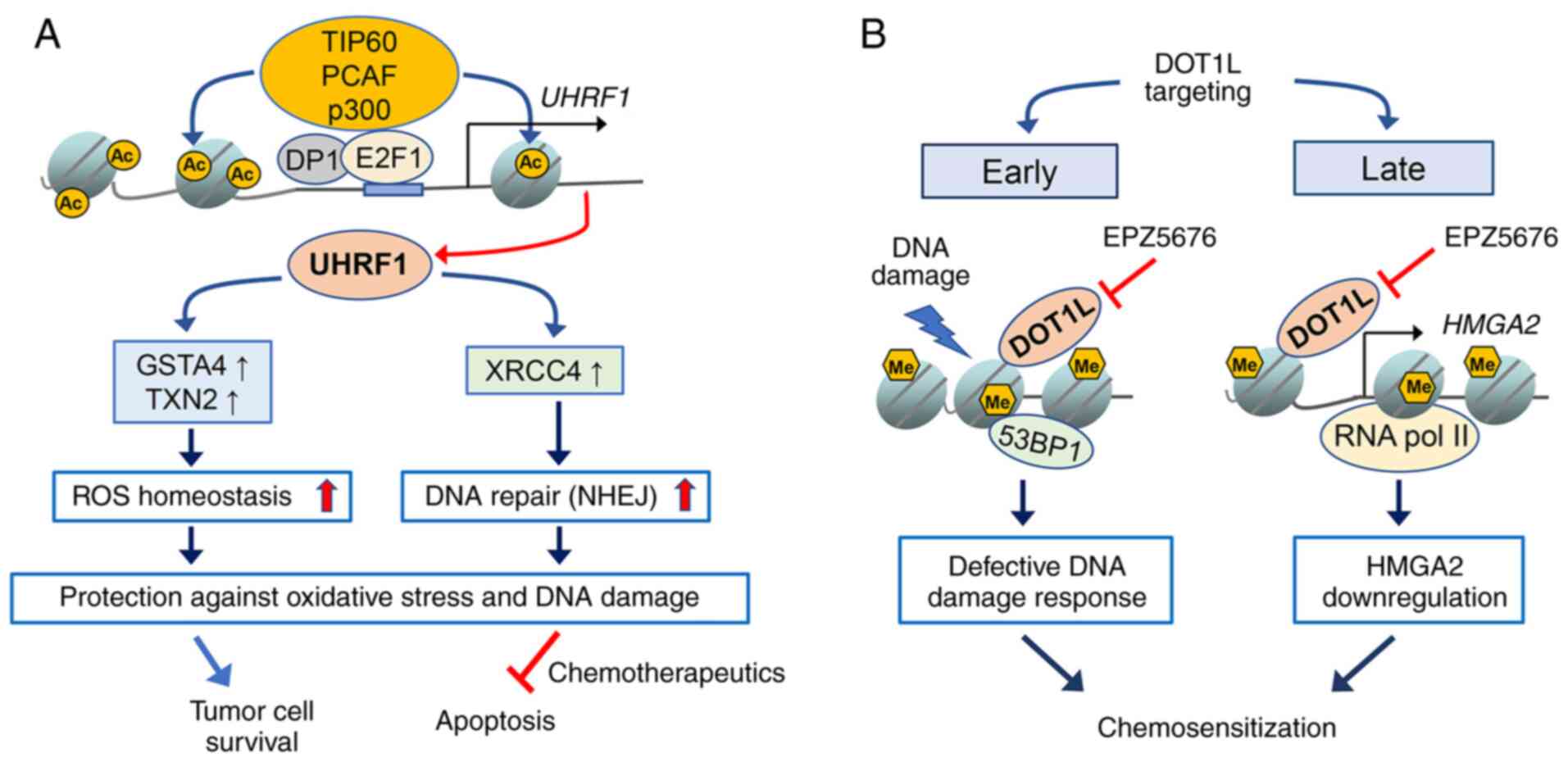
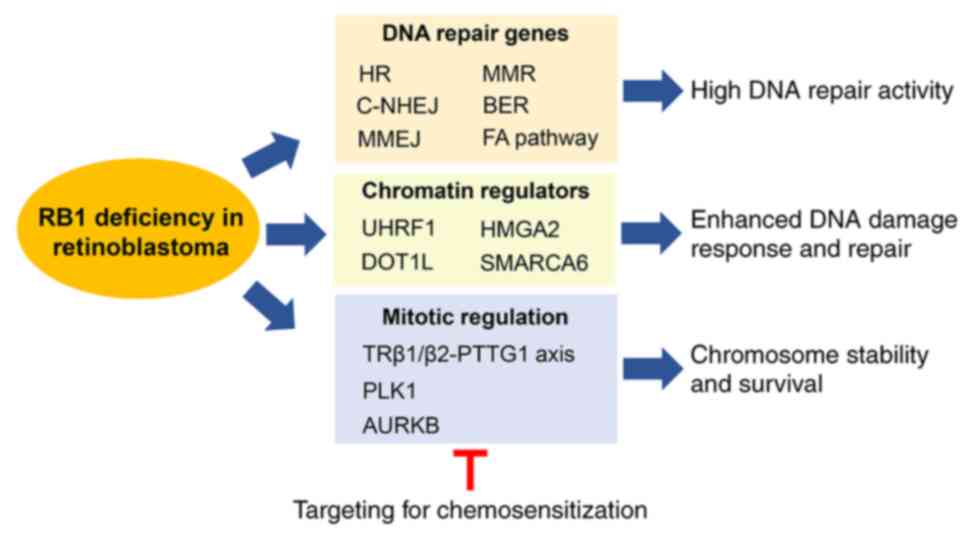
|
1
|
Dimaras H, Corson TW, Cobrinik D, White A,
Zhao J, Munier FL, Abramson DH, Shields CL, Chantada GL, Njuguna F
and Gallie BL: Retinoblastoma. Nat Rev Dis Primers. 1:150212015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Benavente CA and Dyer MA: Genetics and
epigenetics of human retinoblastoma. Annu Rev Pathol. 10:547–562.
2015. View Article : Google Scholar
|
|
3
|
Theriault BL, Dimaras H, Gallie BL and
Corson TW: The genomic landscape of retinoblastoma: A review. Clin
Exp Ophthalmol. 42:33–52. 2014. View Article : Google Scholar
|
|
4
|
Burkhart DL and Sage J: Cellular
mechanisms of tumour suppression by the retinoblastoma gene. Nat
Rev Cancer. 8:671–682. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dick FA, Goodrich DW, Sage J and Dyson NJ:
Non-canonical functions of the RB protein in cancer. Nat Rev
Cancer. 18:442–451. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Talluri S and Dick FA: Regulation of
transcription and chromatin structure by pRB: Here, there and
everywhere. Cell Cycle. 11:3189–3198. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Uchida C: Roles of pRB in the regulation
of nucleosome and chromatin structures. Biomed Res Int.
2016:59597212016. View Article : Google Scholar
|
|
8
|
Knudsen ES, Pruitt SC, Hershberger PA,
Witkiewicz AK and Goodrich DW: Cell cycle and beyond: Exploiting
New RB1 controlled mechanisms for cancer therapy. Trends Cancer.
5:308–324. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Linn P, Kohno S, Sheng J, Kulathunga N, Yu
