Introduction
Esophageal cancer is one of the most prevalent
malignancies in the world and accounted for >500,000 deaths in
2018 (1). Approximately half of all
esophageal cancer cases occur in China (2). Squamous cell carcinoma represents ~90%
of these cases (3). Currently, there
are limited treatment options for esophageal squamous cell
carcinoma (ESCC), resulting in a five-year survival rate of 17.9%
according to the Surveillance, Epidemiology, and End Results
database (4). Definitive radiation
therapy or chemoradiotherapy (CRT) are treatment options for
patients that are not able to undergo surgery (5). However, patients with locally advanced
ESCC demonstrate poor outcomes following radiation therapy, with a
median survival of 16–20 months worldwide (6–8). It is
unclear whether additional treatment results in reduced or lack of
recurrence and metastases following radiation. Therefore,
identification of clinically viable biomarkers that predict
survival following radiation therapy is urgently required. Previous
studies have focused on clinical and pathological factors, such as
sex, age, type, TNM stage, treatment, molecular markers and the
general condition of the patient (9–11).
However, to the best of our knowledge, widely accepted markers that
predict outcomes in patients with ESCC following radiation remain
unidentified.
Circulating tumor (ct)DNA is a tumor-derived DNA
found in the plasma of patients with cancer, that could be used as
a potential biomarker (12). This
non-invasive approach (13) could be
used to monitor tumor burden during treatment, regardless of tumor
heterogeneity (14–17). Using a cancer-personalized profiling
strategy, ctDNA was detected in 100% of patients with stages II–IV
and 50% of patients with stage I non-small cell lung cancer
(18). In a prospective study of 230
patients with early-stage colorectal cancer, ctDNA was assessed at
the first follow-up visit following surgical resection. The results
indicated that recurrence-free survival at 3 years was 0% for the
ctDNA-positive group and 90% for the ctDNA-negative group (19). Previous studies have reported an
interval of 7.9–11.0 months between ctDNA detection and clinical
relapse (14,16,20). In
addition, ctDNA was detected in the plasma of patients with ESCC
and was used for dynamic monitoring (longitudinally monitored
during treatment at different time point) (21).
In the present study, a unique gene panel was used
to evaluate consecutively enrolled patients with ESCC. Plasma
samples were sequenced and analyzed prior to and following
radiation therapy using next-generation sequencing (NGS)
techniques. In addition, ctDNA was monitored using a non-invasive
approach to predict disease outcome following radiation therapy and
identify specific mutations.
Materials and methods
Patient selection and sample
collection
Eligible patients with ESCC were retrospectively
recruited at the Department of Gastrointestinal Oncology, The Fifth
Medical Centre, Chinese People's Liberation Army General Hospital
(Beijing, China). The main inclusion criteria were pathologically
diagnosed ESCC and Eastern Cooperative Oncology Group (ECOG)
performance status ≤2. Only patients who were found to have
unresectable ESCC, as evaluated by a thoracic surgeon and those who
were candidates for definitive radiation therapy or chemoradiation
therapy were included. Patients had received enhanced computed
tomography (CT) scan of the chest and abdomen, and gastroscopy and
endoscopic ultrasonography when suitable, for the evaluation of
primary tumor. The patients were evaluated using the American Joint
Committee on Cancer 7th staging system (22). The patients with ESCC who presented
with other malignant tumors and/or other severe or uncontrolled
diseases were excluded. A total of 29 patients were initially
recruited, and 69 plasma samples were collected from 25 patients
with ESCC, who were enrolled between July 2013 and May 2016
(Table SI). All patients provided
signed informed consent and the study protocol was approved by PLA
307 Hospital Institutional Review Board. Clinical features were
obtained from the patients' medical records. Peripheral blood
samples were collected prior to (pre-radiation or baseline),
during, and following (post-radiation) radiation therapy, as well
as during follow-up visits every three months for ctDNA, CT scan
and tumor marker (CEA, CA19-9, and CA72-4) analyses. CEA, CA19-9
and CA72-4 were routinely tested by chemiluminescence immunoassay
in our hospital and the upper limits of normal were 4.3 ng/ml, 27
and 6.9 U/ml, respectively. Patients were divided according to
baseline ctDNA status (1 vs. >1), abnormal and normal biomarker
levels and according to the positive and negative ctDNA status
post-radiation. Plasma was separated by centrifugation at 1,600 × g
for 10 min, transferred to new microcentrifuge tubes, and
centrifuged at 16,000 × g for 10 min to remove cell debris at room
temperature. Peripheral blood lymphocytes (PBLs) from the first
centrifugation were used for the extraction of germline genomic
DNA.
DNA extraction and quality
control
Cell-free (cf)DNA was isolated from 0.6–1.8 ml
plasma using a QIAamp Circulating Nucleic Acid kit (Qiagen, Inc.).
Baseline PBL DNA was extracted using the QIAamp DNA Blood Mini kit
(Qiagen, Inc.) (23). cfDNA and PBL
DNA concentration levels were measured using a Qubit 3.0
fluorometer and a Qubit dsDNA High Sensitivity Assay kit (both from
Thermo Fisher Scientific Inc.). The fragments of cfDNA were
analyzed on an Agilent 2100 BioAnalyzer using the Agilent High
Sensitivity DNA kit (both from Agilent Technologies, Inc.) and only
samples with a fragment peak at ~170 bp passed quality control.
Qualified PBL DNA was identified using agarose gel
electrophoresis.
Target capture and NGS
A panel, including 180 genes was designed. Briefly,
the panel consisted of top 100 recurrent potential driver genes in
ESCC identified from the COSMIC database (24) (http://cancer.sanger.ac.uk/cosmic), top 100 oncogenes
and tumor suppressor genes (TSG), associated with tumorigenesis and
metastasis in ESCC (25–27), and top 100 genes associated with
other types of cancer were identified from The Cancer Genome Atlas
database (https://cancergenome.nih.gov) (28).
