However,
all patients exhibited a poor response to CCRT and all tumors were
still unresectable after CCRT with FOLFOX. Therefore, the
chemotherapy regimen was changed to irinotecan plus
fluorouracil/leucovorin (FOLFIRI) after FOLFOX-based CCRT (16). The clinical course of the patients
was followed up from the time of cancer diagnosis through to their
last available clinical record. Genetic alterations, such as
microsatellite instability (MSI) and ERCC1 and ERCC2 expression,
were examined. Clinicopathological variables, treatment outcomes
and adverse events were also analyzed. Patients were included in
this study if they had pathologically proven colorectal
adenocarcinoma and a clinical diagnosis of cT4b with cN1 or cN2.
Patients were excluded if they had a history of prior pelvic
irradiation or malignancies other than CRC. A total of two patients
were excluded because one underwent prior pelvic irradiation for
cervical cancer and the other one underwent the same for prostate
cancer. Furthermore, patients with distant metastasis were also not
included in this study. Patient follow-ups were performed by visits
to the clinic until the end of March 2020.
All of the patients underwent pretreatment workups
comprising a physical examination, a history review, chest
radiography, bronchoscopy with tumor biopsy, contrast-enhanced CT
or MRI and routine laboratory tests. The tumor stage was classified
according to the seventh edition of the Cancer Staging Manual and
Handbook of the American Joint Committee on Cancer (8). The Response Evaluation Criteria in
Solid Tumors version 1.1 (RECIST 1.1) guidelines, which are also
routinely used in clinical practice, were used to evaluate the CRC
tumor response (17,18). Side effects were graded according to
the Common Terminology Criteria for Adverse Events, version 3.0
(CTCAE) (http://ctep.cancer.gov/reporting/ctc.html). The
following variables (patient characteristics) were recorded: Age,
sex, tumor location, initial clinical tumor and nodal
classification, tumor size, ERCC1 and ERCC2 overexpression levels
and MSI status in the cancerous specimen by diagnostic biopsy, best
objective response according to the RECIST criteria and adverse
events.
Ethics approval statement
The present study was approved by the ethical and
research committee of Kaohsiung Medical University Hospital
[approval no. KMUHIRB-E(I)-20190182]. This study was conducted in
compliance with institutional review board regulations in
accordance with the Helsinki Declaration of 1975 as revised in
1983. All patients provided written informed consent for the
addition of their sample to the collection, use for scientific
research and added to a specimen bank; patient information was
anonymized prior to the analysis.
Immunohistochemical (IHC) staining of
ERCC1 and ERCC2
All incubations were performed at room temperature
unless otherwise specified. Formalin-fixed and paraffin-embedded
tissue blocks of samples from each patient were used to obtain
4-µm-thick sections, and the sections were deparaffinized in xylene
(10,19). They were then rehydrated in a graded
alcohol series (100, 95 and 75%). Each rehydration step was
performed for 1 min at room temperature. Next, the sections were
washed with tap water for 5 min at room temperature. Antigen
retrieval was performed using target retrieval solution (pH 9.0;
DAKO) in an autoclave (121°C, 1.2 kg/cm2) for 10 min,
and endogenous peroxidase was blocked in the sections by incubating
them in 3% hydrogen peroxide for 5 min. Finally, for antigen
retrieval, the sections were immersed in citrate buffer (ERCC1: pH
9.0, ERCC2: pH 9.0) prior to immunostaining at room temperature.
The sections were incubated for 15 min at room temperature with
antibodies against ERCC1 (dilution, 1/25; cat. no. #ab2356; Abcam)
and ERCC2 (dilution, 1/250; #ab111596; Abcam). Next, the samples
were treated with the DAKO REAL EnVision Detection System-HRP
(DAKO) for 30 min. Finally, the sections were incubated in
3′,3-diaminobenzidine for 5 min, followed by Mayer's hematoxylin
counterstaining, and dehydration was then performed through 2
changes of 95% ethanol and 2 changes of 100% ethanol. Subsequently,
the samples were cleared in 3 changes of xylene and then mounted on
slides for observation by using a microscope. Negative controls
were prepared by replacing the primary antibody with distilled
water.
To improve the accuracy and reduce interobserver
differences, the immunostaining of ERCC1 and ERCC2 was scored by
two independent pathologists (CYC and YTC). They evaluated slides
and scored the extent of immunostaining through light microscopy.
They analyzed gene expression based on the intensity of IHC
staining and the percentage of positive cancerous cells. Samples
with nuclear ERCC1 and cytoplasmic ERCC2 immunostaining were
considered positive. ERCC1 and ERCC2 overexpression was defined as
a score of 2 (positive staining in >50% of cells), whereas
absence of overexpression was defined as a score of 0 or 1.
Pre-operative CCRT and post-operative
chemotherapy or RT regimen
All of the patients received CCRT once the diagnosis
of stage IIIC CRC was confirmed. For pelvic RT, each patient was
placed in a customized thermoplastic immobilization cast. The
primary and boost beams were combined in a single integrated
treatment plan. A total of 19 patients received intensity-modulated
RT. The fractionation scheme was 45 Gy in 25 fractions to the
pelvic lymphatic drainage area with a simultaneous boost of 50 Gy
delivered to primary tumors and also to metastatic lymph nodes for
18 patients. Furthermore, one patient received 49.5 Gy in 27
fractions to the pelvic lymphatic drainage area with a simultaneous
boost of 54 Gy delivered to primary tumors and metastatic lymph
nodes. Three-dimensional conventional RT was delivered to one
patient with a 44 Gy/22 fraction to the pelvis with a boost of 6
Gy/3 fractions to the tumor. RT was delivered without
interruption.
