Introduction
Glioblastoma (GBM) is the most common primary
malignant brain tumor, comprising 16% of all primary brain and
central nervous system neoplasms (1). According to a 2007–2011 statistical
report from the Central Brain Tumor Registry of the United States,
it accounts for 15.4% of primary brain tumors and 45.6% of primary
malignant brain tumors (1). Among
malignant tumors, the incidence of glioblastoma is ~3.19 per
100,000 population, and its risk increases with age, mainly between
the ages of 75 and 84 years old (1).
GBM is characterized with a high mortality and recurrence rate, as
well as low survival rate, with the 5-year survival rate of
patients being ~5% (1). GBM mainly
occurs at the frontal lobe and temporal lobe regardless of an
individual's age (2). Chemotherapy
is a critical process in the postsurgical treatment of glioma, and
temozolomide (TMZ) is currently a major chemotherapeutic drug
(3). However, GBM exhibits high
resistance to TMZ, which can result in relapse, as well as
treatment failure (4). MicroRNAs
(miRNAs/miRs) are short non-coding RNA sequences, typically 19–25
nucleotides in length, first identified in 1993 (5). As important regulatory factors, miRNAs
participate in cell proliferation, differentiation, apoptosis, cell
cycle progression and other biological processes, playing vital
regulatory roles in biological development and tumorigenesis
(6). The miR-181 family has notable
effects on the biological processes of range of tumor cells. Liu
et al (7) demonstrated that
low miR-181b expression in nervous glioma cells has a role as a
tumor suppressor gene, inhibiting the proliferation and migration
of tumor cells, as well as inducing apoptosis. F-box protein is a
type of protein that is widely found in eukaryotes and contains an
F-box domain. In the ubiquitin-proteasome pathway, it specifically
recognizes substrate proteins and participates in cell cycle
regulation, transcription regulation, cell apoptosis, cell signal
transduction and other cellular activities (8). Previous studies have revealed that
miRNAs indirectly participate in cancer progression via mediating
different F-box proteins (8,9). For example, miR-25, miR-27a, miR-92a,
miR-182, miR-223 and miR-503 can promote cancer development by
inhibiting F-box and WD repeat domain-containing 7 in colon,
esophageal, stomach, liver and other types of cancer, while miR-7,
miR-30, miR-203 and miR-340 can inhibit cancer by inhibiting Skp2.
The expression levels of miRNAs of the miR-30 family are
upregulated in cancer cell lines, such as non-small cell lung
cancer and melanoma cells (9).
However, the underlying mechanisms by which miR-181a-5p is involved
in the drug resistance of glioma cells have not been resolved. The
present study investigated the effects of miR-181a-5p on the
invasion and resistance of glioma cells to TMZ, and reported that
miR-181a-5p represents a potential target for overcoming drug
resistance in GBM cells.
Materials and methods
Glioma tissue samples and cells
Glioma tissue samples collected between November
2016 and June 2017 used in the present study were obtained from
Nanfang Hospital and Zhujiang Hospital (Southern Medical
University, Guangzhou, China), and Guangdong 999 Brain Hospital
(Guangzhou, China) after pathological diagnosis, which included 22
grade I–II tumors, 58 grade III–IV tumors (according to the 2007
World Health Organization Classification of Tumours of the Central
Nervous System) (10) and 16
relapsed tumors that had undergone TMZ chemotherapy for 6 months.
All glioma specimens have been examined and diagnosed by
pathological examination at The Department of Pathology of Zhujiang
Hospital and Guangdong 999 Brain Hospital. Tissue samples were
immediately aliquoted into separate test tubes, frozen on dry ice
and stored at −80°C until analysis. Follow-up data was obtained
from reviews of patients' medical records. Prior to surgery, no
patients had received radiation or chemotherapy. The same procedure
occurred for the patients who relapsed. This study was approved by
the Ethics Committee of Hainan Medical University (Haikou, China;
approval no. 201818). The present study was conducted in accordance
with the regulations of the Hainan Institutional Review Board
(affiliated to Hainan Provincial Organization Office). All enrolled
patients provided written informed consent form before craniotomy.
Human nervous glioma cell lines U251 and TMZ-resistant GBM cells
(U251TR) were collected from The Department of Pathology, Nanfang
Hospital, Southern Medical University. In order to verify that
miR-296-3p may at least partially serve a role in multi-drug
resistance in glioblastoma by targeting ether-à-go-go, the
TMZ-resistant GBM U251TR cell line has been previously constructed
from a U251 cell line (11) and
stored in the laboratory of the Department of Laboratory of
Southern Hospital.
Cell culture and transfection
The glioma cell lines, U251 and U251TR, were
cultured in Dulbecco's modified Eagle's medium (DMEM; Gibco; Thermo
Fisher Scientific, Inc.) containing 10% fetal bovine serum (FBS;
HyClone; Cytiva) and 10 µg/ml TMZ (Sigma-Aldrich; Merck KGaA), and
were incubated at 37°C in a humidified 5% CO2-enriched
atmosphere. Human embryonic kidney 293T cells (purchased from the
American Type Culture Collection) were also cultured in the same
way. The glioma cells were transiently transfected using
Lipofectamine® 3000 Transfection reagent (Thermo Fisher
Scientific, Inc.), according to the manufacturer's instructions.
The concentrations of miR-181a-5p mimics, miR-181a-5p inhibitors
and controls were all 100 nmol/l. RNA fragments (miR-181a-5p
mimics/inhibitors) used in transfection assay were purchased from
Shanghai GenePharma Co., Ltd. Cells were divided into 6 groups,
including: Untreated cells as the control group (2 groups, one for
each cell line); cells transfected with miR-181-5p negative control
(NC) as the NC group (2 groups, one for each cell line);
drug-resistant GBM cells (U251TR) transfected with miR-181-5p
mimics as the miR-mimics group; and GBM cells (U251) transfected
with miR-181-5p inhibitors as the miR-inhibitors group.
