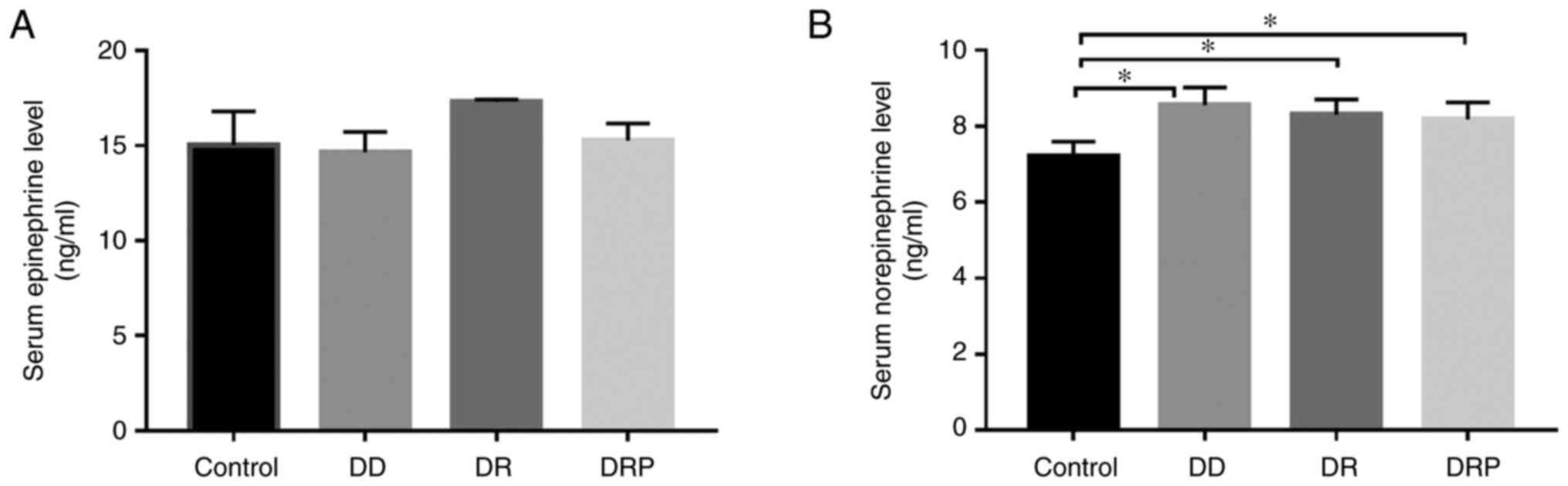
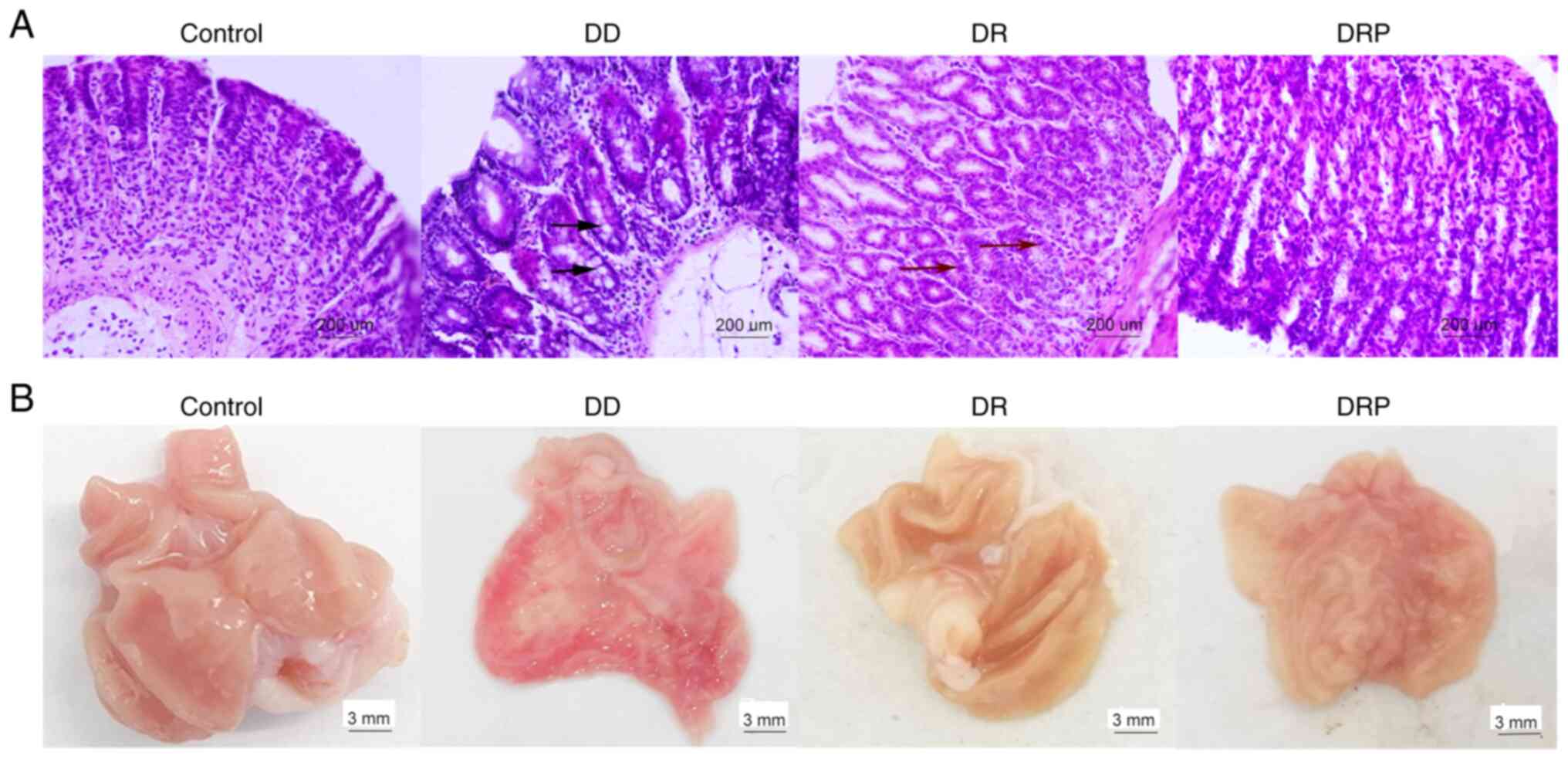