The FOLFOX regimen followed a biweekly schedule
concurrent with RT. Each cycle of FOLFOX consisted of oxaliplatin
(85 mg/m2) through a 2-h infusion concurrently with
folinic acid (400 mg/m2) through a 2-h infusion on day 1
and 5-FU (2,800 mg/m2) through a 46-h infusion repeated
every 2 weeks. The FOLFIRI regimen after CCRT consisted of
leucovorin calcium (calcium folinate), 5-fluorouracil and
irinotecan. Each cycle of FOLFIRI consisted of irinotecan (180
mg/m2) through a 2-h infusion concurrently with folinic
acid (400 mg/m2) through a 2-h infusion on day 1 and
5-FU (2,800 mg/m2) through a 46-h infusion repeated
every 2 weeks. Furthermore, seven patients received bevacizumab
(Avastin; Roche) 5 mg/kg repeated every 2 weeks combined with
FOLFIRI.
After a median post-RT follow-up of 24.3 months
(range, 7.8–37 months), radical protectomy or colectomy was
performed. A total of five patients with negative pathological
margins were treated using capecitabine (Xeloda; Roche) at a dose
of 850 mg/m2 every 12 h between days 1 and 14. This
protocol was applied every 21 days in 6 to 8 cycles and
capecitabine was prescribed for up to 6 months. The other five
patients with negative pathological margins and were treated using
oral tegafur 100 mg-uracil 224 mg (UFUR; TTY Biopharm Co., Ltd.) at
a dose of one capsule three times a day for up to 6 months.
Adjuvant FOLFIRI was administered to four patients (three with
positive circumferential radial margin involvement and one with 0.5
cm circumferential radial margin).
Statistical analysis
Primary endpoints were OS and progression-free
survival (PFS). OS was defined as the period from the date of the
start of treatment to the date of death from any cause or until the
date of the last follow-up. PFS was measured from the start date of
treatment to the date of any type of progression or the final
follow-up. OS and PFS rates were assessed using the Kaplan-Meier
method. Statistical analyses were performed using the SPSS software
package, version 19.0 for Windows (IBM Corp.).
Results
Patient and treatment
characteristics
A total of 20 patients were retrospectively enrolled
after applying the inclusion and exclusion criteria. The median age
was 60.5 years (range, 34–75 years). Table I summarizes the clinical
characteristics of the 20 patients. All of the patients had
overexpression of ERCC2, which was detected using IHC staining.
Furthermore, 18 patients exhibited ERCC1 overexpression, whereas 2
patients did not exhibit ERCC1 overexpression. Only one patient
exhibited high MSI, two patients exhibited low MSI and 17 patients
(85%) exhibited microsatellite stability. According to the RECIST
criteria (16,17), 12 patients (60%) were categorized as
exhibiting partial response (PR), 2 patients (10%) achieved stable
disease and 4 patients had a complete response (CR) (20%). Only two