Additionally, U251 cells were transfected with the mimic to see
whether there was an apoptotic effect. A total of 2×105
cells/well were seeded in a 6-well culture plate and cultured until
the cell density reached 70% at 37°C. According to the
manufacturer's protocol, the transfection reagent was added into
serum-free DMEM, which was added to the culture plate to replace
the complete medium and cultured for 48 h at 37°C. The transfection
efficiency was observed under a fluorescence microscope (data not
shown). The sequences of RNA oligo ribonucleotides were as follows:
miR-181a-5p mimics forward, 5′-AACAUUCAACGCUGUCGGUGAGU-3′ and
reverse, 5′-UCACCGACAGCGUUGAAUGUUUU-3′; miR-181a-5p mimics-NC
forward, 5′-UUCUCCGAACCUGUCACGUTT-3′ and reverse,
5′-ACGUGACACGGUCGGAGAATT-3′; miR-181a-5p inhibitors,
5′-ACUCACCGACAGCGUUGAAUGUU-3′; miR-181a-5p inhibitors-NC,
5′-CAGUACUUUUGUGUAGUACAA-3′.
Reverse transcription-quantitative
(RT-q)PCR analysis
Total RNA was extracted and isolated from tissues
and cells using TRIzol® reagent (Invitrogen; Thermo
Fisher Scientific, Inc.) according to the manufacturer's protocol.
Total RNA was reverse transcribed. The RT mixed reaction solution
was prepared on ice, including the following reagents: 2 µl 5X
PrimeScript® Buffer (Takara Biotechnology Co., Ltd.),
0.5 µl PrimeScript RT Enzyme Mix I, 0.5 µl oligo dT primer (50 µM),
0.5 µl random 6-nucleotide primers (100 µM), template RNA
(4×105 copies), RNase-free double-distilled water up to
10 µl. The reaction conditions were as follows: 37°C for 15 min for
the reverse transcription reaction, and 85°C for 5 sec to
inactivate the reverse transcriptase. The synthesized cDNA was
stored at −20°C until subsequent use. The expression levels of
miR-181a-5p were detected using the SYBR Green Hairpin-it™ miRNAs
qPCR Quantitation kit (Shanghai GenePharma Co., Ltd.), in a Fast
Real-time PCR 7500 System (Applied Biosystems; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocols. The
reaction conditions were as follows: Pre-denaturation at 95°C for
30 sec, followed by 30–40 cycles of denaturation at 95°C for 5 sec,
annealing at 60°C for 30 sec and extension at 72°C for 10 sec. The
U6 gene was used as an internal reference. Each assay was
performed in triplicate and repeated three times. The relative
expression levels were calculated and analyzed using the
2−ΔΔCq method (12).
Primers used in qPCR assays were synthesized by Shanghai GenePharma
Co., Ltd. (miR-181a-5p, cat. no. 91151N61; U6, cat. no.
8301151105).
Chemotherapeutic drug sensitivity of
glioma cells
Cells (1×104) were seeded into 96-well
plates and divided into six groups: Two blank control groups, two
NC groups, miR-181a-5p mimic group and miR-181a-5p inhibitor group,
after transient transfection using Lipofectamine as aforementioned.
After 24 h, the cells were treated with different concentrations of
TMZ, ranging from 10 to 60 µg/ml for 48 h. The assay was undertaken
in five replicate wells for each sample. The spectrophotometric
absorbance of the samples was calculated as an average.
Subsequently, the half maximal inhibitory concentration
(IC50) values of TMZ were respectively calculated.
Cell Counting Kit (CCK)-8 assay
A 96-well plate was used, at a density of
5×104 cells/well. At 24, 48 and 72 h after transfection,
10 µl of CCK-8 solution (Pierce; Thermo Fisher Scientific, Inc.)
was added according to the manufacturer's protocol. After 1 h,
optical density (OD) at 450 nm value was measured using a
microplate reader.
Wound healing assay
Cells were seeded into a 6-well plate at a density
of 5×105 cells/well and cultured for 24 h to create a
confluent monolayer. The cells were serum-starved during this
assay. The confluent monolayer was scraped with a 200-µl pipette
tip along a sterile measuring rule, and washed three times in PBS.
The corresponding images under a light microscope (magnification,
×4) were captured at 0 and 24 h. Finally, ImageJ software v1.52a
(National Institutes of Health) was used for quantification.
Cell invasion and Transwell migration
assays
Cell invasion assays were performed using Transwell
chambers following the manufacturer's instructions. Briefly,
Matrigel (precoating at 37° for 15 min) was diluted using
serum-free DMEM. A total of 200 µl of glioma cell (1×105
cells) suspension was seeded into the upper chamber. Subsequently,
600 µl of media supplemented with 20% FBS was added into the lower
chamber, followed by incubation for 24 h. A cotton-tipped swab was
used to wipe off the cells that did not migrate to the bottom
chamber. Cells were stained with 0.1% Crystal Violet Staining
Solution (Pierce; Thermo Fisher Scientific, Inc.) at room
temperature for 15 min. The chambers were then observed, images
were captured and the number of migrated cells were counted under a
light microscope (magnification, ×10) in six random fields of view.
All assays were independently repeated for three times.
Cell apoptosis assays
Cells (1×105) were trypsinized and
collected by centrifugation at 4°C at 150 × g for 8 min, and
apoptosis was analyzed using an Apoptosis Detection kit (Pierce;
Thermo Fisher Scientific, Inc.). A total of 250 µl binding buffer
was added to the suspended cells, 100 µl of suspension was added
into a 1.5-ml Eppendorf tube, along with 5 µl Annexin V-PE and 10
µl 7-AAD, the samples were gently mixed in the dark and left to
incubate at room temperature for 15 min before the addition of 400
µl of binding buffer, and analyzed using a FASCAria II (BD
Biosciences) flow cytometer and FlowJo software (v7.6.1; FlowJo,
LLC) within 1 h.
