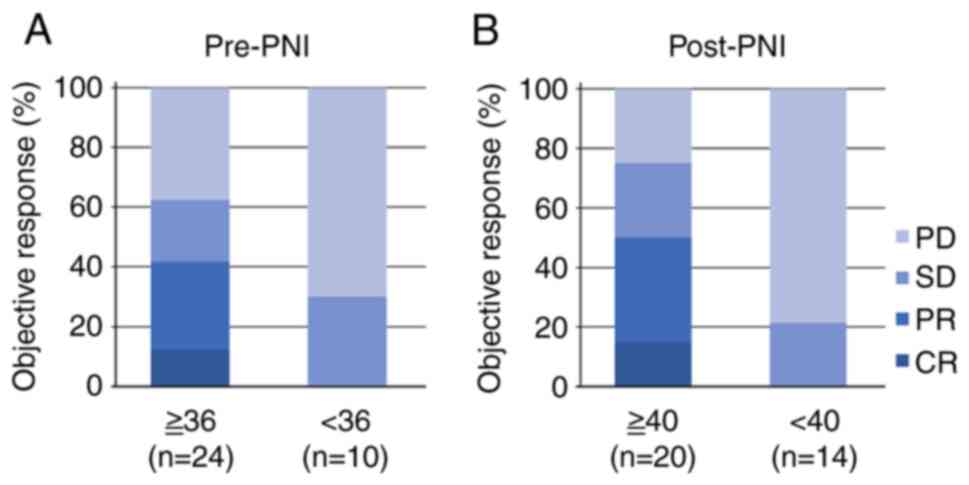
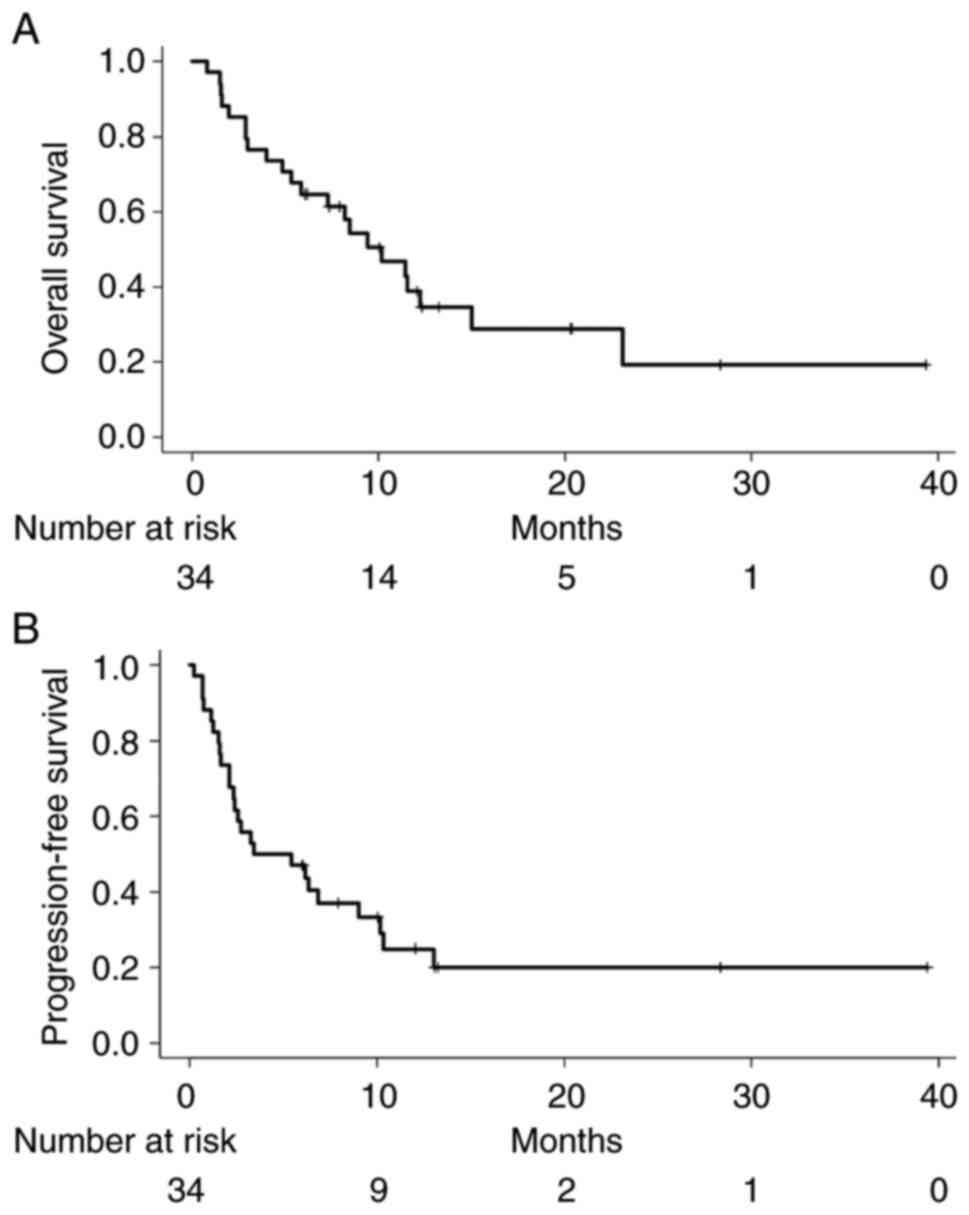
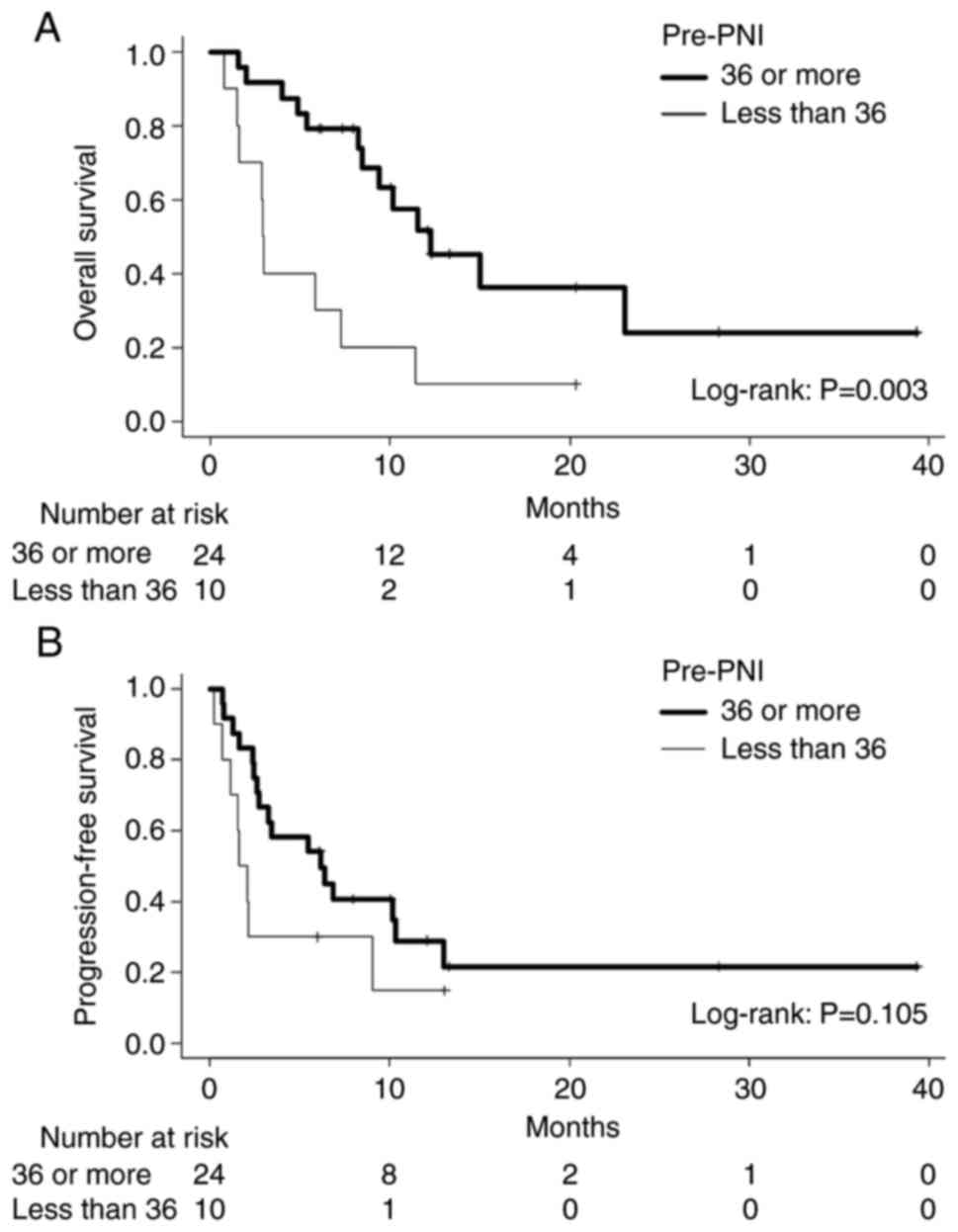
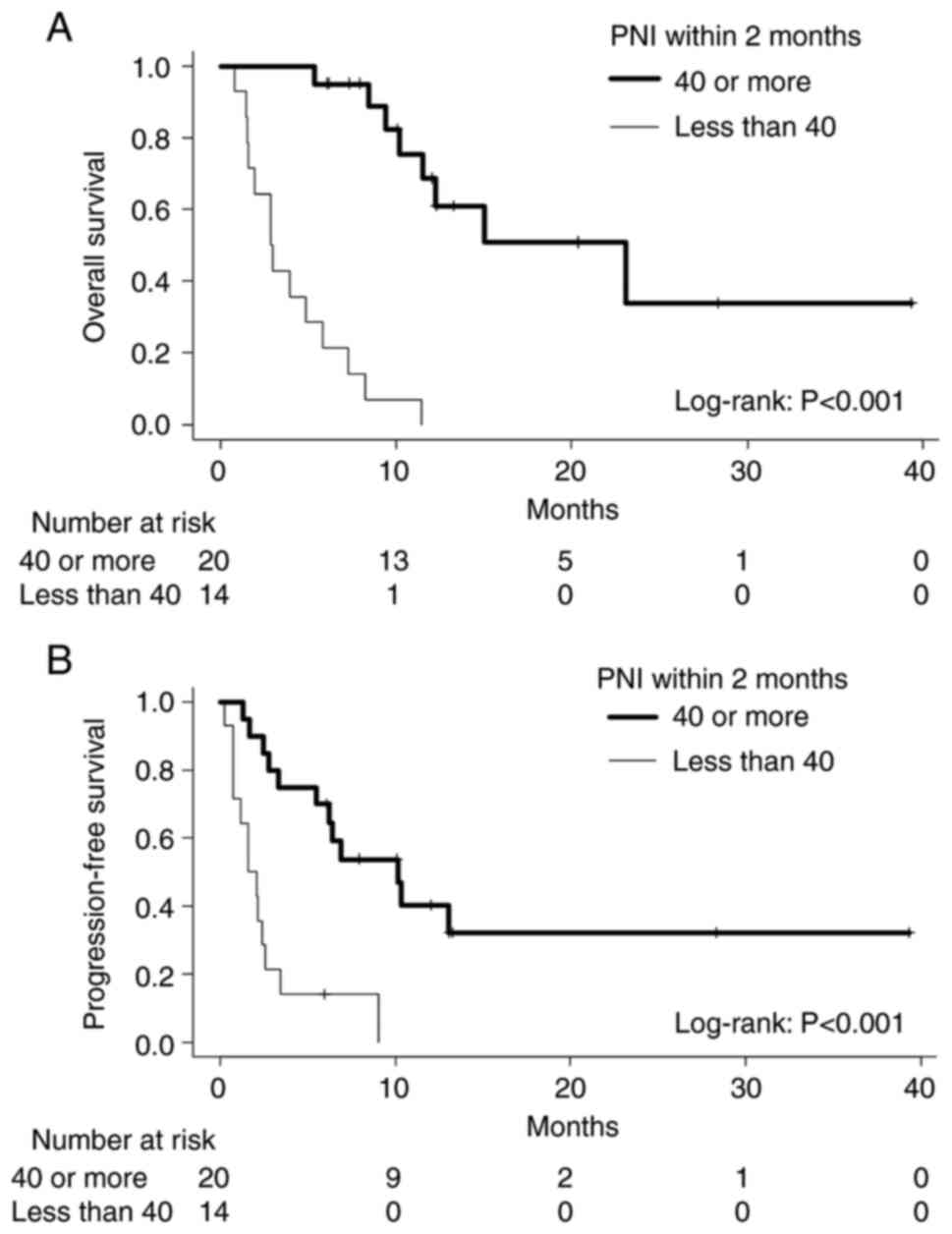
|
1
|
Patel VG, Oh WK and Galsky MD: Treatment
of muscle-invasive and advanced bladder cancer in 2020. CA Cancer J
Clin. 70:404–423. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sternberg CN, Yagoda A, Scher HI, Watson
RC, Ahmed T, Weiselberg LR, Geller N, Hollander PS, Herr HW and
Sogani PC: Preliminary results of M-VAC (methotrexate, vinblastine,
doxorubicin and cisplatin) for transitional cell carcinoma of the
urothelium. J Urol. 133:403–407. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
von der Maase H, Hansen SW, Roberts JT,
Dogliotti L, Oliver T, Moore MJ, Bodrogi I, Albers P, Knuth A,
Lippert CN, et al: Gemcitabine and cisplatin versus methotrexate,
vinblastine, doxorubicin, and cisplatin in advanced or metastatic
bladder cancer: Results of a large, randomized, multinational,
multicenter, phase III study. J Clin Oncol. 18:3068–3077. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bellmunt J, de Wit R, Vaughn DJ, Fradet Y,
