Introduction
Ganglioneuromas (GNs) are highly differentiated
benign tumors that arise from neural crest cells and may develop
anywhere along the sympathetic chain (1). They are most commonly present in the
posterior mediastinum (41.5%) and the retroperitoneum (37.5%)
(2). The tumors are usually
detected incidentally during radiological imaging for unrelated
conditions. However, GN may also present when the tumor has grown
sufficiently in size to the point that it may cause
compression-related symptoms (3,4).
These tumors are frequently non-secretory, but rare cases of
hormone-secreting GNs have also been reported (5). The gold standard for the diagnosis of
GN is histopathological examination, which characteristically
indicates an admixture of ganglion cells and Schwann cells with the
absence of immature elements (neuroblasts) (6). Less commonly, fine-needle aspiration
(FNA) has also been suggested to aid in the diagnosis of GNs
(7). On rare occasions, the
presence of adipose cells has been detected in these tumors
(8). This may result in the
diagnostic confusion of GN via imaging and cytologic diagnostic
approaches with other, more aggressive tumors, such as liposarcoma,
leading to inappropriate management (9).
The present study reported a case of a huge
retroperitoneal GN that was initially diagnosed and managed as
liposarcoma and to acknowledge the possible presence of adipose
cells in sporadic cases of GN. In the writing of the current paper,
the SCARE 2020 Guidelines were taken into account (10).
Case report
Case presentation
A 54-year-old male patient presented to the
Sulaymaniyah Surgical Teaching Hospital (Sulaymaniyah, Iraq) in
