Introduction
Metastatic tumors are usually highly vascularized,
and this increased vascularity aids the dissemination of tumor
cells to promote metastatic events. This high vascularity is
induced via the expression of pro-angiogenic cytokines, which
promote the development of abnormal tumor vasculature. The abnormal
vasculature is characterized by hyperpermeable vessels, increased
vessel diameter and abnormally thickened basement membranes. All
these factors contribute to tumor growth (1–4). Several
anti-angiogenic agents, such as bevacizumab, sunitinib and
vatalanib have been developed to target increased tumor vascularity
(5). The goal of anti-angiogenic
therapy is the obliteration of tumor-induced vasculature with the
aim of decreasing vascular permeability and the perfusion of oxygen
and nutrients to tumor cells. This approach has often been termed
as ‘tumor starvation’. A converse approach to anti-angiogenic
therapy is the use of agents to stabilize abnormal tumor
vasculature by reducing blood vessel diameter and permeability,
controlling vessel perfusion, reducing tumor interstitial pressure
and improving tumor oxygenation with the aim of reducing tumor
hypoxia and thereby controlling metastasis (6–9).
Researchers have even suggested that anti-angiogenic therapy may
transiently normalize tumor vasculature and its microenvironment,
thus enhancing the efficacy of chemoradiotherapy (10). Unfortunately, both these approaches
have failed to achieve a clinically relevant standing despite the
vast amount of research being conducted. The main drawback of these
approaches is that they have been directed at single targets and
have not produced clinically consistent desirable outcomes. In the
present study, the targeting of multiple pro-angiogenic cytokines
to achieve a complete anti-angiogenic outcome is proposed. This
multipronged approach aims to exploit the microRNA (miRNA, miR)
machinery to attain a clinically desirable outcome. The present
study may pave the way for the development of a multi-targeted
strategy, as well as methodologies that could be applicable to
numerous other disease conditions.
The use of RNA molecules to target angiogenesis is
not new (11–14). Various approaches have been developed
to target angiogenesis, including the use of RNA aptamers, miRNAs,
small interfering RNAs (siRNAs) and combinations thereof. Although
promising, the half-life of RNA molecules is very short. In
vitro, natural RNA exhibits a half-life of a few seconds to a
few minutes in various biological fluids, including human serum,
whereas RNAs partially modified at the 20th position have extended
half-lives of 5 sec to 15 h (15).
An RNA-based anti-angiogenesis drug called Macugen has these
modifications; however, although somewhat successful, it is limited
in addressing the problem of angiogenesis (16). The approach used in the present study
is the development of a closed circular RNA having multiple
double-stranded regions coding for miRNAs, which are interrupted by
loops or dumbbells. Using a circular RNA should eliminate
exonuclease activity while double-stranded RNA (dsRNA), being
inherently more stable than single-stranded RNA (ssRNA), should
contribute to an increased half-life. To the best of our knowledge,
this is the first attempt to use closed circular dumbbell RNAs
(dbRNAs) to code for miRNAs/siRNAs with a therapeutic intention.
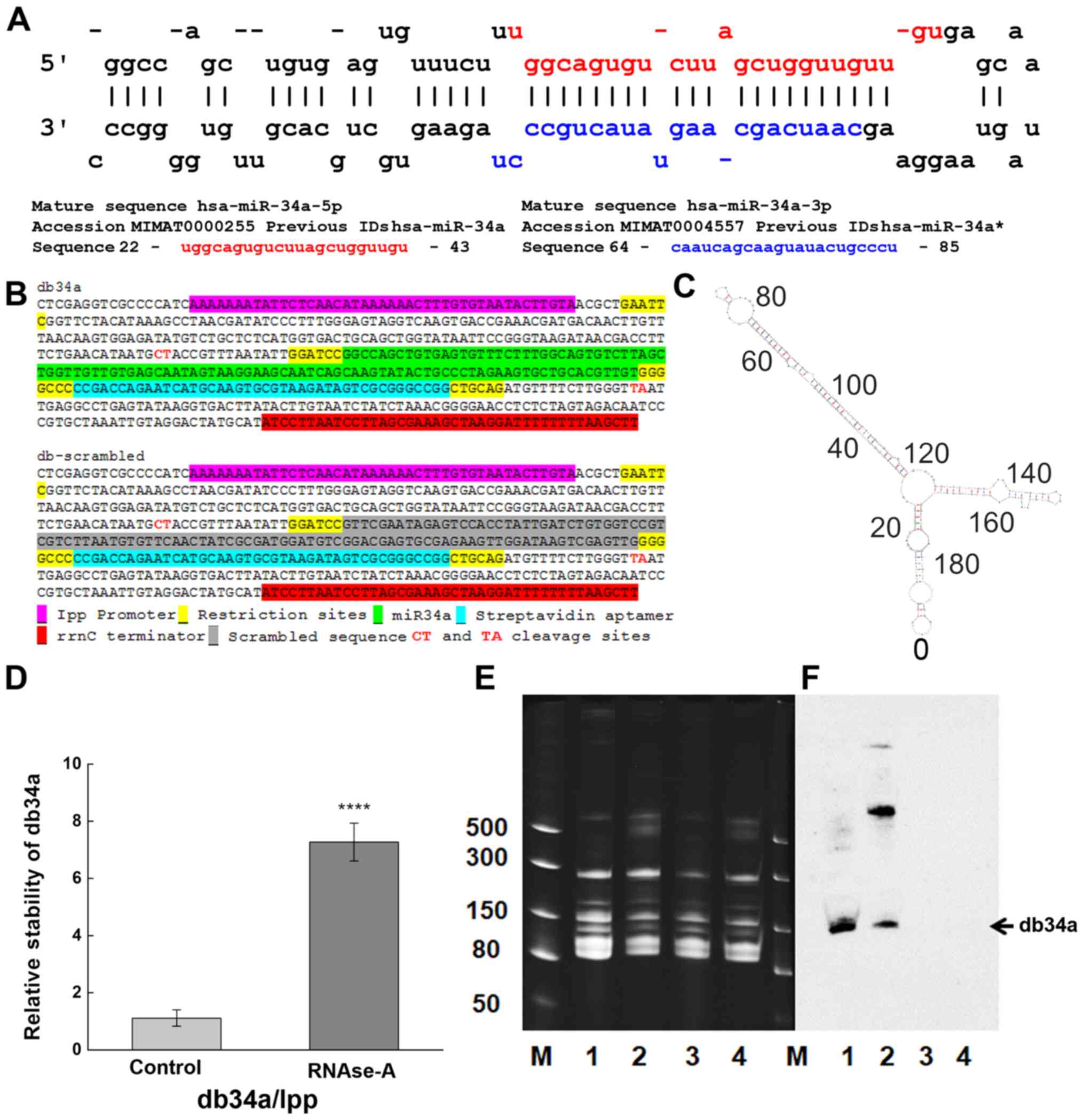
The study uses miR-34a-3p and −5p in a circular form.
Materials and methods
Construction of dbRNAs
The permuted intron-exon (PIE) method was used,
which is an enzyme-free RNA circularization method based on group I
intron self-splicing (17).
Circularization is triggered by the presence of magnesium ions and
guanine nucleotides, and the production of circular RNA can be
conducted using essentially any type of cell (17–19). A
previously described strategy was used, in which recombinant RNA is
disguised as a natural RNA and thus appropriates the host machinery
to evade cellular RNases (20), but
with an added RNA circularization step using the rrnC terminator
(21). Sequences coding for
miR-34a-3p and −5p (db34a) and its scrambled sequence (dbSCR) were
synthesized with the lpp promoter and the rrnC terminator sequences
(Fig. 1A and B), and inserted in
pUC57-Kan plasmids (GenScript Biotech). The JM101Tr E. coli
strain [Δ(lac pro), supE, thi, recA56, srl-300.:Tn10,
(F′, traD36, proAB, lacIq, lacZ, ΔM15)] (20) was used for the isolation of bacterial
RNA. Modeling analysis using the on line mfold tool (http://unafold.rna.albany.edu/?q=mfold/RNA-Folding-Form)
was done to determine the secondary structure of db34a (22).
