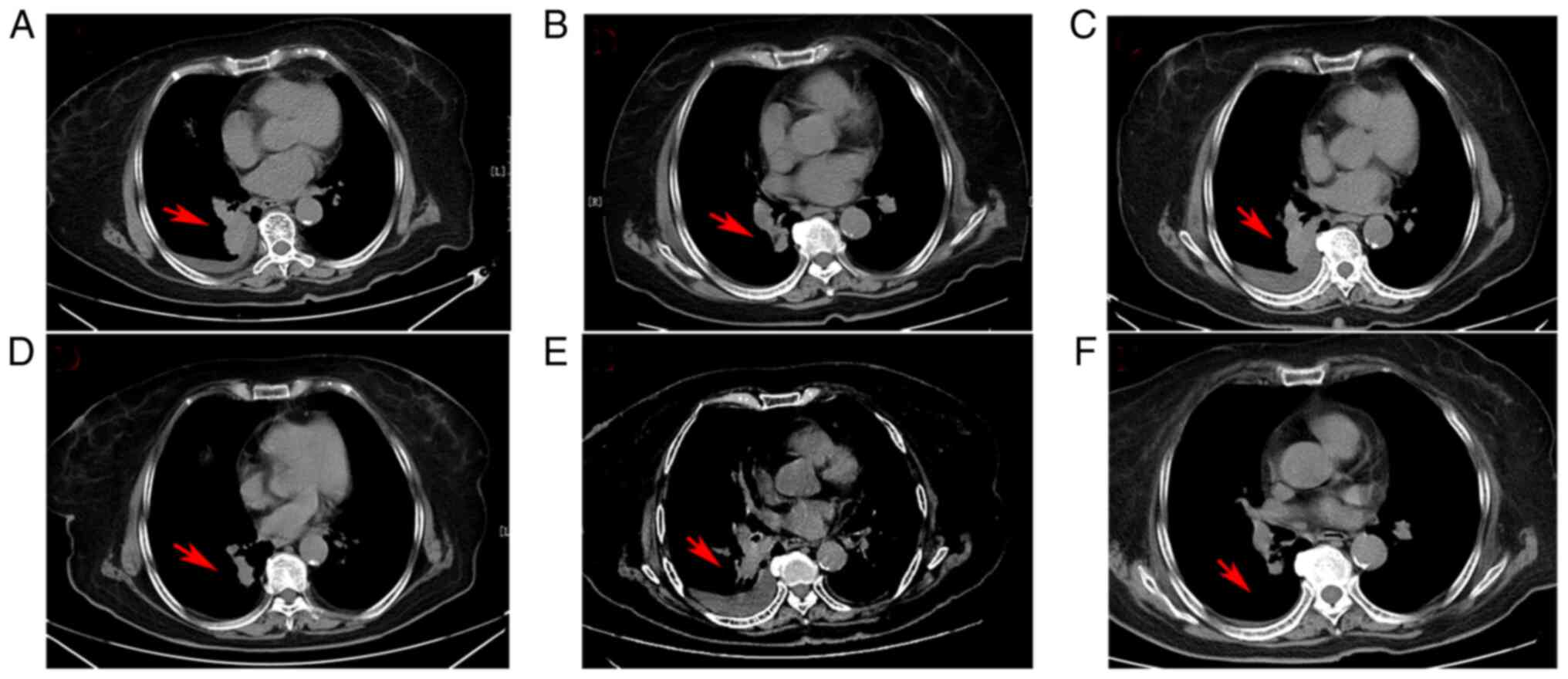
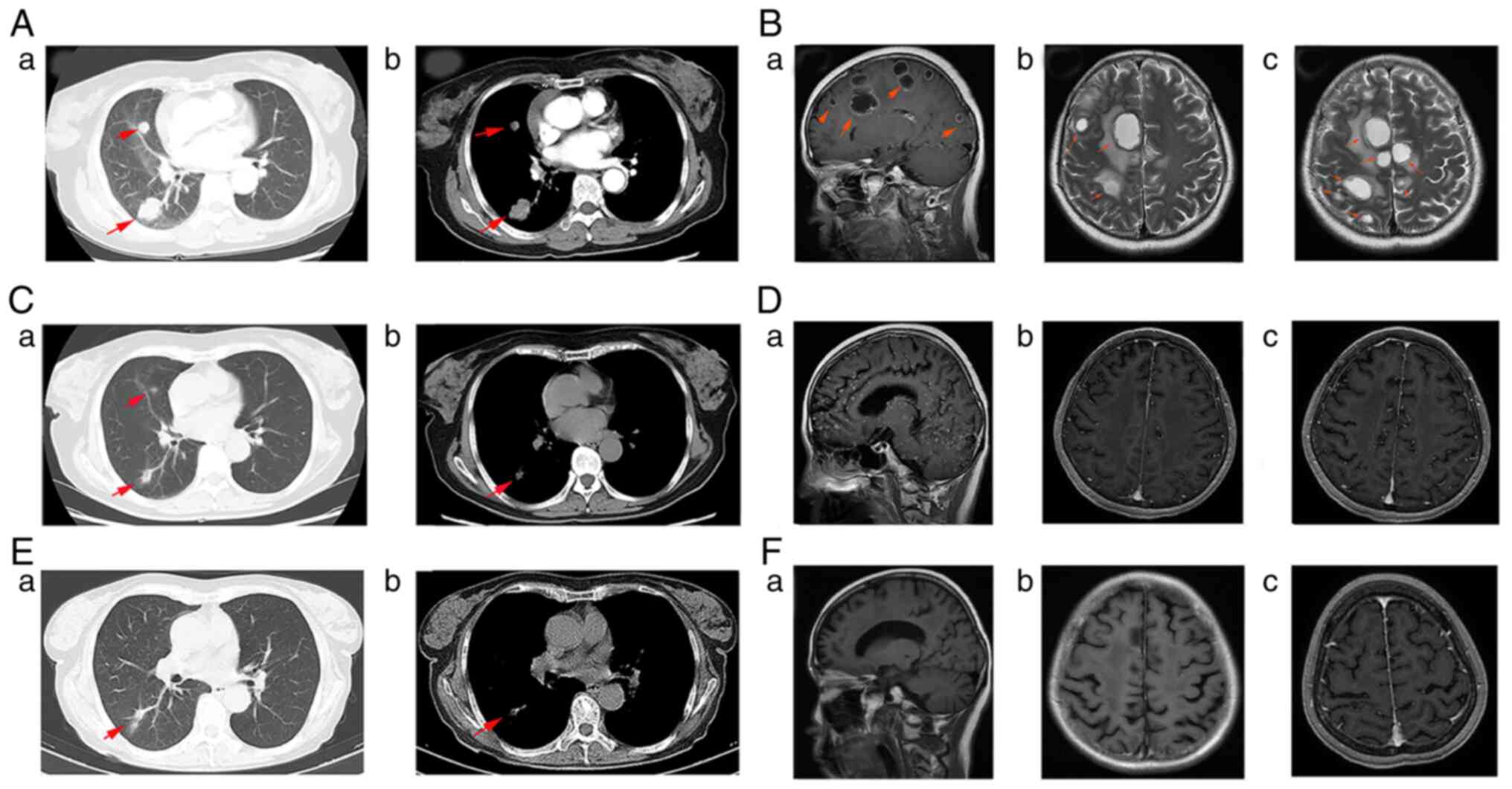