August 2018 with gradually progressing dull backache and abdominal
discomfort for the past six months. The pain radiated to the
anterior thigh, with no relation to daily physical activity. There
were no associated gastrointestinal symptoms. Having assumed a
musculoskeletal origin for his pain, the patient had used different
kinds of non-steroidal anti-inflammatory drugs and other analgesics
without any improvement. The patient had been diagnosed with type 2
diabetes mellitus and was on metformin 500 mg twice daily.
Clinical findings
The patient's vital signs were stable. Physical
examination revealed abdominal distension and mild tenderness on
the right side of the abdomen.
Diagnostic approach
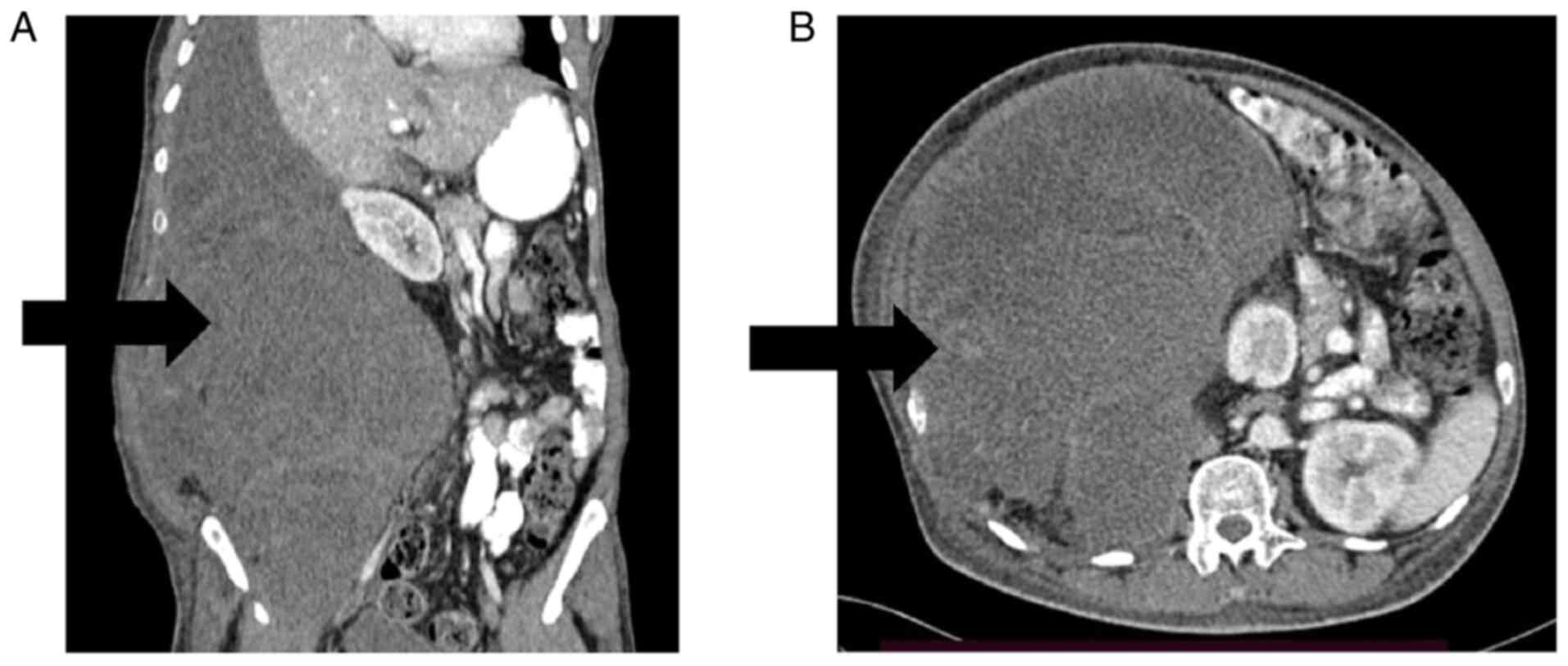
Ultrasound of the abdomen and pelvis revealed a
large right-sided retroperitoneal mass; however, it was unable to
show the detailed characteristics of the mass. Contrast-enhanced
computed tomography (CT) of the abdomen and pelvis indicated a
right-sided multilobulated retroperitoneal mass with the dimensions
of 45×22×23 cm. The mass displaced the right kidney anteromedially
and extended up to the liver. The radiological features were
consistent with sarcoma (Fig. 1).
CT scan of the chest was normal. FNA and core biopsy were performed
on the mass and the pathologic examination was suggestive of myxoid
liposarcoma. The multidisciplinary team (MDT) discussed the case
and a decision was made to start neoadjuvant radiochemotherapy. The
patient received 25 sessions of radiation therapy followed by four
cycles of ifosfamide and doxorubicin according to the National