Western blot assay
Following cell lysis with RIPA lysis buffer
(Beyotime Institute of Biotechnology), a BCA protein concentration
assay kit (Biosharp Life Sciences) and Synergy HTX (BioTek
Instruments, Inc.) microplate reader were used to determine the
protein concentration. Protein loading buffer (5X; Beijing Zoman
Biotechnology Co., Ltd.), was used for equalization of protein
loading. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis
(10% SDS-PAGE) Gel Quick Preparation kit (Beyotime Institute of
Biotechnology) was used for gel production. Samples were boiled at
100°C for 10 min to denature the proteins. Protein samples (100
µg/lane) were separated via SDS-PAGE, and transferred onto
polyvinylidene difluoride membranes (EMD Millipore). The membranes
were blocked with 5% skimmed milk powder for 2 h at room
temperature and then rinsed with TBS-Tween (1% Tween-20) for 3
times (10 min each time). The membrane was incubated with primary
rabbit antibodies against human F-Box protein 11 (FBXO11; 1:500;
cat. no. ab181801; Abcam) and GAPDH (1:1,000; cat. no. AF1186;
Beyotime Institute of Biotechnology) for 24 h on a shaker at 4°C,
followed by incubation with a HRP-labeled goat anti-rabbit
secondary antibody (1:4,000; cat. no. A2028; Beyotime Institute of
Biotechnology) at room temperature for 1 h. Signal was detected
using enhanced chemiluminescence detection reagent (Beyotime
Institute of Biotechnology). Finally, the 4600SF chemiluminescent
imaging system (Tanon) was used to semi-quantify the bands. Three
independent experiments were performed for each analysis.
Bioinformatics analysis
TargetScan (www.targetscan.org) was utilized to predict biological
targets for miR-181a-5p. Of all these hypothetical targets, FBXO11
was selected for further research.
Dual-luciferase reporter assay
The 3′-untranslated region (UTR) and mutant 3′-UTR
of wild-type FBXO11 were amplified by Wuhan GeneCreate Biological
Engineering Co., Ltd., and integrated into the psichenk2.0 (Wuhan
Servicebio Technology Co., Ltd.) vector containing the luciferase
gene. The plasmids were tested and co-transfected into 293T cells
with miR-181a-5p mimics or miR-NC. After 48 h of transfection, the
cells were lysed using a Dual-Luciferase® Reporter Assay
kit (Promega Corporation). In the case of firefly luciferase as the
internal control, the relative light unit (RLU) value measured by
Renilla luciferase was divided by the RLU value measured by
firefly luciferase. According to the obtained ratio, the inhibitory
effect of miRNA on the target gene was compared, and the luciferase
activity was analyzed using a Synergy H1 Hybrid Multi-Mode
Microplate Reader (BioTek Instruments, Inc.).
Statistical analysis
All statistical analyses were conducted using SPSS
19.0 (IBM Corp.) and GraphPad Prism 8.0 software (GraphPad
Software, Inc.). Each experiment was performed in triplicate and
repeated for three times. Data were presented as mean ± standard
deviation. Independent-samples t-tests were used to analyze
