Introduction
Cervical cancer is one of the leading causes of
cancer-related death among women. Worldwide, cervical cancer is the
fourth most frequently occurring malignancy in women, resulting in
530,000 new cases annually and 270,000 deaths (1). The 5-year survival rate depends on the
stage of primary cancer: 91.3% for stage I; 76.6% for stage II;
62.2% for stage III; and 28.3% for stage IV (2). Standard treatment strategies for stage
IB-IIA cervical cancer are surgery and radiation-based therapy.
Based on the post-operative pathological diagnosis,
patients are divided into three groups according to the recurrence
risk: i) High-risk, with lymph node metastasis (LNM) or parametrium
invasion; ii) intermediate-risk, with a deep cervical stromal
invasion (SI), large size or lymphovascular space invasion (LVSI);
and iii) low-risk, with no recurrence risk (3). For patients in the high-risk group,
adjuvant concurrent chemoradiotherapy (CCRT) is recommended to
prevent recurrence (4,5), and in the low-risk group, patients are
usually followed-up without additional therapies (3).
In contrast to adjuvant therapy for patients in the
high-risk group, the effect of adjuvant therapy for patients in the
intermediate-risk group has not yet been fully elucidated.
Worldwide, for intermediate-risk patients, adjuvant radiotherapy
(RT) or CCRT is considered. However, the effect of adjuvant RT/CCRT
is controversial. Certain reports have demonstrated that adjuvant
RT improves progression-free survival (PFS), whereas others
indicate no significant effect on overall survival (OS) (6–10).
According to a Gynecological Oncology Group Trial, which was a
randomized clinical trial of pelvic RT vs. no further therapy for
post-operative intermediate-risk patients, pelvic RT significantly
improved PFS, although severe life-threatening adverse effects were
frequently observed in the RT group (9). Another meta-analysis indicated that no
significant difference was observed in OS between the RT and non-RT
groups (10). In this report,
increased severe adverse effects were also reported in the RT
group.
The effect of adjuvant RT on intermediate-risk
patients has not been fully elucidated. In the present study, the
aim was to evaluate the impact of adjuvant RT on PFS for patients
with stage IB-IIA cervical cancer.
Materials and methods
Patient selection and
clinicopathological findings
The study protocol was approved by the Research
Ethics Committee of the Faculty of Medicine of University of Tokyo
[Tokyo, Japan; approval no. 3084-(7)] and was conducted following the
Declaration of Helsinki (11). A
total of 232 cases of patients with Stage IB and IIA cervical
cancer [diagnosed using the International Federation of Gynecology
and Obstetrics 2008 staging system (12)], initially treated with
extended/radical hysterectomy and pelvic lymphadenectomy between
January 2009 and December 2018 at the University of Tokyo Hospital
(Tokyo, Japan) were retrospectively reviewed. All cases were
identified on the basis of pathological evidence. The histological
types were divided into two groups: Squamous cell carcinoma (SCC)
and non-SCC, which included adenocarcinoma (AC) and adenosquamous
cell carcinoma. Other clinicopathological data [e.g., age at
diagnosis, body mass index, tumor size, SI of cancer, LVSI, the
presence of LNM, post-operative adjuvant therapies (RT or not) and
adverse events] were obtained from the electronic medical record.
Post-operative adjuvant RT was administered at a daily fraction of
1.8 Gy (50.4 Gy/28 fractions in total) and none of the patients
received CCRT. Adverse events were evaluated according to Common
Terminology Criteria for Adverse Events (v.5.0) (13). Patients were excluded if they met
any of the following criteria: i) Histologic cancer type was
neuroendocrine carcinoma; ii) pathological T stage (pT)1a; iii)
pT2b; iv) presence of LNM; or v) a positive surgical margin.
Risk factors for recurrence
The intermediate-risk group was defined according to
the Japan Society of Gynecologic Oncology guidelines (14). Large tumor size (≥4 cm in diameter),
deep SI (≥1/3) and positivity for LVSI were considered risk factors
for recurrence (9,14). In addition to these three risk
factors, the study focused on histology, as several reports have
demonstrated that the non-SCC histological type is a risk factor
for poor prognosis (15–17).
Statistical analysis
Patients were divided into two groups according to
the post-operative adjuvant RT: Patients with adjuvant RT
(RT+) and without (RT−). The
clinicopathological characteristics of the two groups were compared
using an unpaired Student's t-test and the χ2 test. PFS
and OS were estimated using the Kaplan-Meier method and analyzed by
the log-rank test and the univariate Cox-proportional hazard
regression model to calculate hazard ratios (HRs) for each factor.