Comprehensive Cancer Network (NCCN) guidelines (11). However, in the middle of the
cycles, a new CT scan was performed to assess the patient's
response to the treatments, which indicated no reduction in the
mass size. The patient's condition was discussed once again by the
MDT and surgical resection of the mass was determined to be the
best course of action.
Therapeutic intervention and
perioperative diagnosis
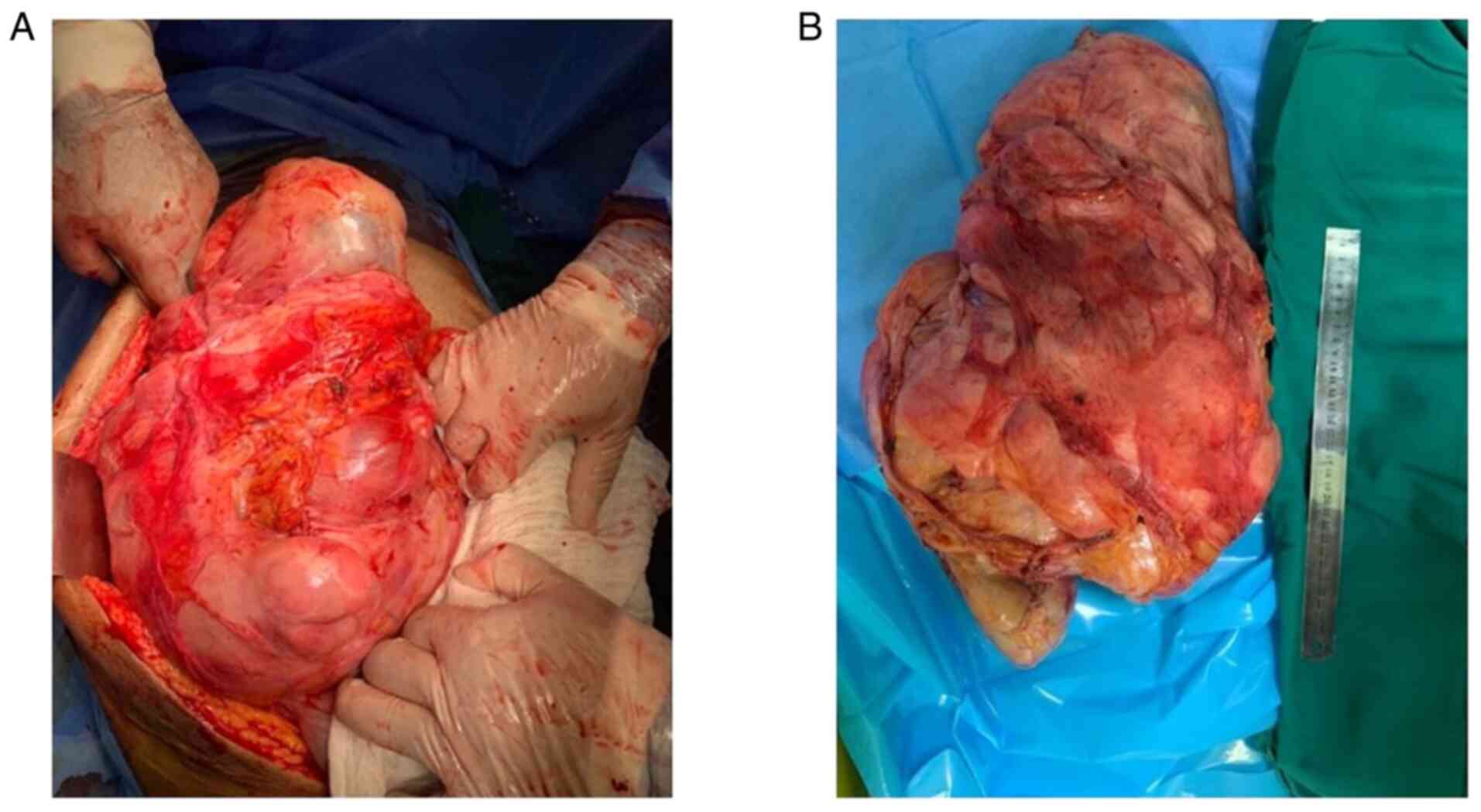
The operation was performed through a midline
laparotomy under general anesthesia and the mass was resected en
bloc (Fig. 2). The
post-operative period was uneventful and the patient was discharged
on the fourth postoperative day. The mass measured 45×35×25 cm and
weighed 10 kilograms. Histopathologic examination according to
standard protocols indicated an encapsulated, hypocellular tumor
composed of ganglion cells within an edematous, collagenous to
myxoid background. Immunohistochemical analysis indicated positive
reaction of the tumor cells to vimentin, S-100 and neuron-specific
enolase (NSE), while stains for smooth muscle actin (SMA) and
calretinin were negative. According to standard protocols, the
following antibodies were used for immunohistochemistry: S100 (cat.
no. Z0311; dilution, 0.3:100; Dako Denmark A/S), NSE (cat. no. BSB
5824; dilution, 0.7:100; Bio SB), Vimentin (cat. no. M0725;
dilution, 1.3:100; Dako Denmark A/S), calretinin (cat. no. M7245;
dilution, 2:100; Dako Denmark A/S) and SMA (cat. no. M0851;
dilution, 1.3:100; Dako Denmark A/S). The overall histology
combined with the immunohistochemical findings were consistent with
a diagnosis of ganglioneuroma (Fig.
3).
