|
1
|
Erratum. Global cancer statistics 2018:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 70:3132020. View Article : Google Scholar
|
|
2
|
Yang F, Ma L, Yang Y, Liu W, Zhao J, Chen
X, Wang M, Zhang H, Cheng S, Shen F, et al: Contribution of
hepatitis B virus infection to the aggressiveness of primary liver
cancer: A clinical epidemiological study in Eastern China. Front
Oncol. 9:3702019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liu YC, Lu LF, Li CJ, Sun NK, Guo JY,
Huang YH, Yeh CT and Chao CCK: Hepatitis B virus X protein induces
RHAMM-dependent motility in hepatocellular carcinoma cells via
PI3K-Akt-Oct-1 signaling. Mol Cancer Res. 18:375–389. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kang I, Kim JA, Kim J, Lee JH, Kim MJ and
Ahn JK: Hepatitis B virus X protein promotes epithelial-mesenchymal
transition of hepatocellular carcinoma cells by regulating SOCS1.
BMB Rep. 55:220–225. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lei Y, Xu X, Liu H, Chen L, Zhou H, Jiang
J, Yang Y and Wu B: HBx induces hepatocellular carcinogenesis
through ARRB1-mediated autophagy to drive the G(1)/S cycle.
Autophagy. 17:4423–4441. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
European Association for the Study of the
Liver. Electronic address, . simpleeasloffice@easloffice.eu;
European Association for the Study of the Liver: EASL clinical