patients exhibited progressive disease (PD). Adverse events noted
during treatment were mostly CTCAE grade 1–2 toxicity. None of the
patients exhibited any grade 4 toxicity. When the patients
experienced CTCAE grade 3 neutropenia, dermatitis, diarrhea,
stomatitis, liver toxicity or painful paresthesia for up to 7 days,
the oxaliplatin dose was reduced by 25%. The median duration
between the last date of radiotherapy and surgery was 32.7 weeks
(range, 10.1–59.3 weeks). The median follow-up time in all patients
was 24.3 months (range, 7.8–37 months).
 | Table I.Demographic and clinicopathological
characteristics of 20 patients with cT4b colorectal cancer. |
Table I.
Demographic and clinicopathological
characteristics of 20 patients with cT4b colorectal cancer.
| Characteristic | Value |
|---|
| Age, years | 60.5 (34–75) |
| Sex |
|
|
Male | 14 (70) |
|
Female | 6 (30) |
| Tumor location |
|
|
Cecum | 1 (5) |
|
Ascending colon | 4 (20) |
|
Descending colon | 2 (10) |
| Sigmoid
colon | 4 (20) |
|
Rectosigmoid colon | 3 (15) |
|
Rectum | 6 (30) |
| Clinical T
classification |
|
|
cT4b | 20 (100) |
| Clinical N
classification |
|
|
cN1 | 7 (35) |
|
cN2 | 13 (65) |
| Tumor size
(cm) |
|
| ≤5 | 2 (10) |
|
>5 | 18 (90) |
| ERCC1
overexpression |
|
|
Yes | 18 (90) |
| No | 2 (10) |
| ERCC2
overexpression |
|
|
Yes | 20 (100) |
| No | 0 (0) |
| MSI |
|
|
High | 1 (5) |
|
Low | 2 (10) |
|
MSS | 17 (85) |
| Best objective
response (RECIST) |
|
|
Complete response | 4 (20) |
| Partial
response | 12 (60) |
| Stable
disease | 2 (10) |
|
Progressive disease | 2 (10) |
| Grade 1/2 adverse
events |
|
|
Nausea/vomiting | 20 (100) |
|
Diarrhea | 8 (40) |
| Skin
itchy rash | 1 (5) |
|
Leukopenia | 16 (80) |
|
Anemia | 17 (85) |
|
Thrombocytopenia | 6 (30) |
| Liver
toxicities | 7 (35) |
| Oral
mucositis | 2 (10) |
|
Peripheral neuropathy | 5 (25) |
| Grade 3 adverse
events |
|
| Skin
itchy rash | 1 (5) |
|
Leukopenia | 3 (15) |
| Liver
toxicity | 1 (5) |
Treatment outcomes and failure
patterns
Of the 20 inoperable patients, the tumors of 14
patients became resectable after the administration of FOLFIRI. Of
the seven patients who received bevacizumab combined with FOLFIRI,
three displayed neoplasms that were converted to resectable tumors,
while the remaining four patients who received bevacizumab combined
with FOLFIRI were still unresectable. A total of six patients
underwent radical proctectomy, 5 patients were treated using
laparoscopic hemicolectomy and 3 patients received laparoscopy
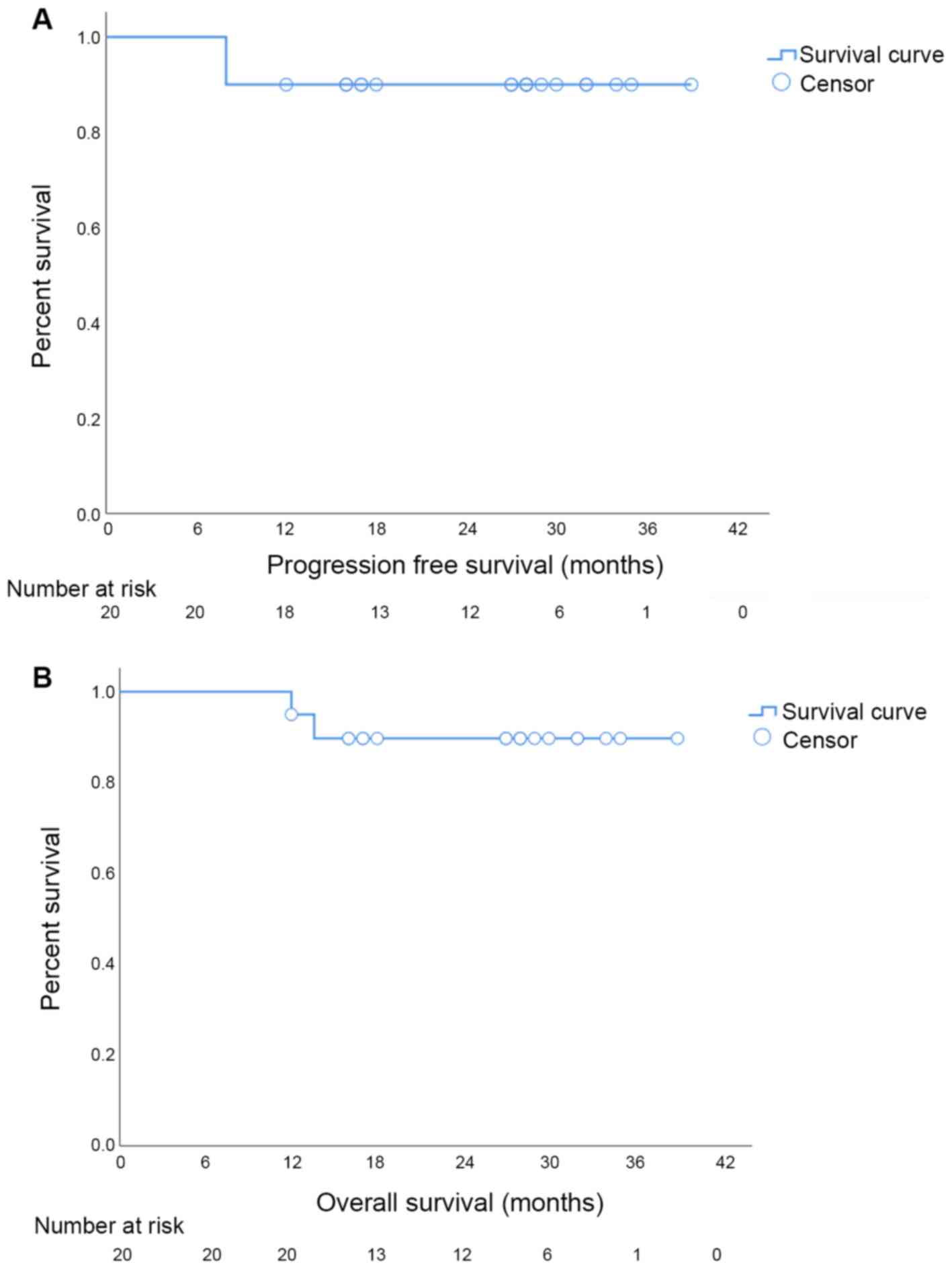
anterior resection. The median OS and PFS were 27.5 months (95% CI:
20.58–28.48) and 27.5 months (95% CI: 19.73–28.37), respectively
(Fig. 1). A total of two patients
developed distant metastases, but no local recurrence was noted.