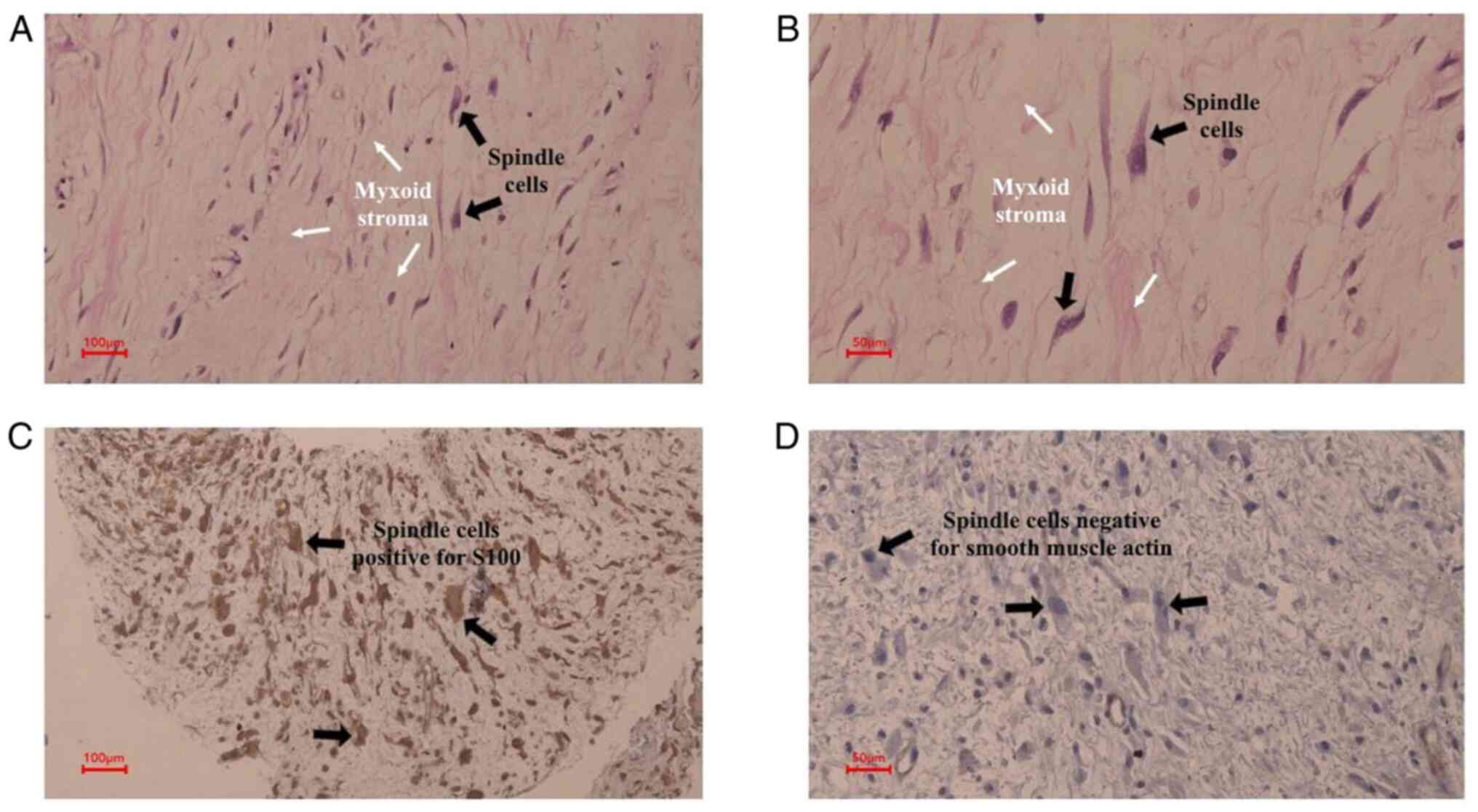 | Figure 3.Histopathologic examination of the
mass. (A) The tumor is hypocellular and is composed of scattered
spindle cells; certain cells had large, round nuclei with fine
chromatin and occasional nucleoli (black arrows), lying within a
loose, edematous, focally myxoid stroma (white arrows) (H&E;
scale bar, 100 µm; magnification, ×20). (B) Magnified window from A
(scale bar, 50 µm; magnification, ×40). (C) The spindle cells,
including the ones with larger nuclei (black arrows), are positive
for S100 in a strong, cytoplasmic and nuclear pattern (scale bar,
100 µm; magnification, ×20). (D) The spindle cells (black arrows)
are negative for smooth muscle actin (scale bar, 50 µm;
magnification, ×40). |
Follow-up and outcome
In October 2020, a follow-up MRI showed 2 recurrent
masses in the right subphrenic area. The patient underwent a second
operation in December 2020. In all aspects, the recurrence was the
same as the primary tumor, radiologically and pathologically. In
May 2021, imaging follow-up showed another recurrence in the same
position and the patient underwent a third operation in July 2021
with similar postoperative histopathological findings.