practice guidelines: Management of hepatocellular carcinoma. J
Hepatol. 69:182–236. 2018. View Article : Google Scholar : PubMed/NCBI
|
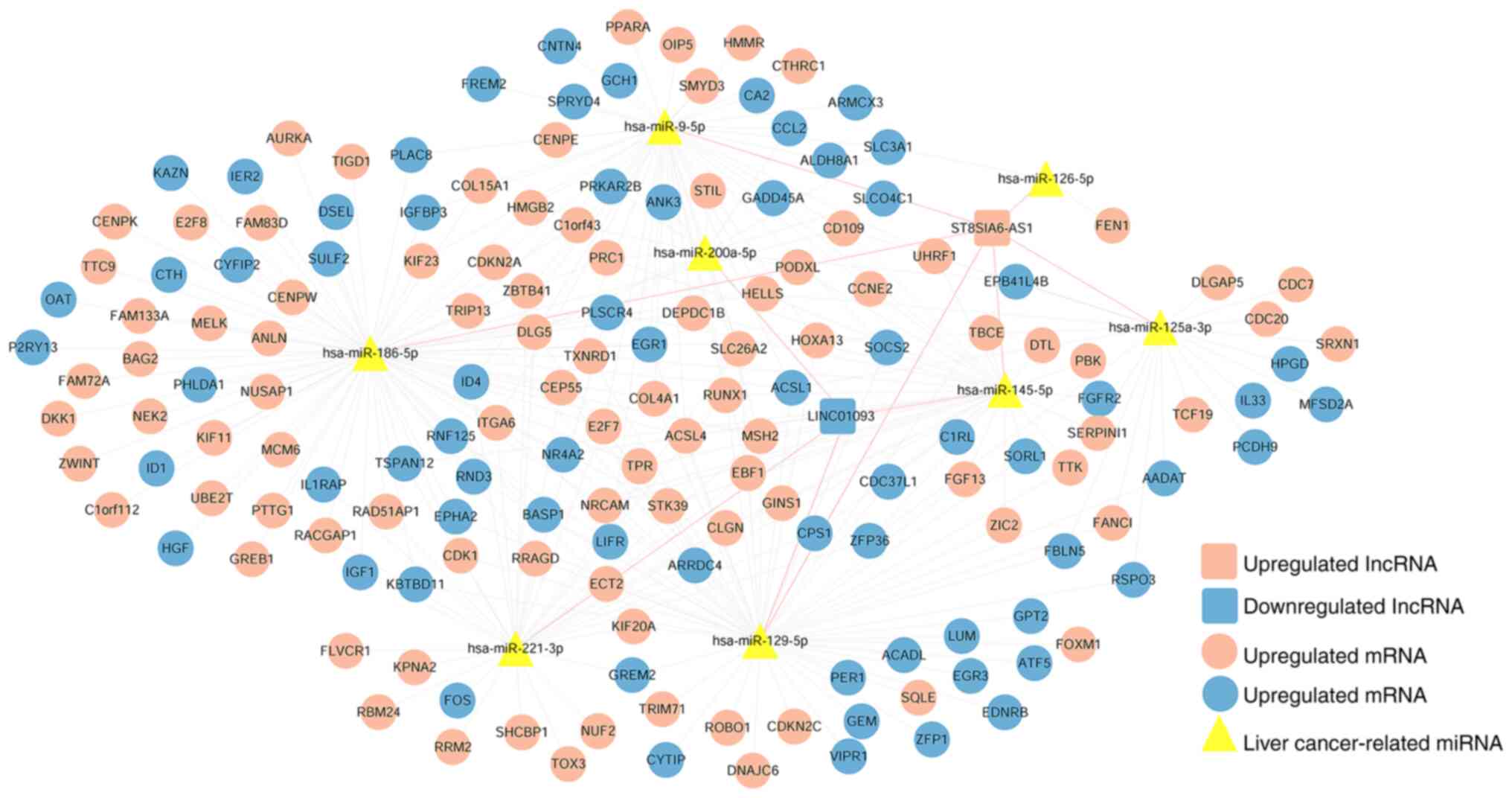
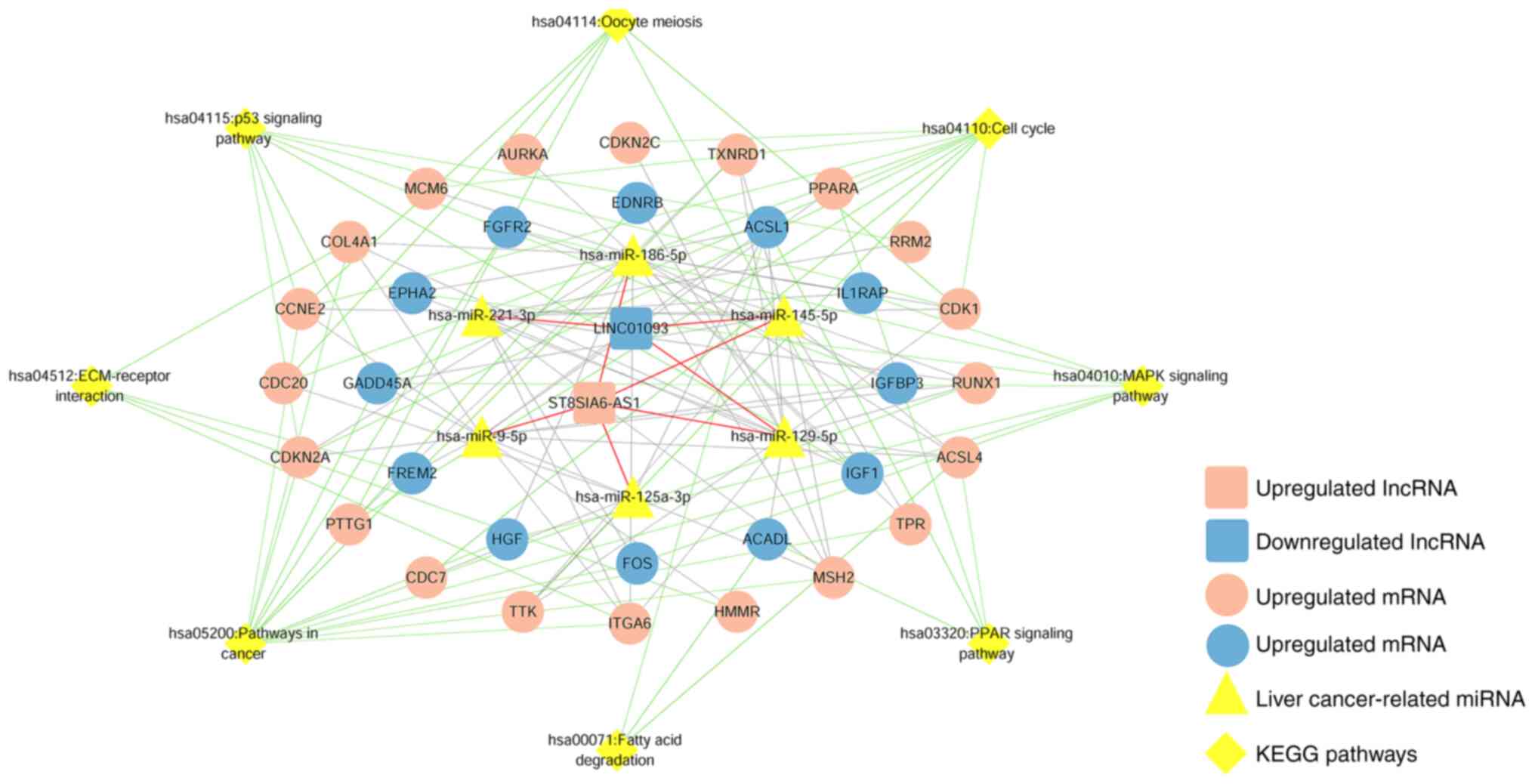
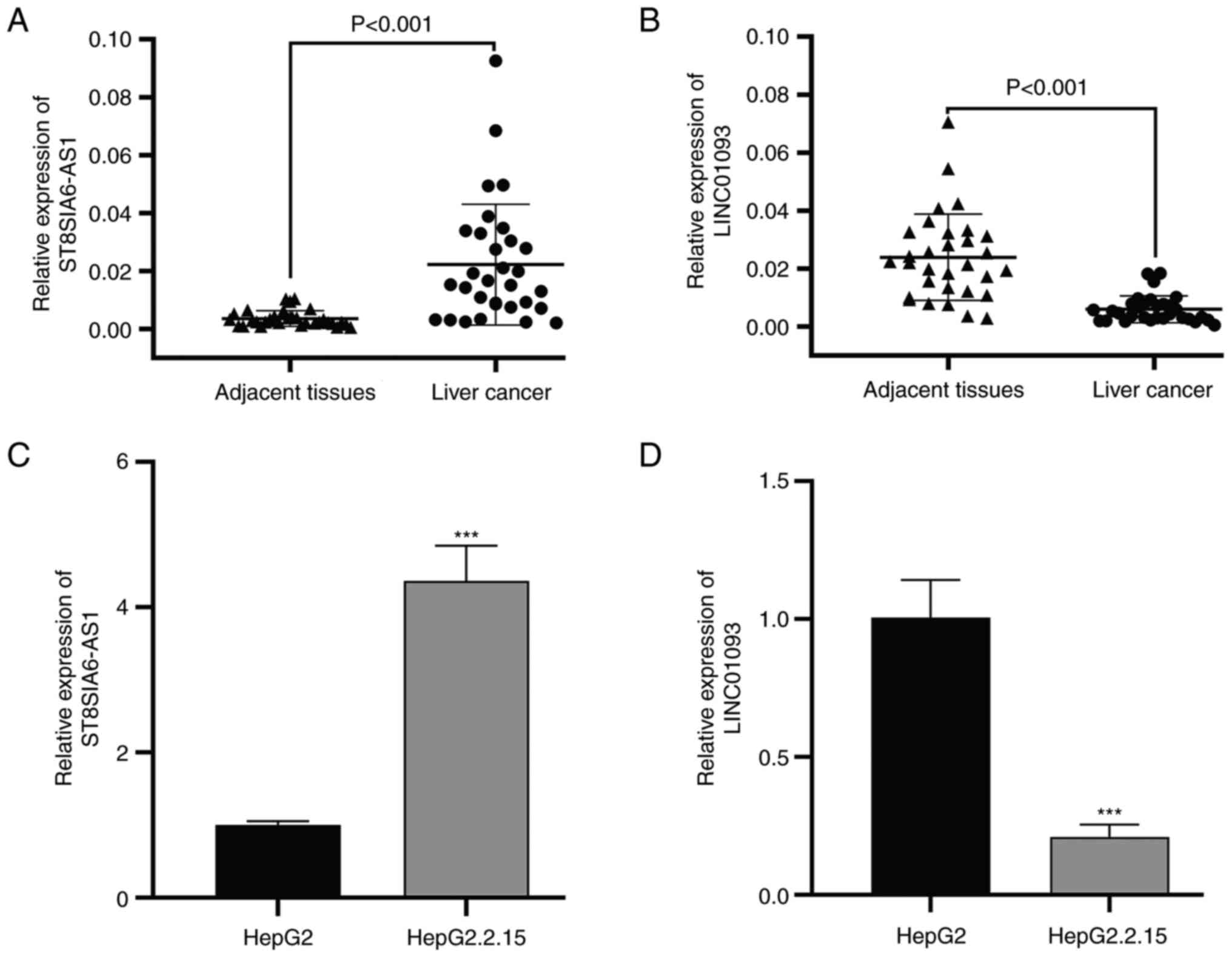
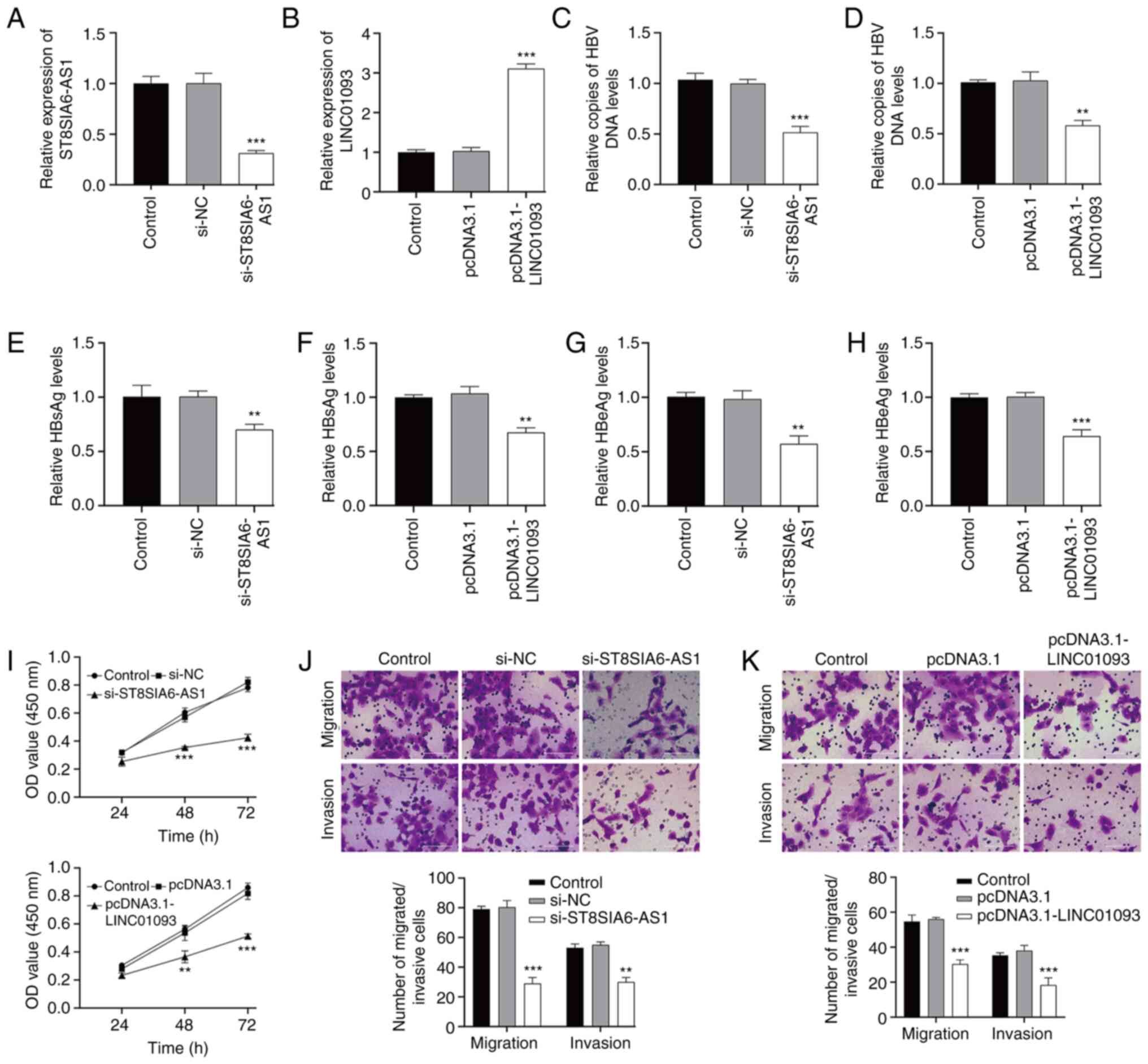
|
7
|
Li LM, Chen C, Ran RX, Huang JT, Sun HL,
Zeng C, Zhang Z, Zhang W and Liu SM: Loss of TARBP2 drives the
progression of hepatocellular carcinoma via miR-145-SERPINE1 axis.
