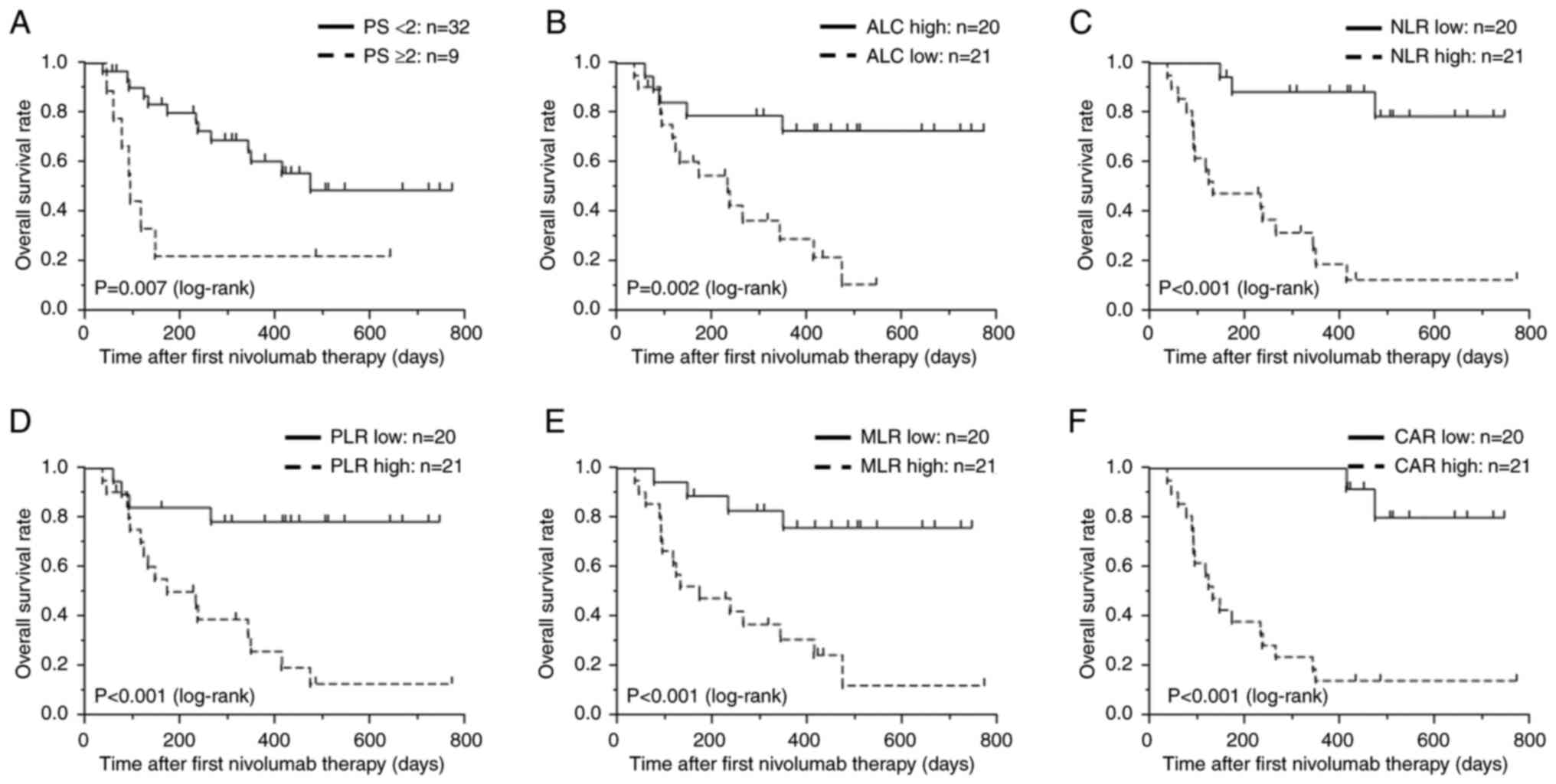
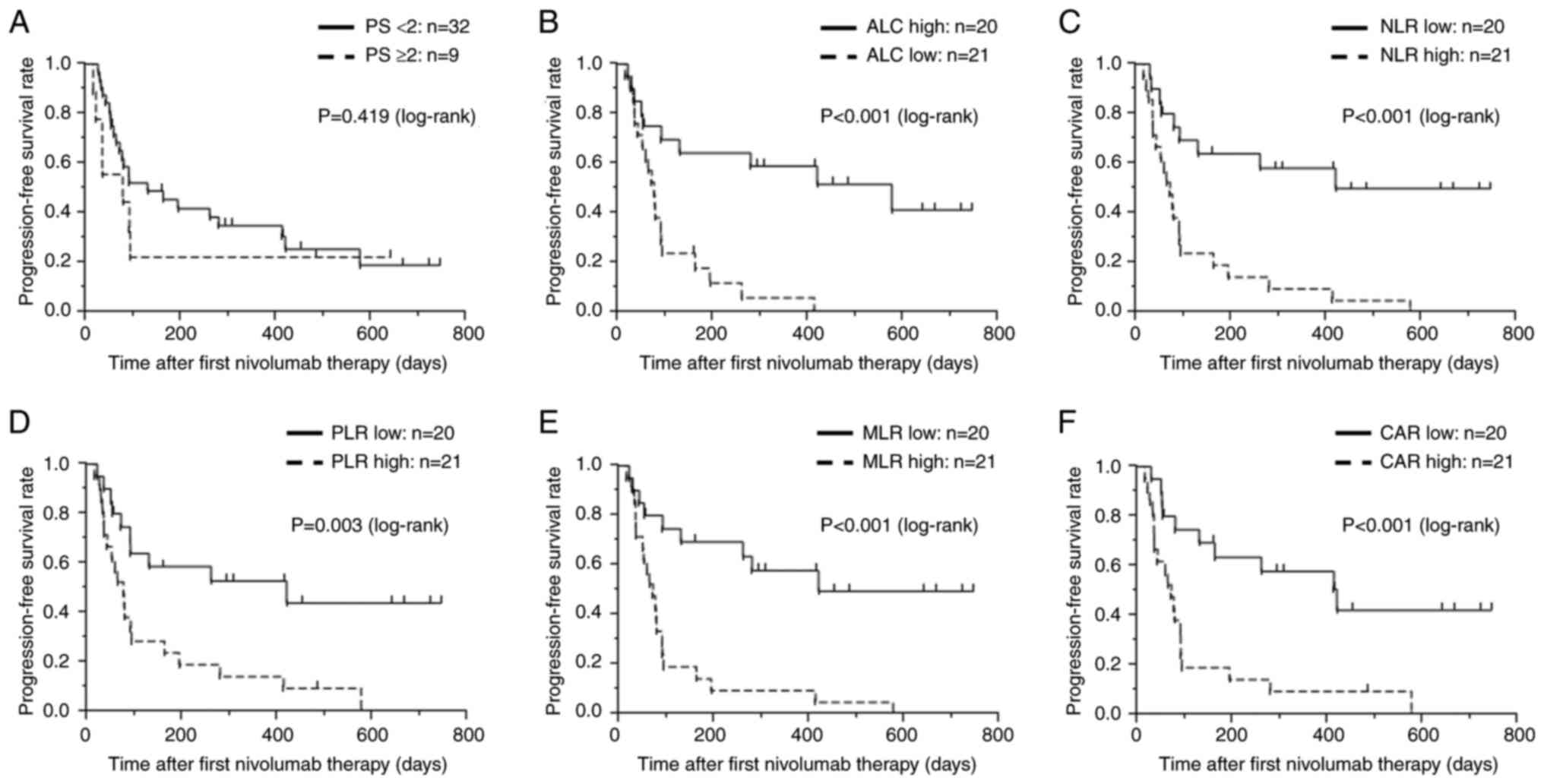
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Watanabe M, Tachimori Y, Oyama T, Toh Y,
Matsubara H, Ueno M, Kono K, Uno T, Ishihara R, Muro K, et al:
Comprehensive registry of esophageal cancer in Japan, 2013.
Esophagus. 18:1–24. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Toh Y, Oki E, Minami K and Okamura T:
Follow-up and recurrence after a curative esophagectomy for
patients with esophageal cancer: The first indicators for
recurrence and their prognostic values. Esophagus. 7:37–43. 2010.
View Article : Google Scholar
|
|
4
|
Miyata H, Yamasaki M, Kurokawa Y,
Takiguchi S, Nakajima K, Fujiwara Y, Konishi K, Mori M and Doki Y:
Survival factors in patients with recurrence after curative
resection of esophageal squamous cell carcinomas. Ann Surg Oncol.