Unfortunately, the mass recurred again in February 2022, and a 6-cm
mass was observed on a CT scan. The patient refused further
intervention and rapid enlargement of the size of the mass was seen
on follow-up. In May 2022, the mass had reached a size of 18 cm.
The patient passed away in June 2022 due to a cardiovascular
event.
Discussion
A literature review was performed through the
CINAHL, PubMed/MEDLINE, Cochrane Library, Web of Science and EMBASE
databases to identify studies published up to January 2022. The
search was performed using the following key words: Ganglioneuroma,
retroperitoneal, retroperitoneum, liposarcoma, misdiagnosis,
mismanagement.
GNs are benign, slow-growing tumors of neural crest
cell origin that arise along the sympathetic chain (1). The tumor was first described in 1870
(6). Less commonly, GN may involve
the medulla of the adrenal glands, the parapharyngeal region and
the visceral ganglia (3). Although
most studies indicate a higher prevalence among females (9,12),
other studies have indicated no gender preference (13). The current case was a male with a
huge GN presenting in the retroperitoneum.
GNs are mostly asymptomatic. Symptoms, when present,
are relatively non-specific and include abdominal, back and neck
pain, vomiting, hemoptysis and shortness of breath due to the
pressure exerted on adjacent orangs by the enlarging tumor.
Referred pain to the lower limbs with paresthesia and numbness to
due to big retroperitoneal ganglioneuroma has also been documented
(2,14–16).
The patient of the present study had abdominal and back pain. Rare
cases of hormonally active GN have been reported in the literature
with symptoms of palpitation, tremor, anxiety, flushing,
diaphoresis, diarrhea and hypertension, which are due to the
secretion of catecholamine, cortisol or vasoactive intestinal
peptide by the tumor (17).
Imaging techniques (CT scan or magnetic resonance
imaging) in patients with retroperitoneal masses are utilized to
describe the tumor's size and location, and to delineate its
anatomic relationship with the neighboring structures, which is
essential for surgical management, as most retroperitoneal tumors
are malignant (18,19). A CT scan of GN generally indicates
a well-demarcated oval mass with low to intermediate attenuation
and punctate calcifications in 20% of cases. These findings,
however, are not diagnostic and are insufficient to make an
accurate diagnosis (20).
Misdiagnosis of GN as sarcoma has been reported via CT scan in
certain cases due to irregular mixed density (9). Workup of the initial CT of the
patient of the present study was also suggestive of sarcoma.
FNA has been suggested to assist in the diagnosis of
GN (7,21). The cytologic features of GN have
been described as reasonably distinctive, provided that the smear
contains both ganglion and spindle cell components. The
ganglioneuroma elements are composed of relatively mature ganglion
cells, Schwann cells and nerve fibers. The ganglion cells may be
identified by their abundant eosinophilic cytoplasm, large nuclei
and prominent nucleoli, and the lipomatous areas consist of mature
adipocytes without atypia (8).
However, in the present case, FNA was suggestive of myxoid
liposarcoma due to the presence of adipocytes and the lack of
spindle cells and ganglion cells in the smear, and due to this
misdiagnosis, chemotherapy and radiation were administered to the
patient. Similar observations have been made in other rare cases of
GN, as showcased by Meng et al (22). Radiological features and even FNA
give crucial clues but are not diagnostic as is histopathological
examination of the specimen.