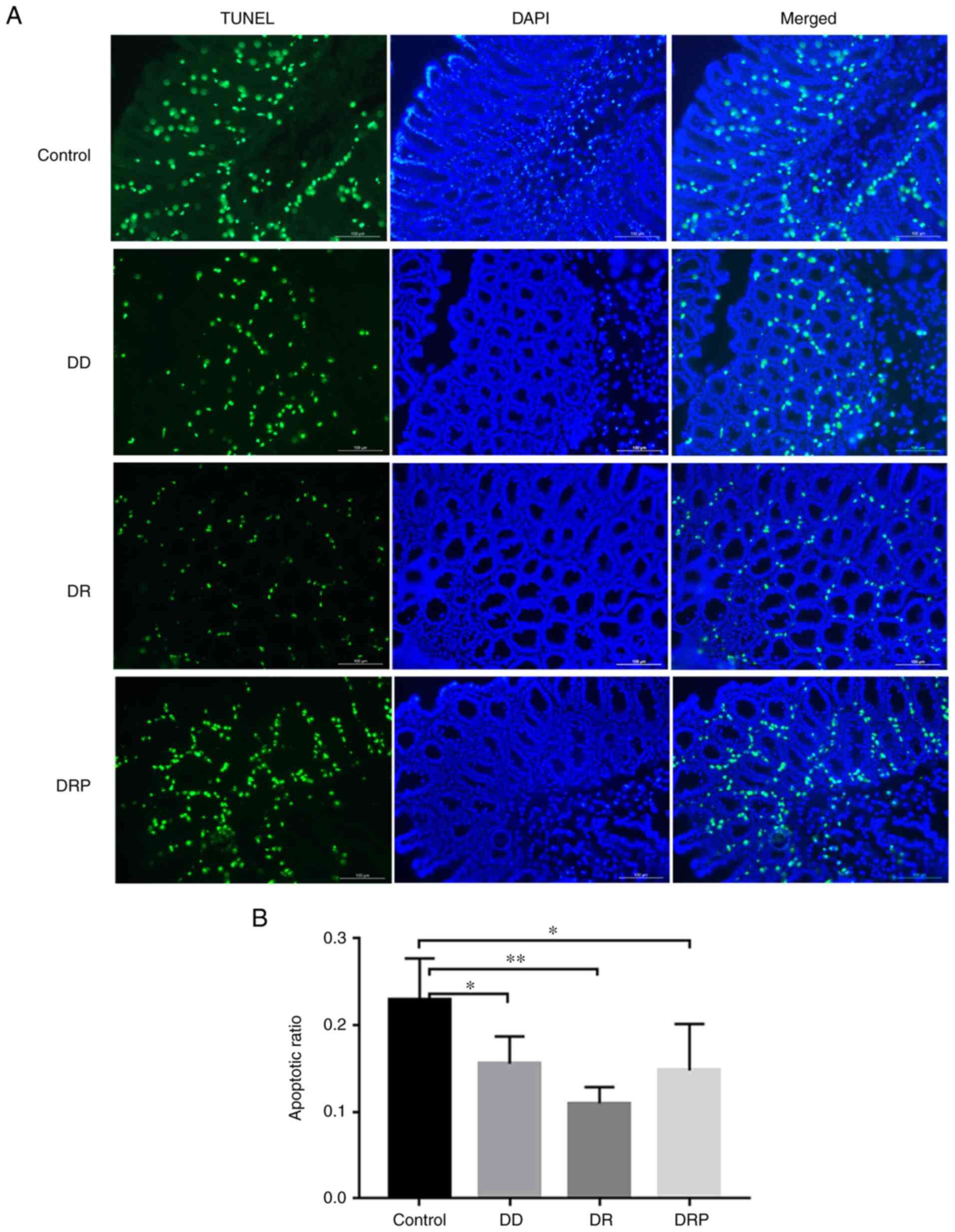
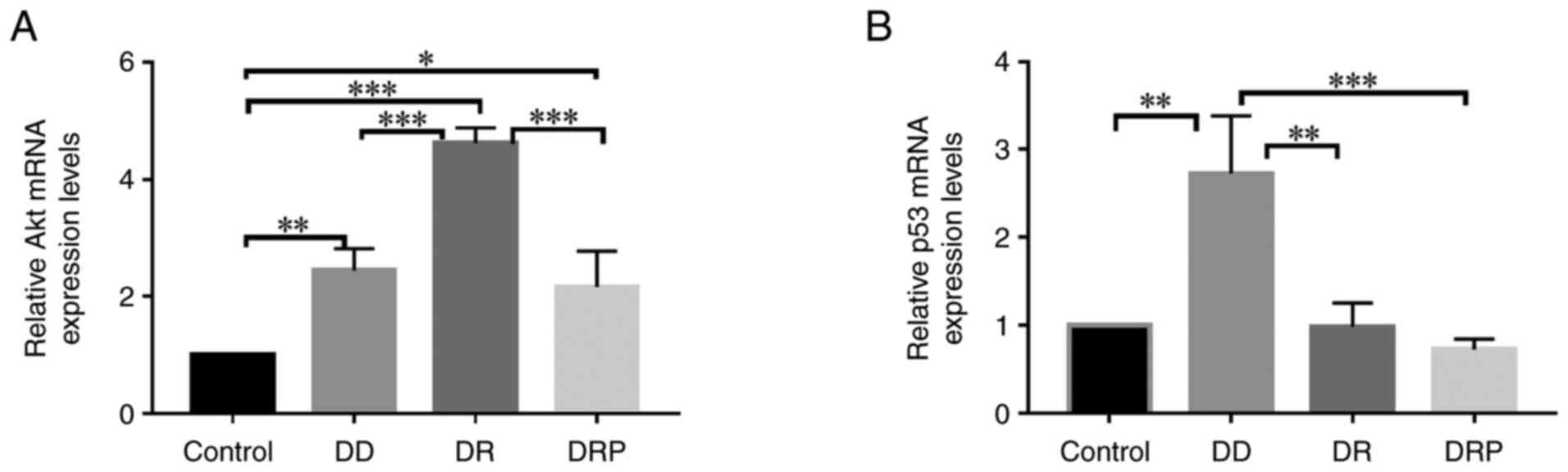