18:3353–3361. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mariette C, Balon JM, Piessen G, Fabre S,
Van Seuningen I and Triboulet JP: Pattern of recurrence following
complete resection of esophageal carcinoma and factors predictive
of recurrent disease. Cancer. 97:1616–1623. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kato K, Cho BC, Takahashi M, Okada M, Lin
CY, Chin K, Kadowaki S, Ahn MJ, Hamamoto Y, Doki Y, et al:
Nivolumab versus chemotherapy in patients with advanced oesophageal
squamous cell carcinoma refractory or intolerant to previous
chemotherapy (ATTRACTION-3): A multicentre, randomised, open-label,
phase 3 trial. Lancet Oncol. 20:1506–1517. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kitagawa Y, Uno T, Oyama T, Kato K, Kato
H, Kawakubo H, Kawamura O, Kusano M, Kuwano H, Takeuchi H, et al:
Esophageal cancer practice guidelines 2017 edited by the Japan
esophageal society: Part 1. Esophagus. 16:1–24. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Brierley JD, Gospodarowicz MK and
Wittekind C: TNM classification of malignant tumours. Union for
International Cancer Control. 8th edition. John Wiley & Sons,
Ltd.; pp. 57–62. 2017
|
|
9
|
Ma X, Zhao K, Guo W, Yang S, Zhu X, Xiang
J, Zhang Y and Li H: Salvage lymphadenectomy versus salvage
radiotherapy/chemoradiotherapy for recurrence in cervical lymph
node after curative resection of esophageal squamous cell
carcinoma. Ann Surg Oncol. 22:624–629. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tsuchida E, Sakai K, Matsumoto Y, Sugita
T, Sasamoto R, Yamanoi T, Sueyama H, Ito T and Sasai K: Concurrent
chemoradiotherapy using low-dose continuous infusion of
5-fluorouracil for postoperative regional lymph node recurrence of
esophageal squamous cell carcinoma. Esophagus. 2:25–31. 2005.
View Article : Google Scholar
|
|
11
|
Nakamura T, Ota M, Narumiya K, Sato T,
Ohki T, Yamamoto M and Mitsuhashi N: Multimodal treatment for lymph
node recurrence of esophageal carcinoma after curative resection.
Ann Surg Oncol. 15:2451–2457. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hironaka S, Tsubosa Y, Mizusawa J, Kii T,
Kato K, Tsushima T, Chin K, Tomori A, Okuno T, Taniki T, et al:
Phase I/II trial of 2-weekly docetaxel combined with cisplatin plus
fluorouracil in metastatic esophageal cancer (JCOG0807). Cancer
Sci. 105:1189–1195. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
National Cancer Institute (NIH), . U.S.
Department of Health and Human Services: Common Terminology
Criteria for Adverse Events (CTCAE) version 4.0. 2009. https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/Archive/CTCAE_4.0_2009-05-29_QuickReference_8.5×11.pdfJanuary
1–2022
|
|
15
|
Hagi T, Kurokawa Y, Kawabata R, Omori T,
Matsuyama J, Fujitani K, Hirao M, Akamaru Y, Takahashi T, Yamasaki
M, et al: Multicentre biomarker cohort study on the efficacy of
nivolumab treatment for gastric cancer. Br J Cancer. 123:965–972.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Karantanos T, Karanika S, Seth B and
Gignac G: The absolute lymphocyte count can predict the overall
survival of patients with non-small cell lung cancer on nivolumab:
A clinical study. Clin Transl Oncol. 21:206–212. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yamada T, Hayashi T, Inokuchi Y, Hayashi
K, Watanabe H, Komori K, Kano K, Shimoda Y, Fujikawa H, Shiozawa M,
et al: Impact of the neutrophil-to-lymphocyte ratio on the survival
of patients with gastric cancer treated with nivolumab monotherapy.
Target Oncol. 15:317–325. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ogata T, Satake H, Ogata M, Hatachi Y,
Inoue K, Hamada M and Yasui H: Neutrophil-to-lymphocyte ratio as a
predictive or prognostic factor for gastric cancer treated with
nivolumab: A multicenter retrospective study. Oncotarget.
9:34520–34527. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang N, Jiang J, Tang S and Sun G:
Predictive value of neutrophil-lymphocyte ratio and
platelet-lymphocyte ratio in non-small cell lung cancer patients
treated with immune checkpoint inhibitors: A meta-analysis. Int
Immunopharmacol. 85:1066772020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rossi S, Toschi L, Finocchiaro G and
Santoro A: Neutrophil and lymphocyte blood count as potential
predictive indicators of nivolumab efficacy in metastatic
non-small-cell lung cancer. Immunotherapy. 12:715–724. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tanoue K, Tamura S, Kusaba H, Shinohara Y,
Ito M, Tsuchihashi K, Shirakawa T, Otsuka T, Ohmura H, Isobe T, et
al: Predictive impact of C-reactive protein to albumin ratio for
recurrent or metastatic head and neck squamous cell carcinoma
receiving nivolumab. Sci Rep. 11:27412021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wolchok JD, Kluger H, Callahan MK, Postow
MA, Rizvi NA, Lesokhin AM, Segal NH, Ariyan CE, Gordon RA, Reed K,
et al: Nivolumab plus ipilimumab in advanced melanoma. N Engl J
Med. 369:122–133. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Carbone DP, Reck M, Paz-Ares L, Creelan B,
Horn L, Steins M, Felip E, van den Heuvel MM, Ciuleanu TE, Badin F,
et al: First-line nivolumab in stage IV or recurrent non-small-cell
lung cancer. N Engl J Med. 376:2415–2426. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Motzer RJ, Tannir NM, McDermott DF,
Frontera OA, Melichar B, Choueiri TK, Plimack ER, Barthélémy P,
Porta C, George S, et al: Nivolumab plus Ipilimumab versus
sunitinib in advanced renal-cell carcinoma. N Engl J Med.