H, Zhang Z, Voon D, Watanabe Y and Takahashi C: Targeting RB1 loss
in cancers. Cancers (Basel). 13:37372021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar
|
|
11
|
Velez-Cruz R and Johnson DG: The
Retinoblastoma (RB) tumor suppressor: Pushing back against genome
instability on multiple fronts. Int J Mol Sci. 18:17762017.
View Article : Google Scholar
|
|
12
|
Cook R, Zoumpoulidou G, Luczynski MT,
Rieger S, Moquet J, Spanswick VJ, Hartley JA, Rothkamm K, Huang PH
and Mittnacht S: Direct involvement of retinoblastoma family
proteins in DNA repair by non-homologous end-joining. Cell Rep.
10:2006–2018. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Velez-Cruz R, Manickavinayaham S, Biswas
AK, Clary RW, Premkumar T, Cole F and Johnson DG: RB localizes to
DNA double-strand breaks and promotes DNA end resection and
homologous recombination through the recruitment of BRG1. Genes
Dev. 30:2500–2512. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Manickavinayaham S, Velez-Cruz R, Biswas
AK, Chen J, Guo R and Johnson DG: The E2F1 transcription factor and
RB tumor suppressor moonlight as DNA repair factors. Cell Cycle.
19:2260–2269. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bester AC, Roniger M, Oren YS, Im MM,
Sarni D, Chaoat M, Bensimon A, Zamir G, Shewach DS and Kerem B:
Nucleotide deficiency promotes genomic instability in early stages
of cancer development. Cell. 145:435–446. 2011. View Article : Google Scholar
|
|
16
|
Longworth MS, Herr A, Ji JY and Dyson NJ:
RBF1 promotes chromatin condensation through a conserved
interaction with the Condensin II protein dCAP-D3. Genes Dev.