Sequencing libraries of both plasma cfDNA and
baseline PBL DNA were constructed using the KAPA LTP DNA Library
Preparation kit#KK8230 (Kapa Biosystems), as previously described
(23). For each sample, at least 30
ng cfDNA and 1.0 µg PBL DNA were used for library input. The
library length was measured using an Agilent 2100 Bioanalyzer
(Agilent Technologies, Inc). The libraries were analyzed by using
an Applied Biosystems 7500 Real-Time PCR system (Thermo Fisher
Scientific Inc.) and 2 n moles of the loading concentration of the
final library was used. Additional 15 pre-capture PCR cycles were
performed on samples close to the minimum input requirement to
generate sufficient PCR product for hybridization. The libraries
were hybridized to custom-designed biotinylated oligonucleotide
probes (Kapa Biosystems) covering ~0.5 Mbp. DNA sequencing was
performed on the HiSeq3000 Sequencing System (Illumina, Inc.) with
2×100-bp paired-end reads using TruSeq PE Cluster Generation Kit v3
and the TruSeq SBS Kit v3 (Illumina, Inc.).
Sequencing data analysis
Terminal adaptor sequences and low-quality reads
were removed from the raw sequencing data. The reads were then
aligned with the human genome build GRCh37 using a Burrows-Wheeler
aligner (29). Picard tools
(http://broadinstitute.github.io/picard/) were used to
mark PCR duplicates. Single nucleotide variants (SNVs), and
insertions and deletions were identified using MuTect (version
1.1.4) (30) and Genome Analysis
Toolkit (version 3.4-46-gbc02625) softwares (31), respectively. Sequencing results of
cfDNA samples were used to identify somatic mutations, while PBL
DNA was used to filter the germline mutations. All candidate
somatic mutations identified using the bioinformatics pipeline were
manually reviewed in the Integrative Genomics Viewer interactive
tool (32) by assessing the quality
of bases the mapping quality of reads and the overall read depth at
each mutation site. A mutation was identified as somatic if: i) a
variant allele fraction (VAF) ≥0.1%; and ii) at least 5
high-quality reads (Phred score, ≥30; mapping quality, ≥30, without
paired-end reads bias) were found. For a specific variant in the
plasma cfDNA, VAF was calculated using the following formula:
VAF=sequencing read count of alternate alleles/(sequencing read
count of reference alleles + sequencing read count of alternate
alleles) ×100%. The mutations were annotated to genes using the
ANNOVAR software (version 77) (33)
to identify the mutated protein-coding position, and filter
intronic and silent changes.
Statistical analysis
Overall survival (OS) was defined as the survival
time from the initiation of radiation therapy to the date of death
from any cause. Progression-free survival (PFS) was defined as the
survival time from the initiation of radiation therapy to the date
of disease progression or death from any cause. Cases without
progression or death events were censored at the date of last
follow-up. Survival curves were determined using the Kaplan-Meier
method and univariate analysis of these calculations were performed
using the log-rank test. Multivariate analysis was performed with
manual backward stepwise Cox regression modeling. The number of
mutations was compared using the Mann-Whitney U test. The Fisher's
exact test was used to compare proportions between the two groups.
All reported P-values were two-sided and P<0.05 was considered
to indicate a statistically significant difference. All statistical
analyses were performed using the SPSS version 21.0 (IBM Corp.) and
PFS and OS were shown by the GraphPad Prism version 5.0 (GraphPad
Software Inc.) softwares.
Results
Patient characteristics
All patients received definitive esophagus radiation
therapy or CRT. The plasma samples were obtained prior to radiation
for all patients and following radiation for 24 patients. The
clinical characteristics of the patients are shown in Table I. The median age at diagnosis was 60
years (range, 40–82 years), and males accounted for 88% of the
patients. A total of 52% of the primary tumors were located in the
upper third of the thoracic esophagus, while 72% of patients were
diagnosed with clinical stage T3 or T4 tumors and with regional LN