significant differences between two groups, while one-way analysis
of variance was utilized to estimate significant differences among
multiple groups. The least significant difference method was used
for multiple comparisons when the variance was equal. The Dunnett's
corrective test was employed when the variance was not equal (in
the chemotherapeutic drug sensitivity experiments of glioma cells).
P<0.05 was considered to indicate a statistically significant
difference.
Results
miR-181a-5p expression is upregulated
in normal glioma cells and downregulated in drug-resistant glioma
cells
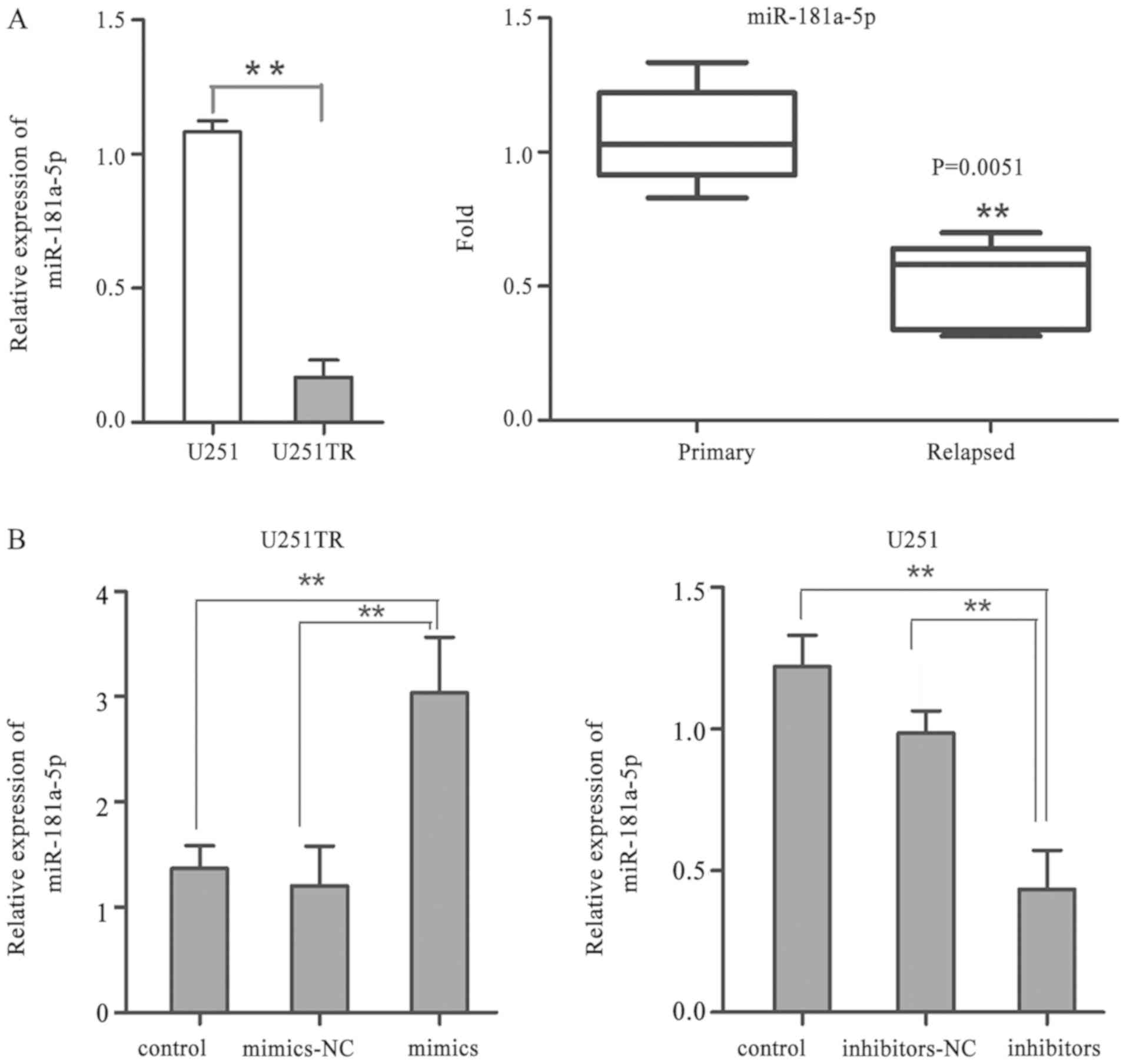
RT-qPCR was employed to detect miR-181a-5p
expression levels in U251 and U251TR cells and 51 glioma tissues
containing primary and relapsed tissues. The data revealed that
miR-181a-5p expression was significantly decreased in U251TR cells
compared with expression in U251 cells (P<0.01; Fig. 1A). It was also revealed that
miR-181a-5p expression was significantly reduced in relapsed GBM
tissues after chemotherapy compared with primary GBM tissues
(0.5080±0.072 vs. 1.06±0.082, respectively; P<0.01; Fig. 1A). Untransfected U251TR and U251
cells were then used as controls in addition to the mimics NC.
miR-181a-5p expression levels were markedly increased in U251TR
cells transfected with miR-181a-5p mimics compared with those in
the NC group (P<0.01). However, the miR-181a-5p expression
levels were significantly decreased in U251 cells transfected with
the miR-181a-5p inhibitors compared with the untransfected control
group (P<0.01) (all Fig. 1B).
miR-181a-5p can inhibit the
proliferation and invasion of glioma cells and promotes the
apoptosis of glioma cells
The present study sought to investigate the
biological functions of miR-181a. The CCK-8 assays indicated that
the miR-181a-5p simulation group had a significantly enhanced
sensitivity to TMZ, with a decreased IC50 value in
transfected U251TR cells compared with untransfected cells
(P<0.01). In contrast, the IC50 value of TMZ in U251
cells transfected with miR-181a-5p inhibitors was notably elevated
compared with the control group (P<0.01) (Fig. 2A). The CCK-8 assay demonstrated that
the proliferation of drug-resistant glioma cells transfected with
the miR-181a-5p mimic was significantly lower compared with that in
the NC group and drug-resistant glioma cells (P<0.05; Fig. 2B). The proliferation rate of normal
glioma cells transfected with miR-181a-5p inhibitor was higher
compared with that in the NC group (P<0.05; Fig. 2B).
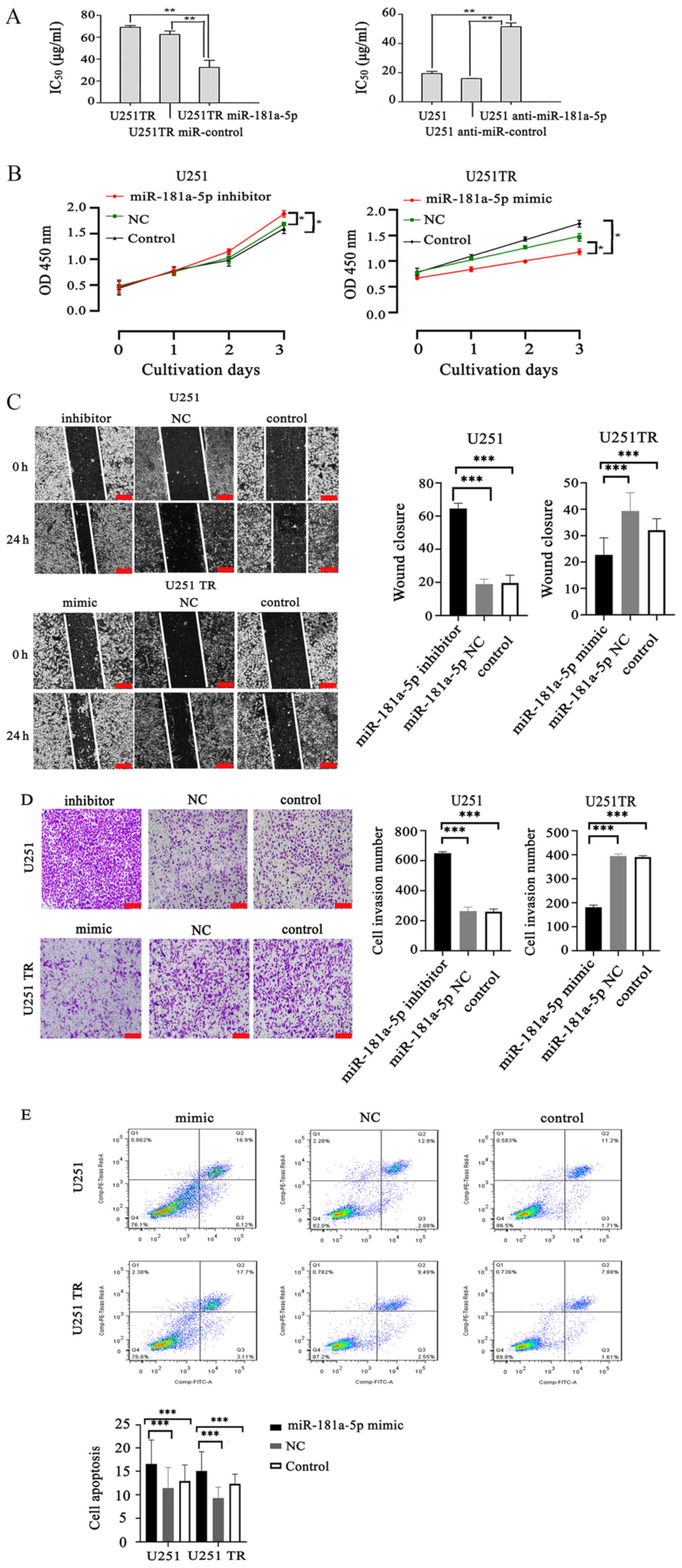 | Figure 2.Effects of miR-181a-5p on the
proliferation, invasion and apoptosis of glioma cells. (A) The
IC50 values of TMZ in glioma U251TR and U251 cells
transfected with miR-181a-5p mimics or inhibitors. **P<0.01. (B)
miR-181a-5p mimics/inhibitors can inhibit or promote the
proliferation of drug-resistant glioma cells/normal glioma cells,
respectively, compared with the NC and untransfected control group.