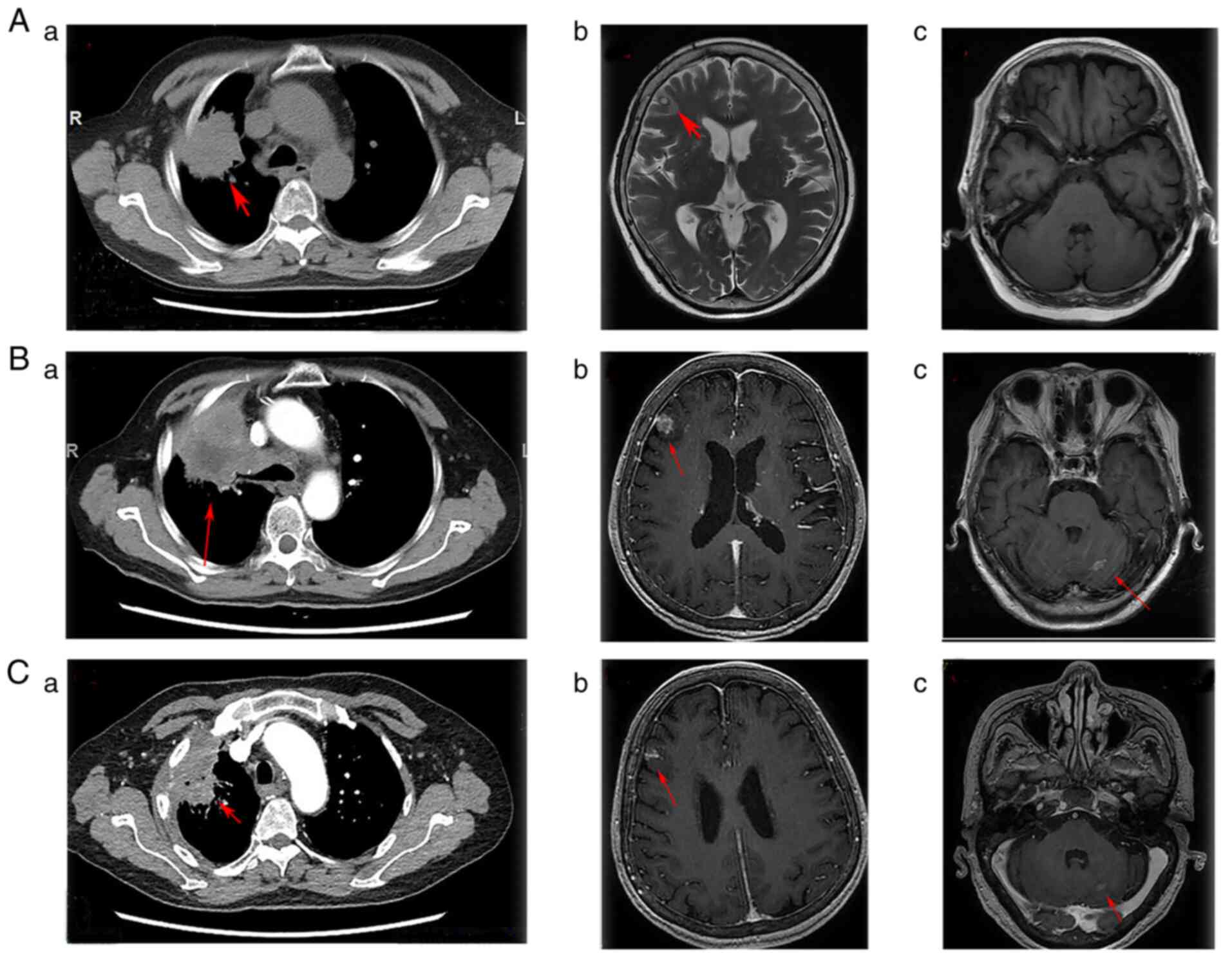
|
1
|
He J, Huang Z, Han L, Gong Y and Xie C:
Mechanisms and management of 3rd-generation EGFR-TKI resistance in
advanced non-small cell lung cancer (Review). Int J Oncol.