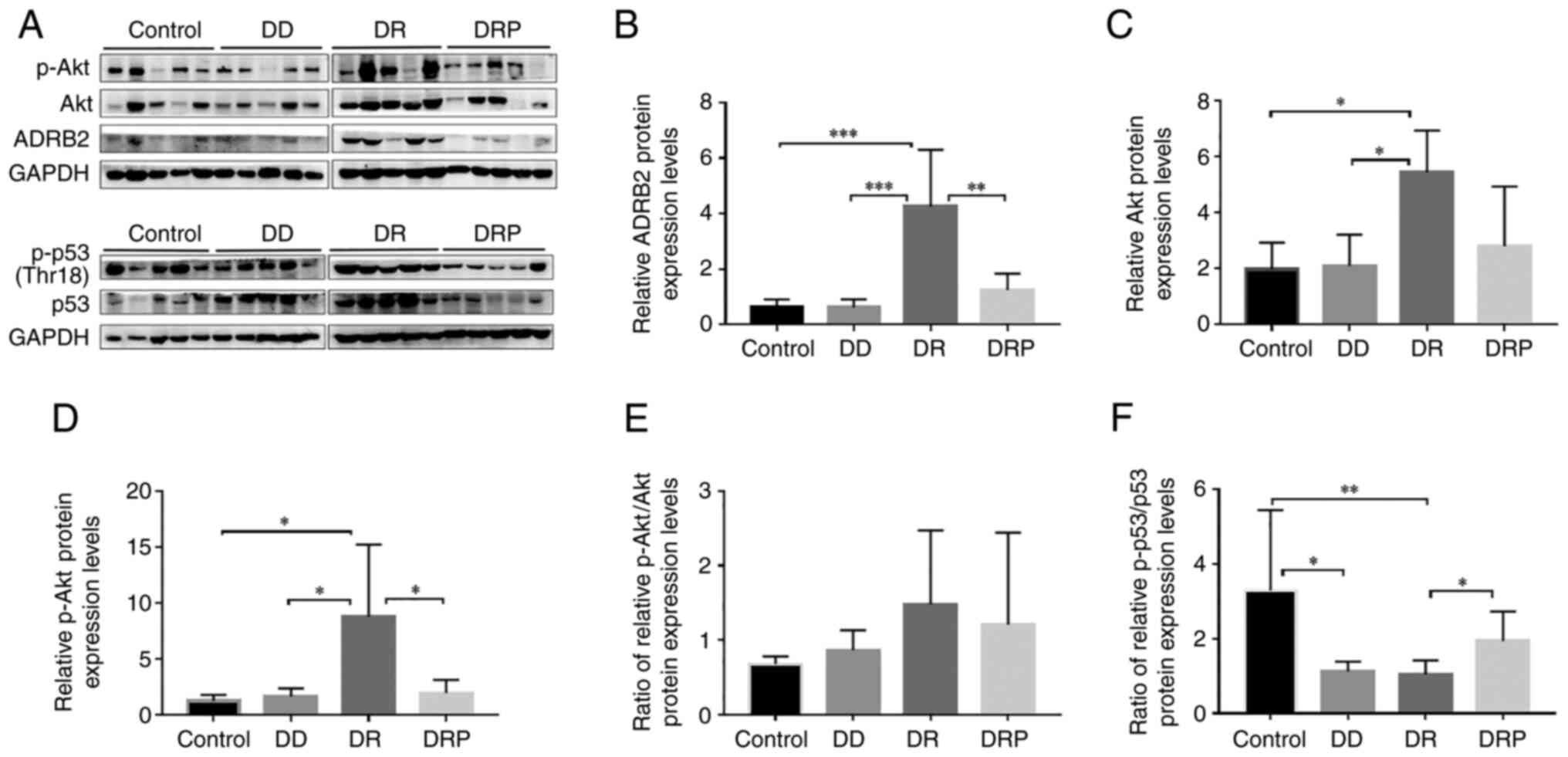
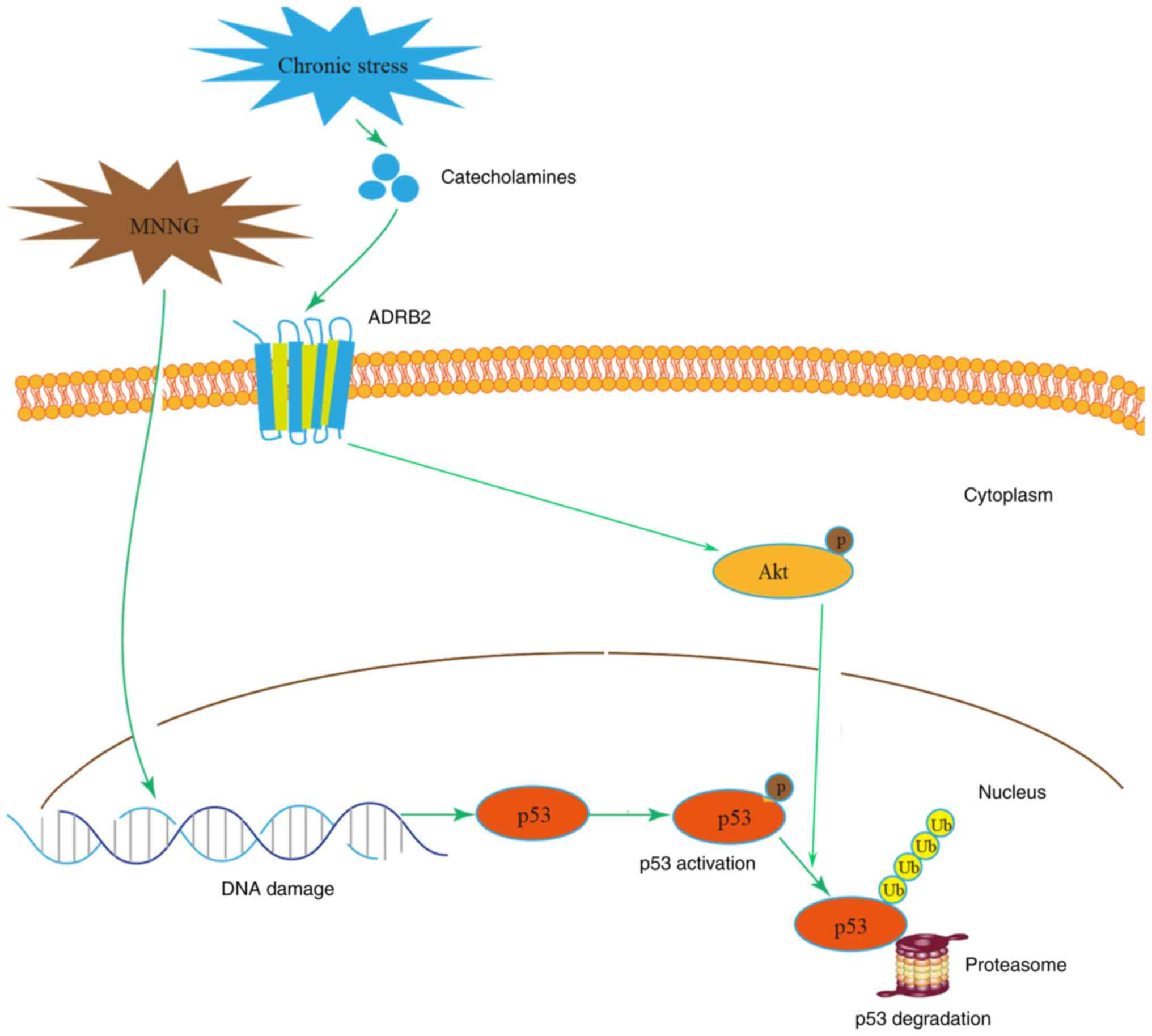
|
1
|
Thrift AP and El-Serag HB: Burden of
gastric cancer. Clin Gastroenterol Hepatol. 18:534–542. 2020.