*P<0.05. (C) Wound healing assay was carried out to determine
the migration of glioblastoma U251TR and U251 cells transfected
with miR-181a-5p mimics/inhibitors, respectively. ***P<0.001.
Scale bar, 200 µm. (D) Transwell migration assays were performed to
determine the effects of miR-181a-5p on the invasive capacity of
the glioma cells. ***P<0.001. Scale bar, 200 µm. (E) miR-181a-5p
induced apoptosis in U251 and U251TR cells. Flow cytometry analysis
showed that overexpression of miR-181a-5p enhanced apoptosis in
U251 and U251TR cells. Analysis indicated that the apoptosis rate
was upregulated in U251 and U251TR cells. ***P<0.001 vs.
control. miR, microRNA; TMZ, temozolomide; IC50, half
maximal inhibitory concentration; NC, negative control. |
The wound healing assay revealed that, compared with
the NC and untransfected control group, the U251 cells transfected
with the miR-181a-5p inhibitor had a significantly faster 24-h
wound closure rate (P<0.001). However, compared with the NC
group, the 24 h wound closure rate of U251TR cells transfected with
the miR-181a-5p mimic group was significantly less (P<0.001;
Fig. 2C). Results of Transwell
migration assays revealed that the miR-181a-5p mimics had
significantly fewer invasive cells compared with the NC group
(P<0.001). In contrast, U251 treatment with miR-181a-5p
inhibitor resulted in a significantly higher number of invasive
cells compared with the NC group (P<0.001; Fig. 2D). Flow cytometry results showed that
compared with the NC and untransfected control group, the
percentage of apoptotic U251 cells was significantly higher in the
miR-181a-5p mimic group (P<0.001; Fig. 2E). For this experiment, U251 cells
were also transfected with the mimic to see whether there was an
apoptotic effect. Additionally, similar results were observed in
U251TR cells (P<0.001; Fig. 2E).
The aforementioned results indicated the inhibitory effects of
miR-181a on glioma cells, especially drug-resistant glioma
cells.
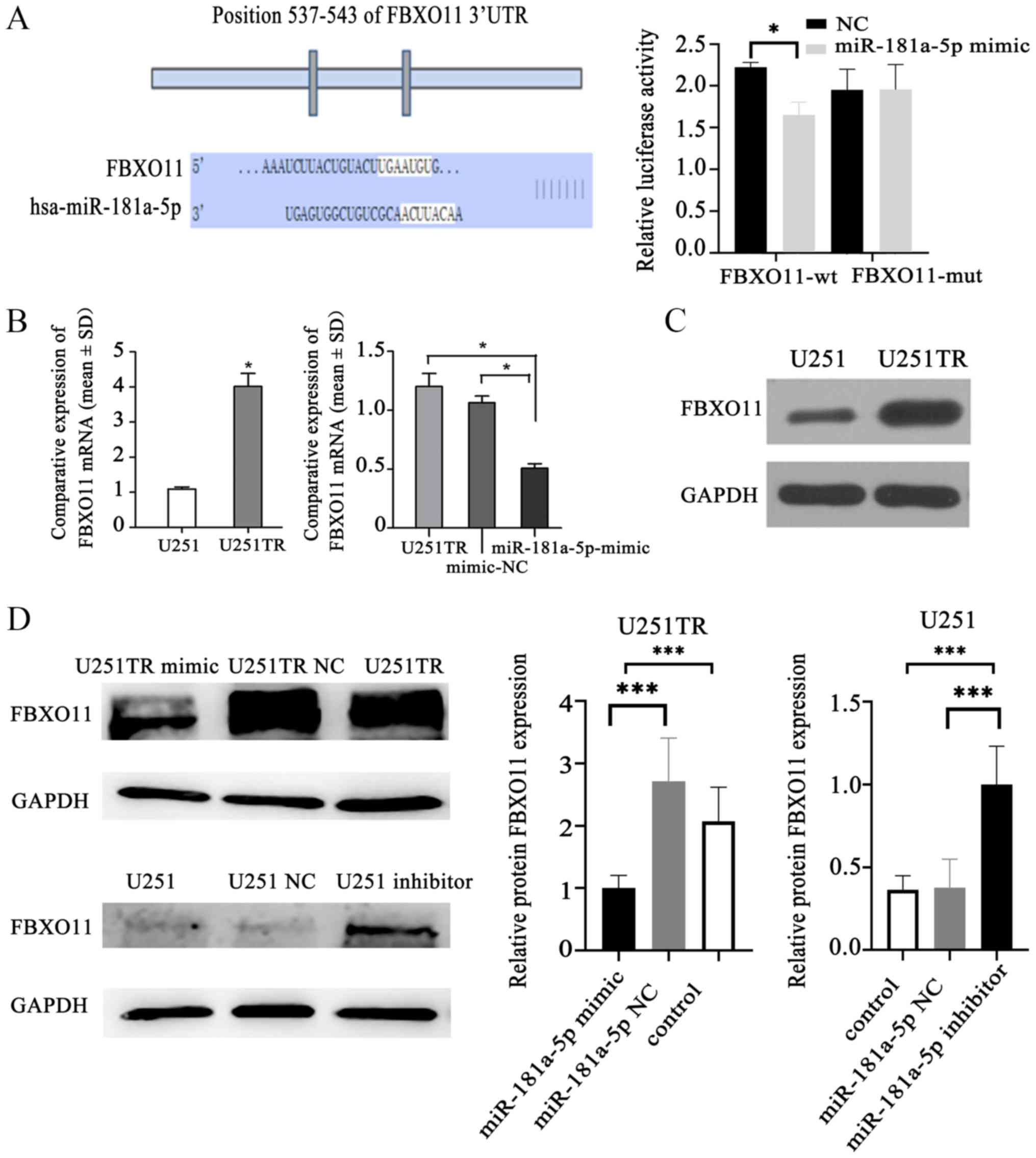
FBXO11 is a downstream target of
miR-181a-5p
According to the TargetScan prediction website,
miR-181a-5p was predicted to bind to FBXO11, the base
complementary region is located at 537–543 of the non-coding region
of FBXO11 gene. In the presence of miR-181-5p mimic, the
luciferase activity of the FBXO11-wt group was significantly lower
than that of the NC group, indicating that miR-181a-5p can target
the 3′UTR end of FBXO11 (P<0.05; Fig. 3A). In glioma-resistant cells, the
expression of FBXO11 was significantly higher compared with in
ordinary glioma cells (P<0.05). When the expression levels of
miR-181a-5p were upregulated, the expression levels of
FBXO11 in glioma-resistant cells were suppressed compared
with untransfected cells (P<0.05) (Fig. 3B). Western blotting demonstrated that
FBXO11 expression was markedly upregulated in drug-resistant cells
compared with non-resistant cells (Fig.
3C). Additionally, FBXO11 expression was downregulated in
drug-resistant glioma cells transfected with miR-181a-5p mimic,
while it was upregulated in glioma cells transfected with
miR-181a-5p inhibitor compared with in the controls (P<0.001;
Fig. 3D).
Discussion
GBM is a common malignant tumor in the brain,
accounting for ~45% of all brain tumors between 2011 and 2014
globally (13,14). Gliomas pose a notable threat to human