involvement. A total of 10 patients (40%) received CRT. The mean
duration of follow-up was 18.0±2.4 months, with a median of 14.7
months. During this period, 14 patients were documented with
disease progression, including local recurrence and/or distant
metastases.
 | Table I.Clinical characteristics of 25
patients with ESCC before radiation. |
Table I.
Clinical characteristics of 25
patients with ESCC before radiation.
| Clinical
characteristics | Value | Percentage |
|---|
| Sex |
|
Male | 22 | 88.0 |
|
Female | 3 | 12.0 |
| Median age, years
(range) | 60 (40–82) |
|
| ECOG performance
status |
| 0 | 3 | 12.0 |
| 1 | 19 | 76.0 |
| 2 | 3 | 12.0 |
| Location |
| Upper
thoracic | 13 | 52.0 |
|
Mid-thoracic | 10 | 40.0 |
| Lower
thoracic | 2 | 8.0 |
|
Differentiation |
|
Well | 1 | 4.0 |
|
Moderate | 15 | 60.0 |
|
Poor | 8 | 32.0 |
|
Unknown | 1 | 4.0 |
| T
stagea |
| T1 | 1 | 4.0 |
| T3 | 17 | 68.0 |
| T4 | 1 | 4.0 |
|
Unknown | 6 | 24.0 |
| N stage |
|
Positive | 18 | 72.0 |
|
Negative | 7 | 28.0 |
| Chemotherapy before
radiation |
|
Yes | 14 | 56.0 |
| No | 11 | 44.0 |
|
Chemoradiotherapy |
|
Yes | 10 | 40.0 |
| No | 15 | 60.0 |
| Tumor
biomarkersb |
|
Normal | 14 | 56.0 |
|
Abnormal | 11 | 44.0 |
Identification of mutations in the
patients with ESCC
NGS was performed on baseline and post-radiation
plasma samples from patients with ESCC. Following quality control
and removal of duplicates, a mean coverage depth of ×1,337 (range,
379–2,222) was achieved. Somatic mutations were recorded, with 59
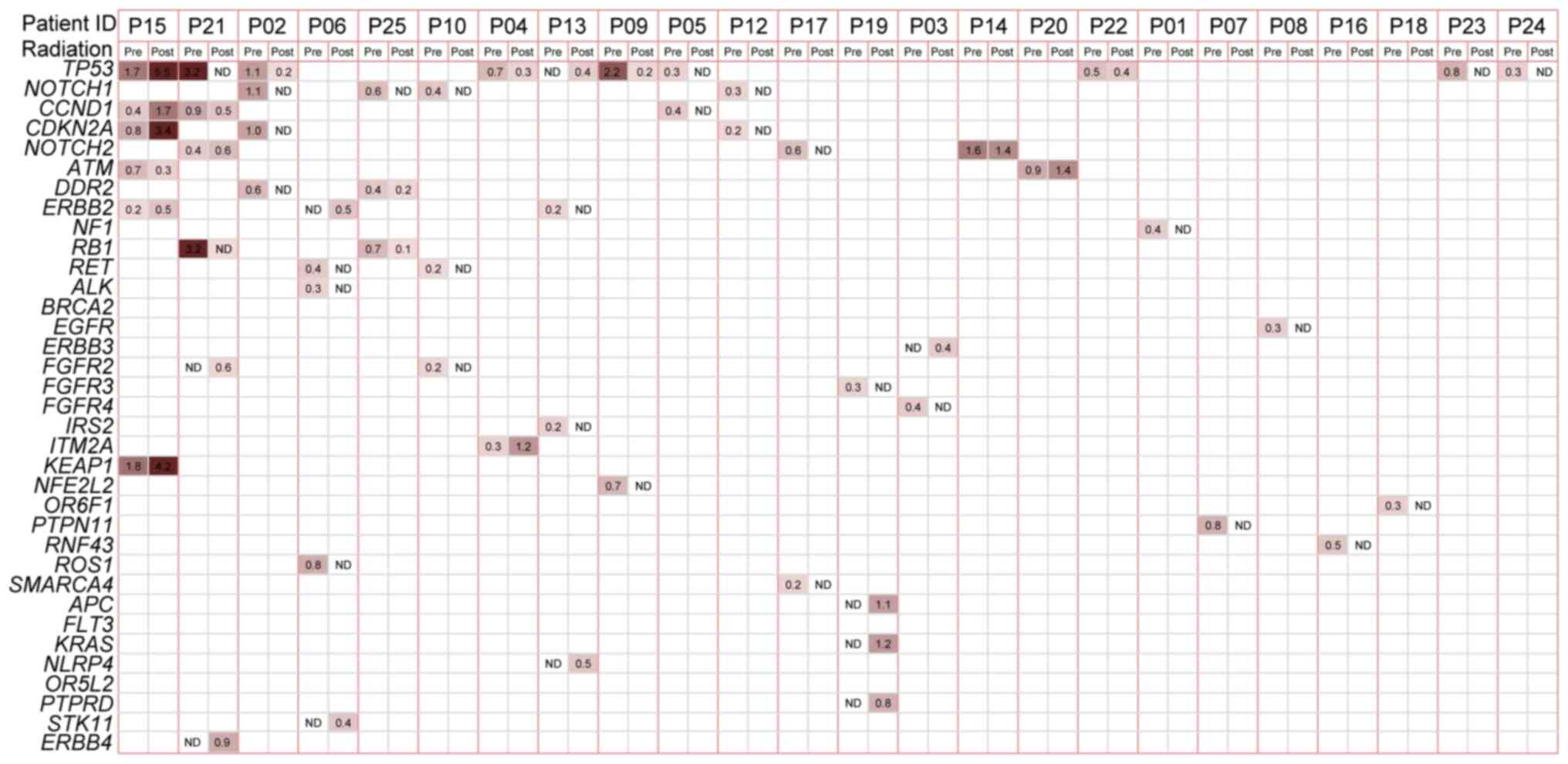
non-silent SNVs identified in 33 genes (Table SII).
The baseline mutation spectrum was analyzed. At
least one somatic mutation was detected per sample. The average
mutation burden at baseline was 1.9 (range, 1–6). A total of 13
patients had one mutation, while 12 patients harbored more than one
mutation. The most frequent mutation was noted in TP53,
which was mutated in 9 (36%) pre-treatment cancer cases. All of the
TP53 variations were either missense or non-sense and were
annotated in the COSMIC (Table SII)
database. Other frequently mutated genes included NOTCH1
(n=4), CCND1 (n=3), CNKN2A (n=3) and NOTCH2
(n=3). Notably, the majority of the mutated genes with a high
prevalence rate were TSGs.
The mutation spectrum in the initial post-radiation
plasma samples was compared with that in the baseline samples
(Fig. 1). Plasma mutations were
altered by radiation therapy. Baseline and matched post-radiation
samples were available for 24 patients, with the exception of one
patient (P11), for whom a post-treatment plasma sample was not
obtained. The mutation burden was lower in the first post-radiation
plasma sample, with an average of 1.1 (range, 0–6) compared with
that in the baseline sample. A total of 15 patients (62.5%)
exhibited a reduced mutation burden following therapy. A total of
10 (41.7%) patients did not exhibit mutations following radiation
(ctDNA-negative). As the most frequently mutated gene, TP53
was tracked following radiation in 5 patients and ATM, CCND1,
ERBB2 and NOTCH2 were also repeatedly found. The most
apparent difference was noted in the NOTCH1 gene, in which
no mutations occurred in the initial post-radiation samples of all
4 patients with baseline mutations. In addition, several mutations
were detected in the first post-radiation samples, whereas these
were not present in the baseline samples. These mutations were the
following: STK11 1062C>G (F354L) in P06 and KRAS
c.38G>A (p.G13D) missense and APC c.4285C>T (p.Q1429*)
truncation in P19.
Association between ctDNA and clinical
features
The association between baseline ctDNA and the
clinical features of the patients was examined. No association was
noted between sex, ECOG performance status, location, or
differentiation and baseline mutation burden (data not shown). A
total of 12 out of 18 patients (67%) with lymph node metastasis
(LNM) (LN-positive) exhibited >1 mutation at baseline compared
with that in patients who were LN-negative and only had one
mutation (Fig. S1A). In addition,
46.2% (6/13) of patients with one mutation and 100% of patients
with more than one mutation were LN-positive (Fisher's exact test,
patients with one mutation vs. those with more than one mutation;
P=0.005).