Front Oncol. 11:6209122021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Qiu L, Wang T, Xu X, Wu Y, Tang Q and Chen
K: Long non-coding RNAs in hepatitis B virus-related hepatocellular
carcinoma: Regulation, functions, and underlying mechanisms. Int J
Mol Sci. 18:25052017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fortes P and Morris KV: Long noncoding
RNAs in viral infections. Virus Res. 212:1–11. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Stojic L, Lun ATL, Mascalchi P, Ernst C,
Redmond AM, Mangei J, Barr AR, Bousgouni V, Bakal C, Marioni JC, et
al: A high-content RNAi screen reveals multiple roles for long
noncoding RNAs in cell division. Nat Commun. 11:18512020.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hou Z, Xu X, Fu X, Tao S, Zhou J, Liu S
and Tan D: HBx-related long non-coding RNA MALAT1 promotes cell
metastasis via up-regulating LTBP3 in hepatocellular carcinoma. Am
J Cancer Res. 7:845–856. 2017.PubMed/NCBI
|
|
12
|
Tao L, Li D, Mu S, Tian G and Yan G:
LncRNA MAPKAPK5_AS1 facilitates cell proliferation in hepatitis B
virus-related hepatocellular carcinoma. Lab Invest. 102:494–504.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Deng Y, Wei Z, Huang M, Xu G, Wei W, Peng
B, Nong S and Qin H: Long non-coding RNA F11-AS1 inhibits
HBV-related hepatocellular carcinoma progression by regulating
NR1I3 via binding to microRNA-211-5p. J Cell Mol Med. 24:1848–1865.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Huang JL, Ren TY, Cao SW, Zheng SH, Hu XM,
Hu YW, Lin L, Chen J, Zheng L and Wang Q: HBx-related long
non-coding RNA DBH-AS1 promotes cell proliferation and survival by
activating MAPK signaling in hepatocellular carcinoma. Oncotarget.
6:33791–33804. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang H, Diab A, Fan H, Mani SKK,
Hullinger R, Merle P and Andrisani O: PLK1 and HOTAIR accelerate
proteasomal degradation of SUZ12 and ZNF198 during hepatitis B
virus-induced liver carcinogenesis. Cancer Res. 75:2363–2374. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang J, Yin G, Bian H, Yang J, Zhou P, Yan
K, Liu C, Chen P, Zhu J, Li Z and Xue T: LncRNA XIST upregulates
TRIM25 via negatively regulating miR-192 in hepatitis B
virus-related hepatocellular carcinoma. Mol Med. 27:412021.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu H, Zhao P, Jin X, Zhao Y, Chen Y, Yan
T, Wang J, Wu L and Sun Y: A 9lncRNA risk score system for
predicting the prognosis of patients with hepatitis B viruspositive
hepatocellular carcinoma. Mol Med Rep. 20:573–583. 2019.PubMed/NCBI
|
|
18
|
Wang SM, Ooi LL and Hui KM: Identification
and validation of a novel gene signature associated with the
recurrence of human hepatocellular carcinoma. Clin Cancer Res.
13:6275–6283. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Melis M, Diaz G, Kleiner DE, Zamboni F,
Kabat J, Lai J, Mogavero G, Tice A, Engle RE, Becker S, et al:
Viral expression and molecular profiling in liver tissue versus
microdissected hepatocytes in hepatitis B virus-associated
hepatocellular carcinoma. J Transl Med. 12:2302014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW,
Shi W and Smyth GK: limma powers differential expression analyses
for RNA-sequencing and microarray studies. Nucleic Acids Res.
43:e472015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Huang DW, Sherman BT and Lempicki RA:
Systematic and integrative analysis of large gene lists using DAVID
bioinformatics resources. Nat Protocols. 4:44–57. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang DW, Sherman BT and Lempicki RA:
Bioinformatics enrichment tools: Paths toward the comprehensive
functional analysis of large gene lists. Nucleic Acids Res.