life and health, which can be manifested as mental disorders, such
as dementia and brain damage as a result of brain tumor hemorrhage,
leading to sudden brain herniation, and even hemiplegia or death,
secondary epilepsy and blindness (15). To date, comprehensive approaches,
including surgical excision, radiotherapy, chemotherapy and
immunotherapy, are major treatment strategies for GBM worldwide.
However, although postoperative patients undergo chemoradiotherapy,
the overall survival time of patients is only ~12 months, owing to
the strong migratory and invasive capabilities of GBM cells
(16). Glioma is characterized by
common recurrence after surgery, drug resistance after
chemotherapy, a high mortality rate and an extremely low survival
rate (17). Among malignant tumors,
the incidence of glioblastoma is ~3.19 per 100,000 population, and
its risk increases with age, mainly between the ages of 75 and 84
years old (1). The 5-year survival
rate of patients is ~5% (1). In
addition, high-grade malignancy, short survival period and drug
resistance during postoperative chemotherapy often lead to the
treatment failure (18). Therefore,
understanding the specific molecular mechanisms underlying the
resistance of glioma to TMZ and understanding of its downstream
regulatory networks have research attracted attention.
TMZ is the most widely used anticancer drug for
chemotherapy of GBM, mainly through influencing various division
phases of tumor cells, especially late G1 phase and
early S phase (19). TMZ can induce
the apoptosis and sensitize tumor tissues to chemotherapy by
interfering with DNA replication and the repair of tumor cells
(19). Nonetheless, the curative
effect of TMZ gradually declines in patients with GBM, indicating
that patients develop a resistance to chemotherapeutics (20). At present, the acknowledged molecular
mechanisms of glioma resistance to TMZ, such as the overexpression
of drug transporter protein, stronger cell repair ability,
repressed apoptosis and obstruction of the blood-brain barrier, are
considered to be involved (21).
Recently, studies have shown that there is a close correlation
between miRNAs and chemotherapy resistance of tumor cells. For
instance, miR-221 is overexpressed in hepatocellular cancer cells
resistant to sorafenib, and then targets caspase-3 to induce drug
resistance of tumor cells through anti-apoptotic pathways (22). However, low expression of
miR-424(322)/503 results in the upregulation of Bcl-2 and the
insulin-like growth factor 1 receptor in breast cancer, which
further induces resistance of breast cancer cells to chemotherapy
(23). In addition, the abnormal
expression of miRNAs modulate the expression of downstream target
genes, and contributes to critical changes in the sensitivity of
tumor cells to chemotherapeutic drugs (24).
miRs are endogenous non-coding RNAs that are 20–25
nucleotides in length. Dysregulation of miRNAs in brain tumors
result in chromosomal aberration, epigenetic modifications and
mutations (25). miRNAs possess
special biological characteristics and an important function
(22–24). For example, in hepatocellular
carcinoma, miR-221 regulates sorafenib resistance by inhibiting
caspase-3-mediated apoptosis (22).
miR-424 is a breast cancer suppressor, and loss of miR-424
expression can enhance resistance to chemotherapy (23,24).