Furthermore, the status of ctDNA in the first
post-radiation plasma samples was further examined. These samples
were collected within 2 months of radiation for 19 patients, within
2–6 months following radiation for 3 patients and within 6 months
following radiation for 2 patients. The patients received no
further treatment between the end of radiation therapy and the
first plasma collection following radiation. A total of 12 out of
17 (71%) LN-positive patients and 29% (2/7) of LN-negative patients
harbored mutations in the first post-radiation plasma sample, and
more mutations were detected in patients with LNM (Fig. S1B). In the 14 ctDNA-positive (at
least one mutation detected) patients, 90.9% (10/11) had documented
disease recurrence, while three patients exhibited no recurrence
status and did not survive following 10 months of disease
progression. In the 10 ctDNA-negative patients, 50% (4/8) had
documented disease recurrence while two patients did not exhibit
recurrence status (data not shown).
Predictive and prognostic values
To determine the prognostic value of ctDNA, the
association of the baseline mutations with patient outcome was
examined. No significant differences were noted for PFS (P=0.221)
or OS (P=0.579) times (Fig. S2A and
B) between patients harboring one and more than one mutation.
The specific baseline mutation of P53 was also evaluated and it was
found not to be a prognostic marker for PFS (P=0.055) and OS
(P=0.147; data not shown).
Subsequently, the association between post-radiation
ctDNA status and PFS and OS times was investigated. Patients who
were ctDNA-positive exhibited a marginally significant reduction in
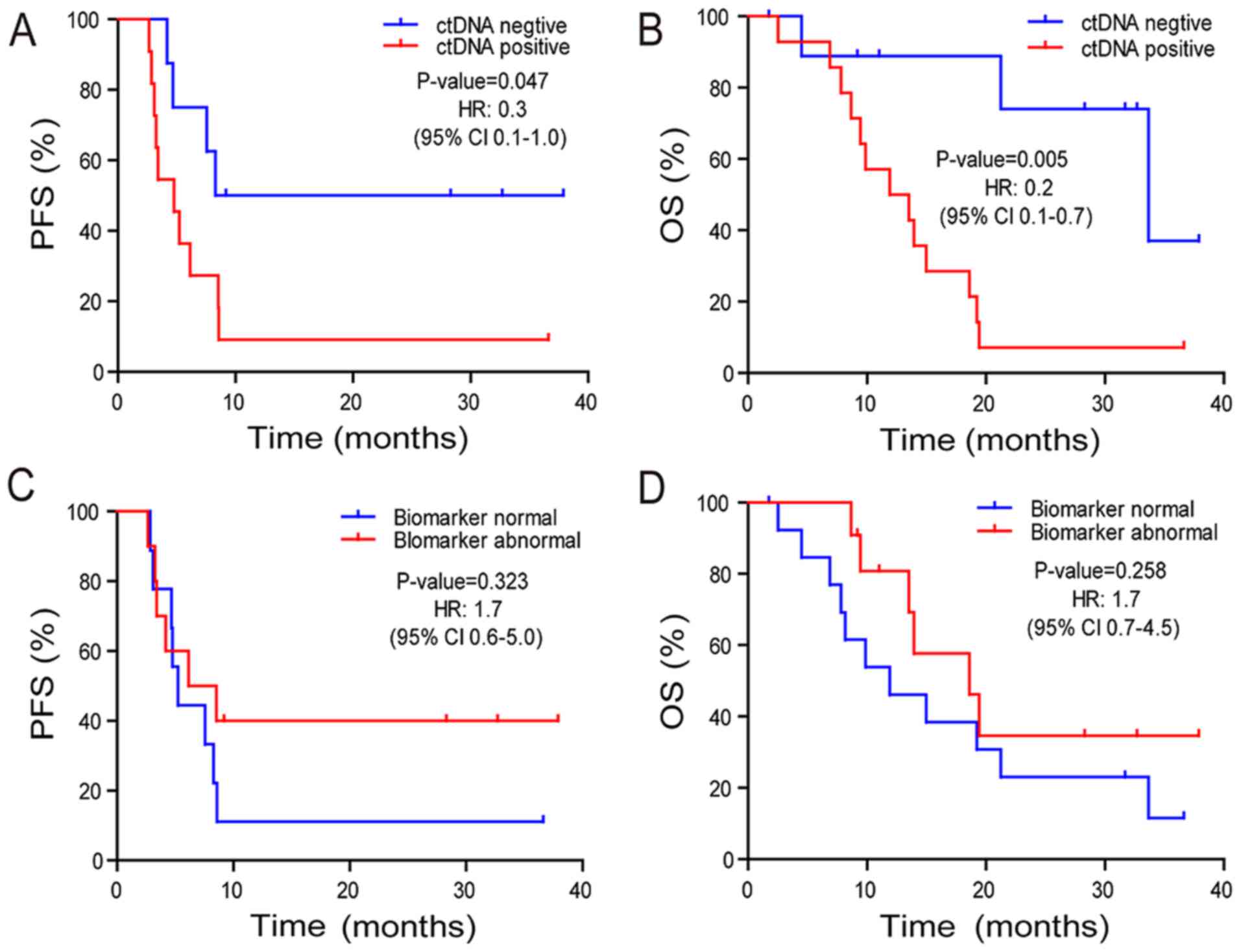
PFS (P=0.047) time and a significantly decreased OS (P=0.005) time
compared with those in patients who were ctDNA-negative (Fig. 2A and B). The median OS time was 33.7
and 11.9 months for the ctDNA-negative and -positive groups,
respectively. The two-year survival rate was 74.1 and 7.1% in the
ctDNA-negative and -positive groups (odds ratio, 24.8; 95% CI,
1.7–1,625.0; P=0.0055) (Fig. 2B).
Abnormal tumor biomarkers CEA, CA19-9, and CA72-4 did not exhibit a
significant association with PFS (P=0.323) or OS (P=0.258) times
(Fig. 2C and D) compared with normal
tumor biomarkers.