Isolation of small RNAs
Purification of small RNA was performed using a
modified RNASwift protocol (23) and
compared with purification performed using RNAzol reagent
(Sigma-Aldrich; Merck KGaA). In the modified RNASwift method,
bacteria expressing the db34a or dbSCR plasmid were grown in
Luria-Bertani (LB) medium (BD Biosciences) with 50 µg/ml of
kanamycin (MilliporeSigma) for 24 h at 37°C. The cultured cells
(1.5×1010 cells) were spun down (3,000 × g for 20 min at
26°C), suspended in 10 ml of LB1 lysis reagent (4% sodium dodecyl
sulfate, pH 7.5, 0.5 M NaCl), and lysed by incubation at 90°C for 4
min. Then, 5 ml of 5 M NaCl was added to the homogenate and the
mixture was centrifuged at 16,000 × g for 25 min at 4°C. The
supernatant was transferred to a new tube, and 6 ml of isopropanol
was added and the mixture was centrifuged at 16,000 × g for 25 min
at 4°C to precipitate out the large RNA. To the remaining
supernatant, 10.5 ml of isopropanol was added and the mixture was
centrifuged 20,000 × g for 25 min at 4°C to precipitate the small
RNAs, which included the dbRNA product. The small RNA was washed
twice with ice-cold 75% ethanol and then dissolved in sterile
water. The purity of the small RNA was verified in a 1% agarose
gel. The quality of RNA was determined using a NanoDrop™
spectrophotometer.
Purification of dbRNAs
The isolated small RNAs were separated on a 15%
urea-polyacrylamide gel (PAGE). The gel was stained with ethidium
bromide (0.5 mg/ml) for 15 min at room temperature and then
destained by washing with distilled water twice. The gel portion
containing the dbRNA was excised with a sharp scalpel and the RNA
was purified using the ZR Small-RNA PAGE Recovery Kit (Zymo
Research Corp.) according to the manufacturer's instructions. RNA
quality and quantity were measured using a NanoDrop
spectrophotometer.
Verification of dbRNA specificity
The isolated dbRNAs were separated on a 15% urea-PAG
(TBE is the running buffer) at 20 mA followed by blotting using the
iBlot™ Gel Transfer Device onto a Novex™ iBlot™ DNA Transfer Stack
with a nylon membrane (both Invitrogen; Thermo Fisher Scientific,
Inc.). Northern blot analysis was conducted using a miR-34a
specific biotinylated probe (sequence:
/5Biosg/AGGGCAGTATACTTGCTGAT) after membrane was incubated with
prehybridization buffer (6X SSC buffer, 10X Denhardt's solution,
0.2% SDS; MilliporeSigma) for 1 h at 42°C on rotating shaker
following 3 day incubation with hybridization buffer (6X SSC
buffer, 5X Denhardt's solution, 0.2% SDS; MilliporeSigma) with
probe at the same conditions. The chemiluminescent signal was
detected using a Pierce™ Chemiluminescent Nucleic Acid Detection
Module kit (Thermo Fisher Scientific, Inc.) according to the
manufacturer's recommendations.
Determining the RNase A resistance of
db34a RNA
To determine the resistance of db34a to RNase A, 1
µg total RNA from bacteria transformed with db34a-expressing
plasmid was isolated and incubated with or without
0.7×10−5 U/µl RNase-A for 24 h in four independent
treatments at room temperature. Since the bacterial lipoprotein
(lpp) promoter (18,24) is a constitutive bacterial promoter
that drives the expression of bacterial lpp, bacterial lpp RNA was
used as the linear control for db34a. First, cDNA was synthesized
using miScript II RT kit (Qiagen GmbH) using conditions: 37°C for
60 min and 95°C for 5 min from untreated and 24 h RNase A treated
samples followed by quantitative PCR (qPCR) with the following
primers: lpp1 forward, CTGTCTTCTGACGTTCAGACTC and lpp reverse,
ACGAGCTGCGTCATCTTTAG. Expression of db34a was measured by qPCR with
miR-34a specific primers [5′UGGCAGUGUCUUAGCUGGUUGU, cat. no.
218300; Hs_miR-34a_1 miScript Primer Assay (forward primer) and
miScript Universal Primer (reverse)] using the miScript SYBR Green
PCR Kit (Qiagen GmbH). The qPCR conditions used were as follows:
Initial heat activation 95°C for 15 min, followed by denaturation
at 94°C for 15 sec, annealing at 55°C for 30 sec and extension at
70°C for 30 sec, for 40 cycles. Four independent experiments with
three replicates in each experiment were performed. ΔCq was
calculating by subtracting the average Cq values of lpp from db34a
for both untreated (control) and RNase A treated cells. ΔΔCq values
were obtained by subtracting the average ΔCq of the control group
from both the control and treatment ΔCq data followed by the
calculation of 2−ΔΔCq (25). The difference between treated and
control groups with reference to db34a/lpp RNA was analyzed using
one-way ANOVA.
Cell culture conditions
The MIA PaCa-2 and PANC-1 pancreatic cancer cell
lines were obtained from American Type Culture Collection (ATCC)
and maintained under the conditions recommended by the supplier.
Cells were grown in tissue culture-treated Petri plates in
RPMI-1640 medium (Thermo Fisher Scientific, Inc.) with 10% FBS (VWR
Corporation) and 1% penicillin-streptomycin (Thermo Fisher
Scientific Inc.) in a humidified 5% CO2 atmosphere at
37°C. To evaluate immune activation, J774A.1 mouse macrophages
obtained from ATCC were used. The macrophages were grown in tissue
culture-treated Petri plates in DMEM (Thermo Fisher Scientific
Inc.) with 10% FBS and 1% penicillin-streptomycin. Human umbilical
vein endothelial cells (HUVECs) were obtained from Thermo Fisher
Scientific, Inc. and cultured in Medium 200 with 1X low serum
growth supplement (Thermo Fisher Scientific Inc.). HUVECs with a
low passage number (3 passages) were used for the tube formation
assay as described in a previous study (26).
Angiogenesis antibody array
An angiogenic antibody array analysis was performed
using a RayBio® Human Angiogenesis Array C1 kit
(AAH-ANG-1; RayBiotech, Inc.) according to the manufacturer's
instructions. In brief, MIA PaCa-2 and PANC-1 cells were seeded
(1.2×106/well, 6-well plate) in tissue culture-treated
Petri plates overnight in RPMI-1640 medium (Thermo Fisher
Scientific Inc.) with 10% FBS (VWR Corporation) and 1%
penicillin-streptomycin (Thermo Fisher Scientific Inc.) in a
humidified 5% CO2 atmosphere at 37°C. The cells were
transfected with 4 µg of db34a or dbSCR using jetPRIME®
transfection reagent (Polyplus-transfection SA at 37°C) or remained
untransfected (control). After a 24-h incubation period, the medium
was replaced with serum-free RPMI-1640 medium (Thermo Fisher
Scientific Inc.) and the plates were incubated for another 24 h.
Conditioned medium was then collected, and the antibody-spotted
membranes from the array kit were incubated with the conditioned
medium according to the manufacturer's protocol. Following
incubation, the membranes were washed, 1 ml diluted
biotin-conjugated antibody mix was added and the membranes were
incubated at room temperature for 2 h, followed by washing and the
addition of diluted HRP-conjugated streptavidin at room temperature
for 2 h. Detection was performed using ECL protocols in which the
membranes were exposed to X-ray films to visualize the binding of
angiogenic factors. The intensities of the spots were quantified
using ImageJ version 1.52a software (National Institutes of
Health), and angiogenic molecules showing a significant difference
in expression levels between the db34a and dbSCR conditioned media
were identified. Significant differences between dbSCR and db34a
conditioned media were detected using one-way ANOVA.