Lee JL, Fong L, Vogelzang NJ, Climent LA, Petrylak DP, Choueiri TK,
et al: Pembrolizumab as second-line therapy for advanced urothelial
carcinoma. N Engl J Med. 376:1015–1026. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yanagisawa T, Mori K, Katayama S,
Mostafaei H, Quhal F, Laukhtina E, Rajwa P, Motlagh RS, Aydh A,
König F, et al: Pretreatment clinical and hematologic prognostic
factors of metastatic urothelial carcinoma treated with
pembrolizumab: A systematic review and meta-analysis. Int J Clin
Oncol. 27:59–71. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ni L, Huang J, Ding J, Kou J, Shao T, Li
J, Gao L, Zheng W and Wu Z: Prognostic nutritional index predicts
response and prognosis in cancer patients treated with immune
checkpoint inhibitors: A systematic review and meta-analysis. Front
Nutr. 9:8230872022. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Johannet P, Sawyers A, Qian Y, Kozloff S,
Gulati N, Donnelly D, Zhong J and Osman I: Baseline prognostic
nutritional index and changes in pretreatment body mass index
associate with immunotherapy response in patients with advanced
cancer. J Immunother Cancer. 8:e0016742020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shimizu T, Miyake M, Hori S, Ichikawa K,
Omori C, Iemura Y, Owari C, Itami Y, Nakai W and Anai S: Clinical
impact of sarcopenia and inflammatory/nutritional markers in
patients with unresectable metastatic urothelial carcinoma treated
with pembrolizumab. Diagnostics (Basel). 10:E3102020. View Article : Google Scholar
|
|
9
|
Ishiyama Y, Kondo T, Nemoto Y, Kobari Y,
Ishihara H, Tachibana H, Yoshida K, Hashimoto Y, Takagi T and
iizuka J: Predictive impact of prognostic nutritional index on
pembrolizumab for metastatic urothelial carcinoma resistant to
platinum-based chemotherapy. Anticancer Res. 41:1607–1614. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Eisenhauer EA, Rherasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Onodera T, Goseki N and Kosaki G:
Prognostic nutritional index in gastrointestinal surgery of
malnourished cancer patients. Nihon Geka Gakkai Zasshi.
85:1001–1005. 1984.(In Japanese). PubMed/NCBI
|
|
12
|
Kanda Y: Investigation of the freely
available easy-to-use software ‘EZR’ for medical statistics. Bone
Marrow Transplant. 48:452–458. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fradet Y, Bellmunt J, Vaughn DJ, Lee JL,
Fong L, Vogelzang NJ, Climent MA, Petrylak DP, Choueiri TK, Necchi
A, et al: Randomized phase III KEYNOTE-045 trial of pembrolizumab
versus paclitaxel, docetaxel, or vinflunine in recurrent advanced
urothelial cancer: Results of >2 years of follow-up. Ann Oncol.
30:970–976. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Davis AA and Patel VG: The role of PD-L1
expression as a predictive biomarker: An analysis of all US Food
and Drug Administration (FDA) approvals of immune checkpoint
inhibitors. J Immunother Cancer. 7:2782019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bellmunt J, de Wit R, Fradet Y, Climent
MA, Petrylak DP, Lee JL, Fong L, Necchi A, Sternberg CN, O'Donnell
PH, et al: Putative biomarkers of clinical benefit with
pembrolizumab in advanced urothelial cancer: Results from the
KEYNOTE-045 and KEYNOTE-052 landmark trials. Clin Cancer Res.
