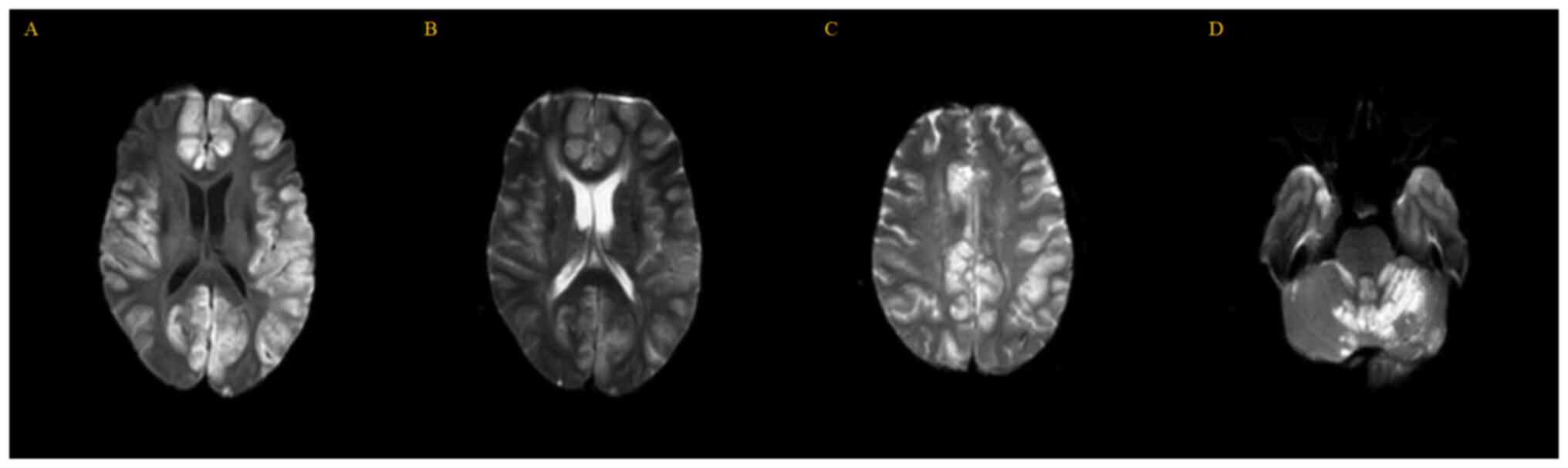
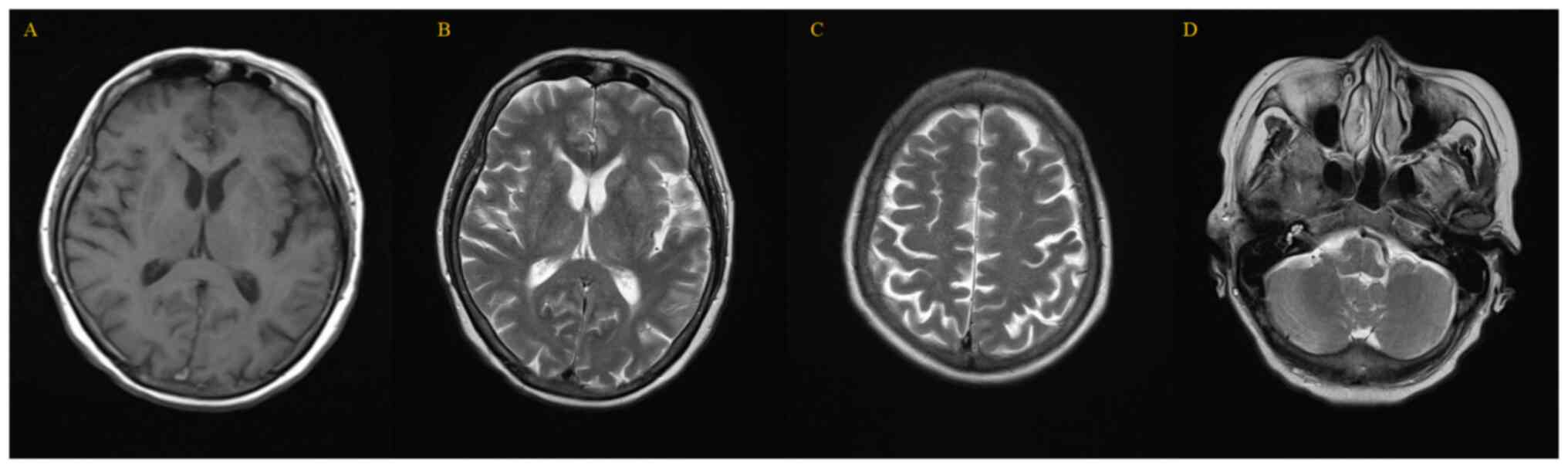
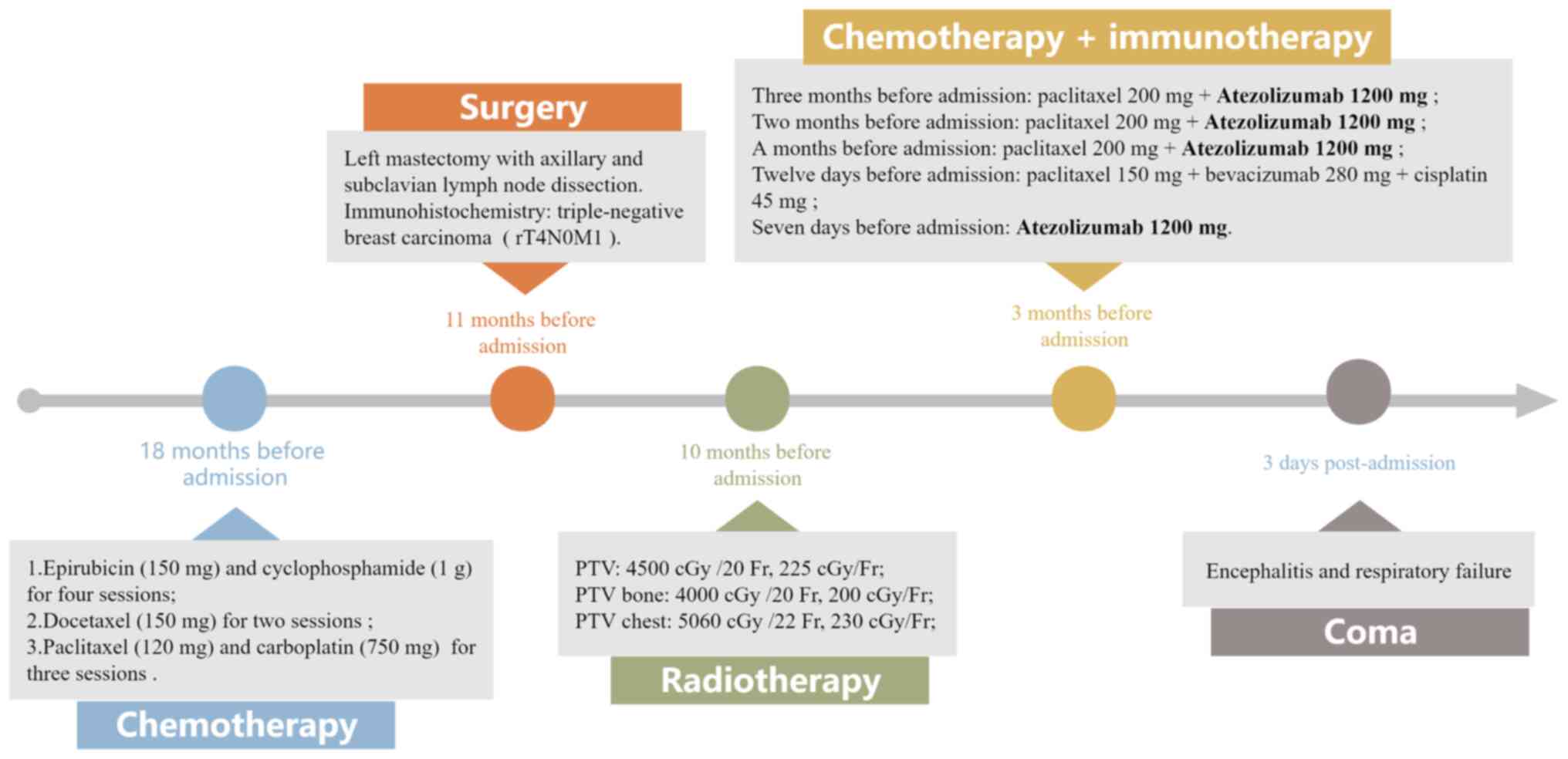
|
1
|
Topalian SL, Taube JM and Pardoll DM:
Neoadjuvant checkpoint blockade for cancer immunotherapy. Science.