Inflammation array
A mouse inflammation array analysis was performed
using a RayBio Mouse Inflammation Array C1 kit (cat. no. AAM-INF-1;
RayBiotech, Inc.) according to the manufacturer's instructions. In
brief, MIA PaCa-2 and PANC-1 cells were seeded
(1.2×106/well; 6-well plate) and cultured overnight in
RPMI-1640 medium (Thermo Fisher Scientific Inc.) supplemented with
10% FBS (VWR Corporation) and 1% penicillin-streptomycin (Thermo
Fisher Scientific Inc.) in a humidified 5% CO2
atmosphere at 37°C. The next day, cells were transfected with 4 µg
of db34a or dbSCR at 37°C using jetPRIME transfection reagent or
remained untransfected (control). After 20 h, the medium was
replaced with serum-free medium RPMI-1640 (Thermo Fisher Scientific
Inc.) and the plates were incubated for another 16 h. Conditioned
medium was then collected after centrifugation at 16,000 × g at 4°C
for 10 min and transferred to freshly grown macrophages. After
overnight incubation at 37°C, the macrophages were lysed with lysis
buffer provided with kit and equal samples from each condition with
regard to total protein quantity (100 µg per sample; total protein
concentration was determined by Pierce 660 nm Protein Assay (Thermo
Fisher Scientific Inc.) were applied to the antibody-spotted
membranes. Following overnight incubation at 4°C, the membranes
were washed and 1 ml diluted biotin-conjugated antibody mix was
added to the membranes, which were kept at room temperature for 2
h. After several washes, the membranes were incubated with diluted
HRP-conjugated streptavidin for 2 h. Detection was performed using
ECL protocols in which the membranes were exposed to X-ray films to
detect the binding of inflammatory factors. Spot intensities were
quantified using ImageJ version 1.52a software. Significant
differences between macrophages grown in dbSCR and db34a
conditioned media were identified using one-way ANOVA.
In vitro angiogenic assay
To determine the anti-angiogenic effect of the dbRNA
molecules, an endothelial cell tube formation assay was conducted
using HUVECs as previously described (26). Briefly, HUVECs were stained with 2
µg/ml cell-permeant fluorescent dye (calcein-AM; Thermo Fisher
Scientific, Inc.) with incubation at 37°C in the dark for ≥30 min.
A Geltrex basement membrane matrix (Thermo Fisher Scientific, Inc;
40 µl) was applied to 96-well plates and allowed to solidify for 30
min at room temperature. The stained HUVECs were trypsinized,
re-suspended in serum-free medium (Gibco Medium 200PR; Gibco;
Thermo Fisher Scientific Inc.) and plated at a density of 20,000
cells/well in 100 µl conditioned medium from the db34a and dbSCR
transfected cells. HUVECs in complete medium (Gibco Medium 200PRF
supplemented with Gibco Low Serum Growth Supplement; Gibco; Thermo
Fisher Scientific Inc.) were used as controls. The plates were
incubated at 37°C in a humidified incubator for 3 h. The formation
of angiogenic network was visualized at ×10 magnification using an
inverted fluorescence microscope and quantified by ImageJ version
1.52a (n=17). The significance of difference of each HUVEC
treatment group compared to control was determined using one-way
ANOVA.
Zebrafish aquaculture
Transgenic VEGFR2:green-reef coral fluorescent
protein (G-RCFP) zebrafish (Danio rerio) that express G-RCFP
under the control of a VEGFR2/KDR promoter were obtained from ZFIN
(y1Tg). The fish were maintained under standard aquaculture
conditions in a circulating tank system constructed as previously
described (27), and according to
protocols approved by the Institutional Animal and Use Committee
(IACUC) of University of Illinois College of Medicine at Peoria.
Water was purified by a reverse osmosis system and supplemented
with sea salts at concentration of 60 mg/l in order to provide the
trace minerals that the fish required. The pH of the water was
maintained between 7.6 and 8.5 by the addition of sodium
bicarbonate. All experimental procedures using zebrafish were
performed with approval from the IACUC of University of Illinois
College of Medicine at Peoria.
In vivo zebrafish angiogenic
assay
Adult male and female zebrafish were placed in
spawning baskets. Newly fertilized eggs were collected soon after
fertilization and rinsed at 28°C in embryo water (Milli-Q water
with 60 mg/l Instant Ocean- Spectrum Brands) and immediately
followed by incubation in 1% methylene blue in embryo water at 28°C
for 24 h. After which the embryos were dechorionated and allowed to
develop in embryo water alone for 24 h at 28°C (48 h post
fertilization). Following this the embryos were incubated in embryo
water alone or embryo water supplemented with 10 pmol db34a or
dbSCR RNA, 5 µM sunitinib or a 1:1,000 dilution of DMSO as vehicle
control at 28°C. After 24 h treatment, the 72 h old embryos were
fixed in 10% buffered formaldehyde at room temperature for 10 min.
The fixed embryos were mounted on glass slides and the completeness
of sub-intestinal vein (SIV) vasculature was observed by the
detection of G-RCFP using fluorescent inverted microscopy. The
significance of differences of each treatment group compared to
control was determined using one-way ANOVA.
Statistical analysis
P<0.05 was considered to indicate a statistically
significant difference. The difference between treated and control
groups with reference to db34a/lpp RNA was analyzed using one-way
ANOVA. For the angiogenesis array, significant differences between
dbSCR and db34a conditioned media were analyzed using one-way
ANOVA. For the macrophage assay, significant differences between
macrophages grown in dbSCR and db34a conditioned media were
analyzed using one-way ANOVA. Data are presented as a mean ±
standard error and calculations done with OriginPro version 8.6
software (OriginLab Corporation)
Results
db34a RNA harboring miR-34a-3p and
miR-34a-5p shows increased stability compared with linear RNA
A circular RNA-expressing plasmid was developed to
express miR-34a. This construct codes for miR-34a and a
streptavidin-binding aptamer (Fig.
1A-C). The JM101Tr E. coli strain was used for the
isolation of bacterial RNA. Upon circularization of the RNA,
minimal degradation was observed, even after exposure to RNAse-A
for 24 h (Fig. 1D). Stability was
quantified using the 2−ΔΔCq method. A statistically
significant difference between the control and RNase A-treated
groups was detected (P=1.41627×10−4) with reference to
linear lpp RNA vs. db34a RNA. The isolated db34a and dbSCR were
characterized using urea-PAG electrophoresis. Using the modified
RNASwift protocol, isolation of the circular RNAs was successfully
achieved, as indicated by the low-molecular-weight bands in the
urea gel (Fig. 1E). To confirm that
the circular forms that were isolated were indeed db34a, northern
blotting was performed using a biotinylated probe specific for
miR-34a. The northern blotting results show the presence of a band
for each db34a group, confirming that the methodology was specific
(Fig. 1F).
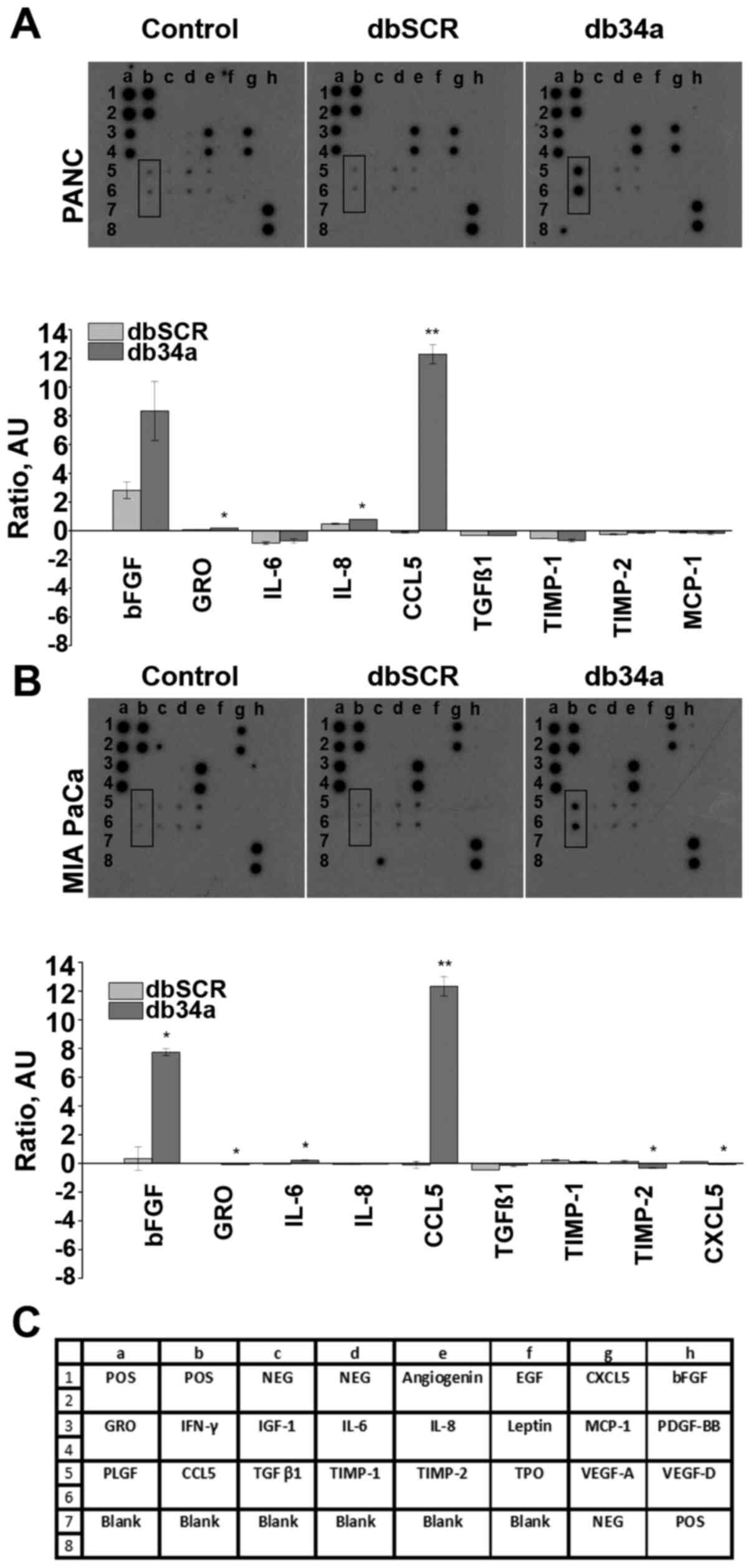
db34a RNA harboring miR-34a-3p and
miR-34a-5p induces the expression of C-C motif chemokine ligand 5
(CCL5) in PANC-1 and MIA PaCa-2 pancreatic cancer cells
To evaluate the expression of pro-angiogenic
molecules, the RayBio Human Angiogenesis Array C1 kit was used.