Furthermore, 18 patients (90%) survived until the end of the
present study. The treatment sequence, OS and PFS are listed in
Table II. All patients received
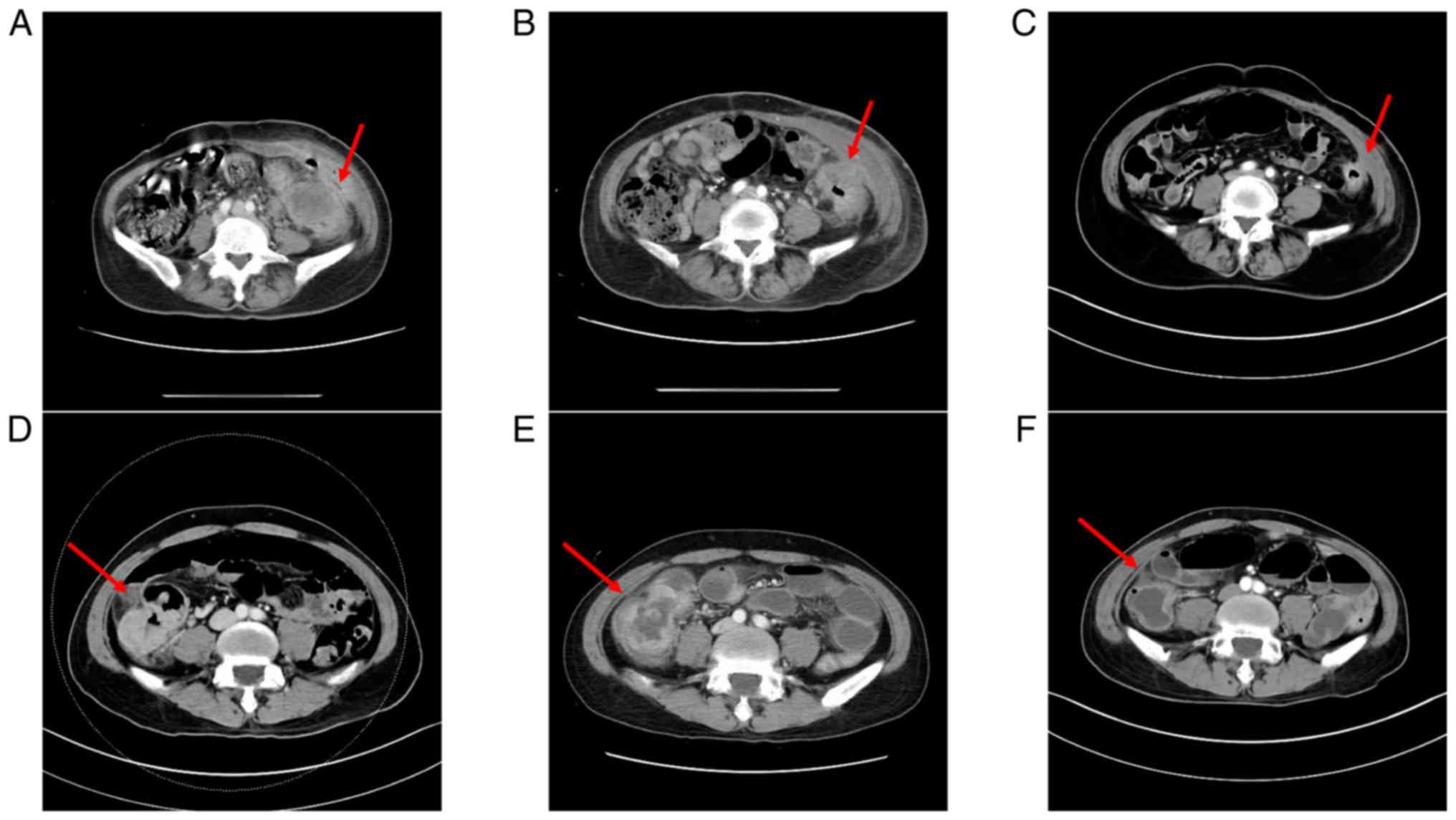
upfront CCRT with FOLFOX. As a poor response was noted on imaging
(Fig. 2), FOLFIRI was added after
CCRT.
 | Table II.Pathological and treatment evaluation
of 20 patients with cT4b colorectal cancer. |
Table II.
Pathological and treatment evaluation
of 20 patients with cT4b colorectal cancer.
| Case no. | Age, years/sex | Tumor location | Primary tumor
resection, yes vs. no | Time intervals
between RT completion date to surgery date, weeks | PFS, months | OS, months | Gene
overexpression | Survival,
yes/no |
|---|
| 1 | 48/M | Sigmoid | No | ND | 8 | 12 | ERCC1 (+)/ERCC2
(+) | No (died of
cancer) |
| 2 | 67/M | Rectum | Yes | 47 | 39 | 39 | ERCC1 (+)/ERCC2
(+) | Yes |
| 3 | 63/M | Rectum | Yes | 27.1 | 35 | 35 | ERCC1 (+)/ERCC2
(+) | Yes |
| 4 | 65/M | Rectum | No | ND | 34 | 34 | ERCC1 (+)/ERCC2
(+) | Yes |
| 5 | 61/M | Cecum | Yes | 13.1 | 32 | 32 | ERCC1 (+)/ERCC2
(+) | Yes |
| 6 | 41/M | Ascending | Yes | 59.3 | 32 | 32 | ERCC1 (+)/ERCC2
(+) | Yes |
| 7 | 60/M | Rectum | Yes | 52.9 | 30 | 30 | ERCC1 (−)/ERCC2
(+) | Yes |
| 8 | 61/M | Descending | No | ND | 8 | 14 | ERCC1 (+)/ERCC2
(+) | No (died of
cancer) |
| 9 | 56/M | Sigmoid | No | ND | 29 | 29 | ERCC1 (+)/ERCC2
(+) | Yes |
| 10 | 61/F | Descending | Yes | 35.1 | 28 | 28 | ERCC1 (+)/ERCC2
(+) | Yes |
| 11 | 56/F | Ascending | Yes | 13.4 | 28 | 28 | ERCC1 (−)/ERCC2
(+) | Yes |
| 12 | 75/M | Rectum | Yes | 39 | 27 | 27 | ERCC1 (+)/ERCC2
(+) | Yes |
| 13 | 39/M | Rectosigmoid | No | ND | 28 | 28 | ERCC1 (+)/ERCC2
(+) | Yes |
| 14 | 53/M | Sigmoid | Yes | 25.3 | 27 | 27 | ERCC1 (+)/ERCC2
(+) | Yes |
| 15 | 70/F | Ascending | Yes | 10.1 | 18 | 18 | ERCC1 (+)/ERCC2
(+) | Yes |
| 16 | 64/F | Sigmoid | Yes | 34.4 | 17 | 17 | ERCC1 (+)/ERCC2
(+) | Yes |
| 17 | 60/F | Rectosigmoid | No | ND | 17 | 17 | ERCC1 (+)/ERCC2
(+) | Yes |
| 18 | 34/F | Rectum | Yes | 30.9 | 16 | 16 | ERCC1 (+)/ERCC2
(+) | Yes |
| 19 | 68/M | Rectosigmoid | Yes | 35.9 | 16 | 16 | ERCC1 (+)/ERCC2
(+) | Yes |
| 20 | 34/M | Ascending | Yes | 29.3 | 12 | 12 | ERCC1 (+)/ERCC2
(+) | Yes |
Among the 14 patients who underwent surgery, 12
patients (85.7%) achieved a pathologic N0 status and 4 patients
achieved a pT0 status. Overall, 4 (28.6%) of the 14 cases that
became resectable after neoadjuvant treatment achieved pathologic
CR (pCR). A total of 12 patients (60%) exhibited an initial CEA
level of >5 ng/ml prior to treatment, but only 5 patients (25%)
exhibited a CEA level of >5 ng/ml at the last follow-up. Among
the 20 patients, after FOLFOX-based CCRT and the FOLFIRI regimen,
only one PD patient exhibited an increase of 21.2% in tumor size
(longest diameter × widest length) (Table III). The remaining 19 patients had
2.3–73.9% reduction in tumor size compared with the size prior to
FOLFIRI administration (Table
III).