View Article : Google Scholar
|
|
2
|
Ferlay J, Colombet M, Soerjomataram I,
Parkin DM, Piñeros M, Znaor A and Bray F: Cancer statistics for the
year 2020: An overview. Int J Cancer. Apr 5–2021.doi:
10.1002/ijc.33588 (Epub ahead of print). View Article : Google Scholar
|
|
3
|
Waldum HL, Kleveland PM and Sørdal ØF:
Helicobacter pylori and gastric acid: An intimate and
reciprocal relationship. Therap Adv Gastroenterol. 9:836–844. 2016.
View Article : Google Scholar
|
|
4
|
Loogna P, Franzén L, Sipponen P and
Domellöf L: Helicobacter pylori,
N-methyl-N'-nitro-N'-nitrosoguanidine, and bile modulate gastric
cell kinetics in experimental cancer. Virchows Arch. 439:653–660.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bagheri V, Memar B, Momtazi AA, Sahebkar
A, Gholamin M and Abbaszadegan MR: Cytokine networks and their
association with Helicobacter pylori infection in gastric
carcinoma. J Cell Physiol. 233:2791–2803. 2018. View Article : Google Scholar
|
|
6
|
Bekaii-Saab T and El-Rayes B: Identifying
and targeting cancer stem cells in the treatment of gastric cancer.
Cancer. 123:1303–1312. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Luo W, Fedda F, Lynch P and Tan D: CDH1
gene and hereditary diffuse gastric cancer syndrome: Molecular and
histological alterations and implications for diagnosis and
treatment. Front Pharmacol. 9:14212018. View Article : Google Scholar
|
|
8
|
Oliveira C, Pinheiro H, Figueiredo J,
Seruca R and Carneiro F: Familial gastric cancer: Genetic
susceptibility, pathology, and implications for management. Lancet
Oncol. 16:e60–e70. 2015. View Article : Google Scholar
|
|
9
|
Ohnishi S, Ma N, Thanan R, Pinlaor S,
Hammam O, Murata M and Kawanishi S: DNA damage in
inflammation-related carcinogenesis and cancer stem cells. Oxid Med
Cell Longev. 2013:3870142013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Costa L, Corre S, Michel V, Le Luel K,
Fernandes J, Ziveri J, Jouvion G, Danckaert A, Mouchet N, Da Silva
Barreira D, et al: USF1 defect drives p53 degradation during
Helicobacter pylori infection and accelerates gastric
carcinogenesis. Gut. 69:1582–1591. 2020. View Article : Google Scholar
|
|
11
|
Ushijima T, Nakajima T and Maekita T: DNA
methylation as a marker for the past and future. J Gastroenterol.
41:401–407. 2006. View Article : Google Scholar
|
|
12
|
Toyoda T, Tsukamoto T, Yamamoto M, Ban H,
Saito N, Takasu S, Shi L, Saito A, Ito S, Yamamura Y, et al: Gene
expression analysis of a Helicobacter pylori-infected and
high-salt diet-treated mouse gastric tumor model: Identification of
CD177 as a novel prognostic factor in patients with gastric cancer.
BMC Gastroenterol. 13:1222013. View Article : Google Scholar
|
|
13
|
Moreno-Villanueva M and Bürkle A: Stress
hormone-mediated DNA damage response-implications for cellular
senescence and tumour progression. Curr Drug Targets. 17:398–404.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Oren M: Decision making by p53: Life,
death and cancer. Cell Death Differ. 10:431–442. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hu W, Feng Z and Levine AJ: The regulation
of multiple p53 stress responses is mediated through MDM2. Genes
Cancer. 3:199–208. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Abraham AG and O'Neill E:
PI3K/Akt-mediated regulation of p53 in cancer. Biochem Soc Trans.
42:798–803. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bálint EE and Vousden KH: Activation and
activities of the p53 tumour suppressor protein. Br J Cancer.
85:1813–1823. 2001. View Article : Google Scholar
|
|
18
|
Tan BX, Liew HP, Chua JS, Ghadessy FJ, Tan
YS, Lane DP and Coffill CR: Anatomy of Mdm2 and Mdm4 in evolution.
J Mol Cell Biol. 9:3–15. 2017. View Article : Google Scholar
|
|
19
|
Wang Y, Sun H, Xiao Z, Zhang G, Zhang D,
Bao X, Li F, Wu S, Gao Y and Wei N: DNA damage and apoptosis
induced by a potent orally podophyllotoxin derivative in breast
cancer. Cell Commun Signal. 16:522018. View Article : Google Scholar
|
|
20
|
Zhu Y, Dai B, Zhang H, Shi G, Shen Y and
Ye D: Long non-coding RNA LOC572558 inhibits bladder cancer cell
proliferation and tumor growth by regulating the AKT-MDM2-p53
signaling axis. Cancer Lett. 380:369–374. 2016. View Article : Google Scholar
|
|
21
|
Chibaya L, Karim B, Zhang H and Jones SN:
Mdm2 phosphorylation by Akt regulates the p53 response to oxidative
stress to promote cell proliferation and tumorigenesis. Proc Natl
Acad Sci USA. 118:e20031931182021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang H, Park S, Zhang H, Park S, Kwon W,
Kim E, Zhang X, Jang S, Yoon D, Choi SK, et al: Targeting AKT with
costunolide suppresses the growth of colorectal cancer cells and
induces apoptosis in vitro and in vivo. J Exp Clin Cancer Res.