22:1011–1024. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Manning AL, Longworth MS and Dyson NJ:
Loss of pRB causes centromere dysfunction and chromosomal
instability. Genes Dev. 24:1364–1376. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Manning AL, Yazinski SA, Nicolay B, Bryll
A, Zou L and Dyson NJ: Suppression of genome instability in
pRB-deficient cells by enhancement of chromosome cohesion. Mol
Cell. 53:993–1004. 2014. View Article : Google Scholar
|
|
19
|
Coschi CH, Ishak CA, Gallo D, Marshall A,
Talluri S, Wang J, Cecchini MJ, Martens AL, Percy V, Welch I, et
al: Haploinsufficiency of an RB-E2F1-Condensin II complex leads to
aberrant replication and aneuploidy. Cancer Discov. 4:840–853.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Coschi CH, Martens AL, Ritchie K, Francis
SM, Chakrabarti S, Berube NG and Dick FA: Mitotic chromosome
condensation mediated by the retinoblastoma protein is
tumor-suppressive. Genes Dev. 24:1351–1363. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ishak CA, Marshall AE, Passos DT, White
CR, Kim SJ, Cecchini MJ, Ferwati S, MacDonald WA, Howlett CJ, Welch
ID, et al: An RB-EZH2 complex mediates silencing of repetitive DNA
Sequences. Mol Cell. 64:1074–1087. 2016. View Article : Google Scholar
|
|
22
|
Gonzalo S, Garcia-Cao M, Fraga MF, Schotta
G, Peters AH, Cotter SE, Eguia R, Dean DC, Esteller M, Jenuwein T
and Blasco MA: Role of the RB1 family in stabilizing histone
methylation at constitutive heterochromatin. Nat Cell Biol.
7:420–428. 2005. View Article : Google Scholar
|
|
23
|
Isaac CE, Francis SM, Martens AL, Julian
LM, Seifried LA, Erdmann N, Binne UK, Harrington L, Sicinski P,
Berube NG, et al: The retinoblastoma protein regulates pericentric
heterochromatin. Mol Cell Biol. 26:3659–3671. 2006. View Article : Google Scholar
|
|
24
|
Montoya-Durango DE, Ramos KA, Bojang P,
Ruiz L, Ramos IN and Ramos KS: LINE-1 silencing by retinoblastoma
proteins is effected through the nucleosomal and remodeling
deacetylase multiprotein complex. BMC Cancer. 16:382016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang J, Benavente CA, McEvoy J,
Flores-Otero J, Ding L, Chen X, Ulyanov A, Wu G, Wilson M, Wang J,
et al: A novel retinoblastoma therapy from genomic and epigenetic
analyses. Nature. 481:329–334. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
McEvoy J, Nagahawatte P, Finkelstein D,
Richards-Yutz J, Valentine M, Ma J, Mullighan C, Song G, Chen X,
Wilson M, et al: RB1 gene inactivation by chromothripsis in human
retinoblastoma. Oncotarget. 5:438–450. 2014. View Article : Google Scholar
|
|
27
|
Davies HR, Broad KD, Onadim Z, Price EA,
Zou X, Sheriff I, Karaa EK, Scheimberg I, Reddy MA, Sagoo MS, et
al: Whole-Genome sequencing of retinoblastoma reveals the diversity
of rearrangements disrupting RB1 and uncovers a treatment-related
mutational signature. Cancers (Basel). 13:7542021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kooi IE, Mol BM, Massink MP, Ameziane N,
Meijers-Heijboer H, Dommering CJ, van Mil SE, de Vries Y, van der
Hout AH, Kaspers GJ, et al: Somatic genomic alterations in
retinoblastoma beyond RB1 are rare and limited to copy number
changes. Sci Rep. 6:252642016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Afshar AR, Pekmezci M, Bloomer MM, Cadenas
NJ, Stevers M, Banerjee A, Roy R, Olshen AB, Van Ziffle J, Onodera
C, et al: Next-Generation sequencing of retinoblastoma identifies
pathogenic alterations beyond RB1 inactivation that correlate with
aggressive histopathologic features. Ophthalmology. 127:804–813.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Francis JH, Richards AL, Mandelker DL,
Berger MF, Walsh MF, Dunkel IJ, Donoghue MTA and Abramson DH:
Molecular changes in retinoblastoma beyond RB1: Findings from
next-generation sequencing. Cancers (Basel). 13:1492021. View Article : Google Scholar
|
|
31
|
Mendonca V, Evangelista AC, P Matta B, M
Moreira MÂ, Faria P, Lucena E and Seuanez HN: Molecular alterations
in retinoblastoma beyond RB1. Exp Eye Res. 211:1087532021.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Corson TW and Gallie BL: One hit, two
hits, three hits, more? Genomic changes in the development of
retinoblastoma. Genes Chromosomes Cancer. 46:617–634. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu J, Ottaviani D, Sefta M, Desbrousses
C, Chapeaublanc E, Aschero R, Sirab N, Lubieniecki F, Lamas G,
Tonon L, et al: A high-risk retinoblastoma subtype with stemness
features, dedifferentiated cone states and neuronal/ganglion cell
gene expression. Nat Commun. 12:55782021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Grobner SN, Worst BC, Weischenfeldt J,
Buchhalter I, Kleinheinz K, Rudneva VA, Johann PD, Balasubramanian
GP, Segura-Wang M, Brabetz S, et al: The landscape of genomic
alterations across childhood cancers. Nature. 555:321–327. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Alexandrov LB, Jones PH, Wedge DC, Sale
JE, Campbell PJ, Nik-Zainal S and Stratton MR: Clock-like
mutational processes in human somatic cells. Nat Genet.