PFS was assessed from the date of diagnosis to the date of first
documentation of disease progression or death from any cause. The
effect of RT was analyzed in each of the following subgroups: i)
Tumor size ≥4 or <4 cm; ii) SI ≥1/3 or <1/3; iii)
LVSI+ or LVSI−; and iv) SCC or non-SCC. A
forest plot was created to visualize the HRs. Based on all
clinicopathological characteristics, propensity scores and
conditional probabilities of receiving adjuvant RT were calculated
via logistic regression analysis. In order to reduce the bias due
to confounding variables between the RT+ and
RT− groups, propensity score matching was performed with
‘EZR (64-bit)’ in R software (version R4.2.2). Standardized
differences for the covariates were calculated to assess the
comparability of the matched cohorts and a normalized difference of
<0.1 was considered to indicate a balance between the cohorts.
Other statistical analyses, including the χ2 test,
log-rank test, Cox-proportional hazard model and Student's t-test,
were performed using JMP software (version 15.0; SAS Institute,
Inc.). P<0.05 was considered to indicate a statistically
significant difference.
Results
Patient characteristics
Of the 232 patients reviewed, 120 were excluded due
to the following reasons: 3 patients had neuroendocrine carcinoma;
20 had pT1a; and 97 had pT2b or were LNM+. None of the
patients presented with positive surgical margins. The remaining
112 patients were included in the subsequent analyses. Among them,
103 patients underwent Okabayashi radical hysterectomy, which
corresponds to a Gynecologic Cancer Group type III hysterectomy
(18), while 9 patients with small
tumor sizes underwent modified radical hysterectomy (19). In the Okabayashi radical
hysterectomies, the cardinal ligaments and anterior layers of the
vesicouterine ligaments were cut and ligated. After mobilization of