Conditioned media collected from untransfected and db34a- or
dbSCR-transfected MIA PaCa-2 and PANC-1 cells were analyzed using
the array kit. From the array data, it was observed that CCL5 was
significantly upregulated in PANC-1 (P=0.0028) and MIA PaCa-2
(P=0.0034) cells transfected with db34a compared with the
respective dbSCR-transfected cells. Interestingly, basic fibroblast
growth factor (bFGF) was also upregulated in PANC-1 and MIA PaCa-2
cells transfected with db34a, however, the upregulation was
significant only in the MIA PaCa-2 cells (P=0.012; Fig. 2).
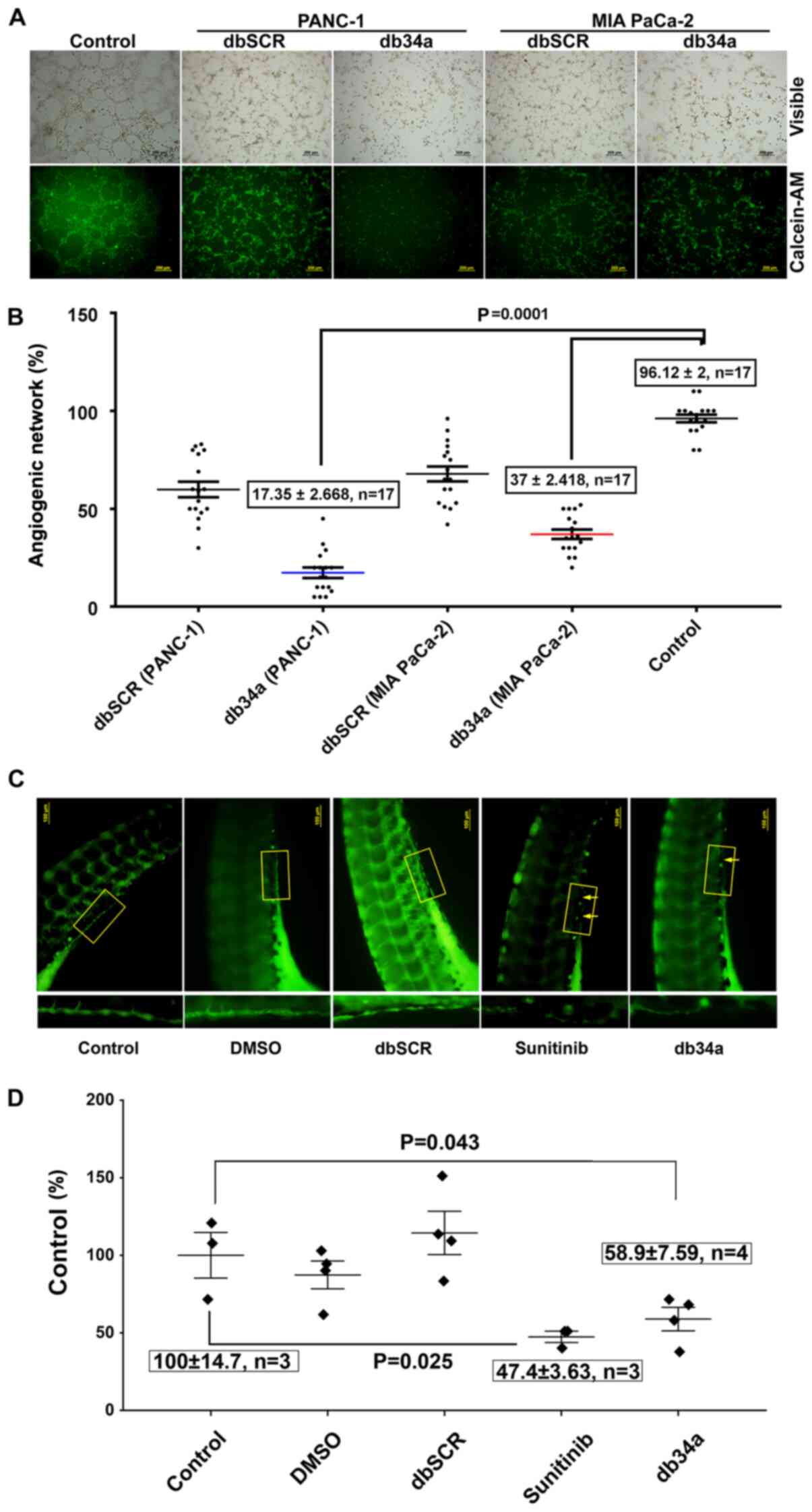
db34a RNA harboring miR-34a-3p and
miR-34a-5p suppresses angiogenic induction in HUVECs
An angiogenic tube formation assay was performed
using HUVECs according to a previously described protocol (26). The assay was used to determine
whether db34a has the ability to alter the angiogenic potential of
PANC-1 and MIA PaCa-2 cells. In this in vitro angiogenic
assay, it was observed that HUVECs treated with conditioned media
from db34a-transfected PANC-1 and MIA PaCa-2 cells exhibited
suppressed angiogenic potential when compared with control (PANC-1,
17.35±2.66%; MIA PaCa-2, 37±2.41%; P=0.0001; Fig. 3A and B).
db34a RNA harboring miR-34a-3p and
miR-34a-5p suppresses angiogenic induction in zebrafish
embryos
Zebrafish strain y1tg that expresses a green
fluorescent protein in the vasculature was used as a model
organism. SIV vasculature was observed 24 h after treatment with 10
pmol db34a or dbSCR RNA, 5 µM sunitinib or a 1:1,000 dilution of
DMSO, and images were captured using a fluorescent microscope
(Fig. 3C). It was observed that the
embryos treated with db34a showed reduced SIV integrity compared
with the controls (58.9±7.59% of the untreated control; P=0.043),
which confirmed the functionality of db34a and indicated the
potential of dbRNA as a therapeutic agent. Positive control embryos
treated with sunitinib also exhibited decreased SIV vasculature
(47.4±3.63% of the untreated control; P=0.025).