40:1142021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang X, Zhang Y, He Z, Yin K, Li B, Zhang
L and Xu Z: Chronic stress promotes gastric cancer progression and
metastasis: An essential role for ADRB2. Cell Death Dis.
10:7882019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Molina-Castro S, Pereira-Marques J,
Figueiredo C, Machado JC and Varon C: Gastric cancer: Basic
aspects. Helicobacter. 22 (Suppl 1):2017.doi: 10.1111/hel.12412.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Qiao G, Chen M, Bucsek MJ, Repasky EA and
Hylander BL: Adrenergic signaling: A targetable checkpoint limiting
development of the antitumor immune response. Front Immunol.
9:1642018. View Article : Google Scholar
|
|
26
|
Wohleb ES, Hanke ML, Corona AW, Powell ND,
Stiner LM, Bailey MT, Nelson RJ, Godbout JP and Sheridan JF:
β-Adrenergic receptor antagonism prevents anxiety-like behavior and
microglial reactivity induced by repeated social defeat. J
Neurosci. 31:6277–6288. 2011. View Article : Google Scholar
|
|
27
|
Sgambato D, Capuano A, Sullo MG, Miranda
A, Federico A and Romano M: Gut-brain axis in gastric mucosal
damage and protection. Curr Neuropharmacol. 14:959–966. 2016.
View Article : Google Scholar
|
|
28
|
Zhi X, Li B, Li Z, Zhang J, Yu J, Zhang L
and Xu Z: Adrenergic modulation of AMPK-dependent autophagy by
chronic stress enhances cell proliferation and survival in gastric
cancer. Int J Oncol. 54:1625–1638. 2019. View Article : Google Scholar
|
|
29
|
Feng Z, Liu L, Zhang C, Zheng T, Wang J,
Lin M, Zhao Y, Wang X, Levine AJ and Hu W: Chronic restraint stress
attenuates p53 function and promotes tumorigenesis. Proc Natl Acad
Sci USA. 109:7013–7018. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tang J, Li Z, Lu L and Cho CH:
β-Adrenergic system, a backstage manipulator regulating tumour
progression and drug target in cancer therapy. Semin Cancer Biol.
23:533–542. 2013. View Article : Google Scholar
|
|
31
|
Kim-Fuchs C, Le CP, Pimentel MA,
Shackleford D, Ferrari D, Angst E, Hollande F and Sloan EK: Chronic
stress accelerates pancreatic cancer growth and invasion: A
critical role for beta-adrenergic signaling in the pancreatic
microenvironment. Brain Behav Immun. 40:40–47. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Le CP, Nowell CJ, Kim-Fuchs C, Botteri E,
Hiller JG, Ismail H, Pimentel MA, Chai MG, Karnezis T, Rotmensz N,
et al: Chronic stress in mice remodels lymph vasculature to promote
tumour cell dissemination. Nat Commun. 7:106342016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Partecke LI, Speerforck S, Käding A,
Seubert F, Kühn S, Lorenz E, Schwandke S, Sendler M, Keßler W,
Trung DN, et al: Chronic stress increases experimental pancreatic
cancer growth, reduces survival and can be antagonised by
beta-adrenergic receptor blockade. Pancreatology. 16:423–433. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Thaker PH, Han LY, Kamat AA, Arevalo JM,
Takahashi R, Lu C, Jennings NB, Armaiz-Pena G, Bankson JA, Ravoori
M, et al: Chronic stress promotes tumor growth and angiogenesis in
a mouse model of ovarian carcinoma. Nat Med. 12:939–944. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lu YJ, Geng ZJ, Sun XY, Li YH, Fu XB, Zhao
XY and Wei B: Isoprenaline induces epithelial-mesenchymal
transition in gastric cancer cells. Mol Cell Biochem. 408:1–13.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shi M, Liu D, Duan H, Han C, Wei B, Qian
L, Chen C, Guo L, Hu M, Yu M, et al: Catecholamine up-regulates
MMP-7 expression by activating AP-1 and STAT3 in gastric cancer.
Mol Cancer. 9:2692010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Liang S, Wang T, Hu X, Luo J, Li W, Wu X,
Duan Y and Jin F: Administration of Lactobacillus helveticus NS8
improves behavioral, cognitive, and biochemical aberrations caused
by chronic restraint stress. Neuroscience. 310:561–577. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Naert G, Ixart G, Maurice T,
Tapia-Arancibia L and Givalois L: Brain-derived neurotrophic factor
and hypothalamic–pituitary–adrenal axis adaptation processes in a
depressive-like state induced by chronic restraint stress. Mol Cell
Neurosci. 46:55–66. 2011. View Article : Google Scholar
|
|
39
|
Huang T, Xiao Y, Zhang Y, Wang C, Chen X,
Li Y, Ge Y and Gao J: miR-223 ameliorates thalamus
hemorrhage-induced central poststroke pain via targeting NLRP3 in a
mouse model. Exp Ther Med. 23:3532022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Murata I, Sugai T, Murakawa Y, Miyamoto Y,
Kobayashi J, Inoue Y and Kanamoto I: Salvianolic acid B improves
the survival rate, acute kidney dysfunction, inflammation and
NETosis-mediated antibacterial action in a crush syndrome rat
model. Exp Ther Med. 23:3202022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Dixon MF, Genta RM, Yardley JH and Correa
P: Classification and grading of gastritis. The updated Sydney
system. International workshop on the histopathology of gastritis,
Houston 1994. Am J Surg Pathol. 20:1161–1181. 1996. View Article : Google Scholar
|
|
42
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Gunathilake M, Lee J, Choi IJ, Kim YI and
Kim J: Association between bacteria other than Helicobacter
pylori and the risk of gastric cancer. Helicobacter.