47:1402–1407. 2015. View Article : Google Scholar
|
|
36
|
Polski A, Xu L, Prabakar RK, Gai X, Kim
JW, Shah R, Jubran R, Kuhn P, Cobrinik D, Hicks J and Berry JL:
Variability in retinoblastoma genome stability is driven by age and
not heritability. Genes Chromosomes Cancer. 59:584–590. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kato MV, Shimizu T, Ishizaki K, Kaneko A,
Yandell DW, Toguchida J and Sasaki MS: Loss of heterozygosity on
chromosome 17 and mutation of the p53 gene in retinoblastoma.
Cancer Lett. 106:75–82. 1996. View Article : Google Scholar
|
|
38
|
Kondo Y, Kondo S, Liu J, Haqqi T, Barnett
GH and Barna BP: Involvement of p53 and WAF1/CIP1 in
gamma-irradiation-induced apoptosis of retinoblastoma cells. Exp
Cell Res. 236:51–56. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Xu XL, Fang Y, Lee TC, Forrest D,
Gregory-Evans C, Almeida D, Liu A, Jhanwar SC, Abramson DH and
Cobrinik D: Retinoblastoma has properties of a cone precursor tumor
and depends upon cone-specific MDM2 signaling. Cell. 137:1018–1031.
2009. View Article : Google Scholar
|
|
40
|
Xu XL, Singh HP, Wang L, Qi DL, Poulos BK,
Abramson DH, Jhanwar SC and Cobrinik D: Rb suppresses human
cone-precursor-derived retinoblastoma tumours. Nature. 514:385–388.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Laurie NA, Donovan SL, Shih CS, Zhang J,
Mills N, Fuller C, Teunisse A, Lam S, Ramos Y, Mohan A, et al:
Inactivation of the p53 pathway in retinoblastoma. Nature.
444:61–66. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chakraborty S, Khare S, Dorairaj SK,
Prabhakaran VC, Prakash DR and Kumar A: Identification of genes
associated with tumorigenesis of retinoblastoma by microarray
analysis. Genomics. 90:344–353. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ganguly A and Shields CL: Differential
gene expression profile of retinoblastoma compared to normal
retina. Mol Vis. 16:1292–1303. 2010.PubMed/NCBI
|
|
44
|
Kapatai G, Brundler MA, Jenkinson H,
Kearns P, Parulekar M, Peet AC and McConville CM: Gene expression
profiling identifies different sub-types of retinoblastoma. Br J
Cancer. 109:512–525. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Rajasekaran S, Nagarajha Selvan LD, Dotts
K, Kumar R, Rishi P, Khetan V, Bisht M, Sivaraman K, Krishnakumar
S, Sahoo D, et al: Non-coding and coding transcriptional profiles
are significantly altered in pediatric retinoblastoma tumors. Front
Oncol. 9:2212019. View Article : Google Scholar
|
|
46
|
Aubry A, Pearson JD, Huang K, Livne-Bar I,
Ahmad M, Jagadeesan M, Khetan V, Ketela T, Brown KR, Yu T, et al:
Functional genomics identifies new synergistic therapies for
retinoblastoma. Oncogene. 39:5338–5357. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Markey MP, Bergseid J, Bosco EE, Stengel
K, Xu H, Mayhew CN, Schwemberger SJ, Braden WA, Jiang Y, Babcock
GF, et al: Loss of the retinoblastoma tumor suppressor:
Differential action on transcriptional programs related to cell
cycle control and immune function. Oncogene. 26:6307–6318. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Mayhew CN, Carter SL, Fox SR, Sexton CR,
Reed CA, Srinivasan SV, Liu X, Wikenheiser-Brokamp K, Boivin GP,
Lee JS, et al: RB loss abrogates cell cycle control and genome
integrity to promote liver tumorigenesis. Gastroenterology.