 | Table III.Treatment outcomes in 20 patients
with cT4b colorectal cancer. |
Table III.
Treatment outcomes in 20 patients
with cT4b colorectal cancer.
| Case no. | Sex | Age, years | Pre-CCRT tumor
size, cm | Pre-FOLFIRI therapy
tumor size, cm | Post-FOLFIRI
therapy tumor size, cm | Tumor size
reduction, % (post-FOLFIRI minus pre-FOLFIRI) | Post-resection
tumor risk | Best overall
response (RECIST) |
|---|
| 1 | Male | 48 | 7.3×5.2 | 8.0×5.3 | 9.7×7.5 | +21.2 | Unresectable | PD |
| 2 | Male | 67 | 6.0×3.7 | 6.5×3.6 | 2.5×1.5 | −61.5 | Tumor margin
negative | PR |
| 3 | Male | 63 | 6.7×3.4 | 4.6×2.8 | 1.2×1.0 | −73.9 | Pathological
CR | CR |
| 4 | Male | 65 | 7.6×5.8 | 6.1×4.0 | 4.6×2.4 | −24.6 | Unresectable | SD |
| 5 | Male | 61 | 7.2×5.3 | 6.3×5.1 | 4.6×2.7 | −27.0 | Tumor margin
negative | PR |
| 6 | Male | 41 | 6.3×3.2 | 4.4×2.5 | 4.3×2.3 | −2.3 | Tumor margin
positive (CRM involved) | PR |
| 7 | Male | 60 | 10.9×9.6 | 6.8×5.5 | 3.7×3.2 | −45.6 | Tumor margin
negative | PR |
| 8 | Male | 61 | 6.0×5.2 | 5.3×3.5 | 3.7×3.1 | −30.2 | Unresectable (due
to peritoneal carcinomatosis) | PD |
| 9 | Male | 56 | 15.4×10.3 | 9.8×6.8 | 6.8×3.1 | −30.6 | Unresectable (due
to tumor invasion to urinary bladder and cecum persisting) | PR |
| 10 | Female | 61 | 8.9×4.8 | 7.5×2.7 | 3.0×1.3 | −60.0 | Tumor margin
positive (CRM involved) | PR |
| 11 | Female | 56 | 5.1×3.2 | 5.6×3.6 | 3.6×2.5 | −35.7 | Tumor margin
negative | PR |
| 12 | Male | 75 | 5.6×2.7 | 3.5×2.5 | 1.0×1.0 | −71.4 | Pathological
CR | CR |
| 13 | Male | 39 | 11.6×5.4 | 10.9×3.7 | 9.6×3.6 | −11.9 | Unresectable | SD |
| 14 | Male | 53 | 8.6×4.7 | 6.7×3.4 | 2.7×1.2 | −59.7 | Pathological
CR | CR |
| 15 | Female | 70 | 7.9×3.0 | 6.0×4.0 | 4.5×2.4 | −25.0 | Tumor margin
negative | PR |
| 16 | Female | 64 | 5.5×5.5 | 4.9×2.1 | 1.5×1.0 | −69.4 | Tumor margin
positive (CRM involved) | PR |
| 17 | Female | 60 | 7.9×2.8 | 6.5×2.7 | 5.1×2.3 | −21.5 | Unresectable | PR |
| 18 | Female | 34 | 5.5×3.5 | 4.8×1.9 | 3.2×1.2 | −33.0 | Tumor margin
negative (CRM 0.5 cm) | PR |
| 19 | Male | 68 | 4.0×3.5 | 5.4×3.0 | 3.0×0.9 | −44.0 | Pathological
CR | CR |
| 20 | Male | 34 | 3.9×1.2 | 4.9×3.0 | 2.5×1.2 | −49.0 | Tumor margin
negative | PR |
Discussion
T4b CRC is associated with a poor prognosis due to
the direct local extension or infiltration into surrounding
structures or organs, thus causing unresectability or a high
incidence of nodal and distant metastases (12,20,21). As
it involves adhesion to the adjacent organs, complete resection of
cT4b CRC is difficult, as the organs may be damaged during
resection. A growing body of clinical evidence suggests that the
combination of neoadjuvant chemotherapy, specifically pre-operative
CCRT, curative surgery and adjuvant chemotherapy, has a favorable
prognostic effect on patients with locally advanced CRC (22). In the present study, unresectable
tumors in 14 of the 20 patients with CRC (70%), who exhibited a
poor response toward CCRT with FOLFOX, were converted to resectable
tumors after undergoing FOLFIRI. Gao et al (13) analyzed the Surveillance, Epidemiology
and End Results (SEER) dataset between 1973 and 2008 in their
study. They determined an incidence of 4.4% for pathological T4b
CRC. Rectal cancers (21.3%) were reported to have a lower risk of
developing a real pathological invasion (pathological T4) than
colon cancers (48.8%) (23). The
principle of ‘en bloc’ resection is to resect all invaded organs
along with the colon tumor, which may be performed in 65–91.1% of
cases (23,24). For unresectable locally advanced
disease, pre-operative chemotherapy is delivered to reduce the size
of primary tumors and convert them to resectable tumors. The
randomized phase III FOxTROT trial of locally advanced, operable
colon cancer demonstrated that 3 cycles of pre-operative FOLFOX
resulted in significant downstaging compared with post-operative
chemotherapy (25). The patients
with cT4b colon cancer treated with neoadjuvant chemotherapy had a
23% lower risk of death at 3 years than the patients who received
post-operative chemotherapy (26).