26:e128362021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Machlowska J, Baj J, Sitarz M, Maciejewski
R and Sitarz R: Gastric cancer: Epidemiology, risk factors,
classification, genomic characteristics and treatment strategies.
Int J Mol Sci. 21:40122020. View Article : Google Scholar
|
|
45
|
Blair VR, McLeod M, Carneiro F, Coit DG,
D'Addario JL, van Dieren JM, Harris KL, Hoogerbrugge N, Oliveira C,
van der Post RS, et al: Hereditary diffuse gastric cancer: Updated
clinical practice guidelines. Lancet Oncol. 21:e386–e397. 2020.
View Article : Google Scholar
|
|
46
|
Kuligowski J, Sanjuan-Herraez D,
Vázquez-Sánchez MA, Brunet-Vega A, Pericay C, Ramírez-Lázaro MJ,
Lario S, Gombau L, Junquera F, Calvet X and Quintás G: Metabolomic
analysis of gastric cancer progression within the Correa's cascade
using ultraperformance liquid chromatography-mass spectrometry. J
Proteome Res. 15:2729–2738. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Han F, Li CF, Cai Z, Zhang X, Jin G, Zhang
WN, Xu C, Wang CY, Morrow J, Zhang S, et al: The critical role of
AMPK in driving Akt activation under stress, tumorigenesis and drug
resistance. Nat Commun. 9:47282018. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Sun X, Chen L and He Z: PI3K/Akt-Nrf2 and
Anti-inflammation effect of macrolides in chronic obstructive
pulmonary disease. Curr Drug Metabolism. 20:301–304. 2019.
View Article : Google Scholar
|
|
49
|
Zhao Y, Hu X, Liu Y, Dong S, Wen Z, He W,
Zhang S, Huang Q and Shi M: ROS signaling under metabolic stress:
Cross-talk between AMPK and AKT pathway. Mol Cancer. 16:792017.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kastenhuber ER and Lowe SW: Putting p53 in
context. Cell. 170:1062–1078. 2017. View Article : Google Scholar
|
|
51
|
Lee JT and Gu W: The multiple levels of
regulation by p53 ubiquitination. Cell Death Differ. 17:86–92.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Liu Y, Deisenroth C and Zhang Y:
RP-MDM2-p53 pathway: Linking ribosomal biogenesis and tumor
surveillance. Trends Cancer. 2:191–204. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Manning BD and Toker A: AKT/PKB signaling:
Navigating the network. Cell. 169:381–405. 2017. View Article : Google Scholar
|
|
54
|
Agac D, Estrada LD, Maples R, Hooper LV
and Farrar JD: The beta2-adrenergic receptor controls inflammation
by driving rapid IL-10 secretion. Brain BehavImmun. 74:176–185.
2018. View Article : Google Scholar
|
|
55
|
Choy C, Raytis JL, Smith DD, Duenas M,
Neman J, Jandial R and Lew MW: Inhibition of β2-adrenergic receptor
reduces triple-negative breast cancer brain metastases: The
potential benefit of perioperative beta-blockade. Oncol Rep.
35:3135–3142. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Matheis F, Muller PA, Graves CL, Gabanyi
I, Kerner ZJ, Costa-Borges D, Ahrends T, Rosenstiel P and Mucida D:
Adrenergic signaling in muscularis macrophages limits
infection-induced neuronal loss. Cell. 180:64–78.e16. 2020.
View Article : Google Scholar
|
|
57
|
Mohammadpour H, MacDonald CR, Qiao G, Chen
M, Dong B, Hylander BL, McCarthy PL, Abrams SI and Repasky EA: β2
adrenergic receptor-mediated signaling regulates the
immunosuppressive potential of myeloid-derived suppressor cells. J
Clin Invest. 129:5537–5552. 2019. View Article : Google Scholar
|
|
58
|
Nakai A and Suzuki K: Adrenergic control
of lymphocyte trafficking and adaptive immune responses. Neurochem
Int. 130:1043202019. View Article : Google Scholar
|
|
59
|
Wirth K and Scheibenbogen C: A unifying
hypothesis of the pathophysiology of myalgic
Encephalomyelitis/Chronic fatigue syndrome (ME/CFS): Recognitions
from the finding of autoantibodies against ss2-adrenergic
receptors. Autoimmun Rev. 19:1025272020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Ohyama W, Okada E, Fujiishi Y, Narumi K
and Yasutake N: In vivo rat glandular stomach and colon
micronucleus tests: Kinetics of micronucleated cells, apoptosis,
and cell proliferation in the target tissues after a single oral
administration of stomach- or colon-carcinogens. Mutat Res.