In addition to the aforementioned data, the
histopathological variables were integrated with the ctDNA status
in the initial post-radiation plasma samples and a multivariate
analysis was conducted. According to the univariate analysis, only
ctDNA status was significantly associated with OS time (Table II). Multivariate Cox proportional
hazard regression analysis revealed that the presence of ctDNA in
the initial post-radiation plasma sample was an independent
prognostic factor for patients with ESCC (P=0.011; Table III).
 | Table II.Univariate analysis of clinical
characteristics affecting OS time. |
Table II.
Univariate analysis of clinical
characteristics affecting OS time.
| Clinical
characteristics | Number (n=25) | OS (95% CI) | P-value |
|---|
| Sex |
|
| 0.373 |
|
Male | 22 | 14.97
(7.75–22.19) |
|
|
Female | 3 | 9.87 |
|
| ECOG performance
status |
|
| 0.174 |
|
0/1 | 22 | 18.62
(10.86–26.38) |
|
| 2 | 3 | 9.87
(9.18–10.56) |
|
| Tumor location |
|
| 0.482 |
| Upper
1/3 | 13 | 14.97
(3.73–26.21) |
|
| Middle
1/3 | 10 | 13.52
(1.34–25.70) |
|
| Lower
1/3 | 2 | 9.44 |
|
|
Differentiation |
|
| 0.221 |
|
Well |
|
|
|
|
Moderate | 15 | 11.91
(5.22–18.60) |
|
|
Poor | 8 | 21.11
(6.01–35.17) |
|
|
Unknown | 1 |
|
|
| N stage |
|
| 0.131 |
|
Positive | 18 | 12.93
(7.63–18.22) |
|
|
Negative | 7 | 23.07
(7.96–36.20) |
|
| Tumor
biomarkers |
|
| 0.258 |
|
Normal | 14 | 11.91
(3.91–19.91) |
|
|
Abnormal | 11 | 18.62
(8.50–28.74) |
|
| Chemotherapy before
radiation |
|
| 0.350 |
|
Yes | 14 | 14.97
(6.27–23.67) |
|
| No | 11 | 13.95
(0.94–26.96) |
|
|
Chemoradiotherapy |
|
| 0.552 |
|
Yes | 10 | 18.62
(10.80–26.44) |
|
| No | 15 | 13.52
(4.82–22.22) |
|
| ctDNA status in
first post-treatment plasma |
|
| 0.005 |
|
Negative | 10 | 33.68
(15.66–51.70) |
|
|
Positive | 14 | 11.91
(5.22–18.60) |
|
 | Table III.Multivariate analysis of clinical
characteristics affecting OS. |
Table III.
Multivariate analysis of clinical
characteristics affecting OS.
| Clinical
characteristics | HR (95% CI) | P-value |
|---|
| ECOG (0/1 vs.
2) | 0.691
(0.176–2.710) | 0.596 |
| N stage (negative
vs. positive) | 0.466
(0.110–1.983) | 0.302 |
| ctDNA status in
first post-radiation plasma (negative vs. positive) | 0.183
(0.049–0.678) | 0.011 |
ctDNA dynamic monitoring
ctDNA may also be used as a longitudinal monitoring
biomarker during and following treatment to evaluate disease
progression (Fig. S3). Mutations in
both P1 and P10 were undetectable following radiation therapy,
which was consistent with the clinical findings, demonstrating that
both patients were disease-free during follow-up. The frequency of
the NOTCH2 mutation in P14 was decreased following
radiation; however, it was still detectable and was increased
afterwards, which was consistent with multiple LNMs detected by CT
scan, at that time point. Elevated VAF values were detected in
several driver genes (TP53, KEAP1 and CDKN2A) in P15
following radiation; however, disease progression was detected from
imaging analysis 4 months following radiation.
Discussion
Optimal treatment for the upper esophagus or locally
advanced disease remains problematic for patients with ESCC
(34). Esophagectomy can cause
serious postoperative complications, such as anastomotic leakage
and pneumonia (35,36). Clinical trial results, that compared
chemoradiation followed by surgery with chemoradiation alone,
indicated similar median survival rates and 2-year survival rates,
of no more than 40% for both groups (7). These data suggested that the majority
of the patients who received surgery remained vulnerable to local
recurrence or distant metastasis. Thus, how to select the right
patient for surgery is essential. The establishment of a
multi-disciplinary and more effective treatment modality for
patients with ESCC would improve disease outcomes. The development
of a viable marker that could predict outcome following radiation
therapy would facilitate more informed treatment decisions.
ctDNA is a highly sensitive biomarker, that has been
used in multiple types of cancer, including melanoma (37), breast cancer (38) and colorectal cancer (39). ctDNA reflects tumor burden and
dynamic alterations, that can be monitored non-invasively during
treatment (40). The presence of
ctDNA following curative therapy indicated minimal residual disease
and predicted recurrence (19,20). In
ESCC, Luo et al (21)
designed a panel of 90 genes, which accounted for at least one
mutation in 94% of patients and provided promising results for
ctDNA detection. However, the prognostic value of ctDNA was not
evaluated in ESCC. Therefore, to the best of our knowledge, the
present study was the first to report ctDNA detection in a cohort
of patients with locally advanced ESCC. The results demonstrated
that the mutations found in the plasma ctDNA, following radiation
therapy, were the most efficacious independent prognostic factors
for patients with ESCC. Both LNM and baseline mutation number were
not prognostic factors in the present study. Therefore, the outcome
from radiation was affected by the ctDNA biomarker following
radiation and was not associated with the pre-radiation
disease.
A marginally significant difference in PFS was
observed for patients who were ctDNA-positive and -negative,
following radiation. Furthermore, the high recurrence rate of
patients who were ctDNA-positive indicated a positive predictive
value of ctDNA, suggesting that they should be monitored carefully.
In contrast to the patients who were ctDNA-positive, 50% of the
patients who were ctDNA-negative exhibited disease recurrence,
which indicated a considerably lower negative predictive value
(NPV) of ctDNA. However, 3 out of 4 patients with ctDNA-negative
status exhibited a relatively long OS time (>20 months).
Therefore, ctDNA negativity was not a strong predictor of prolonged
PFS time; however, it may be an indicator of longer survival time.