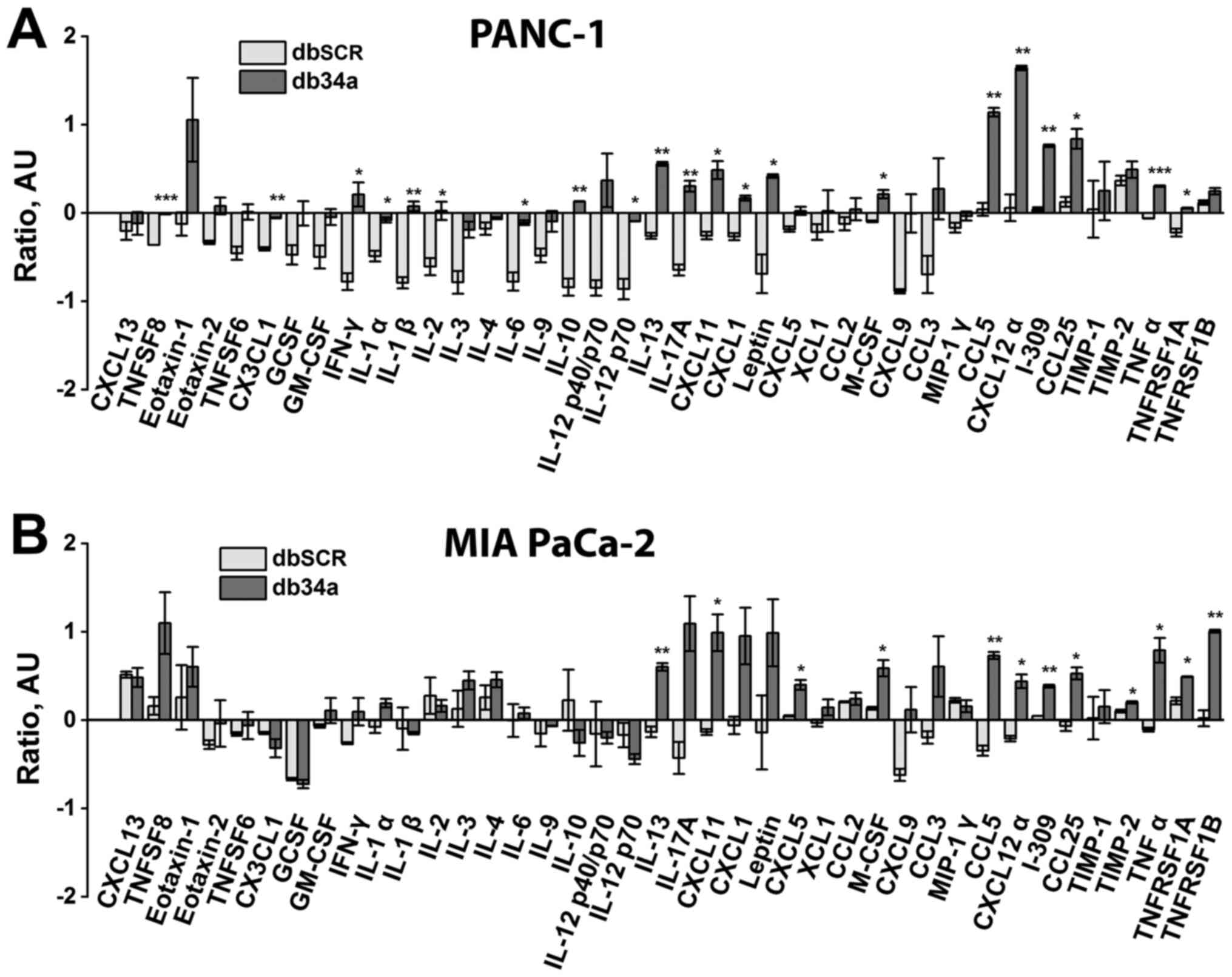
db34a RNA harboring miR-34a-3p and
miR-34a-5p induces the PANC-1 and MIA PaCa-2 pancreatic cancer
cell-mediated activation of macrophage inflammatory markers
To determine whether db34a induces immune
activation, conditioned media from PANC-1 and MIA PaCa-2 cells
transfected with db34a were collected and the ability of the
conditioned media to activate an inflammatory response in a mouse
macrophage cell line was determined. It was observed that
conditioned media from PANC-1 and MIA PaCa-2 cells transfected with
db34a significantly induced the expression of interleukin (IL)-13,
CCL5 and C-X-C motif chemokine ligand 12 α (CXCL12α) among other
inflammatory response genes (Table I
and Fig. 4).
 | Table I.Cytokines significantly different
between macrophages treated with conditioned media from PANC-1 and
MIA PaCa-2 cell lines transfected with db34a compared with
dbSCR. |
Table I.
Cytokines significantly different
between macrophages treated with conditioned media from PANC-1 and
MIA PaCa-2 cell lines transfected with db34a compared with
dbSCR.
| Cytokine | PANC-1 (fold change
vs. control) | P-value | MIA PaCa-2 (fold
changes vs. control) | P-value |
|---|
| CD30 ligand
(TNFSF8) | 1.543449 |
4.61297×10−5 | 1.808521 | – |
| Fractalkine
(CX3CL1) | 1.591752 | 0.00309 | 0.798687 | – |
| IFN-γ | 5.434441 | 0.02768 | 1.484371 | – |
| IL-1α (IL-1
F1) | 1.793997 | 0.02553 | 1.287388 | – |
| IL-1β (IL-1
F2) | 5.106111 | 0.0092 | 0.943795 | – |
| IL-2 | 2.601422 | 0.04669 | 0.909556 | – |
| IL-6 | 3.973113 | 0.02473 | 1.081643 | – |
| IL-10 | 7.051837 | 0.00978 | 0.605812 | – |
| IL-12 p70 | 6.513021 | 0.02201 | 0.673057 | – |
| IL-13 | 2.100802 | 0.00251 | 1.849357 | 0.00994 |
| IL-17A | 3.668945 | 0.00847 | 3.655409 | 0.03355 |
| I-TAC (CXCL11) | 1.996696 | 0.02114 | 2.297556 | – |
| KC (CXCL1) | 1.596054 | 0.0123 | 2.07834 | – |
| Leptin | 4.536913 | 0.0375 | 2.313414 | – |
| LIX (CXCL5) | 1.252327 | – | 1.334515 | 0.02446 |
| M-CSF | 1.339565 | 0.02349 | 1.40063 | 0.03974 |
| RANTES (CCL5) | 2.056593 | 0.00633 | 2.646111 | 0.00422 |
| SDF-1α
(CXCL12α) | 2.493808 | 0.00911 | 1.820613 | 0.01686 |
| I-309
(TCA-3/CCL1) | 1.689148 | 0.00128 | 1.319467 | 0.00263 |
| TECK (CCL25) | 1.633533 | 0.02981 | 1.637408 | 0.02115 |
| TIMP-2 | 1.091021 | – | 1.089587 | 0.04152 |
| TNF-α | 1.387676 |
4.64829×10−4 | 2.003669 | 0.02406 |
| TNF RI
(TNFRSF1A) | 1.355374 | 0.02447 | 1.223076 | 0.02037 |
| TNF RII
(TNFRSF1B) | 1.114493 | – | 1.962633 | 0.00873 |
Discussion
Despite decades of research, the treatment of
pancreatic ductal adenocarcinoma continues to be a major challenge.
According to the American Cancer Society, for all stages of
pancreatic cancer combined, the 1-year survival rate is ~20% and
the 5-year survival rate is 9% (28). One of the major factors that
contributes to this poor prognosis is late detection, as
early-stage pancreatic cancer is usually asymptomatic (29). New surgical techniques and evolving
therapeutics have achieved only modest outcomes (30). A clearer understanding of targetable
molecular pathways is required to design therapeutic agents and
achieve a desired clinical outcome. Non-coding RNAs such as miRNAs
play important roles in the regulation of gene expression. Unlike
siRNAs, miRNAs target multiple genes and are usually process
specific (31). Most biological
processes are in some way regulated by miRNAs, which are small RNA
molecules ~22 nucleotides in length that probably function as
antisense regulators of other RNAs (32). The mechanisms of miRNA action have
been elucidated, although not completely (33). Their capacity to regulate mRNAs and
other miRNAs is being reported with increasing frequency, and they
have become powerful tools for gene regulation. The use of RNA
molecules to target angiogenesis continues to be explored (11–16).