the ureters, the posterior layers of the vesicouterine ligaments
were cut and ligated (20). In the
modified radical hysterectomy, the posterior layers of the
vesicouterine ligaments were preserved to protect pelvic nerves
(19). The median number of
resected lymph nodes was 39 (interquartile range [IQR], 11–106),
and there were no differences in the number of lymph nodes by
surgical procedure. The patients' clinicopathological
characteristics according to adjuvant RT are summarized in Table I. Patients in the RT+
group had significantly larger tumors, higher incidence of deep SI
and/or higher frequency of LVSI than those in the RT−
group. There was no difference in the age of the patients between
the groups; the median ages were 46 years old (IQR, 28–74) and 45
years old (IQR, 22–74) in the RT+ and RT−
groups, respectively.
 | Table I.Patients' clinicopathological
characteristics. |
Table I.
Patients' clinicopathological
characteristics.
| Characteristic | RT+
(n=41) | RT−
(n=71) | P-value |
|---|
| Age, years | 46 (28–74) | 45 (22–74) | 0.839a |
| FIGO stage |
|
| 0.883b |
| IB | 36 (88) | 63 (89) |
|
|
IIA | 5 (12) | 8 (11) |
|
| Histology |
|
| 0.203b |
|
Non-SCC | 14 (34) | 33 (47) |
|
|
SCC | 27 (66) | 38 (54) |
|
| Tumor size, cm |
|
|
<0.001b |
|
<4 | 13 (32) | 56 (79) |
|
| ≥4 | 28 (68) | 15 (21) |
|
| SI |
|
|
<0.001b |
|
<1/3 | 2 (4.9) | 37 (52) |
|
|
≥1/3 | 39 (95) | 34 (48) |
|
| LVSI |
|
|
<0.001b |
|
Positive | 25 (61) | 23 (32) |
|
|
Negative | 16 (39) | 48 (68) |
|
Prognosis
Among the 112 patients, 15 (13.4%) relapsed and 4
(3.6%) died. First, the PFS according to the four risk factors for
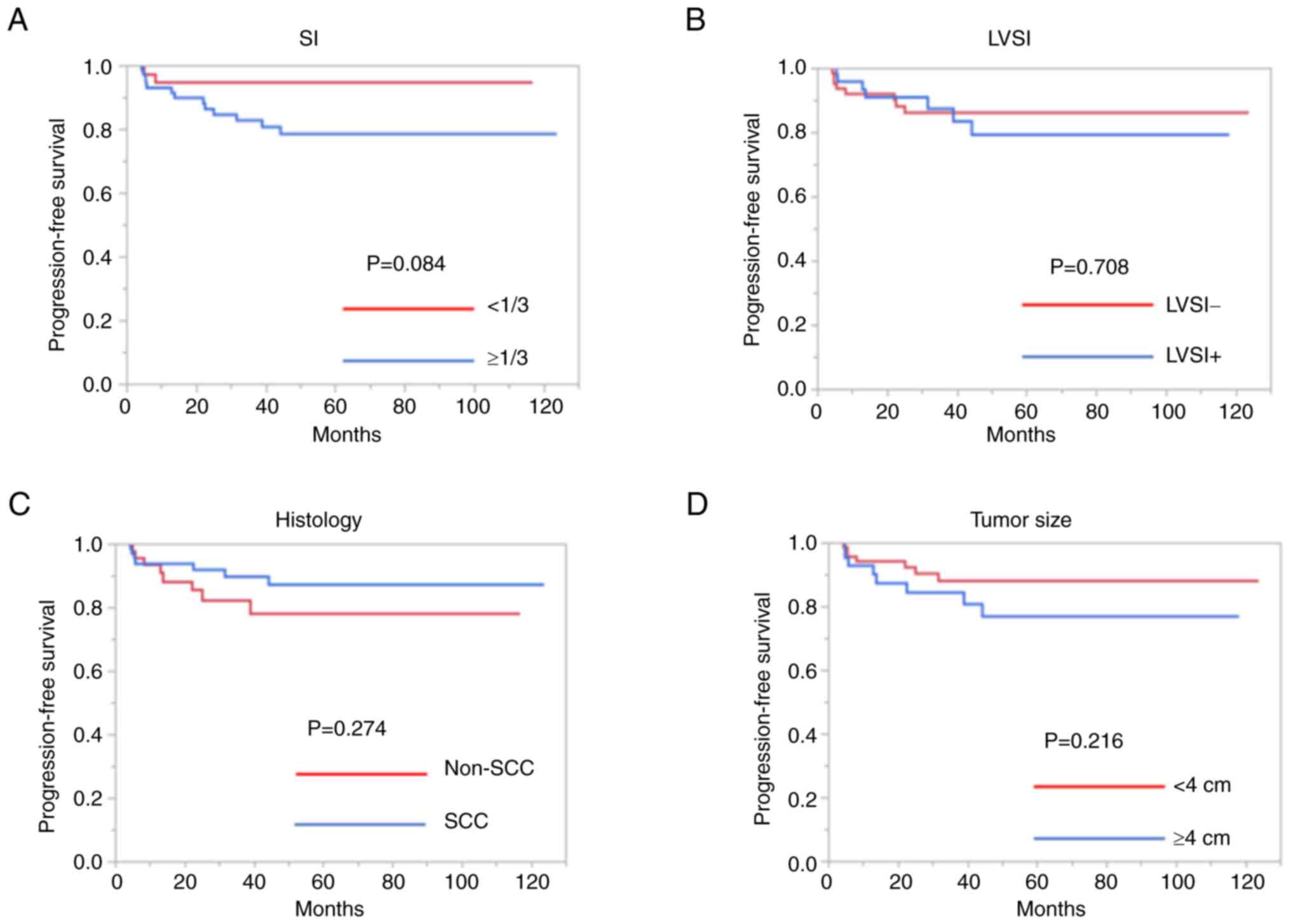
recurrence was compared. As presented in Fig. 1, although patients with deep SI
tended to have a poorer PFS, no significant differences were
observed among these groups (SI, P=0.084; tumor size, P=0.216;
LVSI, P=0.708; histology, P=0.274).