37:1–13. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Deist TM, Dankers F, Valdes G, Wijsman R,
Hsu IC, Oberije C, Lustberg T, van Soest J, Hoebers F, Jochems A,
et al: Machine learning algorithms for outcome prediction in
(chemo) radiotherapy: An empirical comparison of classifiers. Med
Phys. 45:3449–3459. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Goeman JJ: L1 penalized estimation in the
Cox proportional hazards model. Biom J. 52:70–84. 2010.PubMed/NCBI
|
|
25
|
Tolosi L and Lengauer T: Classification
with correlated features: Unreliability of feature ranking and
solutions. Bioinformatics. 27:1986–1994. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wu J, Zhang H, Li L, Hu M, Chen L, Xu B
and Song Q: A nomogram for predicting overall survival in patients
with low-grade endometrial stromal sarcoma: A population-based
analysis. Cancer Commun (Lond). 40:301–312. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jiang Q, Wang Y, Hao Y, Juan L, Teng M,
Zhang X, Li M, Wang G and Liu Y: miR2Disease: A manually curated
database for microRNA deregulation in human disease. Nucleic Acids
Res. 37:D98–D104. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Paraskevopoulou MD, Vlachos IS, Karagkouni
D, Georgakilas G, Kanellos I, Vergoulis T, Zagganas K, Tsanakas P,
Floros E, Dalamagas T and Hatzigeorgiou AG: DIANA-LncBase v2:
Indexing microRNA targets on non-coding transcripts. Nucleic Acids
Res. 44:D231–D238. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li JH, Liu S, Zhou H, Qu LH and Yang JH:
starBase v2.0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA
interaction networks from large-scale CLIP-Seq data. Nucleic Acids
Res. 42:D92–D97. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shannon P, Markiel A, Ozier O, Baliga NS,
Wang JT, Ramage D, Amin N, Schwikowski B and Ideker T: Cytoscape: A
software environment for integrated models of biomolecular
interaction networks. Genome Res. 13:2498–2504. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ridruejo E: Does hepatitis B virus therapy
reduce the risk of hepatocellular carcinoma? Expert Opin Drug Saf.
14:439–451. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bisceglie AMD: Hepatitis B and
hepatocellular carcinoma. Hepatology. 49:S56–S60. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chauhan R and Lahiri N: Tissue- and
serum-associated biomarkers of hepatocellular carcinoma. Biomarkers
Cancer. 8:37–55. 2016.PubMed/NCBI
|
|
35
|
Pettinelli P, Arendt BM, Teterina A,
McGilvray I, Comelli EM, Fung SK, Fischer SE and Allard JP: Altered
hepatic genes related to retinol metabolism and plasma retinol in
patients with non-alcoholic fatty liver disease. PLoS One.
13:e02057472018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Liping X, Jia L, Qi C, Liang Y, Dongen L
and Jianshuai J: Cell Cycle Genes are potential diagnostic and
prognostic biomarkers in hepatocellular carcinoma. Biomed Res Int.
2020:62061572020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lee CH, Olson P and Evans RM: Minireview:
Lipid metabolism, metabolic diseases, and peroxisome
proliferator-activated receptors. Endocrinology. 144:2201–2207.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Oyefiade A, Erdman L, Goldenberg A, Malkin
D, Bouffet E, Taylor MD, Ramaswamy V, Scantlebury N and Law N: PPAR
and GST polymorphisms may predict changes in intellectual
functioning in medulloblastoma survivors. J Neurooncol. 142:39–48.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Chen SZ, Ling Y, Yu LX, Song YT, Chen XF,
Cao QQ, Yu H, Chen C, Tang JJ, Fan ZC, et al: 4-phenylbutyric acid
promotes hepatocellular carcinoma via initiating cancer stem cells
through activation of PPAR-α. Clin Transl Med. 11:e3792021.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang Y, Zhu P, Luo J, Wang J, Liu Z, Wu W,
Du Y, Ye B, Wang D, He L, et al: LncRNA HAND2-AS1 promotes liver
cancer stem cell self-renewal via BMP signaling. EMBO J.