Based on a higher recurrence rate and worse OS time, patients who
are ctDNA-positive may require additional treatments following
radiation, including chemotherapy or surgery. In addition, it could
be hypothesized that immunotherapy performed in non-small cell lung
cancer (41,42) could also be applied in ESCC (43) in the future. Furthermore, due to the
poor NPV, the ctDNA assays require further optimization by addition
of other genes, notably those associated with radiation resistance,
such as OBSCN and DGKK (44).
Patients with ESCC, in the present study, indicated
heterogeneous mutant gene profiles. The most prevalent mutated
genes encoded tumor suppressor proteins. The absence of hot-spot
mutations increases the suitability of using NGS and a large gene
panel for the identification of molecular markers. A notable
difference was noted in the number of mutations in the
post-radiation samples was the disappearance of NOTCH1
mutations, which were only detected in baseline samples. This
result suggested that cancer cell populations harboring
NOTCH1 mutations may be more sensitive to radiation therapy,
as previously described (44).
Future large-scale studies should aim to investigate the efficacy
of radiation in patients harboring NOTCH1 mutations and
notably clonal NOTCH1. The presence of new mutations after
radiation, such as STK11, which have been previously
associated with radioresistance (45), warrants future evaluation, with
respect to their association with disease progression.
The VAF values of the mutations, in this study, were
low, reflecting the low plasma concentration of ctDNA. This may be
due to the relatively early stage of tumors, that were used in the
present study, which included locally advanced cancers lacking
metastases at baseline. Plasma ctDNA concentration has been
previously associated with tumor size (15) and stage (12). The analysis of tumor tissue volume,
measured using CT, and plasma ctDNA VAF indicated that tumor size
was associated with mean plasma VAF of clonal SNVs (15). In a previous study, the
quantification of tumor mutations in each patient, using digital
droplet PCR revealed that patients with stage I disease rarely
exhibited >10 copies per 5 ml plasma. By contrast, patients with
advanced prostate, ovarian, or colorectal cancer exhibited a median
concentration of 100–1,000 copies per 5 ml plasma (12). In addition, the amount of ctDNA
released from different tissue-derived tumors into the blood was
distinct. For example, a higher number of ctDNA copies was detected
per 5 ml plasma in patients with advanced colorectal cancer
compared with that in patients with advanced gastroesophageal
cancer (12). We found that the
general VAF may be relatively low in ESCC compared with that in
colorectal and ovarian cancers (46). Nevertheless, the use of integrated
digital error suppression technology, based on a molecular barcode,
ensures that ctDNA mutations can be detected at VAFs as low as
0.1%, such as in lung cancer (47).
High heterogeneity among patients with ESCC has been
reported in several large-scale genomic studies (25,26,48,49).
Several hundreds of somatic mutations exist; however, no single
mutation has been found to be widespread among all patients. The
limitations of the present study can be summarized as follows:
Firstly, the small sample size and the small number of mutations
detected prevented analysis of the significance of individual genes
(such as TP53), including prognostic value, presence of
local recurrence or metastases and predictive value of chemotherapy
or radiation therapy. In addition, the small number of baseline
mutations and LNM led to poor statistical power with respect to the
detection of significant differences in disease prognosis.
Secondly, since the present study was retrospective, several
inconsistencies may lead to potentially substantial confounding
recurrence outcomes, such as treatment heterogeneity (including the
concurrent therapy with radiation and different chemotherapy
regimens) and the heterogeneity of time points at which the ctDNA
was acquired following therapy. These deviations could be corrected
in a future prospective study including a larger cohort of
patients.
In patients with locally advanced ESCC, ctDNA was
detectable prior to and following radiation therapy. The ctDNA
status in the first post-radiation plasma samples could be used as
an independent prognostic factor for patients with locally advanced
ESCC.
Supplementary Material
Supporting Data
Supporting Data
Supporting Data
Acknowledgements
The abstract of the study has been presented in the
2018 54th American Society of Clinical Oncology annual meeting in
Chicago, United States of America, June 1st to June 5th and
published as an abstract no. e16053 in Journal of Clinical Oncology
2018 36:15_suppl, e16053.
Funding
The present study was supported by the Application
of Clinical Features of Capital City (grant no.
Z141107002514105).
Availability of data and materials
The details of the mutations in each patient are
presented in Table SII. The other
datasets used and/or analyzed during the current study are
available from the corresponding author upon reasonable
request.
Author's contributions
JMX, RJ and CHZ conceived the project and designed
experiments. CHZ, RRL, YZ and HEC contributed to the acquisition of
the data. RJ, CHZ and PSL contributed to the raw data collection
and organization. PSL, LPC, YHG, YFG and XY performed the
experiments and analyzed the data. RJ, PSL and JMX wrote the
manuscript with assistance and feedback of all the other
co-authors. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The study was approved by the PLA 307 Hospital
Institutional Review Board. All patients provided signed informed
consent.
Patient consent for publication
All patients whose data were included in the current
study provided informed consent for publication.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Feng RM, Zong YN, Cao SM and Xu RH:
Current cancer situation in China: Good or bad news from the 2018
global cancer statistics? Cancer Commun (Lond). 39:222019.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Worni M, Martin J, Gloor B, Pietrobon R,
D'Amico TA, Akushevich I and Berry MF: Does surgery improve
outcomes for esophageal squamous cell carcinoma? An analysis using
the surveillance epidemiology and end results registry from 1998 to
2008. J Am Coll Surg. 215:643–651. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen Y, Ye J, Zhu Z, Zhao W, Zhou J, Wu C,
Tang H, Fan M, Li L, Lin Q, et al: Comparing paclitaxel plus
fluorouracil versus cisplatin plus fluorouracil in
chemoradiotherapy for locally advanced esophageal squamous cell
cancer: A randomized, multicenter, phase III clinical trial. J Clin
Oncol. 37:1695–1703. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rawat S, Kumar G, Kakria A, Sharma MK and
Chauhan D: Chemoradiotherapy in the management of locally advanced
squamous cell carcinoma esophagus: Is surgical resection required?