As up to 45% of patients with T4 CRC develop distant metastases,
chemotherapy not only results in size regression but also
eradicates micrometastatic disease (27). Furthermore, up to 65% of patients
with T4 CRC develop nodal dissemination (28), and pre-operative CCRT enhances local
RT sensitization and eradication of micrometastases. For locally
advanced rectal cancer, pre-operative CCRT also has the potential
to increase the rate of sphincter preservation. In the present
study, 14 (70%) and 4 patients (20%) with both ERCC1 and ERCC2
overexpression reached PR and pCR after receiving FOLFIRI as a
second-line therapy, respectively. In the present study, the
conversion to resectability rate (70%) was higher than that
observed in previous studies. Population-based data from the SEER
dataset from January 1992 to December 2004 on 109,953 patients with
colon cancer were compared with NCDb data of 134,206 patients; the
5-year OS rate in the T4bN1-2 patients was 15.8–27.9% (14). Other studies have reported that the
5-year relative survival rate in T4b with nodal involvement was
15.7–38.5% (11–13). A negative resection margin (R0)
resulted in a 5-year local control rate for primary locally
advanced CRC of up to 89% and 5-year OS of up to 66% (29). In the present study, 11 out of 14
patients had R0 after conversion to resectability and only 3 had
positive resection margins.
The use of peri-operative pelvic RT in the treatment
of patients with stage III unresectable CRC continues to develop.
Well-established neoadjuvant RT protocols for treating T4 colon
tumors are not available (22). At
present, several cohorts of patients do not respond to
pre-operative therapies, resulting in inoperable status and poor
survival. The present study aimed to incorporate the detection of
ERCC expression for prescribing precision medicine. Pre-clinical
and clinical trials involving ERCC have demonstrated the importance
of understanding the pharmacogenetics prior to treatment (30–32).
Protein products of ERCC genes are responsible for
nucleotide excision repair (NER) of damaged DNA. Oxaliplatin
induces adducts, which are not processed by the mismatch repair
mechanism. They are predominantly repaired by components of the NER
and base excision repair pathways (33). NER removes a DNA segment with
adducts, followed by restoration of that DNA segment (34). ERCC1 is an excision nuclease in the
NER pathway that is involved in oxaliplatin metabolism (35). High ERCC1 expression levels are
associated with increased platinum drug resistance (36). Shirota et al (37) first reported that a high level of
intratumoral ERCC1 mRNA was associated with poor prognosis in CRC
patients who received oxaliplatin-based chemotherapy. A marked
increase in ERCC1 protein expression levels was also noted in
patients with C/T or T/T genotypes (70% vs. 20%; P<0.01), which
was associated with a significantly lower response to FOLFOX-4
(36.4% vs. 57.5%; P=0.01), shorter PFS (7 vs. 13 months; P<0.01)
and shorter OS (16 months vs. 25 months; P<0.01) (38). The ERCC1 rs11615 polymorphism with
the T allele was indicated to be associated with a significant
increase in the risk of shorter PFS and OS in patients with CRC
treated with oxaliplatin-based chemotherapy in a recent
meta-analysis (9). However, certain
studies have not determined any correlation between ERCC1 mRNA
expression and FOLFOX activity (39,40). An
increase in ERCC protein expression was noted in the patients of
the present study, which may account for a poor response to
FOLFOX-based CCRT treatment; this result is compatible with the
findings of most previous studies (10,19,36).
However, 6 patients (30%) had unresectable tumors that were not
conversion to resectable tumors by neoadjuvant treatment. Further
validation of the ERCC1 for the FOLFIRI regimen in patients with
resistance to CCRT and FOLFOX is mandatory.
The present study has certain limitations. Given its
nonrandomized retrospective design, a selection bias that may have
affected the present results is likely. In addition, only 20
patients from a single institution were analyzed in the present
small-sample study. Despite these drawbacks, the conclusions may
provide a path for conversion to resectable tumor by using FOLFIRI
in patients with cT4b CRC that did not sufficiently respond to
FOLFOX-based CCRT. The underlying mechanisms of action require
further investigation.
In conclusion, the present retrospective study
suggested that overexpression of ERCC is an indicator of poor
response to FOLFOX-based CCRT and indicated the potential advantage
of FOLFORI as a second-line neoadjuvant treatment in cT4b CRC that
do not respond to FOLFOX-based CCRT. FOLFIRI may potentiate the
antitumor response and thereby improve the efficacy of
peri-operative treatment for patients with cT4b CRC. The underlying
mechanisms of action of FOLFIRI in conversion to resectable tumors
by neoadjuvant treatment require additional prospective clinical
trials to confirm its validity.
Acknowledgements
Not applicable.