133:976–984. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Black EP, Huang E, Dressman H, Rempel R,
Laakso N, Asa SL, Ishida S, West M and Nevins JR: Distinct gene
expression phenotypes of cells lacking Rb and Rb family members.
Cancer Res. 63:3716–3723. 2003.PubMed/NCBI
|
|
50
|
Ren B, Cam H, Takahashi Y, Volkert T,
Terragni J, Young RA and Dynlacht BD: E2F integrates cell cycle
progression with DNA repair, replication, and G(2)/M checkpoints.
Genes Dev. 16:245–256. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Bracken AP, Ciro M, Cocito A and Helin K:
E2F target genes: Unraveling the biology. Trends Biochem Sci.
29:409–417. 2004. View Article : Google Scholar
|
|
52
|
Mun JY, Baek SW, Park WY, Kim WT, Kim SK,
Roh YG, Jeong MS, Yang GE, Lee JH, Chung JW, et al: E2F1 promotes
progression of bladder cancer by modulating RAD54L involved in
homologous recombination repair. Int J Mol Sci. 21:90252020.
View Article : Google Scholar
|
|
53
|
Sakthivel KM and Hariharan S: Regulatory
players of DNA damage repair mechanisms: Role in cancer
chemoresistance. Biomed Pharmacother. 93:1238–1245. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Hosoya N and Miyagawa K: Targeting DNA
damage response in cancer therapy. Cancer Sci. 105:370–388. 2014.
View Article : Google Scholar
|
|
55
|
O'Connor MJ: Targeting the DNA damage
response in cancer. Mol Cell. 60:547–560. 2015. View Article : Google Scholar
|
|
56
|
Jeggo PA and Downs JA: Roles of chromatin
remodellers in DNA double strand break repair. Exp Cell Res.
329:69–77. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Lee C and Kim JK: Chromatin regulators in
retinoblastoma: Biological roles and therapeutic applications. J
Cell Physiol. 236:2318–2332. 2021. View Article : Google Scholar
|
|
58
|
Bronner C, Krifa M and Mousli M:
Increasing role of UHRF1 in the reading and inheritance of the
epigenetic code as well as in tumorogenesis. Biochem Pharmacol.
86:1643–1649. 2013. View Article : Google Scholar
|
|
59
|
Alhosin M, Omran Z, Zamzami MA, Al-Malki
AL, Choudhry H, Mousli M and Bronner C: Signalling pathways in
UHRF1-dependent regulation of tumor suppressor genes in cancer. J
Exp Clin Cancer Res. 35:1742016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Tian Y, Paramasivam M, Ghosal G, Chen D,
Shen X, Huang Y, Akhter S, Legerski R, Chen J, Seidman MM, et al:
UHRF1 contributes to DNA damage repair as a lesion recognition
factor and nuclease scaffold. Cell Rep. 10:1957–1966. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Liang CC, Zhan B, Yoshikawa Y, Haas W,
Gygi SP and Cohn MA: UHRF1 is a sensor for DNA interstrand
crosslinks and recruits FANCD2 to initiate the Fanconi anemia
pathway. Cell Rep. 10:1947–1956. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Zhang H, Liu H, Chen Y, Yang X, Wang P,
Liu T, Deng M, Qin B, Correia C, Lee S, et al: A cell
cycle-dependent BRCA1-UHRF1 cascade regulates DNA double-strand
break repair pathway choice. Nat Commun. 7:102012016. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Mancini M, Magnani E, Macchi F and
Bonapace IM: The multi-functionality of UHRF1: Epigenome
maintenance and preservation of genome integrity. Nucleic Acids
Res. 49:6053–6068. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Muto M, Kanari Y, Kubo E, Takabe T,
Kurihara T, Fujimori A and Tatsumi K: Targeted disruption of Np95
gene renders murine embryonic stem cells hypersensitive to DNA
damaging agents and DNA replication blocks. J Biol Chem.
277:34549–34555. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
He H, Lee C and Kim JK: UHRF1 depletion
sensitizes retinoblastoma cells to chemotherapeutic drugs via
downregulation of XRCC4. Cell Death Dis. 9:1642018. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Kim JK, Kan G, Mao Y, Wu Z, Tan X, He H
and Lee C: UHRF1 downmodulation enhances antitumor effects of
histone deacetylase inhibitors in retinoblastoma by augmenting
oxidative stress-mediated apoptosis. Mol Oncol. 14:329–346. 2020.