J Gastrointest Cancer. 44:277–284. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bedenne L, Michel P, Bouche O, Milan C,
Mariette C, Conroy T, Pezet D, Roullet B, Seitz JF, Herr JP, et al:
Chemoradiation followed by surgery compared with chemoradiation
alone in squamous cancer of the esophagus: FFCD 9102. J Clin Oncol.
25:1160–1168. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Conroy T, Galais MP, Raoul JL, Bouché O,
Gourgou-Bourgade S, Douillard JY, Etienne PL, Boige V, Martel-Lafay
I, Michel P, et al: Definitive chemoradiotherapy with FOLFOX versus
fluorouracil and cisplatin in patients with oesophageal cancer
(PRODIGE5/ACCORD17): Final results of a randomised, phase 2/3
trial. Lancet Oncol. 15:305–314. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chen MF, Yang YH, Lai CH, Chen PC and Chen
WC: Outcome of patients with esophageal cancer: A nationwide
analysis. Ann Surg Oncol. 20:3023–3030. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen MF, Chen PT, Lu MS, Lee CP and Chen
WC: Survival benefit of surgery to patients with esophageal
squamous cell carcinoma. Sci Rep. 7:461392017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Stahl M, Lehmann N, Walz MK, Stuschke M
and Wilke H: Prediction of prognosis after trimodal therapy in
patients with locally advanced squamous cell carcinoma of the
oesophagus. Eur J Cancer. 48:2977–2982. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bettegowda C, Sausen M, Leary RJ, Kinde I,
Wang Y, Agrawal N, Bartlett BR, Wang H, Luber B, Alani RM, et al:
Detection of circulating tumor DNA in early- and late-stage human
malignancies. Sci Transl Med. 6:224ra242014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Murtaza M, Dawson SJ, Pogrebniak K, Rueda
OM, Provenzano E, Grant J, Chin SF, Tsui DWY, Marass F, Gale D, et
al: Multifocal clonal evolution characterized using circulating
tumour DNA in a case of metastatic breast cancer. Nat Commun.
6:87602015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Olsson E, Winter C, George A, Chen Y,
Howlin J, Tang MH, Dahlgren M, Schulz R, Grabau D, van Westen D, et
al: Serial monitoring of circulating tumor DNA in patients with
primary breast cancer for detection of occult metastatic disease.
EMBO Mol Med. 7:1034–1047. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Abbosh C, Birkbak NJ, Wilson GA,
Jamal-Hanjani M, Constantin T, Salari R, Le Quesne J, Moore DA,
Veeriah S, Rosenthal R, et al: Phylogenetic ctDNA analysis depicts
early-stage lung cancer evolution. Nature. 545:446–451. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Reinert T, Scholer LV, Thomsen R, Tobiasen
H, Vang S, Nordentoft I, Lamy P, Kannerup AS, Mortensen FV,
Stribolt K, et al: Analysis of circulating tumour DNA to monitor
disease burden following colorectal cancer surgery. Gut.
65:625–634. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pectasides E, Stachler MD, Derks S, Liu Y,
Maron S, Islam M, Alpert L, Kwak H, Kindler H, Polite B, et al:
Genomic heterogeneity as a barrier to precision medicine in
gastroesophageal adenocarcinoma. Cancer Discov. 8:37–48. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Newman AM, Bratman SV, To J, Wynne JF,
Eclov NC, Modlin LA, Liu CL, Neal JW, Wakelee HA, Merritt RE, et
al: An ultrasensitive method for quantitating circulating tumor DNA
with broad patient coverage. Nat Med. 20:548–554. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tie J, Wang Y, Tomasetti C, Li L, Springer
S, Kinde I, Silliman N, Tacey M, Wong HL, Christie M, et al:
Circulating tumor DNA analysis detects minimal residual disease and
predicts recurrence in patients with stage II colon cancer. Sci
Transl Med. 8:346ra922016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Garcia-Murillas I, Schiavon G, Weigelt B,
Ng C, Hrebien S, Cutts RJ, Cheang M, Osin P, Nerurkar A, Kozarewa
I, et al: Mutation tracking in circulating tumor DNA predicts
relapse in early breast cancer. Sci Transl Med. 7:302ra1332015.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Luo H, Li H, Hu Z, Wu H, Liu C, Li Y,
Zhang X, Lin P, Hou Q, Ding G, et al: Noninvasive diagnosis and
monitoring of mutations by deep sequencing of circulating tumor DNA
in esophageal squamous cell carcinoma. Biochem Biophys Res Commun.
471:596–602. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Edge SB, Byrd DR, Compton CC, Fritz AG,
Greene FL and Trotti A: AJCC Cancer Staging Manual. (7th edition).
Springer; New York, NY: pp. 103–115. 2010
|
|
23
|
Yang X, Chu Y, Zhang R, Han Y, Zhang L, Fu
Y, Li D, Peng R, Li D, Ding J, et al: Technical validation of a
next-generation sequencing assay for detecting clinically relevant
levels of breast cancer-related single-nucleotide variants and copy
number variants using simulated Cell-Free DNA. J Mol Diagn.
19:525–536. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Forbes SA, Beare D, Boutselakis H, Bamford
S, Bindal N, Tate J, Cole CG, Ward S, Dawson E, Ponting L, et al:
COSMIC: Somatic cancer genetics at high-resolution. Nucleic Acids
Res. 45:D777–D783. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Song Y, Li L, Ou Y, Gao Z, Li E, Li X,
Zhang W, Wang J, Xu L, Zhou Y, et al: Identification of genomic
alterations in oesophageal squamous cell cancer. Nature. 509:91–95.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gao YB, Chen ZL, Li JG, Hu XD, Shi XJ, Sun
ZM, Zhang F, Zhao ZR, Li ZT, Liu ZY, et al: Genetic landscape of
esophageal squamous cell carcinoma. Nat Genet. 46:1097–1102. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Qin HD, Liao XY, Chen YB, Huang SY, Xue
WQ, Li FF, Ge XS, Liu DQ, Cai Q, Long J, et al: Genomic
characterization of esophageal squamous cell carcinoma reveals
critical genes underlying tumorigenesis and poor prognosis. Am J
Hum Genet. 98:709–727. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cancer Genome Atlas Research Network, ;
Weinstein JN, Collisson EA, Mills GB, Shaw KR, Ozenberger BA,
Ellrott K, Shmulevich I, Sander C and Stuart JM: The Cancer Genome
Atlas Pan-Cancer analysis project. Nat Genet. 45:1113–1120. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li H and Durbin R: Fast and accurate short
read alignment with Burrows-Wheeler transform. Bioinformatics.