755:141–147. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Cai T, Zhang C, Zhao Z, Li S, Cai H, Chen
X, Cai D, Liu W, Yan Y, Xie K, et al: The gastric mucosal
protective effects of Astragaloside IV in mnng-induced GPL rats.
Biomed Pharmacother. 104:291–299. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Crabtree JE, Jeremy AH, Duval C, Dixon MF,
Danjo K, Carr IM, Pritchard DM and Robinson PA: Effects of EGFR
inhibitor on Helicobacter pylori induced gastric epithelial
pathology in vivo. Pathogens. 2:571–590. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Kidd M, Tang LH, Modlin IM, Zhang T, Chin
K, Holt PR and Moss SF: Gastrin-mediated alterations in gastric
epithelial apoptosis and proliferation in a mastomys rodent model
of gastric neoplasia. Digestion. 62:143–151. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Scotiniotis IA, Rokkas T, Furth EE, Rigas
B and Shiff SJ: Altered gastric epithelial cell kinetics in
Helicobacter pylori-associated intestinal metaplasia:
Implications for gastric carcinogenesis. Int J Cancer. 85:192–200.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Lipshitz HD, Claycomb JM and Smibert CA:
Post-transcriptional regulation of gene expression. Methods.
126:1–2. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Pu M, Chen J, Tao Z, Miao L, Qi X, Wang Y
and Ren J: Regulatory network of miRNA on its target: Coordination
between transcriptional and post-transcriptional regulation of gene
expression. Cell Mol Life Sci. 76:441–451. 2019. View Article : Google Scholar
|
|
67
|
Fang MZ, Mar W and Cho MH: Cadmium affects
genes involved in growth regulation during two-stage transformation
of Balb/3T3 cells. Toxicology. 177:253–265. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Cross B, Chen L, Cheng Q, Li B, Yuan ZM
and Chen J: Inhibition of p53 DNA binding function by the MDM2
protein acidic domain. J Biol Chem. 286:16018–16029. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Li J and Kurokawa M: Regulation of MDM2
stability after DNA damage. J Cell Physiol. 230:2318–2327. 2015.
View Article : Google Scholar
|
|
70
|
Hara MR, Kovacs JJ, Whalen EJ, Rajagopal
S, Strachan RT, Grant W, Towers AJ, Williams B, Lam CM, Xiao K, et
al: A stress response pathway regulates DNA damage through
beta2-adrenoreceptors and β-arrestin-1. Nature. 477:349–353. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Hara MR, Sachs BD, Caron MG and Lefkowitz
RJ: Pharmacological blockade of a β(2)AR-β-arrestin-1 signaling
cascade prevents the accumulation of DNA damage in a behavioral
stress model. Cell Cycle. 12:219–224. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Meek DW: Regulation of the p53 response
and its relationship to cancer. Biochem J. 469:325–346. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Chen H, Zhang W, Cheng X, Guo L, Xie S, Ma
Y, Guo N and Shi M: β2-AR activation induces chemoresistance by
modulating p53 acetylation through upregulating Sirt1 in cervical
cancer cells. Cancer Sci. 108:1310–1317. 2017. View Article : Google Scholar
|
|
74
|
Yang G, Zhang G, Pittelkow MR, Ramoni M
and Tsao H: Expression profiling of UVB response in melanocytes
identifies a set of p53-target genes. J Invest Dermatol.
126:2490–2506. 2006. View Article : Google Scholar
|
|
75
|
Berkelman T: Fluorescent western blotting:
High sensitivity detection of multiple targets. Curr Protoc
Pharmacol. 88:e722020. View Article : Google Scholar
|
|
76
|
Gopal A and Herr AE: Multiplexed in-gel
microfluidic immunoassays: Characterizing protein target loss
during reprobing of benzophenone-modified hydrogels. Sci Rep.
9:153892019. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Litovchick L: Stripping of the immunoblot
for reprobing. Cold Spring Harb Protoc. 2020:0984912020. View Article : Google Scholar
|
|
78
|
Sennepin AD, Charpentier S, Normand T,
Sarre C, Legrand A and Mollet LM: Multiple reprobing of Western
blots after inactivation of peroxidase activity by its substrate,
hydrogen peroxide. Anal Biochem. 393:129–131. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Ferguson RE, Carroll HP, Harris A, Maher
ER, Selby PJ and Banks RE: Housekeeping proteins: A preliminary
study illustrating some limitations as useful references in protein
expression studies. Proteomics. 5:566–571. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Ruan W and Lai M: Actin, a reliable marker
of internal control? Clin Chim Acta. 385:1–5. 2007. View Article : Google Scholar
|
|
81
|
Hotamisligil GS and Davis RJ: Cell
signaling and stress responses. Cold Spring Harb Perspect Biol.
8:a0060722016. View Article : Google Scholar
|
|
82
|
Wang P, Hao X, Li X, Yan Y, Tian W, Xiao
L, Wang Z and Dong J: Curcumin inhibits adverse psychological
stress-induced proliferation and invasion of glioma cells via
down-regulating the ERK/MAPK pathway. J Cell Mol Med. 25:7190–7203.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Yong HY, Koh MS and Moon A: The p38 MAPK
inhibitors for the treatment of inflammatory diseases and cancer.
Expert Opin Investig Drugs. 18:1893–1905. 2009. View Article : Google Scholar : PubMed/NCBI
|