Thus, they not only could be specific diagnostic markers for
certain malignant tumors and hereditary diseases, but also
indicators of invasion, migration, drug resistance or prognosis of
malignant tumors (26,27). A previous study analyzed the results
of a glioma-related GeneChip and found that there are ~22
differentially expressed miRNAs, and that some miRNAs promote the
proliferation of GBM cells (28).
miR-181a is a highly conserved member of the miR-181 family and
acts as an early brain tumor marker, as well as target-regulating
factor (29).
Glioma is the most common malignant tumor of the
central nervous system, and the prognosis of patients is still very
limited. miRNAs have been a research hotspot in recent years, and
the mechanism of miR-181a-5p regulating the drug resistance of
glioma remains unknown. The present study demonstrated that
miR-181a-5p could regulate glioma resistance to TMZ and inhibit the
migration and invasion capacities of glioma cells. Moreover, the
results indicated that the expression of FBXO11 was reduced in
U251TR cells via overexpression of miR-181a-5p, which indicated
that increased expression of miR-181a-5p can downregulate FBXO11
expression. However, in U251 cells with depleted miR-181a-5p
expression, FBXO11 expression was increased, demonstrating that
increased miR-181a-5p expression could upregulate FBXO11
expression. In an experiment that analyzed the expression levels of
miR-181a-5p in glioma resistant cells and relapsed glioma tissues
after chemotherapy, the average expression levels of miR-181a-5p
were significantly lower after chemotherapy in relapsed glioma
cells compared with the tumor tissue. Further studies are required
to determine if miR-181a-5p expression is higher in patients who
relapse compared with patients who do not relapse. It would be
valuable to understand the differences between patients that had
relapsed with different grades. The present study suggested that
miR-181a-5p and FBXO11 serve an important role in regulating the
resistance to chemotherapy in glioma; however, research on
miR-181a-5p and glioma drug resistance remains unclear. Future
studies should determine the association between miR-181a-5p and
glioma drug resistance, clarify its specific mechanism of action
and clinical significance, improve the theoretical basis of glioma
drug resistance and provide new targets for glioma drug resistance
treatment.
Furthermore, the present results suggested that
miR-181a-5p increased the chemotherapeutic sensitivity of glioma
probably by affecting apoptosis. Drug resistance, similar to
tumorigenesis and progression, is a complex process involving
altered expression of several genes, multipath changes and
multi-step accumulation (30).
Hence, molecular mechanisms of drug resistance should be further
explored. According to the current experimental results, the
mechanism of apoptosis could be further explored to look for
pathway genes associated with apoptosis genes.
At present, application of miRNA in the
drug-resistant treatment of GBM has become an important research
avenue and has an extensive clinical prospect. In summary,
miR-181a-5p inhibits the proliferation and invasion of
drug-resistant GBM cells. At present, the treatment of GBM cells
mainly depends on chemotherapy, and the increase of drug resistance
is a global problem (4). Several
studies have revealed that miRNA is involved in the process of
glioma resistance (23–25). Collectively, the present findings
demonstrated that miR-181a-5p is a tumor suppressor gene in GBM,
potentially providing a theoretical basis for treatment of
drug-resistant GBM in clinical practice.
Acknowledgements
Not applicable.
Funding
The present study was financially supported by Key
Research and Development Project of Hainan Province (grant no.
ZDYF2018157), the National Natural Science Foundation of China
(grant no. 81760541), the China Postdoctoral Science Foundation
funded project (grant no. 2017M613272XB) and the Natural Science
Foundation of Guangxi Province (grant nos. 2017GXNSFBA198001 and
2019GXNSFAA245089).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XW, LM and QL conceived and designed the study. SL,
MG and XL designed the experiments. HL and YC performed the
experiments and analyzed the data. XW and XK performed the
pathological analysis. XW and SL wrote, edited and revised the
manuscript. All authors have read and approved the final version of
the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Hainan Medical University (Haikou, China; approval no.
201818). This study was conducted in accordance with the
regulations of the Hainan Institutional Review Board (affiliated to
Hainan Provincial Organization Office). All enrolled patients
signed the written informed consent form before surgery.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Strom QT, Gittleman H, Liao P, Rouse C,
Chen Y, Dowling J, Wolinsky Y, Kruchko C and Barnholtz-Sloan J:
CBTRUS statistical report: Primary brain and central nervous system
tumors diagnosed in the United States in 2007–2011. Neuro Oncol. 16
(Suppl 4):iv1–iv63. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bradley CA: Glioblastoma: Stem
cells-masters of their fates. Nat Rev Cancer. 17:574–575. 2017.
View Article : Google Scholar
|
|
3
|
McCarthy DJ, Komotar RJ, Starke RM and
Connolly ES: Randomized trial for short-term radiation therapy with
temozolomide in elderly patients with glioblastoma. Neurosurgery.
81:N21–N23. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Garros-Regulez L, Aldaz P, Arrizabalaga O,
Moncho-Amor V, Carrasco-Garcia E, Manterola L, Moreno-Cugnon L,
Barrena C, Villanua J, Ruiz I, et al: mTOR inhibition decreases
SOX2-SOX9 mediated glioma stem cell activity and temozolomide
resistance. Expert Opin Ther Targets. 20:393–405. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Slattery ML, Lee FY, Pellatt AJ, Mullany
E, Stevens JR, Samowitz WS, Wolff RK and Herrick JS: Infrequently
expressed miRNAs in colorectal cancer tissue and tumor molecular
phenotype. Mod Pathol. 31:2092018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Irani S: miRNAs signature in head and neck
squamous cell carcinoma metastasis: A literature review. J Dent
(Shiraz). 17:71–83. 2016.PubMed/NCBI
|
|
7
|
Liu YS, Lin HY, Lai SW, Huang CY, Huang
BR, Chen PY, Wei KC and Lu DY: miR-181b modulates EGFR-dependent
VCAM-1 expression and monocyte adhesion in glioblastoma. Oncogene.