View Article : Google Scholar
|
|
67
|
Sharma V, Collins LB, Chen TH, Herr N,
Takeda S, Sun W, Swenberg JA and Nakamura J: Oxidative stress at
low levels can induce clustered DNA lesions leading to NHEJ
mediated mutations. Oncotarget. 7:25377–25390. 2016. View Article : Google Scholar
|
|
68
|
Karanjawala ZE, Murphy N, Hinton DR, Hsieh
CL and Lieber MR: Oxygen metabolism causes chromosome breaks and is
associated with the neuronal apoptosis observed in DNA
double-strand break repair mutants. Curr Biol. 12:397–402. 2002.
View Article : Google Scholar
|
|
69
|
Mao Y, Sun Y, Wu Z, Zheng J, Zhang J, Zeng
J, Lee C and Kim JK: Targeting of histone methyltransferase DOT1L
plays a dual role in chemosensitization of retinoblastoma cells and
enhances the efficacy of chemotherapy. Cell Death Dis. 12:11412021.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Feng Q, Wang H, Ng HH, Erdjument-Bromage
H, Tempst P, Struhl K and Zhang Y: Methylation of H3-lysine 79 is
mediated by a new family of HMTases without a SET domain. Curr
Biol. 12:1052–1058. 2002. View Article : Google Scholar
|
|
71
|
Wood K, Tellier M and Murphy S: DOT1L and
H3K79 methylation in transcription and genomic stability.
Biomolecules. 8:112018. View Article : Google Scholar
|
|
72
|
Wakeman TP, Wang Q, Feng J and Wang XF:
Bat3 facilitates H3K79 dimethylation by DOT1L and promotes DNA
damage-induced 53BP1 foci at G1/G2 cell-cycle phases. EMBO J.
31:2169–2181. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Kari V, Raul SK, Henck JM, Kitz J, Kramer
F, Kosinsky RL, Ubelmesser N, Mansour WY, Eggert J, Spitzner M, et
al: The histone methyltransferase DOT1L is required for proper DNA
damage response, DNA repair, and modulates chemotherapy
responsiveness. Clin Epigenetics. 11:42019. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Liu W, Deng L, Song Y and Redell M: DOT1L
inhibition sensitizes MLL-rearranged AML to chemotherapy. PLoS One.
9:e982702014. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Chau KY, Manfioletti G, Cheung-Chau KW,
Fusco A, Dhomen N, Sowden JC, Sasabe T, Mukai S and Ono SJ:
Derepression of HMGA2 gene expression in retinoblastoma is
associated with cell proliferation. Mol Med. 9:154–165. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Palmieri D, Valentino T, D'Angelo D, De
Martino I, Postiglione I, Pacelli R, Croce CM, Fedele M and Fusco
A: HMGA proteins promote ATM expression and enhance cancer cell
resistance to genotoxic agents. Oncogene. 30:3024–3035. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Natarajan S, Hombach-Klonisch S, Droge P
and Klonisch T: HMGA2 inhibits apoptosis through interaction with
ATR-CHK1 signaling complex in human cancer cells. Neoplasia.
15:263–280. 2013. View Article : Google Scholar
|
|
78
|
Nalini V, Deepa PR, Raguraman R, Khetan V,
Reddy MA and Krishnakumar S: Targeting HMGA2 in Retinoblastoma
Cells in vitro using the aptamer strategy. Ocul Oncol Pathol.
2:262–269. 2016. View Article : Google Scholar
|
|
79
|
Kollarovic G, Topping CE, Shaw EP and
Chambers AL: The human HELLS chromatin remodelling protein promotes
end resection to facilitate homologous recombination and
contributes to DSB repair within heterochromatin. Nucleic Acids
Res. 48:1872–1885. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Costelloe T, Louge R, Tomimatsu N,
Mukherjee B, Martini E, Khadaroo B, Dubois K, Wiegant WW, Thierry
A, Burma S, et al: The yeast Fun30 and human SMARCAD1 chromatin
remodellers promote DNA end resection. Nature. 489:581–584. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Zocchi L, Mehta A, Wu SC, Wu J, Gu Y, Wang
J, Suh S, Spitale RC and Benavente CA: Chromatin remodeling protein
HELLS is critical for retinoblastoma tumor initiation and
progression. Oncogenesis. 9:252020. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Manning AL and Dyson NJ: RB: Mitotic
implications of a tumour suppressor. Nat Rev Cancer. 12:220–226.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Pappas L, Xu XL, Abramson DH and Jhanwar
SC: Genomic instability and proliferation/survival pathways in
RB1-deficient malignancies. Adv Biol Regul. 64:20–32. 2017.