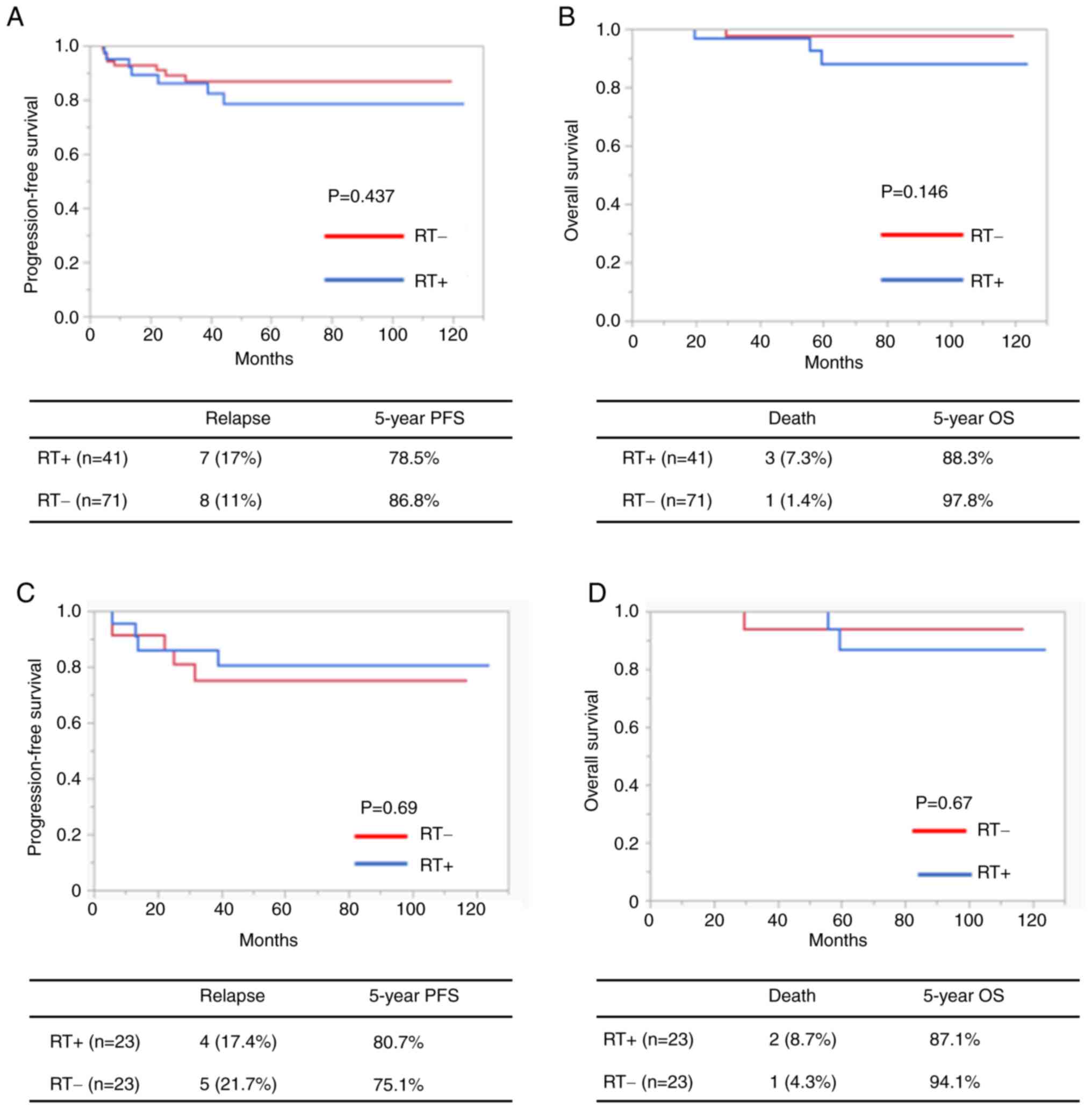
The overall prognoses were also compared between the
RT+ and RT− groups. There was no significant
difference either in the OS or PFS between the RT+ and
RT− groups (5-year OS rate, 88.3 vs. 97.8%,
respectively; 5-year PFS rate, 78.5 vs. 86.8%, respectively;
Fig. 2A and B).
Since the patients in the RT+ group had
more recurrence risk factors compared to those in the
RT− group, the PFS and OS in a propensity score-matched
cohort were subsequently compared. Of the 112 patients with
intermediate risks, 23 RT− patients were matched with 23
RT+ patients. For all covariates except for the age of
patients, the absolute standardized difference was <0.1 after
matching, implying sufficiently balanced treatment and
non-treatment groups (Table SI).
Kaplan-Meier curves based on adjuvant RT after propensity score
matching are depicted in Fig. 2C and
D. No significant differences were recorded between the groups.
The 5-year PFS rates were 75.1 and 80.7% in the non-treatment and
treatment groups, respectively (P=0.69; Fig. 2C).