However, the approach described in the present study, utilizing a
circular RNA molecule with two miRs is novel and, to the best of
our knowledge, has not been described previously. The approach was
to develop a closed circular RNA coding for miRNAs. A circular RNA
is not readily degraded by exonucleases and the inherent higher
stability of dsRNAs compared with ssRNAs may contribute to an
increased half-life (34). Increased
stability of db34a compared with linear RNA was observed in the
present study. To the best of our knowledge, the present study is
the first to use closed circular dbRNAs to code for both miR34-3p
and −5p simultaneously with a therapeutic intention.
The present study aimed to demonstrate that circular
RNA harboring miRs are functional and have significant therapeutic
potential using dbRNA encoding miR-34a-3p and −5p. To construct the
circular RNA expressing miR-34a-3p and −5p, the PIE method was
used, which is an enzyme-free RNA circularization method based on
group I intron self-splicing (17),
which can be conducted using essentially any type of cell. Cells
transcribe more RNA than they accumulate (35). This indicates that RNA is constantly
being degraded and that a dynamic process occurs to balance RNA
synthesis and degradation. The use of siRNA therapeutically is not
new but has the limitations of low half-life and questionable in
vivo efficacy (36,37), which is mainly due to the effects of
exonuclease RNases. The present study circumvents the problem of a
low half-life by designing dbRNAs that should not be degraded by
the exonuclease activity of RNases. A previous study demonstrated
the stability of a 35mer FITC-labeled RNA in the presence of
circular dbRNA/DNA chimeric oligonucleotides to RNase H. This
induced stability is most likely due to the competition of RNA/DNA
chimeric with the 35mer FITC-labeled RNA (38). The stability experiments conducted in
the present study demonstrate that circular RNA exposed to RNase A
for 24 h was more stable than linear RNA driven by the same lpp
promoter (18,24). Gene silencing by siRNA/miRNAs can be
used as a therapeutic approach for the treatment of un-druggable
targets. Researchers have also shown that dumbbell-shaped DNA
minimal vectors can be used for small hairpin RNA expression
(39,40). These studies also demonstrated the
use of a dumbbell DNA vector to produce hairpin RNAs that can be
used therapeutically. Another study described the generation of
dumbbell-shaped nano-circular RNAs for RNA interference, using a
chemical method of synthesis involving the enzyme-based
circularization of small RNA molecules (41). By contrast, the present study reveals
the development of a larger dbRNA and is unique in that it uses a
bacterial system for large-scale dbRNA production.
Although not much is known about miR-34a-3p,
miR-34a-5p is well studied and has been shown to be involved in
tumor suppression, angiogenic suppression and cancer stem cell
suppression (42–49); hsa-miR-34a-3p was previously known as
hsa-miR-34a* and hsa-miR-34a-5p was previously known hsa-miR-34a.
It was recently demonstrated that normal cells do show detectable
expression levels of miR34a (50).
Normal cell lines were not used in the present study and this is a
limitation of the study. Since normal cells do show detectable
levels of miR34a, the addition of db34a to normal cells should not,
in theory, induce a notable perturbance in the existing molecular
pathways of normal cells; this however requires further study. To
demonstrate the possibility of using circular RNA therapeutically,
in the present study circular miR-34a-3p and −5p were transfected
into pancreatic cancer cells and the expression of pro-angiogenic
molecules was determined. It was observed that in the two
pancreatic cancer cell lines transfected with db34a, the expression
levels of bFGF and CCL5 were significantly increased. miR-34a-5p
expression is known to be increased by the activation of the PI3K
signaling pathway (51) and,
notably, mir-34a-5p has been shown to suppress the PI3K signaling
pathway (52–56). These contradictory observations
indicates the possibility of the existence of a tight regulatory
apparatus that is not yet clearly understood. Interestingly, TNF is
predicted as the target of hsa-miR-34a-3p by the miRDB online
database, and TNF is a strong promoter of angiogenesis (57). This indicates that hsa-miR-34a-3p may
be more therapeutically relevant in angiogenic suppression. It
should be noted that both miR-34a-3p and miR-34a-5p are processed
from the same pre-miR. This is consistent with the observation that
angiogenesis was suppressed significantly in the in vitro
and in vivo angiogenic assays of the present study. This
observation appears contrary to the expected outcome; however,
miR-34a-5p is a known angiogenic suppressor (43) and the quantification of other
relevant angiogenesis-associated molecules will provide a clearer
mechanistic profile of this angiogenesis modulation. Every
biological process in an oncogenic system is tightly regulated;
therefore, targeting a process rather than a single molecule would
not be clinically relevant as compensatory pathways can be
activated to achieve the oncogenic phenotype. The overexpression of
bFGF may be one such compensatory response, which is seen in both
pancreatic cell lines.
The present study also demonstrated that CCL5 is
significantly upregulated in PANC-1 and MIA PaCa-2 cells
transfected with db34a. CCL5, also known as regulated on
activation, normal T cell expressed and secreted (RANTES), is a
chemokine that is involved in T-cell activation. RANTES has been
identified as a major HIV-suppressive factor. Studies have shown
that recombinant human RANTES induces a dose-dependent inhibition
of different strains of HIV-1, HIV-2 and simian immunodeficiency
virus (SIV) (58,59). The expression of CCL5 in MIA PaCa-2
and PANC-1 cells transfected with db34a RNA indicates the
involvement of a conserved pathway. Additional research is
necessary to determine whether transfection with db34a can also
induce the overexpression of CCL5 in other types of cells. In a
recent study, it was shown that CCL5 and CCR5 expression levels are
increased in pancreatic cancer tissue sections, and the
overexpression of CCL5 and CCR5 increases the invasiveness and
metastatic potential of pancreatic cancer cells (60). It has also been reported that
progranulin (PGRN) expression levels correlate with a poor
prognosis for melanoma patients, and PGRN inhibits CCL5 gene
expression at the transcriptional level, showing that CCL5 is
responsible for the recruitment of activated natural killer cells
to the tumor microenvironment (61).
Another study highlighted the importance of CCL5, demonstrating
that the blockade of 1,4-α-glucan-branching enzyme in lung cancer
cells promoted the secretion of CCL5, which induced the recruitment
of CD8+ T lymphocytes to the tumor microenvironment
(62). CCL5 expression has also been
shown to restore the immune surveillance in antigen-expressing
MYC;CTNNB1 hepatocellular carcinoma tumors (63). This indicates that CCL5 is necessary
for tumor cells to be visible to the immune system; especially,
CCL5 expression is necessary for immune recognition. However, it is
also important to note that CCL5 was recently identified as a
signature of poor prognosis for oral squamous cell carcinoma,
indicating its multifaceted role (64). Interestingly, another study observed
that the expression of CCL5 in breast cancer cells was associated
with lymph node status and tumor node-metastasis stage (65). The conflicting roles of CCL5, and
whether its overexpression is a predisposing factor for poor
prognosis or a response to poor prognosis remain unclear. The
association of CCL5 with other immune-associated molecules may
provide clearer insight, and CCL5 expression alone should not be
used as a measure of prognosis.
Furthermore, the present study investigated whether
the transfection of pancreatic cancer cells with db34a induces the
activation of immune cells. It was observed that the conditioned
medium from db34a-transfected pancreatic cancer cells significantly
induced the expression of IL-13, CCL5 and CXCL12α among other
inflammatory response genes in mouse macrophage cell lysates.
Tumor-associated macrophages (TAMs) are present in high numbers in
the microenvironment of solid tumors and have been studied for
their potential as targets for cancer therapy (66). TAMs suppress host immune responses
and also promote tumor cell proliferation, angiogenesis, invasion
and metastasis (67). The role of
TAMs remains unclear; however, most evidence suggests that TAMs
promote tumor progression. From the data in the present study,
especially the overexpression of IL-13, it appears that the J774A.1
macrophages exposed to conditioned medium from db34a-transfected
pancreatic cancer cells appeared more closely associated with the
M1 phenotype than the M2 phenotype (data not shown) (68). Further investigation is required to
decipher the influence of db34a on macrophages.
In conclusion, the present data suggest that the
supplementation of miR-34a-3p and −5p as a circular pre-miR form is
a viable method of miR replacement therapy.