59:902021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Soria JC, Ohe Y, Vansteenkiste J,
Reungwetwattana T, Chewaskulyong B, Lee KH, Dechaphunkul A, Imamura
F, Nogami N, Kurata T, et al: Osimertinib in Untreated EGFR-Mutated
Advanced non-small-cell lung cancer. N Engl J Med. 378:113–125.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schmid S, Li JJN and Leighl NB: Mechanisms
of osimertinib resistance and emerging treatment options. Lung
Cancer. 147:123–129. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lu S, Dong X, Jian H, Chen J, Chen G, Sun
Y, Ji Y, Wang Z, Shi J, Lu J, et al: AENEAS: A randomized phase III
trial of aumolertinib versus gefitinib as first-line therapy for
locally advanced or metastaticnon-small-cell lung cancer with EGFR
Exon 19 Deletion or L858R Mutations. J Clin Oncol. Sep
20–2022.(Epub ahead of print).
|
|
5
|
Deeks ED: Furmonertinib: First Approval.
Drugs. 81:1775–1780. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jian H, Wang K, Cheng Y, Ding L, Wang Y,
Shi Z, Zhang L, Wang Y and Lu S: Phase I trial of a third
generation EGFR mutant-selective inhibitor (D-0316) in patients
with advanced non-small cell lung cancer. Oncologist. 27:163–e213.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lv W, Cheng H, Shao D, Wei Y, Zhu W, Wu K,
Jiang W, Hu L, Sha Z, Zhong B and Pei X: Treatment patterns and
survival of patients with advanced non-small cell lung cancer
guided by comprehensive genomic profiling: Real-World
single-institute study in China. Front Oncol. 11:6307172021.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chang Y, Liu S, Jiang Y, Hua L and Wen L:
Effective treatment of pulmonary adenocarcinoma harboring triple
EGFR mutations of L858R, T790M, cis-G796s/cis-C797s by osimertinib,
brigatinib, and bevacizumab combination therapy: A case report.
Respir Med Case Rep. 36:1015822022.PubMed/NCBI
|
|
9
|
Wang Y, Han R, Zhu M, He T and He Y: Case
report: Durable response to the combination of brigatinib and
cetuximab plus icotinib in a NSCLC patient harboring EGFR
L858R-T790M-cis-G796S and L718Q resistance mutations following
progression with osimertinib. Front Oncol. 12:8753132022.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang F, Markovic SN, Molina JR,
Halfdanarson TR, Pagliaro LC, Chintakuntlawar AV, Li R, Wei J, Wang
L, Liu B, et al: Association of sex, age, and eastern cooperative
oncology group performance status with survival benefit of cancer
immunotherapy in randomized clinical trials: A systematic review
and meta-analysis. JAMA Netw Open. 3:e20125342020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chansky K, Detterbeck FC, Nicholson AG,
Rusch VW, Vallieres E, Groome P, Kennedy C, Krasnik M, Peake M,
Shemanski L, et al: The IASLC lung cancer staging project: External
validation of the revision of the TNM stage groupings in the eighth
edition of the TNM classification of lung cancer. J Thorac Oncol.
12:1109–1121. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fang C, Liu T, Liang W, Feng S, Su Z, Tang
H, Huang H and Chen Z: Clinical pharmacist participation in
selecting and dosing targeted drugs for a patient with ALK-positive
non-small cell lung cancer: A case report. Ann Transl Med.
9:14882021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ma P, Huang R, Gu Y, Fang Y, Wu X, Chen
DS, Zhang HW, Gao W and Shu Y: Savolitinib monotherapy exerted
significant benefit in a non-small cell lung cancer patient with
osimertinib resistance harboring primary EGFR L858R mutation and
MET amplification: A case report. Anticancer Drugs. Aug
10–2022.(Epub ahead of print). View Article : Google Scholar
|
|
14
|
Lim JU: Overcoming osimertinib resistance
in advanced non-small cell lung cancer. Clin Oncol (R Coll Radiol).