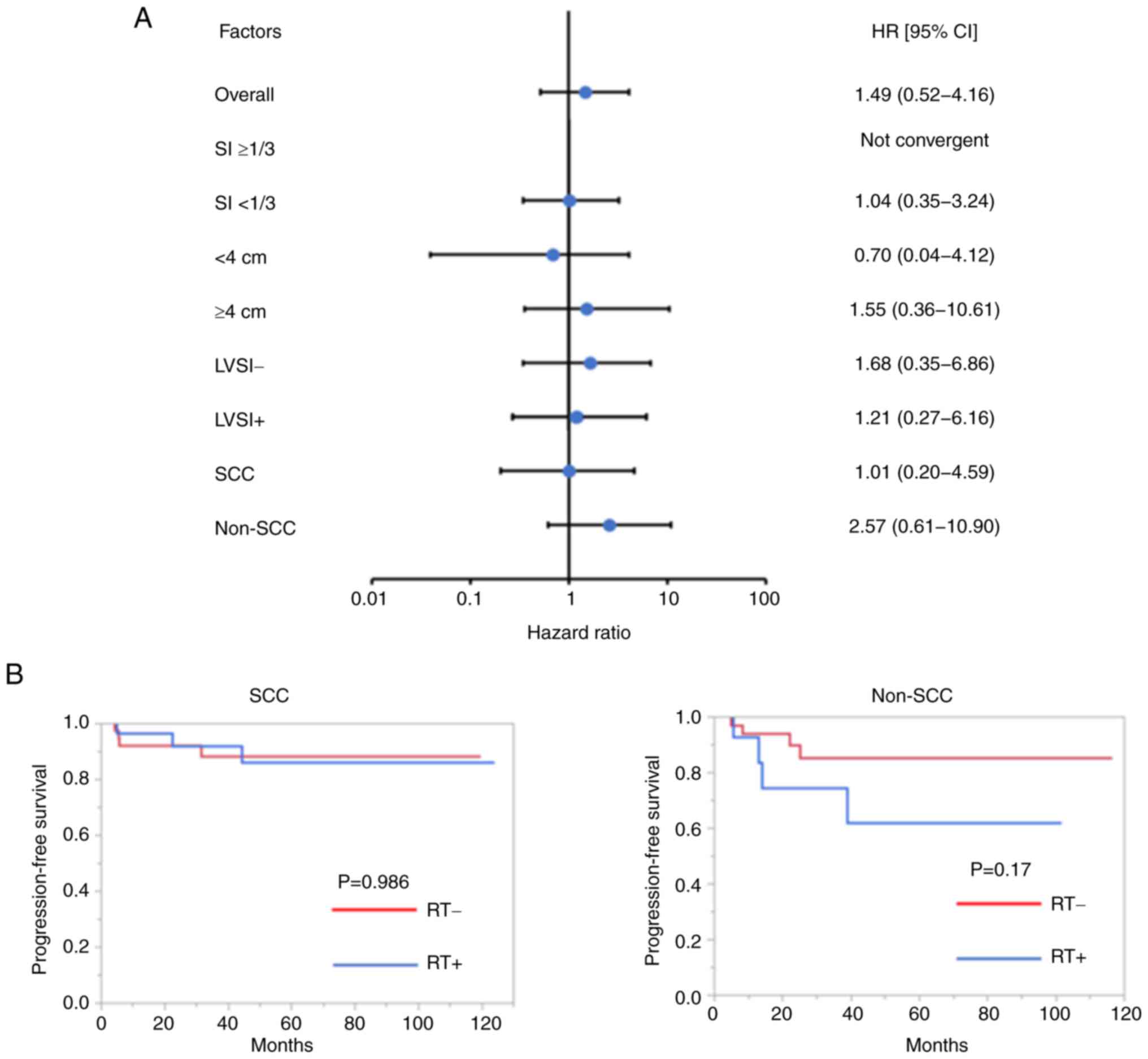
The effect of RT in each of the following subgroups
was subsequently evaluated: i) Tumor size ≥4 or <4 cm; ii) SI
≥1/3 or <1/3; iii) LVSI+ or LVSI−; and iv)
SCC or non-SCC by the univariate Cox-proportional hazard regression
model (Fig. 3A). Although no
significant differences were observed between RT+ and
RT− groups in each subgroup, RT had a different impact
on HR for PFS between the SCC and non-SCC groups (Fig. 3A). In the SCC group, no difference
was observed in the RT+ and RT− groups. In
the non-SCC group, although no significant difference could be
observed, patients in the RT+ group exhibited a slight
trend toward poorer prognosis compared to those in the
RT− group (SCC, P=0.986; non-SCC, P=0.17; Fig. 3B). The clinicopathological
characteristics of each pathology according to adjuvant RT are
provided in Table II. In both the
SCC and non-SCC groups, patients with RT had more risk factors of
recurrence, such as large tumor size, deep MI and LVS, than those
without RT.
 | Table II.Patients' clinicopathological
characteristics for each pathology type. |
Table II.