25:1754–1760. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Cibulskis K, Lawrence MS, Carter SL,
Sivachenko A, Jaffe D, Sougnez C, Gabriel S, Meyerson M, Lander ES
and Getz G: Sensitive detection of somatic point mutations in
impure and heterogeneous cancer samples. Nat Biotechnol.
31:213–219. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
McKenna A, Hanna M, Banks E, Sivachenko A,
Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly
M and DePristo MA: The genome analysis toolkit: A MapReduce
framework for analyzing next-generation DNA sequencing data. Genome
Res. 20:1297–1303. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Robinson JT, Thorvaldsdottir H, Winckler
W, Guttman M, Lander ES, Getz G and Mesirov JP: Integrative
genomics viewer. Nat Biotechnol. 29:24–26. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang K, Li M and Hakonarson H: ANNOVAR:
Functional annotation of genetic variants from high-throughput
sequencing data. Nucleic Acids Res. 38:e1642010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Oh D and Kim JH: The current evidence on
neoadjuvant therapy for locally advanced esophageal squamous cell
carcinoma. Korean J Thorac Cardiovasc Surg. 53:160–167. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wu PC and Posner MC: The role of surgery
in the management of oesophageal cancer. Lancet Oncol. 4:481–488.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Stahl M, Stuschke M, Lehmann N, Meyer HJ,
Walz MK, Seeber S, Klump B, Budach W, Teichmann R, Schmitt M, et
al: Chemoradiation with and without surgery in patients with
locally advanced squamous cell carcinoma of the esophagus. J Clin
Oncol. 23:2310–2317. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tsao SC, Weiss J, Hudson C, Christophi C,
Cebon J, Behren A and Dobrovic A: Monitoring response to therapy in
melanoma by quantifying circulating tumour DNA with droplet digital
PCR for BRAF and NRAS mutations. Sci Rep. 5:111982015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Dawson SJ, Tsui DW, Murtaza M, Biggs H,
Rueda OM, Chin SF, Dunning MJ, Gale D, Forshew T, Mahler-Araujo B,
et al: Analysis of circulating tumor DNA to monitor metastatic
breast cancer. N Engl J Med. 368:1199–1209. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Diehl F, Schmidt K, Choti MA, Romans K,
Goodman S, Li M, Thornton K, Agrawal N, Sokoll L, Szabo SA, et al:
Circulating mutant DNA to assess tumor dynamics. Nat Med.
14:985–990. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang Y, Zhao C, Chang L, Jia R, Liu R,
Zhang Y, Gao X, Li J, Chen R, Xia X, et al: Circulating tumor DNA
analyses predict progressive disease and indicate
trastuzumab-resistant mechanism in advanced gastric cancer.
EBioMedicine. 43:261–269. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Antonia SJ, Villegas A, Daniel D, Vicente
D, Murakami S, Hui R, Kurata T, Chiappori A, Lee KH, de Wit M, et
al: Overall survival with durvalumab after chemoradiotherapy in
stage III NSCLC. N Engl J Med. 379:2342–2350. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Antonia SJ, Villegas A, Daniel D, Vicente
D, Murakami S, Hui R, Yokoi T, Chiappori A, Lee KH, de Wit M, et
al: Durvalumab after chemoradiotherapy in stage III non-small-cell
lung cancer. N Engl J Med. 377:1919–1929. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kelly RJ, Lockhart AC, Jonker DJ, Melichar
B, Andre T, Chau I, Clarke SJ, Cleary JM, Doki Y, Franke FA, et al:
CheckMate 577: A randomized, double-blind, phase 3 study of
nivolumab (Nivo) or placebo in patients (Pts) with resected lower
esophageal (E) or gastroesophageal junction (GEJ) cancer. J Clin
Oncol. 35 (4 Suppl):TPS2122017. View Article : Google Scholar
|
|
44
|
Yang L, Zhang X, MacKay M, Foox J, Hou Q,
Zheng X, Zhou R, Huang M, Jing Z, Mason CE and Wu S: Identification
of radioresponsive genes in esophageal cancer from longitudinal and
single cell exome sequencing. Int J Radiat Oncol Biol Phys.
108:1103–1114. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
He Q, Li J, Dong F, Cai C and Zou X: LKB1
promotes radioresistance in esophageal cancer cells exposed to
radiation, by suppression of apoptosis and activation of autophagy
via the AMPK pathway. Mol Med Rep. 16:2205–2210. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Phallen J, Sausen M, Adleff V, Leal A,
Hruban C, White J, Anagnostou V, Fiksel J, Cristiano S, Papp E, et
al: Direct detection of early-stage cancers using circulating tumor
DNA. Sci Transl Med. 9:eaan24152017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Newman AM, Lovejoy AF, Klass DM, Kurtz DM,
Chabon JJ, Scherer F, Stehr H, Liu CL, Bratman SV, Say C, et al:
Integrated digital error suppression for improved detection of
circulating tumor DNA. Nat Biotechnol. 34:547–555. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Lin DC, Hao JJ, Nagata Y, Xu L, Shang L,
Meng X, Sato Y, Okuno Y, Varela AM, Ding LW, et al: Genomic and
molecular characterization of esophageal squamous cell carcinoma.
Nat Genet. 46:467–473. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Hao JJ, Lin DC, Dinh HQ, Mayakonda A,
Jiang YY, Chang C, Jiang Y, Lu CC, Shi ZZ, Xu X, et al: Spatial
intratumoral heterogeneity and temporal clonal evolution in
esophageal squamous cell carcinoma. Nat Genet. 48:1500–1507. 2016.
View Article : Google Scholar : PubMed/NCBI
|