33:619–626. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cui Q, Hu Y, Cui Q, Wu D, Mao Y, Ma D and
Liu H: Osimertinib rechallenge with bevacizumab vs. chemotherapy
plus bevacizumab in EGFR-Mutant NSCLC patients with osimertinib
resistance. Front Pharmacol. 12:7467072021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Satoh H and Kagohashi K: Response to
erlotinib and bevacizumab combination therapy after acquired
resistance to osimertinib in patients with non-small cell lung
cancer. Anticancer Drugs. 33:320–322. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lu S, Wang Q, Zhang G, Dong X, Yang CT,
Song Y, Chang GC, Lu Y, Pan H, Chiu CH, et al: Efficacy of
Aumolertinib (HS-10296) in Patients With Advanced EGFR T790M+
NSCLC: Updated post-national medical products administration
approval results from the APOLLO registrational trial. J Thorac
Oncol. 17:411–422. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang Y, Zhang Y, Niu W, Ge X, Huang F,
Pang J, Li X, Wang Y, Gao W, Fan F, et al: Experimental study of
almonertinib crossing the blood-brain barrier in EGFR-Mutant NSCLC
brain metastasis and spinal cord metastasis models. Front
Pharmacol. 12:7500312021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yang F, Zhang W, Shang X, Liu N, Ma X, Qin
J, Zhang Y, Liu Y and Wang X: Comparison of the efficacy and safety
of first-line treatments based on clinicopathological
characteristics for patients with advanced epidermal growth factor
receptor mutated non-small-cell lung cancer: A systematic review
and network meta-analysis. Crit Rev Oncol Hematol. 177:1037602022.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Shen G, Shi L, Tian X, Huang D, Chen H,
Gao C, Shen X and Zhang H: Case report: Response to almonertinib in
a patient with metastatic NSCLC resistant to osimertinib due to
acquired EGFR L718Q Mutation. Front Pharmacol. 12:7318952021.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wu L, Zhong W, Li A, Qiu Z, Xie R, Shi H
and Lu S: Successful treatment of EGFR T790M-mutant non-small cell
lung cancer with almonertinib after osimertinib-induced
interstitial lung disease: A case report and literature review. Ann
Transl Med. 9:9502021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang Q, Liu H and Yang J: Aumolertinib
effectively reduces clinical symptoms of an EGFR L858R-Mutant
non-small cell lung cancer case coupled with osimertinib-induced
carditoxicity: Case report and review. Front Endocrinol.
13:8339292022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yang JC, Camidge DR, Yang CT, Zhou J, Guo
R, Chiu CH, Chang GC, Shiah HS, Chen Y, Wang CC, et al: Safety,
efficacy, and pharmacokinetics of almonertinib (HS-10296) in
pretreated patients With EGFR-Mutated advanced NSCLC: A
multicenter, open-label, phase 1 trial. J Thorac Oncol.
15:1907–1918. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li Y, Meng L, Ma Y, Li Y, Xing X, Guo C
and Dong Z: Determination of osimertinib, aumolertinib, and
furmonertinib in human plasma for therapeutic drug monitoring by
UPLC-MS/MS. Molecules. 27:44742022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Deng Y, Yang C, Liu W, Cai S and Guo X:
Aumolertinib-based comprehensive treatment for an uncommon site of
EGFR exon 20 insertion mutations with multiple metastases non-small
cell lung cancer: A case report. Anticancer Drugs. 33:406–412.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yang GJ, Li J, Xu HY, Sun Y, Liu L, Li HS,
Yang L, Zhang Y, Li GH and Wang Y: Osimertinib for Chinese advanced
non-small cell lung cancer patients harboring diverse EGFR exon 20
insertion mutations. Lung Cancer. 152:39–48. 2021. View Article : Google Scholar : PubMed/NCBI
|