Funding
This work was supported by grants received as funds
from the Ministry of Science and Technology (grant nos. MOST
109-2314-B-037-035, MOST 109-2314-B-037-040, MOST
109-2314-B-037-046-MY3, MOST108- 2314-B-037-021-MY3), the Ministry
of Health and Welfare (grant nos. MOHW107-TDU-B-212-123006,
MOHW107-TDU-B-212-114026B, MOHW108-TDU-B- 212-133006 and
MOHW109-TDU-B-212-124026), the Health and Welfare Surcharge of
Tobacco Products, and Kaohsiung Medical University Hospital (grant
nos. KMUH108-8R66, KMUH108-8R34, KMUH108-8R35, KMUH108-8M33,
KMUH108-8M35, KMUH108-8M36, KMUHS10801, KMUHSA10804, KMUHS10807,
KMUH-DK109001-3 and KMUH-DK109005-3), as well as the Center for
Cancer Research (grant no. KMU-TC108A04) and the Research Center
Grant (grant no. KMU-TC108B07) of Kaohsiung Medical University
(Kaohsiung, Taiwan). In addition, this study was supported by the
Grant of Taiwan Precision Medicine Initiative, Academia Sinica,
Taiwan, R.O.C.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
JYW designed the study. MYH performed the
statistical analysis, participated in the interpretation of data,
and wrote and revised the manuscript. HHL drafted the manuscript.
JYW, CWH, CJM, HLT and TCY recruited patients for the study and
treated them. MYH, HHL and CMH are radiation oncologists who
contributed to the provision of RT. CYC and YTC were the
pathologists who scored the IHC staining. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
This present study was approved by the Institutional
Review Board of Kaohsiung Medical University Hospital [Kaohsiung,
Taiwan; no. KMUHIRB-E(I)-20190182].
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Douaiher J, Ravipati A, Grams B, Chowdhury
S, Alatise O and Are C: Colorectal cancer-global burden, trends,
and geographical variations. J Surg Oncol. 115:619–630. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Torre LA, Islami F, Siegel RL, Ward EM and
Jemal A: Global cancer in women: Burden and trends. Cancer
Epidemiol Biomarkers Prev. 26:444–457. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ministry of Health Welfare: The cause of
death in Taiwan in 2016. http://www.mohw.gov.tw/cp-16-33598-1.htmlAugust
25–2017
|
|
6
|
National Comprehensive Cancer Network:
Rectal Cancer. NCCN Clinical Practice Guidelines in Oncology (NCCN
Guidelines). version 2. 2019.
|
|
7
|
Hawkins AT, Ford MM, Geiger TM, Hopkins
MB, Kachnic LA, Muldoon RL and Glasgow SC: Neoadjuvant radiation
for clinical T4 colon cancer: A potential improvement to overall
survival. Surgery. 165:469–475. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Edge SB, Byrd DR, Compton CC, Fritz AG,
Greene FL and Trotti A: AJCC Cancer Staging Manual. 7th. New York;
Springer-Verlag: 2010
|
|
9
|
Ma SC, Zhao Y, Zhang T, Ling XL and Zhao
D: Association between the ERCC1 rs11615 polymorphism and clinical
outcomes of oxaliplatin-based chemotherapies in gastrointestinal
cancer: A meta-analysis. Onco Targets Ther. 8:641–648.
2015.PubMed/NCBI
|
|
10
|
Huang MY, Tsai HL, Lin CH, Huang CW, Ma
CJ, Huang CM, Chai CY and Wang JY: Predictive value of ERCC1,
ERCC2, and XRCC1 overexpression for stage III colorectal cancer
patients receiving FOLFOX-4 adjuvant chemotherapy. J Surg Oncol.
108:457–464. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Amshel C, Avital S, Miller A, Sands L,
Marchetti F and Hellinger M: T4 rectal cancer: Analysis of patient
outcome after surgical excision. Am Surg. 71:901–903; discussion
904. 2005.PubMed/NCBI
|
|
12
|
Diaconescu M, Burada F, Mirea CS, Moraru
E, Ciorbagiu MC, Obleaga CV and Vilcea ID: T4 Colon cancer-current
management. Curr Health Sci J. 44:5–13. 2018.PubMed/NCBI
|
|
13
|
Gao P, Song YX, Wang ZN, Xu YY, Tong LL,
Sun JX, Yu M and Xu HM: Is the prediction of prognosis not improved
by the seventh edition of the TNM classification for colorectal
cancer? Analysis of the surveillance, epidemiology, and end results
(SEER) database. BMC Cancer. 13:1232013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gunderson LL, Jessup JM, Sargent DJ,
Greene FL and Stewart AK: Revised TN categorization for colon
cancer based on national survival outcomes data. J Clin Oncol.
28:264–271. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Huang C-M, Huang M-Y, Ma C-J, Yeh Y-S,
Tsai H-L, Huang C-W, Huang C-J and Wang J-Y: Neoadjuvant FOLFOX
chemotherapy combined with radiotherapy followed by radical
resection in patients with locally advanced colon cancer. Radiat
Oncol. 12:482017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lu CY, Huang CW, Wu IC, Tsai HL, Ma CJ,
Yeh YS, Chang SF, Huang ML and Wang JY: Clinical implication of
UGT1A1 promoter polymorphism for irinotecan dose escalation in
metastatic colorectal cancer patients treated with bevacizumab
combined with FOLFIRI in the first-line setting. Transl Oncol.
8:474–479. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tirkes T, Hollar MA, Tann M, Kohli MD,
Akisik F and Sandrasegaran K: Response criteria in oncologic
imaging: Review of traditional and new criteria. Radiographics.
33:1323–1341. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Huang MY, Huang JJ, Huang CM, Lin CH, Tsai
HL, Huang CW, Chai CY, Lin CY and Wang JY: Relationship between
expression of proteins ERCC1, ERCC2, and XRCC1 and clinical
outcomes in patients with rectal cancer treated with FOLFOX-based
preoperative chemoradiotherapy. World J Surg. 41:2884–2897. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chang H, Yu X, Xiao WW, Wang QX, Zhou WH,
Zeng ZF, Ding PR, Li LR and Gao YH: Neoadjuvant chemoradiotherapy
followed by surgery in patients with unresectable locally advanced
colon cancer: A prospective observational study. Onco Targets Ther.