367:eaax01822020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ribas A and Wolchok JD: Cancer
immunotherapy using checkpoint blockade. Science. 359:1350–1355.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Möhn N, Beutel G, Gutzmer R, Ivanyi P,
Satzger I and Skripuletz T: Neurological immune related adverse
events associated with nivolumab, ipilimumab, and pembrolizumab
therapy-review of the literature and future outlook. J Clin Med.
8:17772019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pan PC and Haggiagi A: Neurologic
immune-related adverse events associated with immune checkpoint
inhibition. Curr Oncol Rep. 21:1082019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Darnell EP, Mooradian MJ, Baruch EN,
Yilmaz M and Reynolds KL: Immune-related adverse events (irAEs):
Diagnosis, management, and clinical pearls. Curr Oncol Rep.
22:392020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Williams TJ, Benavides DR, Patrice KA,
Dalmau JO, de Ávila AL, Le DT, Lipson EJ, Probasco JC and Mowry EM:
Association of autoimmune encephalitis with combined immune
checkpoint inhibitor treatment for metastatic cancer. JAMA Neurol.
73:928–933. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Levine JJ, Somer RA, Hosoya H and
Squillante C: Atezolizumab-induced encephalitis in metastatic
bladder cancer: A case report and review of the literature. Clin
Genitourin Cancer. 15:e847–e849. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Laserna A, Tummala S, Patel N, El Hamouda
DEM and Gutiérrez C: Atezolizumab-related encephalitis in the
intensive care unit: Case report and review of the literature. SAGE
Open Med Case Rep. 6:2050313X187924222018.PubMed/NCBI
|
|
9
|
Arakawa M, Yamazaki M, Toda Y, Saito R,
Ozawa A, Kosaihira S and Kimura K: Atezolizumab-induced
encephalitis in metastatic lung cancer: A case report and
literature review. eNeurologicalSci. 14:49–50. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Robert L, Langner-Lemercier S, Angibaud A,
Sale A, Thepault F, Corre R, Lena H and Ricordel C: Immune-related
encephalitis in two patients treated with immune checkpoint
inhibitor. Clin Lung Cancer. 21:e474–e477. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yamaguchi Y, Nagasawa H, Katagiri Y and
Wada M: Atezolizumab-associated encephalitis in metastatic lung
adenocarcinoma: A case report. J Med Case Rep. 14:882020.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tatsumi S, Uryu K, Iwasaki S and Harada H:
A case of anti-CRMP5 paraneoplastic neurological syndrome induced
by atezolizumab for small cell lung cancer. Intern Med. Aug
12–2020.(Epub ahead of print).
|
|
13
|
Nader R, Tannoury E, Rizk T and Ghanem H:
Atezolizumab-induced encephalitis in a patient with metastatic
breast cancer: A case report and review of neurological adverse
events associated with checkpoint inhibitors. Autops Case Rep.
11:e20212612021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Özdirik B, Jost-Brinkmann F, Savic LJ,
Mohr R, Tacke F, Ploner CJ, Roderburg C and Müller T: Atezolizumab
and bevacizumab-induced encephalitis in advanced hepatocellular
carcinoma: Case report and literature review. Medicine (Baltimore).