378:1277–1290. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kang YK, Boku N, Satoh T, Ryu MH, Chao Y,
Kato K, Chung HC, Chen JS, Muro K, Kang WK, et al: Nivolumab in
patients with advanced gastric or gastro-oesophageal junction
cancer refractory to, or intolerant of, at least two previous
chemotherapy regimens (ONO-4538-12, ATTRACTION-2): A randomised,
double-blind, placebo-controlled, phase 3 trial. Lancet.
390:2461–2471. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Satoh T, Kato K, Ura T, Hamamoto Y, Kojima
T, Tsushima T, Hironaka S, Hara H, Iwasa S, Muro K, et al:
Five-year follow-up of nivolumab treatment in Japanese patients
with esophageal squamous-cell carcinoma (ATTRACTION-1/ONO-4538-07).
Esophagus. 18:835–843. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Takahashi M, Kato K, Okada M, Chin K,
Kadowaki S, Hamamoto Y, Doki Y, Kubota Y, Kawakami H, Ogata T, et
al: Nivolumab versus chemotherapy in Japanese patients with
advanced esophageal squamous cell carcinoma: A subgroup analysis of
a multicenter, randomized, open-label, phase 3 trial
(ATTRACTION-3). Esophagus. 18:90–99. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ueda K, Suekane S, Kurose H, Ogasawara N,
Hiroshige T, Chikui K, Uemura K, Nakiri M, Nishihara K, Matsuo M
and Igawa T: Absolute lymphocyte count is an independent predictor
of survival in patients with metastatic renal cell carcinoma
treated with nivolumab. Jpn J Clin Oncol. 52:179–186. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Haratani K, Hayashi H, Chiba Y, Kudo K,
Yonesaka K, Kato R, Kaneda H, Hasegawa Y, Tanaka K, Takeda M and
Nakagawa K: Association of immune-related adverse events with
nivolumab efficacy in non-small-cell lung cancer. JAMA Oncol.
4:374–378. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kato K, Doki Y, Ura T, Hamamoto Y, Kojima
T, Tsushima T, Hironaka S, Hara H, Kudo T, Iwasa S, et al:
Long-term efficacy and predictive correlates of response to
nivolumab in Japanese patients with esophageal cancer. Cancer Sci.
111:1676–1684. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yarchoan M, Hopkins A and Jaffee EM: Tumor
mutational burden and response rate to PD-1 inhibition. N Engl J
Med. 377:2500–2501. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pavan A, Calvetti L, Dal Maso A, Attili I,
Bianco PD, Pasello G, Guarneri V, Aprile G, Conte P and Bonanno L:
Peripheral blood markers identify risk of immune-related toxicity
in advanced non-small cell lung cancer treated with
immune-checkpoint inhibitors. Oncologist. 24:1128–1136. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Okadome K, Baba Y and Baba H: Prognostic
nutritional index, tumor-infiltrating lymphocytes, and prognosis in
patients with esophageal cancer. Ann Surg. 271:693–700. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lee KH, Kim EY, Yun JS, Park YL, Do SI,
Chae SW and Park CH: The prognostic and predictive value of
tumor--infiltrating lymphocytes and hematologic parameters in
patients with breast cancer. BMC Cancer. 18:9382018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lim SH, Hong M, Ahn S, Choi YL, Kim KM, Oh
D, Ahn YC, Jung SH, Ahn MJ, Park K, et al: Changes in tumour
expression of programmed death-ligand 1 after neoadjuvant
concurrent chemoradiotherapy in patients with squamous oesophageal
cancer. Eur J Cancer. 52:1–9. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yan T, Cui H, Zhou Y, Yang B, Kong P,
Zhang Y, Liu Y, Wang B, Cheng Y, Li J, et al: Multi-region
sequencing unveils novel actionable targets and spatial
heterogeneity in esophageal squamous cell carcinoma. Nat Commun.
10:16702019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Jiao R, Zheng X, Sun Y, Feng Z, Song S and
Ge H: IDO1 Expression increased after neoadjuvant therapy predicts
poor pathologic response and prognosis in esophageal squamous cell
carcinoma. Front Oncol. 10:10992020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Tan Q, Zhang C, Yang W, Liu Y, Heyilimu P,
Feng D, Xing L, Ke Y and Lu Z: Isolation of T cell receptor
specifically reactive with autologous tumour cells from
tumour-infiltrating lymphocytes and construction of T cell receptor
engineered T cells for esophageal squamous cell carcinoma. J
Immunother Cancer. 7:2322019. View Article : Google Scholar : PubMed/NCBI
|