Acknowledgements
Assistance for the maintenance of Zebrafish culture
at the Laboratory Animal Care Facility, UICOMP, LACF-Peoria IL, by
Laboratory Animal Care Supervisor Ms. Angela Daniels and,
Laboratory Animal Care Technician Ms. Stephanie Sampson is
acknowledged.
Funding
The Department of Internal Medicine, University of
Illinois College of Medicine at Peoria; McElroy Foundation
(Springfield, IL, USA) (grant no, 096726) and gift from The Theresa
Tracy Strive to Survive Foundation (East Peoria, IL, USA) supported
the present study.
Availability of data and materials
The plasmid constructs db34a and dbSCR are available
from the corresponding author upon reasonable request in accordance
with institutional MTA approvals and all applicable rules and
regulations. The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
MG designed and performed experiments and reviewed
the manuscript. LD validated the constructs, designed the RNA
stability experiments, confirmed dbRNA stability and quantified
data. LG and PS were involved in designing the concept of the
study. SB was involved in RNA experiments and data collection. JAN
was involved in zebrafish data collection. CSG designed, and
developed the concept and experiments, and wrote the manuscript.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
All experimental procedures using zebrafish were
performed with approval from the Institutional Animal and Use
Committee of University of Illinois College of Medicine at
Peoria.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Boehm T, Folkman J, Browder T and O'Reilly
MS: Antiangiogenic therapy of experimental cancer does not induce
acquired drug resistance. Nature. 390:404–407. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zetter BR: Angiogenesis and tumor
metastasis. Annu Rev Med. 49:407–424. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Folkman J: Antiangiogenic gene therapy.
Proc Natl Acad Sci USA. 95:9064–9066. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Donmez G, Sullu Y, Baris S, Yildiz L,
Aydin O, Karagoz F and Kandemir B: Vascular endothelial growth
factor (VEGF), matrix metalloproteinase-9 (MMP-9), and
thrombospondin-1 (TSP-1) expression in urothelial carcinomas.
Pathol Res Pract. 205:854–7. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Neves KB, Montezano AC, Lang NN and Touyz
RM: Vascular toxicity associated with anti-angiogenic drugs. Clin
Sci (Lond). 134:2503–20. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mattison R, Jumonville A, Flynn PJ,
Moreno-Aspitia A, Erlichman C, LaPlant B and Juckett MB: A phase II
study of AZD2171 (cediranib) in the treatment of patients with
acute myeloid leukemia or high-risk myelodysplastic syndrome. Leuk
Lymphoma. 56:2061–2066. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hong DS, Garrido-Laguna I, Ekmekcioglu S,
Falchook GS, Naing A, Wheler JJ, Fu S, Moulder SL, Piha-Paul S,
Tsimberidou AM, et al: Dual inhibition of the vascular endothelial
growth factor pathway: A phase 1 trial evaluating bevacizumab and
AZD2171 (cediranib) in patients with advanced solid tumors. Cancer.
120:2164–2173. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liu JF, Tolaney SM, Birrer M, Fleming GF,
Buss MK, Dahlberg SE, Lee H, Whalen C, Tyburski K, Winer E, et al:
A phase 1 trial of the poly(ADP-ribose) polymerase inhibitor
olaparib (AZD2281) in combination with the anti-angiogenic
cediranib (AZD2171) in recurrent epithelial ovarian or
triple-negative breast cancer. Eur J Cancer. 49:2972–2978. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Climent-Salarich M, Quintavalle M,
Miragoli M, Chen J, Condorelli G and Elia L: TGFβ triggers
miR-143/145 transfer from smooth muscle cells to endothelial cells,
thereby modulating vessel stabilization. Circ Res. 116:1753–1764.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang G and Chen L: Tumor vasculature and
microenvironment normalization: A possible mechanism of
antiangiogenesis therapy. Cancer Biother Radiopharm. 23:661–667.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Barbas AS and White RR: The development
and testing of aptamers for cancer. Curr Opin Investig Drugs.
10:572–578. 2009.PubMed/NCBI
|
|
12
|
Guglielmelli T, Bringhen S and Palumbo A:
Update on the use of defibrotide. Expert Opin Biol Ther.
12:353–361. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gatto B and Cavalli M: From proteins to
nucleic acid-based drugs: The role of biotech in anti-VEGF therapy.
Anticancer Agents Med Chem. 6:287–301. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kanwar JR, Mahidhara G and Kanwar RK:
Antiangiogenic therapy using nanotechnological-based delivery
system. Drug Discov Today. 16:188–202. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Friedman AD, Kim D and Liu R: Highly
stable aptamers selected from a 20-fully modified fGmH RNA library
for targeting biomaterials. Biomaterials. 36:110–23. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Inoue M, Kadonosono K, Arakawa A, Yamane S
and Ishibashi T: Long-term outcome of intravitreal pegaptanib
sodium as maintenance therapy in japanese patients with neovascular
age-related macular degeneration. Jpn J Ophthalmol. 59:173–178.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Puttaraju M and Been MD: Circular
ribozymes generated in Escherichia coli using group I
self-splicing permuted intron-exon sequences. J Biol Chem.
271:26081–26087. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Umekage S and Kikuchi Y: In vivo circular
RNA production using a constitutive promoter for high-level
expression. J Biosci Bioeng. 108:354–356. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ford E and Ares M Jr: Synthesis of
circular RNA in bacteria and yeast using RNA cyclase ribozymes
derived from a group I intron of phage T4. Proc Natl Acad Sci USA.
91:3117–3121. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ponchon L and Dardel F: Recombinant RNA
technology: The tRNA scaffold. Nat Methods. 4:571–576. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Umekage S and Kikuchi Y: In vitro and in
vivo production and purification of circular RNA aptamer. J
Biotechnol. 139:265–272. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zuker M: Mfold web server for nucleic acid
folding and hybridization prediction. Nucleic Acids Res.
31:3406–3415. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nwokeoji AO, Kilby PM, Portwood DE and
Dickman MJ: RNASwift: A rapid, versatile RNA extraction method free
from phenol and chloroform. Anal Biochem. 512:36–46. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen QX, Wang WP, Zeng S, Urayama S and Yu
AM: A general approach to high-yield biosynthesis of chimeric RNAs
bearing various types of functional small RNAs for broad
applications. Nucleic Acids Res. 43:3857–3869. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-delta delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gnanamony M, Antony R, Fernandez KS, Jaime
L, Lin J, Joseph PA and Gondi CS: Chronic radiation exposure of
neuroblastoma cells reduces nMYC copy number. Oncol Lett.
14:3363–3370. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kim S, Carlson R, Zafreen L, Rajpurohit SK
and Jagadeeswaran P: Modular, easy-to-assemble, low-cost zebrafish
facility. Zebrafish. 6:269–274. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jelski W and Mroczko B: Biochemical
diagnostics of pancreatic cancer-present and future. Clin Chim
Acta. 498:47–51. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
McGuigan A, Kelly P, Turkington RC, Jones
C, Coleman HG and McCain RS: Pancreatic cancer: A review of
clinical diagnosis, epidemiology, treatment and outcomes. World J
Gastroenterol. 24:4846–4861. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhou LY, Qin Z, Zhu YH, He ZY and Xu T:
Current RNA-based therapeutics in clinical trials. Curr Gene Ther.
19:172–196. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ambros V: microRNAs: Tiny regulators with
great potential. Cell. 107:823–826. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Toscano-Garibay JD and Aquino-Jarquin G:
Transcriptional regulation mechanism mediated by miRNA-DNA•DNA
triplex structure stabilized by Argonaute. Biochim Biophys Acta.
1839:1079–1083. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li J, Mohammed-Elsabagh M, Paczkowski F
and Li Y: Circular nucleic acids: Discovery, functions and
applications. Chembiochem. 21:1547–1566. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Houseley J and Tollervey D: The many
pathways of RNA degradation. Cell. 136:763–776. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ryther RC, Flynt AS, Phillips JA III and
Patton JG: siRNA therapeutics: Big potential from small RNAs. Gene
Ther. 12:5–11. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bakhtiyari S, Haghani K, Basati G and
Karimfar MH: siRNA therapeutics in the treatment of diseases. Ther
Deliv. 4:45–57. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Park WS, Miyano-Kurosaki N, Abe T, Takai K
and Takaku H: Properties of circular dumbbell RNA/DNA chimeric
oligonucleotides containing antisense phosphodiester
oligonucleotides. Nucleic Acids Symp Ser. 225–226. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yu H, Jiang X, Tan KT, Hang L and Patzel
V: Efficient production of superior dumbbell-shaped DNA minimal
vectors for small hairpin RNA expression. Nucleic Acids Res.