View Article : Google Scholar
|
|
84
|
Salehi F, Kovacs K, Scheithauer BW, Lloyd
RV and Cusimano M: Pituitary tumor-transforming gene in endocrine
and other neoplasms: A review and update. Endocr Relat Cancer.
15:721–743. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Holland AJ and Cleveland DW: Boveri
revisited: Chromosomal instability, aneuploidy and tumorigenesis.
Nat Rev Mol Cell Biol. 10:478–487. 2009. View Article : Google Scholar
|
|
86
|
Gong X, Du J, Parsons SH, Merzoug FF,
Webster Y, Iversen PW, Chio LC, Van Horn RD, Lin X, Blosser W, et
al: Aurora A kinase inhibition is synthetic lethal with loss of the
RB1 tumor suppressor gene. Cancer Discov. 9:248–263. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Oser MG, Fonseca R, Chakraborty AA, Brough
R, Spektor A, Jennings RB, Flaifel A, Novak JS, Gulati A, Buss E,
et al: Cells lacking the RB1 tumor suppressor gene are
hyperdependent on Aurora B kinase for survival. Cancer Discov.
9:230–247. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Willems E, Dedobbeleer M, Digregorio M,
Lombard A, Lumapat PN and Rogister B: The functional diversity of
Aurora kinases: A comprehensive review. Cell Div. 13:72018.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Borah NA, Sradhanjali S, Barik MR, Jha A,
Tripathy D, Kaliki S, Rath S, Raghav SK, Patnaik S, Mittal R and
Reddy MM: Aurora Kinase B expression, its regulation and
therapeutic targeting in human retinoblastoma. Invest Ophthalmol
Vis Sci. 62:162021. View Article : Google Scholar
|
|
90
|
Singh L, Pushker N, Sen S, Singh MK,
Chauhan FA and Kashyap S: Prognostic significance of polo-like
kinases in retinoblastoma: Correlation with patient outcome,
clinical and histopathological parameters. Clin Exp Ophthalmol.
43:550–557. 2015. View Article : Google Scholar
|
|
91
|
Ma H, Nie C, Chen Y, Li J, Xie Y, Tang Z,
Gao Y, Ai S, Mao Y, Sun Q and Lu R: Therapeutic Targeting PLK1 by
ON-01910.Na is effective in local treatment of retinoblastoma.
Oncol Res. 28:745–761. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Takaki T, Trenz K, Costanzo V and
Petronczki M: Polo-like kinase 1 reaches beyond
mitosis-cytokinesis, DNA damage response, and development. Curr
Opin Cell Biol. 20:650–660. 2008. View Article : Google Scholar
|
|
93
|
Sun J, Xi HY, Shao Q and Liu QH:
Biomarkers in retinoblastoma. Int J Ophthalmol. 13:325–341. 2020.
View Article : Google Scholar
|
|
94
|
Chai P, Jia R, Li Y, Zhou C, Gu X, Yang L,
Shi H, Tian H, Lin H, Yu J, et al: Regulation of epigenetic
homeostasis in uveal melanoma and retinoblastoma. Prog Retin Eye
Res. Dec 1–2021.(Epub ahead of print). View Article : Google Scholar
|
|
95
|
Chan HS, Gallie BL, Munier FL and Beck
Popovic M: Chemotherapy for retinoblastoma. Ophthalmol Clin North
Am. 1855–63. (viii)2005. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Gombos DS, Hungerford J, Abramson DH,
Kingston J, Chantada G, Dunkel IJ, Antoneli CB, Greenwald M, Haik
BG, Leal CA, et al: Secondary acute myelogenous leukemia in
patients with retinoblastoma: Is chemotherapy a factor?