28:2050–2060. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ogihara K, Kikuchi E, Shigeta K, Okabe T,
Hattori S, Yamashita R, Yoshimine S, Shirotake S, Nakazawa R,
Matsumoto K, et al: The pretreatment neutrophil-to-lymphocyte ratio
is a novel biomarker for predicting clinical responses to
pembrolizumab in platinum-resistant metastatic urothelial carcinoma
patients. Urol Oncol. 38:602.e1–602.e10. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kobayashi T, Ito K, Kojima T, Maruyama S,
Mukai S, Tsutsumi M, Miki J, Okuno T, Yoshio Y, Matsumoto H, et al:
Pre-pembrolizumab neutrophil-to-lymphocyte ratio (NLR) predicts the
efficacy of second-line pembrolizumab treatment in urothelial
cancer regardless of the pre-chemo NLR. Cancer Immunol Immunother.
71:461–471. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kurashina R, Ando K, Inoue M, Izumi K,
Maruyama R, Mitani K, Takenobu H, Haruta M, Iizuka T, Kamijo T, et
al: Platelet-to-lymphocyte ratio predicts the efficacy of
pembrolizumab in patients with urothelial carcinoma. Anticancer
Res. 42:1131–1136. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yamamoto Y, Yatsuda J, Shimokawa M, Fuji
N, Aoki A, Sakano S, Yamamoto M, Suga A, Tei Y, Yoshihiro S, et al:
Prognostic value of pre-treatment risk stratification and
post-treatment neutrophil/lymphocyte ratio change for pembrolizumab
in patients with advanced urothelial carcinoma. Int J Clin Oncol.
26:169–177. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tomioka-Inagawa R, Nakane K, Enomoto T,
Tomioka M, Taniguchi T, Ishida T, Ozawa K, Takagi K, Ito H,
Takeuchi S, et al: The impact of Neutrophil-to-Lymphocyte ratio
after two courses of pembrolizumab for oncological outcomes in
patients with metastatic urothelial carcinoma. Biomedicines.
10:16092022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fujiwara M, Fujiwara R, Urasaki T, Oguchi
T, Komai Y, Numao N, Yamamoto S, Yonese J and Yuasa T: Early serum
and hematological responses to pembrolizumab therapy as predictors
of survival in metastatic urothelial cancer. Anticancer Res.
42:2045–2051. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Uchimoto T, Nakamura K, Komura K,
Fukuokaya W, Yano Y, Nishimura K, Kinoshita S, Nishio K, Fukushima
T, Nakamori K, et al: Prognostic value of the fluctuation in the
neutrophil-lymphocyte ratio at 6 weeks of pembrolizumab treatment
is specific to the clinical response in metastatic urothelial
carcinoma. Urol Oncol. 40:344.e11–344.e17. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ryman JT and Meibohm B: Pharmacokinetics
of monoclonal antibodies. CPT Pharmacometrics Syst Pharmacol.
6:576–588. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Turner DC, Kondic AG, Anderson KM,
Robinson AG, Garon EB, Riess JW, Jain L, Mayawala K, Kang J,
Ebbinghaus SW, et al: Pembrolizumab Exposure-response assessments
challenged by association of cancer cachexia and catabolic
clearance. Clin Cancer Res. 24:5841–5849. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Delyon J, Mateus C, Lefeuvre D, Lanoy E,
Zitvogel L, Chaput N, Roy S, Eggermont AM, Routier E and Robert C:
Experience in daily practice with ipilimumab for the treatment of
patients with metastatic melanoma: An early increase in lymphocyte
and eosinophil counts is associated with improved survival. Ann
Oncol. 24:1697–1703. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Khunger M, Patil PD, Khunger A, Li M, Hu
B, Rakshit S, Basu A, Pennell N, Stevenson JP, Elson P, et al:
Post-treatment changes in hematological parameters predict response
to nivolumab monotherapy in non-small cell lung cancer patients.
PLoS One. 13:e01977432018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Karantanos T, Karanika S, Seth B and
Gignac G: The absolute lymphocyte count can predict the overall
survival of patients with non-small cell lung cancer on nivolumab:
A clinical study. Clin Transl Oncol. 21:206–212. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ueda K, Suekane S, Kurose H, Ogasawara N,
Hiroshige T, Chikui K, Uemura K, Nakiri M, Nishihara K, Matsuo M,
et al: Absolute lymphocyte count is an independent predictor of
survival in patients with metastatic renal cell carcinoma treated
with nivolumab. Jpn J Clin Oncol. 52:179–186. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lee YJ, Park YS, Lee HW, Park TY, Lee JK
and Heo EY: Peripheral lymphocyte count as a surrogate marker of
immune checkpoint inhibitor therapy outcomes in patients with
non-small-cell lung cancer. Sci Rep. 12:6262022. View Article : Google Scholar : PubMed/NCBI
|