38:e1011102019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Jing GY, Zheng XZ and Ji XX: lncRNA
HAND2-AS1 overexpression inhibits cancer cell proliferation in
hepatocellular carcinoma by downregulating RUNX2 expression. J Clin
Lab Anal. 35:e237172021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Fei Q, Song F, Jiang X, Hong H, Xu X, Jin
Z, Zhu X, Dai B, Yang J, Sui C and Xu M: LncRNA ST8SIA6-AS1
promotes hepatocellular carcinoma cell proliferation and resistance
to apoptosis by targeting miR-4656/HDAC11 axis. Cancer Cell Int.
20:2322020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Zhang X, Xu S, Hu C, Fang K, Zhou J, Guo
Z, Zhu G and Li L: LncRNA ST8SIA6-AS1 promotes hepatocellular
carcinoma progression by regulating MAGEA3 and DCAF4L2 expression.
Biochem Biophys Res Commun. 533:1039–1047. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yin Z, Ma T, Yan J, Shi N, Zhang C, Lu X,
Hou B and Jian Z: LncRNA MAGI2-AS3 inhibits hepatocellular
carcinoma cell proliferation and migration by targeting the
miR-374b-5p/SMG1 signaling pathway. J Cell Physiol.
234:18825–18836. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Fang G, Wang J, Sun X, Xu R, Zhao X, Shao
L, Sun C and Wang Y: LncRNA MAGI2-AS3 is downregulated in the
distant recurrence of hepatocellular carcinoma after surgical
resection and affects migration and invasion via ROCK2. Ann
Hepatol. 19:535–540. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wei H, Tang Q, Wang A, Zhang Y, Qin Z, Li
W, Xu Z, Wang J and Pu J: lncRNA MAGI2-AS3 exerts antioncogenic
roles in hepatocellular carcinoma via regulating the
miR-519c-3p/TXNIP Axis. J Oncol. 2021:55473452021. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zhang L, Wang Y, Sun J, Ma H and Guo C:
LINC00205 promotes proliferation, migration and invasion of HCC
cells by targeting miR-122-5p. Pathol Res Pract. 215:1525152019.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Cheng T, Yao Y, Zhang S, Zhang XN, Zhang
AH, Yang W and Hou CZ: LINC00205, a YY1-modulated lncRNA, serves as
a sponge for miR-26a-5p facilitating the proliferation of
hepatocellular carcinoma cells by elevating CDK6. Eur Rev Med
Pharmacol Sci. 25:6208–6219. 2021.PubMed/NCBI
|
|
49
|
Liu C, Tang L, Xu M, Lin Y, Shen J, Zhou
L, Ho L, Lu J and Ai X: LncRNA RUSC1-AS1 contributes to the
progression of hepatocellular carcinoma cells by modulating
miR-340-5p/CREB1 axis. Am J Transl Res. 13:1022–1036.
2021.PubMed/NCBI
|
|
50
|
He J, Zuo Q, Hu B, Jin H, Wang C, Cheng Z,
Deng X, Yang C, Ruan H, Yu C, et al: A novel, liver-specific long
noncoding RNA LINC01093 suppresses HCC progression by interaction
with IGF2BP1 to facilitate decay of GLI1 mRNA. Cancer Lett.
450:98–109. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Qin SJ, Zhou HZ, Xu NS, Yang HC and Chen
PX: The diagnostic value of serum ST8SIA6-AS1 as biomarker in
hepatocellular carcinoma. Clin Lab. 66:doi:
10.7754/Clin.Lab.2020.200231. 2020. View Article : Google Scholar
|
|
52
|
Kuai J, Zheng L, Yi X, Liu Z, Qiu B, Lu Z
and Jiang Y: ST8SIA6-AS1 promotes the development of hepatocellular
carcinoma cells through miR-338-3p/NONO axis. Digestive Liver Dis.
53:1192–1200. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Zhang B, Liu Z, Liu J, Cao K, Shan W, Wen
Q and Wang R: Long non-coding RNA ST8SIA6-AS1 promotes the
migration and invasion of hypoxia-treated hepatocellular carcinoma
cells through the miR-338/MEPCE axis. Oncol Rep. 45:73–82. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Mou Y, Wang D, Xing R, Nie H, Mou Y, Zhang
Y and Zhou X: Identification of long noncoding RNAs biomarkers in
patients with hepatitis B virus-associated hepatocellular
carcinoma. Cancer Biomark. 23:95–106. 2018. View Article : Google Scholar : PubMed/NCBI
|