Patients' clinicopathological
characteristics for each pathology type.
|
| SCC (n=65) |
| Non-SCC (n=47) |
|
|---|
|
|
|
|
|
|
|---|
| Characteristic | RT+
(n=27) | RT−
(n=38) | P-value | RT+
(n=14) | RT−
(n=33) | P-value |
|---|
| Age, years | 47 (28–74) | 45.5 (22–74) | 0.642a | 44 (32–68) | 44 (31–71) | 0.474a |
| FIGO stage |
|
| 0.471b |
|
| 0.472b |
| IB | 21 (78) | 31 (82) |
| 12 (86) | 30 (91) |
|
|
IIA | 6 (22) | 7 (18) |
| 2 (4.3) | 3 (9.1) |
|
| Tumor size, cm |
|
|
<0.001c |
|
| 0.0044c |
|
<4 | 8 (30) | 30 (79) |
| 5 (58) | 26 (79) |
|
| ≥4 | 19 (70) | 8 (21) |
| 9 (64) | 7 (21) |
|
| SI |
|
| 0.011c |
|
|
<0.001c |
|
<1/3 | 2 (7.4) | 13 (34) |
| 0 (0) | 24 (73) |
|
|
≥1/3 | 25 (93) | 25 (66) |
| 14 (100) | 9 (27) |
|
| LVSI |
|
| 0.080b |
|
| 0.016c |
|
Positive | 17 (63) | 16 (42) |
| 8 (57) | 7 (21) |
|
|
Negative | 10 (37) | 22 (58) |
| 6 (43) | 26 (79) |
|
Adverse events
The adverse events between the RT+ and
RT− groups were compared. The number of adverse events,
including gastrointestinal disorders, bowel obstruction, lymphedema
and dysuria, tended to be higher in the RT+ group. In
particular, gastrointestinal disorders and lymphedema were observed
significantly more frequent in the RT+ group compared to
the RT− group (P<0.001 and P=0.0001, respectively;
Table III). In addition, it is
noteworthy that, although there were no significant differences
between the two groups, RT tended to increase grade 3 and 4 adverse
events, including bowel obstruction, dysuria and lymphedema.
 | Table III.Adverse events according to adjuvant
RT. |
Table III.
Adverse events according to adjuvant
RT.
|
| RT+
(n=41) | RT−
(n=71) | P-value |
|---|
|
|
|
|
|
|---|
| Adverse events | All grades | Grade 3 and 4 | All grades | Grade 3 and 4 | All grades | Grade 3 and 4 |
|---|
| Gastrointestinal
disorders | 12 (29) | 0 | 0 | 0 | <0.001 | - |
| Bowel
obstruction | 3 (7.3) | 3 (7.3) | 1 (1.4) | 1 (1.4) | 0.14 | 0.14 |
| Dysuria | 8 (20) | 2 (4.9) | 14 (20) | 0 | 0.59 | 0.13 |
| Lymphedema | 27 (66) | 5 (12) | 19 (27) | 3 (4.2) | 0.0001 | 0.12 |
Discussion
From the overall analysis, post-operative adjuvant
RT did not improve the survival outcomes of patients with cervical
cancer in the intermediate-risk group. In addition, survival
analyses using a propensity score-matched cohort did not
demonstrate any differences between PFS and OS. However, the
positivity for risk factors of recurrence was much higher in the
RT+ group than that in the RT− group,
suggesting that the prognosis of relatively higher-risk patients
may be improved by adjuvant RT. Further research to compare the
prognosis between RT+ and RT− groups in
patients with cervical cancer in the intermediate-risk group with
relatively higher risks is warranted to confirm the hypothesis.
In the present study, it was investigated which
patients may benefit from adjuvant RT based on subgroup analyses.
Due to the small sample size, statistically significant findings
were not achieved. However, the results suggested that the effect
of adjuvant RT may be dependent on histology. For the patients with
SCC, there was no difference in relapse rate or PFS between the
RT+ and RT− groups. By contrast, for the
patients with non-SCC, those who received adjuvant RT tended to
have poorer prognoses compared to those without adjuvant RT. The
results suggest that, in terms of reducing recurrent risk after
surgery, patients with SCC may benefit from adjuvant RT, whereas
the effect of adjuvant RT may be limited in patients with non-SCC.
In general, sensitivity to RT depends on cancer histology, as SCC
is more sensitive to RT than AC (21). Several reports have demonstrated
that, in the case of initial treatment by definitive RT, AC is
relatively resistant to RT compared to SCC (22–24).
The differential effect of adjuvant RT on SCC and non-SCC may be
due to the differences in radiosensitivity between SCC and AC.
In the present analysis, the frequency of adverse
events was much higher in the RT+ group than in the
RT− group, which concurs with previous studies (8,10). In
order to prevent harmful effects from treatment, it is important to
select patients who are likely to benefit from post-operative RT.
Although the results suggest a differential effect of RT according
to histology type, further research is required to prove this
hypothesis.