36:5006–5022. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Skaar JR, Pagan JK and Pagano M: SnapShot:
F box proteins I. Cell. 137:1160–1160.e1. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wu ZH and Pfeffer LM: MicroRNA regulation
of F-box proteins and its role in cancer. Semin Cancer Biol.
36:80–87. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Louis DN, Ohgaki H, Wiestler OD, Cavenee
WK, Burger PC, Jouvet A, Scheithauer BW and Kleihues P: The 2007
WHO classification of tumours of the central nervous system. Acta
Neuropathol. 114:97–109. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bai Y, Liao H, Liu T, Zeng X, Xiao F, Luo
L, Guo H and Guo L: miR-296-3p regulates cell growth and multi-drug
resistance of human glioblastoma by targeting ether-à-go-go (EAG1).
Eur J Cancer. 49:710–724. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wick W, Gorlia T, Bendszus M, Taphoorn M,
Sahm F, Harting I, Brandes AA, Taal W, Domont J, Idbaih A, et al:
Lomustine and bevacizumab in progressive glioblastoma. N Engl J
Med. 377:1954–1963. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cavaliere R, Wen PY and Schiff D: Novel
therapies for malignant gliomas. Neurol Clin. 25:1141–1171. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chrastina J, Novak Z, Brazdil M and
Hermanova M: Glioblastoma multiforme in a patient with isolated
hemimegalencephaly. J Neurol Surg Rep. 76:e160–e163. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ding Z, Roos A, Kloss J, Dhruv H, Peng S,
Pirrotte P, Eschbacher JM, Tran NL and Loftus JC: A novel signaling
complex between TROY and EGFR mediates glioblastoma cell invasion.
Mol Cancer Res. 16:322–332. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Moinuddin FM, Hirano H, Shinsato Y, Higa
N, Arita K and Furukawa T: ATP7B expression in human glioblastoma
is related to temozolomide resistance. Oncol Lett. 14:7777–7782.
2017.PubMed/NCBI
|
|
18
|
Guo G, Gong K, Ali S, Ali N, Shallwani S,
Hatanpaa KJ, Pan E, Mickey B, Burma S, Wang DH, et al: A
TNF-JNK-Axl-ERK signaling axis mediates primary resistance to EGFR
inhibition in glioblastoma. Nat Neurosci. 20:1074–1084. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hochhauser D, Glynne-Jones R, Potter V,
Grávalos C, Doyle TJ, Pathiraja K, Zhang Q, Zhang L and Sausville
EA: A phase II study of temozolomide in patients with advanced
aerodigestive tract and colorectal cancers and methylation of the
O6-methylguanine-DNA methyltransferase promoter. Mol Cancer Ther.
12:809–818. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bocangel DB, Finkelstein S, Schold SC,
Bhakat KK, Mitra S and Kokkinakis DM: Multifaceted resistance of
gliomas to temozolomide. Clin Cancer Res. 8:2725–2734.
2002.PubMed/NCBI
|
|
21
|
Thomas AA, Brennan CW, DeAngelis LM and
Omuro AM: Emerging therapies for glioblastoma. JAMA Neurol.
71:1437–1444. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fornari F, Pollutri D, Patrizi C, La Bella
T, Marinelli S, Gardini AC, Marisi G, Toaldo MB, Baglioni M,
Salvatore V, et al: In hepatocellular carcinoma miR-221 modulates
sorafenib resistance through inhibition of caspase-3-mediated
apoptosis. Clin Cancer Res. 23:3953–3965. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rodriguez-Barrueco R, Nekritz EA, Bertucci
F, Yu J, Sanchez-Garcia F, Zeleke TZ, Gorbatenko A, Birnbaum D,
Ezhkova E, Cordon-Cardo C, et al: miR-424(322)/503 is a breast
cancer tumor suppressor whose loss promotes resistance to
chemotherapy. Genes Dev. 31:553–566. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Carta A, Chetcuti R and Ayers D: An
introspective update on the influence of miRNAs in breast carcinoma
and neuroblastoma chemoresistance. Genet Res Int.
2014:7430502014.PubMed/NCBI
|
|
25
|
Shahar T, Granit A, Zrihan D, Canello T,
Charbit H, Einstein O, Rozovski U, Elgavish S, Ram Z, Siegal T and
Lavon I: Expression level of miRNAs on chromosome 14q32.31 region
correlates with tumor aggressiveness and survival of glioblastoma
patients. J Neurooncol. 130:413–422. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
López-Ochoa S, Ramírez-García M,
Castro-Sierra E and Arenas-Huertero F: Analysis of chromosome 17
miRNAs and their importance in medulloblastomas. Biomed Res Int.
2015:7175092015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ying W, Riopel M, Bandyopadhyay G, Dong Y,
Birmingham A, Seo JB, Ofrecio JM, Wollam J, Hernandez-Carretero A,
Fu W, et al: Adipose tissue macrophage-derived exosomal miRNAs can
modulate in vivo and in vitro insulin sensitivity. Cell.
171:372–384.e12. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Piwecka M, Rolle K, Belter A, Barciszewska
AM, Żywicki M, Michalak M, Nowak S, Naskręt-Barciszewska MZ and
Barciszewski J: Comprehensive analysis of microRNA expression
profile in malignant glioma tissues. Mol Oncol. 9:1324–1340. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen G, Zhu W, Shi D, Lv L, Zhang C, Liu P
and Hu W: MicroRNA-181a sensitizes human malignant glioma U87MG
cells to radiation by targeting Bcl-2. Oncol Rep. 23:997–1003.
2010.PubMed/NCBI
|
|
30
|
Wilhelm I and Krizbai IA: In vitro models
of the blood-brain barrier for the study of drug delivery to the
brain. Mol Pharm. 11:1949–1963. 2014. View Article : Google Scholar : PubMed/NCBI
|