Ophthalmology. 114:1378–1383. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Mulvihill A, Budning A, Jay V, Vandenhoven
C, Heon E, Gallie BL and Chan HS: Ocular motility changes after
subtenon carboplatin chemotherapy for retinoblastoma. Arch
Ophthalmol. 121:1120–1124. 2003. View Article : Google Scholar
|
|
98
|
Zoumpoulidou G, Alvarez-Mendoza C, Mancusi
C, Ahmed RM, Denman M, Steele CD, Tarabichi M, Roy E, Davies LR,
Manji J, et al: Therapeutic vulnerability to PARP1,2 inhibition in
RB1-mutant osteosarcoma. Nat Commun. 12:70642021. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Basavapathruni A, Jin L, Daigle SR, Majer
CR, Therkelsen CA, Wigle TJ, Kuntz KW, Chesworth R, Pollock RM,
Scott MP, et al: Conformational adaptation drives potent, selective
and durable inhibition of the human protein methyltransferase
DOT1L. Chem Biol Drug Des. 80:971–980. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Waters NJ: Preclinical Pharmacokinetics
and pharmacodynamics of pinometostat (EPZ-5676), a First-in-Class,
small Molecule S-Adenosyl methionine competitive inhibitor of
DOT1L. Eur J Drug Metab Pharmacokinet. 42:891–901. 2017. View Article : Google Scholar
|
|
101
|
Stein EM, Garcia-Manero G, Rizzieri DA,
Tibes R, Berdeja JG, Savona MR, Jongen-Lavrenic M, Altman JK,
Thomson B, Blakemore SJ, et al: The DOT1L inhibitor pinometostat
reduces H3K79 methylation and has modest clinical activity in adult
acute leukemia. Blood. 131:2661–2669. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Shields CL, Manjandavida FP, Lally SE,
Pieretti G, Arepalli SA, Caywood EH, Jabbour P and Shields JA:
Intra-arterial chemotherapy for retinoblastoma in 70 eyes: Outcomes
based on the international classification of retinoblastoma.
Ophthalmology. 121:1453–1460. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Munier FL, Gaillard MC, Balmer A, Soliman
S, Podilsky G, Moulin AP and Beck-Popovic M: Intravitreal
chemotherapy for vitreous disease in retinoblastoma revisited: From
prohibition to conditional indications. Br J Ophthalmol.
96:1078–1083. 2012. View Article : Google Scholar
|
|
104
|
Ghassemi F, Shields CL, Ghadimi H,
Khodabandeh A and Roohipoor R: Combined intravitreal melphalan and
topotecan for refractory or recurrent vitreous seeding from
retinoblastoma. JAMA Ophthalmol. 132:936–941. 2014. View Article : Google Scholar
|
|
105
|
Lee JE and Kim MY: Cancer epigenetics:
Past, present and future. Semin Cancer Biol. Mar 31–2021.(Epub
ahead of print). View Article : Google Scholar
|
|
106
|
He L and Lomberk G: Collateral Victim or
Rescue Worker?-The role of histone methyltransferases in DNA damage
repair and their targeting for therapeutic opportunities in cancer.
Front Cell Dev Biol. 9:7351072021. View Article : Google Scholar
|
|
107
|
Unoki M, Nishidate T and Nakamura Y:
ICBP90, an E2F-1 target, recruits HDAC1 and binds to methyl-CpG
through its SRA domain. Oncogene. 23:7601–7610. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Ganesan A, Arimondo PB, Rots MG, Jeronimo
C and Berdasco M: The timeline of epigenetic drug discovery: From
reality to dreams. Clin Epigenetics. 11:1742019. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Chun AW, Cosenza SC, Taft DR and Maniar M:
Preclinical pharmacokinetics and in vitro activity of ON 01910.Na,
a novel anti-cancer agent. Cancer Chemother Pharmacol. 65:177–186.
2009. View Article : Google Scholar
|
|
110
|
Ohnuma T, Lehrer D, Ren C, Cho SY, Maniar
M, Silverman L, Sung M, Gretz HF III, Benisovich V, Navada S, et
al: Phase 1 study of intravenous rigosertib (ON 01910.Na), a novel
benzyl styryl sulfone structure producing G2/M arrest and
apoptosis, in adult patients with advanced cancer. Am J Cancer Res.
3:323–338. 2013.PubMed/NCBI
|
|
111
|
O'Neil BH, Scott AJ, Ma WW, Cohen SJ,
Aisner DL, Menter AR, Tejani MA, Cho JK, Granfortuna J, Coveler L,
et al: A phase II/III randomized study to compare the efficacy and
safety of rigosertib plus gemcitabine versus gemcitabine alone in
patients with previously untreated metastatic pancreatic cancer.
Ann Oncol. 26:1923–1929. 2015. View Article : Google Scholar
|
|
112
|
Bowles DW, Diamond JR, Lam ET, Weekes CD,
Astling DP, Anderson RT, Leong S, Gore L, Varella-Garcia M, Vogler
BW, et al: Phase I study of oral rigosertib (ON 01910.Na), a dual
inhibitor of the PI3K and Plk1 pathways, in adult patients with
advanced solid malignancies. Clin Cancer Res. 20:1656–1665. 2014.
View Article : Google Scholar : PubMed/NCBI
|