There are two hypotheses for the histogenesis of fat
in GN: First, spontaneous degeneration of the tumor leads to fatty
replacement in the tumor, which is a reasonable explanation in the
present case, considering the large size and the time it took to
reach that size. The second hypothesis is that due to the origin of
GNs from neural crest cells, which are regarded as ectomesenchyme,
they may have the potential to undergo lipometaplasia and
differentiate into adipocytes (22).
Although GNs represent the benign end of the
spectrum compared to their ganglion tumor counterparts,
ganglioneuroblastoma, and neuroblastoma constitute the malignant
end of the spectrum. Among a total of 49 patients with GN, two
cases of malignant transformation and metastasis have been
documented (13). This leads to a
controversy regarding GN management in circumstances where complete
resection of the tumor imposes a high risk of mortality and
morbidity on the patient.
Surgical removal of GN is curative in most cases,
with a low incidence of recurrence (2). Xiao et al (9) demonstrated that among 32 patients
with GN, two patients chose surveillance instead of an operation
and the tumor remained stable during follow-up in both cases. Among
the other 30 patients, four had the tumor incompletely resected and
they still did not exhibit any signs of progression or malignancy
during follow-up (9). However, the
present case exhibited a recurrence of GN in the subhepatic area
two years after the operation. Histopathology remains the gold
standard for diagnosing GN, which characteristically indicates an
admixture of Schwann cells and ganglion cells in the absence of
immature cells (neuroblasts). Immunohistochemistry further supports
the diagnosis (6). What made the
diagnosis difficult in the present case was that the tumor enlarged
rapidly and the FNA indicated atypical cells, which was misleading
to the pathologist.
There were certain limitations to this report, as no
molecular analysis was performed. For GN, no molecular test
appeared to be available; however, a test for liposarcoma exists,
but there was no access to it at our hospital.
In conclusion, preoperative diagnosis of GN is
difficult due to radiological confusion with other tumors and a
lack of specific clinical manifestations, leading to misdiagnosis
and mismanagement. FNA may help with the diagnosis, but the current
case demonstrated the importance of taking aspirates from multiple
sites, particularly for large tumors, due to the confounding
presence of adipocytes in rare cases of GN. Histopathological
examination is the only method for a definitive diagnosis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
RB: Surgeon performing the operation, major
contribution to the conception of the study, literature review,
final approval of the manuscript. FHK: Literature review, writing
the manuscript, final approval of the manuscript, major
contribution to the conception of the study. RMA: Pathological
examination, major contribution to the conception of the study and
revision of the manuscript. DSH and DMH: Acquisition of data and
revision of the manuscript.. TAH, IA and AMS: Major contribution to
the conception of the study and revision and final revision of the
manuscript. RB and FHK confirm the authenticity of all the raw data
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient and patient's family provided written
informed consent for the publication of the patient's data and
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Yang Y, Ren M, Yuan Z, Li K, Zhang Z,
Zhang J, Xie L and Yang Z: Thoracolumbar paravertebral giant
ganglioneuroma and scoliosis: A case report and literature review.
World J Surg Oncol. 14:652016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Silveira CRS, Vieira CGM, Pereira BM,
Junior LE, Gerson G, Távora DGF and Chhabra A: Magnetic resonance
neurography in the diagnosis of a retroperitoneal ganglioneuroma:
Case report and literature review. Radiol Case Rep. 13:380–385.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yam B, Walczyk K, Mohanty SK, Coren CV and
Katz DS: Radiology-pathology conference: Incidental posterior
mediastinal ganglioneuroma. Clin Imaging. 33:390–394. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hussain MH, Iqbal Z, Mithani MS and Khan
MN: Retroperitoneal ganglioneuroma in a patient presenting with
vague abdominal pain. Cureus. 12:e91332020.PubMed/NCBI
|
|
5
|
Shah A, Thummar D, Mehta M, Shah D,
Suryanarayan U and Anand D: Hormone-secreting ganglioneuroma: A
rare entity. J Radiat Cancer Res. 12:131–133. 2021. View Article : Google Scholar
|
|
6
|
Spinelli C, Rossi L, Barbetta A, Ugolini C
and Strambi S: Incidental ganglioneuromas: A presentation of 14
surgical cases and literature review. J Endocrinol Invest.