The present study has certain limitations. First,
the retrospective design of the present study may be linked to
potential treatment bias. Herein, patients in the RT+
group had markedly higher recurrence risks than those in the
RT− group. Even in a propensity score-matched cohort,
there were only a small number of patients with deep SI, which made
it difficult to compare those patients. Although a clear treatment
effect of RT in intermediate-risk patients was not apparent, the
results should be interpreted with caution, as an increase in the
number of risk factors is known to be associated with a worse
prognosis (7,25). Further research with multi-center or
nationwide data is warranted to validate the findings. As another
limitation, only one institution was involved in the present study,
which led to a relatively small sample size. Further studies in a
larger population are warranted to confirm the findings.
In conclusion, the present study has demonstrated
that the impact of post-operative RT for stage IB and IIA cervical
cancer is limited and is accompanied by increased adverse events.
Although there were no statistically significant differences, the
findings suggested that histology may influence the effects of
post-operative RT for intermediate-risk patients with cervical
cancer.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Mr. Katsuhiko Nara,
Pharmaceutical Department of the University of Tokyo Hospital
(Tokyo, Japan) for helping to create the forest plot.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
AT and MT conceptualized the study. CI, HH, AN, SE
and YM obtained the data. CI and AT analyzed the data. CI, AT, TT,
KS, MM, MT and YO interpreted the data. CI and AT wrote the
original draft. TT, MT, KS, MM and YO reviewed and edited the
manuscript. CI and AT confirm the authenticity of all the raw data.
All authors have read and approved the final version of the
manuscript. YO supervised the study.
Ethics approval and consent to
participate
This study was approved by the Research Ethics
Committee of the Faculty of Medicine of University of Tokyo [Tokyo,
Japan; approval no. 3084-(7)] and
all methods were performed in accordance with relevant guidelines
and regulations. The Research Ethics Committee of the Faculty of
Medicine of the University of Tokyo (Tokyo, Japan) waived the
requirement to obtain informed consent due to the retrospective
nature of the study. This study was performed by the opt-out method
on the hospital website.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
RT
|
radiotherapy
|
|
OS
|
overall survival
|
|
PFS
|
progression-free survival
|
|
SCC
|
squamous cell carcinoma
|
|
LNM
|
lymph node metastasis
|
|
SI
|
stromal invasion
|
|
LVSI
|
lymphovascular space invasion
|
|
CCRT
|
concurrent chemoradiotherapy
|
|
AC
|
adenocarcinoma
|
References
|
1
|
Small W Jr, Bacon MA, Bajaj A, Chuang LT,
Fisher BJ, Harkenrider MM, Jhingran A, Kitchener HC, Mileshkin LR,
Viswanathan AN and Gaffney DK: Cervical cancer: A global health
crisis. Cancer. 123:2404–2412. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Song S, Song C, Kim HJ, Wu HG, Kim JH,
Park NH, Song YS, Kim JW, Kang SB and Ha SW: 20 year experience of
postoperative radiotherapy in IB-IIA cervical cancer patients with
intermediate risk factors: Impact of treatment period and
concurrent chemotherapy. Gynecol Oncol. 124:63–67. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
National Comprehensive Cancer Network, .
NCCN Clinical Practice Guidelines in Oncology (NCCN
Guidelines®): Cervical Cancer. National Comprehensive
Cancer Network, Inc.; 2018
|
|
4
|
Peters WA III, Liu PY, Barrett RJ II,
Stock RJ, Monk BJ, Berek JS, Souhami L, Grigsby P, Gordon W Jr and
Alberts DS: Concurrent chemotherapy and pelvic radiation therapy
compared with pelvic radiation therapy alone as adjuvant therapy
after radical surgery in high-risk early-stage cancer of the
cervix. J Clin Oncol. 18:1606–1613. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rosa DD, Medeiros LR, Edelweiss MI,
Pohlmann PR and Stein AT: Adjuvant platinum-based chemotherapy for
early state cervical cancer. Cochrane Database Syst Rev.
6:CD0053422012.PubMed/NCBI
|
|
6
|
Landoni F, Maneo A, Colombo A, Placa F,
Milani R, Perego P, Favini G, Ferri L and Mangioni C: Randomised
study of radical surgery versus radiotherapy for stage Ib-IIa
cervical cancer. Lancet. 350:535–540. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sartori E, Tisi G, Chiudinelli F, La Face
B, Franzini R and Pecorelli S: Early stage cervical cancer:
Adjuvant treatment in negative lymph node cases. Gynecol Oncol. 107
(1 Suppl 1):S170–S174. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ryu SY, Park SI, Nam BH, Cho CK, Kim K,
Kim BJ, Kim MH, Choi SC, Lee ED and Lee KH: Is adjuvant
chemoradiotherapy overtreatment in cervical cancer patients with
intermediate risk factors? Int J Radiat Oncol Biol Phys.