100:e263772021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nishijima H, Kon T, Seino Y, Yagihashi N,
Suzuki C, Nakamura T, Tanaka H, Sakamoto Y, Wakabayashi K and
Tomiyama M: Bilateral thalamic lesions associated with
atezolizumab-induced encephalitis: A follow-up report with autopsy
findings. Neurology. 98:204–205. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brahmer JR, Lacchetti C, Schneider BJ,
Atkins MB, Brassil KJ, Caterino JM, Chau I, Ernstof MS, Gardner JM,
Ginex P, et al: Management of immune-related adverse events in
patients treated with immune checkpoint inhibitor therapy: American
society of clinical oncology clinical practice guideline. J Clin
Oncol. 36:1714–1768. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Velasco R, Villagrán M, Jové M, Simó M,
Vilariño N, Alemany M, Palmero R, Martínez-Villacampa MM, Nadal E
and Bruna J: Encephalitis induced by immune checkpoint inhibitors:
A systematic review. JAMA Neurol. 78:864–873. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Giuliano AE, Edge SB and Hortobagyi GN:
Eighth edition of the AJCC cancer staging manual: Breast cancer.
Ann Surg Oncol. 25:1783–1785. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mehta R GP trainee and Chinthapalli K
consultant neurologist: Glasgow coma scale explained. BMJ.
365:l12962019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Trinh S, Le A, Gowani S and La-Beck N:
Management of immune-related adverse events associated with immune
checkpoint inhibitor therapy: A minireview of current clinical
guidelines. Asia Pac J Oncol Nurs. 6:154–160. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Duong SL, Barbiero FJ, Nowak RJ and
Baehring JM: Neurotoxicities associated with immune checkpoint
inhibitor therapy. J Neurooncol. 152:265–277. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Haanen JB, Carbonnel F, Robert C, Kerr KM,
Peters S, Larkin J and Jordan K; ESMO Guidelines Committee, :
Management of toxicities from immunotherapy: ESMO clinical practice
guidelines for diagnosis, treatment and follow-up. Ann Oncol. 28
(Suppl 4):iv119–iv142. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Touat M, Talmasov D, Ricard D and Psimaras
D: Neurological toxicities associated with immune-checkpoint
inhibitors. Curr Opin Neurol. 30:659–668. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Vogrig A, Muñiz-Castrillo S, Joubert B,
Picard G, Rogemond V, Marchal C, Chiappa AM, Chanson E, Skowron F,
Leblanc A, et al: Central nervous system complications associated
with immune checkpoint inhibitors. J Neurol Neurosurg Psychiatry.
91:772–778. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schmid P, Rugo HS, Adams S, Schneeweiss A,
Barrios CH, Iwata H, Diéras V, Henschel V, Molinero L, Chui SY, et
al: Atezolizumab plus nab-paclitaxel as first-line treatment for
unresectable, locally advanced or metastatic triple-negative breast
cancer (IMpassion130): Updated efficacy results from a randomised,
double-blind, placebo-controlled, phase 3 trial. Lancet Oncol.
21:44–59. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Das S and Johnson DB: Immune-related
adverse events and anti-tumor efficacy of immune checkpoint
inhibitors. J Immunother Cancer. 7:3062019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Arriola E, Wheater M, Galea I, Cross N,
Maishman T, Hamid D, Stanton L, Cave J, Geldart T, Mulatero C, et
al: Outcome and biomarker analysis from a multicenter phase 2 study
of ipilimumab in combination with carboplatin and etoposide as
first-line therapy for extensive-stage SCLC. J Thorac Oncol.
11:1511–1521. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kumar V, Chaudhary N, Garg M, Floudas CS,
Soni P and Chandra AB: Current diagnosis and management of immune
related adverse events (irAEs) induced by immune checkpoint
inhibitor therapy. Front Pharmacol. 8:492017. View Article : Google Scholar : PubMed/NCBI
|