11:409–418. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li L and Ma BB: Colorectal cancer in
Chinese patients: Current and emerging treatment options. Onco
Targets Ther. 7:1817–1828. 2014.PubMed/NCBI
|
|
22
|
National Comprehensive Cancer Network:
Colon cancer. NCCN Clinical Practice Guidelines in Oncology (NCCN
Guidelines). version 2. 2019.
|
|
23
|
Gezen C, Kement M, Altuntas YE, Okkabaz N,
Seker M, Vural S, Gumus M and Oncel M: Results after multivisceral
resections of locally advanced colorectal cancers: An analysis on
clinical and pathological t4 tumors. World J Surg Oncol. 10:392012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lehnert T, Methner M, Pollok A, Schaible
A, Hinz U and Herfarth C: Multivisceral resection for locally
advanced primary colon and rectal cancer: An analysis of prognostic
factors in 201 patients. Ann Surg. 235:217–225. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Foxtrot Collaborative Group, : Feasibility
of preoperative chemotherapy for locally advanced, operable colon
cancer: The pilot phase of a randomised controlled trial. Lancet
Oncol. 13:1152–1160. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Dehal A, Graff-Baker AN, Vuong B, Fischer
T, Klempner SJ, Chang SC, Grunkemeier GL, Bilchik AJ and Goldfarb
M: Neoadjuvant chemotherapy improves survival in patients with
clinical T4b colon cancer. J Gastrointest Surg. 22:242–249. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Grossmann I, Klaase JM, Avenarius JK, de
Hingh IH, Mastboom WJ and Wiggers T: The strengths and limitations
of routine staging before treatment with abdominal CT in colorectal
cancer. BMC Cancer. 11:4332011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Takano S, Kato J, Yamamoto H, Shiode J,
Nasu J, Kawamoto H, Okada H and Shiratori Y: Identification of risk
factors for lymph node metastasis of colorectal cancer.
Hepatogastroenterology. 54:746–750. 2007.PubMed/NCBI
|
|
29
|
Larkin JO and O'Connell PR: Multivisceral
resection for T4 or recurrent colorectal cancer. Dig Dis. 30 (Suppl
2):S96–S101. 2012. View Article : Google Scholar
|
|
30
|
Olaussen KA, Dunant A, Fouret P, Brambilla
E, André F, Haddad V, Taranchon E, Filipits M, Pirker R, Popper HH,
et al: DNA repair by ERCC1 in non-small-cell lung cancer and
cisplatin-based adjuvant chemotherapy. N Engl J Med. 355:983–991.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Grimminger PP, Shi M, Barrett C, Lebwohl
D, Danenberg KD, Brabender J, Vigen CL, Danenberg PV, Winder T and
Lenz HJ: TS and ERCC-1 mRNA expressions and clinical outcome in
patients with metastatic colon cancer in CONFIRM-1 and −2 clinical
trials. Pharmacogenomics J. 12:404–411. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lee SM, Falzon M, Blackhall F, Spicer J,
Nicolson M, Chaudhuri A, Middleton G, Ahmed S, Hicks J, Crosse B,
et al: Randomized prospective biomarker trial of ERCC1 for
comparing platinum and nonplatinum therapy in advanced
non-small-cell lung cancer: ERCC1 trial (ET). J Clin Oncol.
35:402–411. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Raymond E, Faivre S, Chaney S, Woynarowski
J and Cvitkovic E: Cellular and molecular pharmacology of
oxaliplatin. Mol Cancer Ther. 1:227–235. 2002.PubMed/NCBI
|
|
34
|
Rosell R, Mendez P, Isla D and Taron M:
Platinum resistance related to a functional NER pathway. J Thorac
Oncol. 2:1063–1066. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gossage L and Madhusudan S: Current status
of excision repair cross complementing-group 1 (ERCC1) in cancer.
Cancer Treat Rev. 33:565–577. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Noda E, Maeda K, Inoue T, Fukunaga S,
Nagahara H, Shibutani M, Amano R, Nakata B, Tanaka H, Muguruma K,
et al: Predictive value of expression of ERCC 1 and GST-p for
5-fluorouracil/oxaliplatin chemotherapy in advanced colorectal
cancer. Hepatogastroenterology. 59:130–133. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shirota Y, Stoehlmacher J, Brabender J,
Xiong YP, Uetake H, Danenberg KD, Groshen S, Tsao-Wei DD, Danenberg
PV and Lenz HJ: ERCC1 and thymidylate synthase mRNA levels predict
survival for colorectal cancer patients receiving combination
oxaliplatin and fluorouracil chemotherapy. J Clin Oncol.
19:4298–4304. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chang PM, Tzeng CH, Chen PM, Lin JK, Lin
TC, Chen WS, Jiang JK, Wang HS and Wang WS: ERCC1 codon 118 C->T
polymorphism associated with ERCC1 expression and outcome of
FOLFOX-4 treatment in Asian patients with metastatic colorectal
carcinoma. Cancer Sci. 100:278–283. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Basso M, Strippoli A, Orlandi A, Martini
M, Calegari MA, Schinzari G, Di Salvatore M, Cenci T, Cassano A,
Larocca LM and Barone C: KRAS mutational status affects
oxaliplatin-based chemotherapy independently from basal mRNA ERCC-1
expression in metastatic colorectal cancer patients. Br J Cancer.
108:115–120. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ishibashi K, Okada N, Ishiguro T, Kuwabara
K, Ohsawa T, Yokoyama M, Kumamoto K, Haga N, Mori T, Yamada H, et
al: The expression of thymidylate synthase (TS) and excision repair
complementing-1 (ERCC-1) protein in patients with unresectable
colorectal cancer treated with mFOLFOX6 therapy. Gan To Kagaku
Ryoho. 37:2532–2535. 2010.(In Japanese). PubMed/NCBI
|