79:794–799. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sedlis A, Bundy BN, Rotman MZ, Lentz SS,
Muderspach LI and Zaino RJ: A randomized trial of pelvic radiation
therapy versus no further therapy in selected patients with stage
IB carcinoma of the cervix after radical hysterectomy and pelvic
lymphadenectomy: A Gynecologic Oncology Group Study. Gynecol Oncol.
73:177–183. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rogers L, Siu SS, Luesley D, Bryant A and
Dickinson HO: Radiotherapy and chemoradiation after surgery for
early cervical cancer. Cochrane Database Syst Rev.
5:CD0075832012.PubMed/NCBI
|
|
11
|
World Medical Association, . World medical
association declaration of Helsinki: Ethical principles for medical
research involving human subjects. JAMA. 310:2191–2194. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim HS and Song YS: International
federation of gynecology and obstetrics (FIGO) staging system
revised: What should be considered critically for gynecologic
cancer? J Gynecol Oncol. 20:135–136. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
U.S. Department of Health and Human
Services, . Common Terminology Criteria for Adverse Events Version
5.0 (CTCAE v5.0). https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5×7.pdfNovember
27–2017
|
|
14
|
Ebina Y, Mikami M, Nagase S, Tabata T,
Kaneuchi M, Tashiro H, Mandai M, Enomoto T, Kobayashi Y, Katabuchi
H, et al: Japan society of gynecologic oncology guidelines 2017 for
the treatment of uterine cervical cancer. Int J Clin Oncol.
24:1–19. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gien LT, Beauchemin MC and Thomas G:
Adenocarcinoma: A unique cervical cancer. Gynecol Oncol.
116:140–146. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jung EJ, Byun JM, Kim YN, Lee KB, Sung MS,
Kim KT and Jeong DH: Cervical adenocarcinoma has a poorer prognosis
and a higher propensity for distant recurrence than squamous cell
carcinoma. Int J Gynecol Cancer. 6:1228–1236. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu SY, Huang EY and Lin H: Optimal
treatments for cervical adenocarcinoma. Am J Cancer Res.
9:1224–1234. 2019.PubMed/NCBI
|
|
18
|
Marin F, Plesca M, Bordea CI, Moga MA and
Blidaru A: Types of radical hysterectomies: From Thoma Ionescu and
Wertheim to present day. J Med Life. 7:172–176. 2014.PubMed/NCBI
|
|
19
|
Kunieda F, Kasamatsu T, Arimoto T, Onda T,
Toita T, Shibata T, Fukuda H and Kamura T; Gynecologic Cancer Study
Group of the Japan Clinical Oncology Group, : Non-randomized
confirmatory trial of modified radical hysterectomy for patients
with tumor diameter 2 cm or less FIGO Stage IB1 uterine cervical
cancer: Japan Clinical Oncology Group Study (JCOG1101). Jpn J Clin
Oncol. 45:123–126. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Konishi I: The Okabayashi radical
hysterectomy: Basic principle and step-by-step procedure. Surg J (N
Y). 7 (Suppl 2):S57–S69. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Turner JE: Basic information for radiation
therapy referral. Q Bull Northwest Univ Med Sch. 35:76–86.
1961.PubMed/NCBI
|
|
22
|
Hu K, Wang W, Liu X, Meng Q and Zhang F:
Comparison of treatment outcomes between squamous cell carcinoma
and adenocarcinoma of cervix after definitive radiotherapy or
concurrent chemoradiotherapy. Radiat Oncol. 13:2492018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lee YJ, Kim YT, Kim S, Lee B, Lim MC, Kim
JW and Won YJ: Prognosis of cervical cancer in the era of
concurrent chemoradiation from national database in Korea: A
comparison between squamous cell carcinoma and adenocarcinoma. PLoS
One. 10:e01448872015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Galic V, Herzog TJ, Lewin SN, Neugut AI,
Burke WM, Lu YS, Hershman DL and Wright JD: Prognostic significance
of adenocarcinoma histology in women with cervical cancer. Gynecol
Oncol. 125:287–291. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shigeta S, Shimada M, Tsuji K, Nagai T,
Tanase Y, Matuo K, Kamiura S, Iwata T, Yokota H and Mikami M: Risk
assessment in the patients with uterine cervical cancer harboring
intermediate risk factors after radical hysterectomy: A
multicenter, retrospective analysis by the Japanese gynecologic
oncology group. Int J Clin Oncol. 27:1507–1515. 2022. View Article : Google Scholar : PubMed/NCBI
|