43:e1202015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jiang X, Yu H, Teo CR, Tan GS, Goh SC,
Patel P, Chua YK, Hameed NB, Bertoletti A and Patzel V: Advanced
design of dumbbell-shaped genetic minimal vectors improves
non-coding and coding RNA expression. Mol Ther. 24:1581–1591. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Abe N, Abe H, Nagai C, Harada M,
Hatakeyama H, Harashima H, Ohshiro T, Nishihara M, Furukawa K,
Maeda M, et al: Synthesis, structure, and biological activity of
dumbbell-shaped nanocircular RNAs for RNA interference. Bioconjug
Chem. 22:2082–2092. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Misso G, Di Martino MT, De Rosa G, Farooqi
AA, Lombardi A, Campani V, Zarone MR, Gullà A, Tagliaferri P,
Tassone P and Caraglia M: Mir-34: A new weapon against cancer? Mol
Ther Nucleic Acids. 3:e1942014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li XJ, Ren ZJ and Tang JH: MicroRNA-34a: A
potential therapeutic target in human cancer. Cell Death Dis.
5:e13272014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kofman AV, Kim J, Park SY, Dupart E,
Letson C, Bao Y, Ding K, Chen Q, Schiff D, Larner J and Abounader
R: microRNA-34a promotes DNA damage and mitotic catastrophe. Cell
Cycle. 12:3500–3511. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Kumar B, Yadav A, Lang J, Teknos TN and
Kumar P: Dysregulation of microRNA-34a expression in head and neck
squamous cell carcinoma promotes tumor growth and tumor
angiogenesis. PLoS One. 7:e376012012. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Tivnan A, Tracey L, Buckley PG, Alcock LC,
Davidoff AM and Stallings RL: MicroRNA-34a is a potent tumor
suppressor molecule in vivo in neuroblastoma. BMC Cancer.
11:332011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Gallardo E, Navarro A, Vinolas N, Marrades
RM, Diaz T, Gel B, Quera A, Bandres E, Garcia-Foncillas J, Ramirez
J and Monzo M: miR-34a as a prognostic marker of relapse in
surgically resected non-small-cell lung cancer. Carcinogenesis.
30:1903–1909. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wei JS, Song YK, Durinck S, Chen QR, Cheuk
AT, Tsang P, Zhang Q, Thiele CJ, Slack A, Shohet J and Khan J: The
MYCN oncogene is a direct target of miR-34a. Oncogene.
27:5204–5213. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Bommer GT, Gerin I, Feng Y, Kaczorowski
AJ, Kuick R, Love RE, Zhai Y, Giordano TJ, Qin ZS, Moore BB, et al:
p53-mediated activation of miRNA34 candidate tumor-suppressor
genes. Curr Biol. 17:1298–1307. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Bozgeyik E, Tepe NB and Bozdag Z:
Identification of microRNA expression signature for the diagnosis
and prognosis of cervical squamous cell carcinoma. Pathol Res
Pract. 216:1531592020. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Barnes PJ, Baker J and Donnelly LE:
Cellular senescence as a mechanism and target in chronic lung
diseases. Am J Respir Crit Care Med. 200:556–564. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhai L, Zhao Y, Liu Z, Wu J and Lin L:
mRNA expression profile analysis reveals a C-MYC/miR-34a pathway
involved in the apoptosis of diffuse large B-cell lymphoma cells
induced by yiqichutan treatment. Exp Ther Med. 20:2157–2165.
2020.PubMed/NCBI
|
|
53
|
Jiang C, Cheng Z, Jiang T, Xu Y and Wang
B: MicroRNA-34a inhibits cell invasion and epithelial-mesenchymal
transition via targeting AXL/PI3K/AKT/snail signaling in
nasopharyngeal carcinoma. Genes Genomics. 42:971–978. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Wang S, Tang Q, Ge F and Guo Q: Typhae
pollen polysaccharides protect hypoxia-induced PC12 cell injury via
regulation of miR-34a/SIRT1. Int J Immunopathol Pharmacol.
34:20587384209100052020. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhang YM, Wu QM, Chang LY and Liu JC:
miR-34a and miR-125a-5p inhibit proliferation and metastasis but
induce apoptosis in hepatocellular carcinoma cells via repressing
the MACC1-mediated PI3K/AKT/mTOR pathway. Neoplasma. 67:1042–1053.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Tao H, Cheng L and Yang R: Downregulation
of miR-34a promotes proliferation and inhibits apoptosis of rat
osteoarthritic cartilage cells by activating PI3K/akt pathway. Clin
Interv Aging. 15:373–385. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Mahdavi Sharif P, Jabbari P, Razi S,
Keshavarz-Fathi M and Rezaei N: Importance of TNF-alpha and its
alterations in the development of cancers. Cytokine.
130:1550662020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Scarlatti G, Tresoldi E, Bjorndal A,
Fredriksson R, Colognesi C, Deng HK, Malnati MS, Plebani A,
Siccardi AG, Littman DR, et al: In vivo evolution of HIV-1
co-receptor usage and sensitivity to chemokine-mediated
suppression. Nat Med. 3:1259–1265. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Cocchi F, DeVico AL, Garzino-Demo A, Arya
SK, Gallo RC and Lusso P: Identification of RANTES, MIP-1 alpha,
and MIP-1 beta as the major HIV-suppressive factors produced by
CD8+ T cells. Science. 270:1811–5. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Singh SK, Mishra MK, Eltoum IA, Bae S,
Lillard JW Jr and Singh R: CCR5/CCL5 axis interaction promotes
migratory and invasiveness of pancreatic cancer cells. Sci Rep.
8:13232018. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Voshtani R, Song M, Wang H, Li X, Zhang W,
Tavallaie MS, Yan W, Sun J, Wei F and Ma X: Progranulin promotes
melanoma progression by inhibiting natural killer cell recruitment
to the tumor microenvironment. Cancer Lett. 465:24–35. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Li L, Yang L, Cheng S, Fan Z, Shen Z, Xue
W, Zheng Y, Li F, Wang D, Zhang K, et al: Lung
adenocarcinoma-intrinsic GBE1 signaling inhibits anti-tumor
immunity. Mol Cancer. 18:1082019. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ruiz de Galarreta M, Bresnahan E,
Molina-Sanchez P, Lindblad KE, Maier B, Sia D, Puigvehi M, Miguela
V, Casanova-Acebes M, Dhainaut M, et al: β-catenin activation
promotes immune escape and resistance to anti-PD-1 therapy in
hepatocellular carcinoma. Cancer Discov. 9:1124–1141. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Wang J, Wang Y, Kong F, Han R, Song W,
Chen D, Bu L, Wang S, Yue J and Ma L: Identification of a six-gene
prognostic signature for oral squamous cell carcinoma. J Cell
Physiol. 235:3056–3068. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
An G, Wu F, Huang S, Feng L, Bai J, Gu S
and Zhao X: Effects of CCL5 on the biological behavior of breast
cancer and the mechanisms of its interaction with tumorassociated
macrophages. Oncol Rep. 42:2499–2511. 2019.PubMed/NCBI
|
|
66
|
Komohara Y, Fujiwara Y, Ohnishi K and
Takeya M: Tumor-associated macrophages: Potential therapeutic
targets for anti-cancer therapy. Adv Drug Deliv Rev. 99:180–185.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Cui R, Yue W, Lattime EC, Stein MN, Xu Q
and Tan XL: Targeting tumor-associated macrophages to combat
pancreatic cancer. Oncotarget. 7:50735–50754. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Bosurgi L, Cao YG, Cabeza-Cabrerizo M,
Tucci A, Hughes LD, Kong Y, Weinstein JS, Licona-Limon P, Schmid
ET, Pelorosso F, et al: Macrophage function in tissue repair and
remodeling requires IL-4 or IL-13 with apoptotic cells. Science.
356:1072–1076. 2017. View Article : Google Scholar : PubMed/NCBI
|