38:547–554. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Domanski HA: Fine-needle aspiration of
ganglioneuroma. Diagn Cytopathol. 32:363–366. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Adachi S, Kawamura N, Hatano K, Kakuta Y,
Takada T, Hara T and Yamaguchi S: Lipomatous ganglioneuroma of the
retroperitoneum. Pathol Int. 58:183–186. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xiao J, Zhao Z, Li B and Zhang T: Primary
retroperitoneal ganglioneuroma: A retrospective cohort study of 32
patients. Front Surg. 8:6424512021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Agha RA, Franchi T, Sohrabi C, Mathew G
and Kerwan A; SCARE Group, : The SCARE 2020 guideline: Updating
consensus Surgical CAse REport (SCARE) guidelines. Int J Surg.
84:226–230. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Von Mehren M, Randall RL, Benjamin RS,
Boles S, Bui MM, Ganjoo KN, George S, Gonzalez RJ, Heslin MJ, Kane
JM, et al: Soft tissue sarcoma, version 2.2018, NCCN clinical
practice guidelines in oncology. J Natl Compr Canc Netw.
16:536–563. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
De Bernardi B, Gambini C, Haupt R, Granata
C, Rizzo A, Conte M, Tonini GP, Bianchi M, Giuliano M, Luksch R, et
al: Retrospective study of childhood ganglioneuroma. J Clin Oncol.
26:1710–1716. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Geoerger B, Hero B, Harms D, Grebe J,
Scheidhauer K and Berthold F: Metabolic activity and clinical
features of primary ganglioneuromas. Cancer. 91:1905–1913. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Esen HK, Esen O and Irsi C:
Retroperitoneal ganglioneuroma: Mimicking an ovarian mass in a
child. Pak J Med Sci. 31:724–726. 2015.PubMed/NCBI
|
|
15
|
Hayat J, Ahmed R, Alizai S and Awan MU:
Giant ganglioneuroma of the posterior mediastinum. Interact
Cardiovasc Thorac Surg. 13:344–345. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Papaetis GS, Georgiadis CP, Tsitskari MA,
Constantinou PG and Antoniou AP: Retroperitoneal ganglioneuroma
causing chronic lower back and leg pain in an 80-year-old man: A
case report. Ann Med Surg (Lond). 61:101–103. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Erem C, Fidan M, Civan N, Cobanoglu U,
Kangul F, Nuhoglu I and Alhan E: Hormone-secreting large adrenal
ganglioneuroma in an adult patient: A case report and review of
literature. Blood Press. 23:64–69. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zografos GN, Farfaras A, Vasiliadis G,
Pappa T, Aggeli C, Vasilatou E, Kaltsas G and Piaditis G:
Laparoscopic resection of large adrenal tumors. JSLS. 14:364–368.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Al-Ali MHM, Salih AM, Ahmed OF, Kakamad
FH, Mohammed SH, Hassan MN, Sidiq SH, Mustafa MQ, Najar KA and
Abdullah IY: Retroperitoneal lipoma; a benign condition with
frightening presentation. Int J Surg Case Rep. 57:63–66. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Duffy S, Jhaveri M, Scudierre J, Cochran E
and Huckman M: MR imaging of a posterior mediastinal
ganglioneuroma: Fat as a useful diagnostic sign. AJNR Am J
Neuroradiol. 26:2658–2662. 2005.PubMed/NCBI
|
|
21
|
Jain M, Shubha BS, Sethi S, Banga V and
Bagga D: Retroperitoneal ganglioneuroma: Report of a case diagnosed
by fine-needle aspiration cytology, with review of the literature.
Diagn Cytopathol. 21:194–196. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Meng QD, Ma XN, Wei H, Pan RH, Jiang W and
Chen FS: Lipomatous ganglioneuroma of the retroperitoneum. Asian J
Surg. 39:116–119. 2016. View Article : Google Scholar : PubMed/NCBI
|