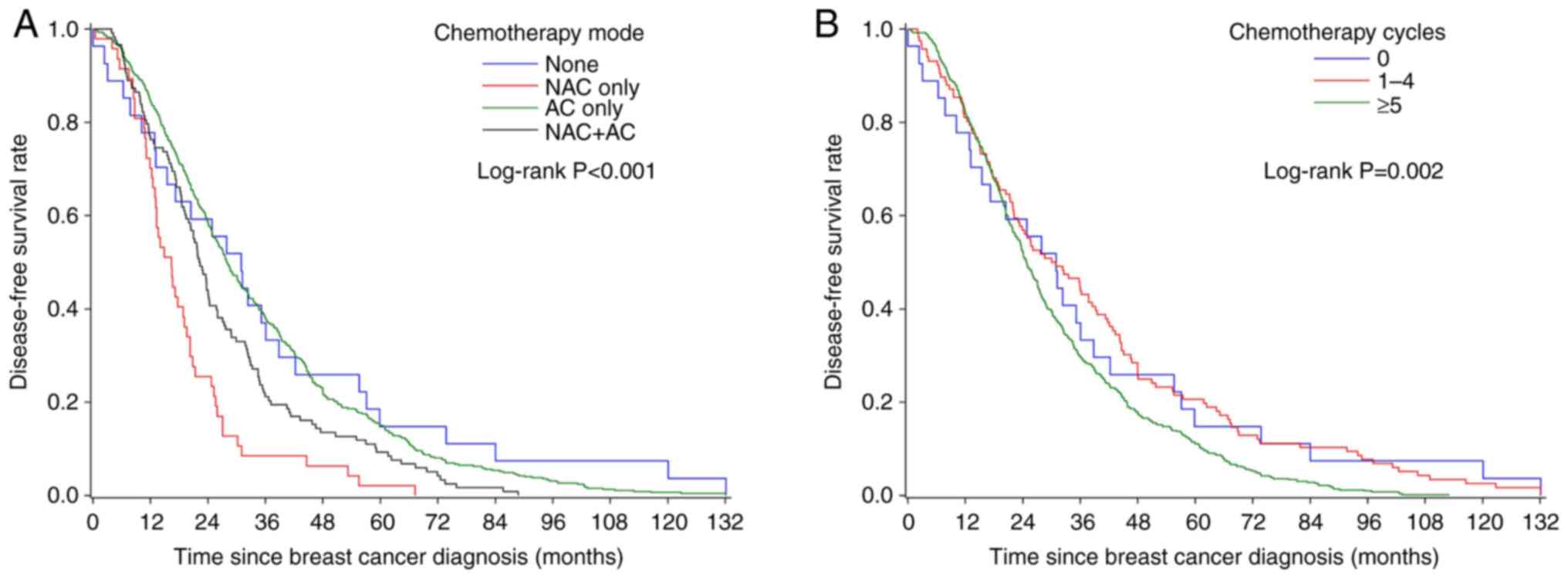
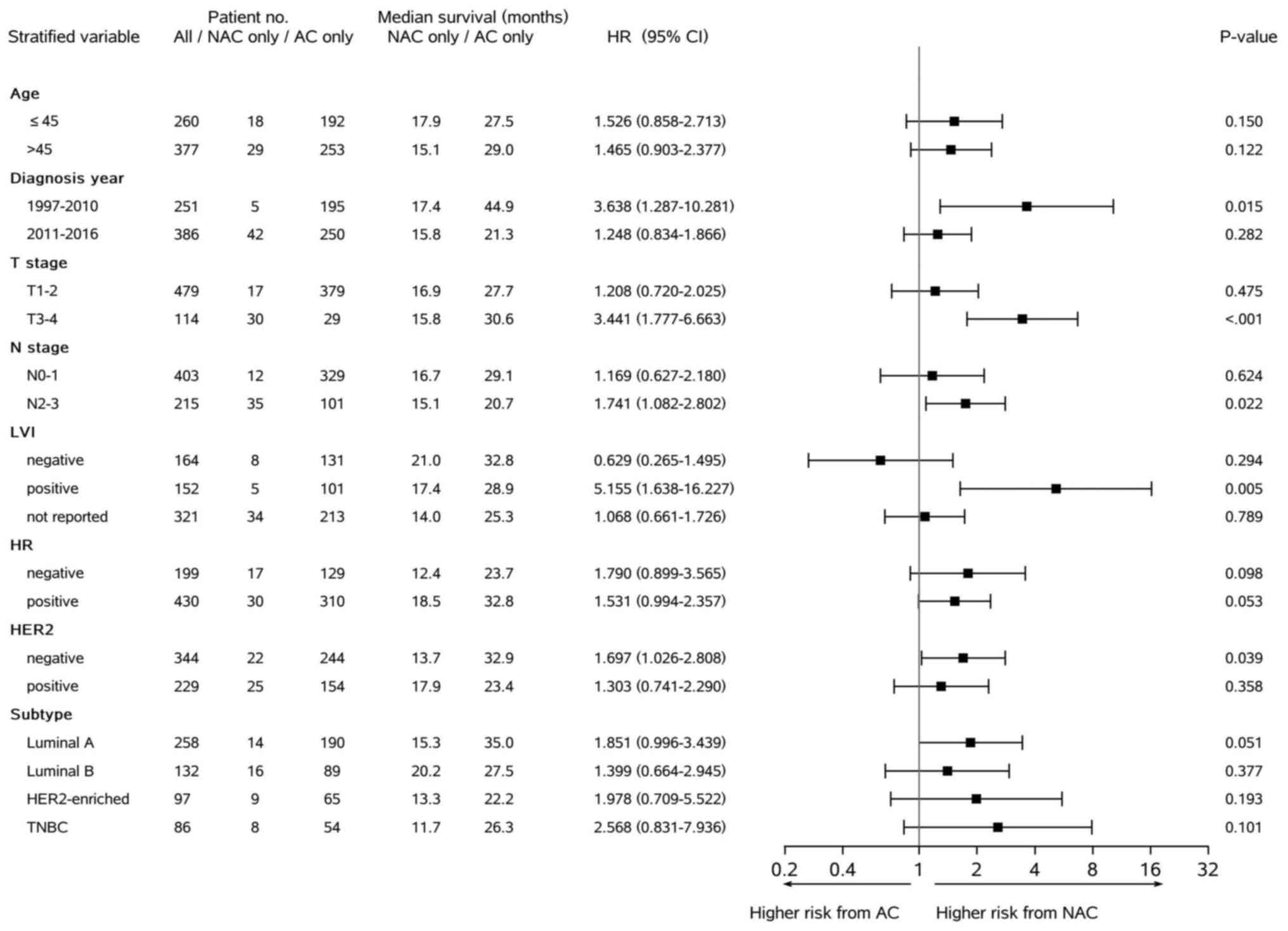
|
1
|
Mougalian SS, Soulos PR, Killelea BK,
Lannin DR, Abu-Khalaf MM, DiGiovanna MP, Sanft TB, Pusztai L, Gross
CP and Chagpar AB: Use of neoadjuvant chemotherapy for patients
with stage I to III breast cancer in the United States. Cancer.
121:2544–2552. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Caparica R, Brandão M and Piccart M:
Systemic treatment of patients with early breast cancer: Recent
updates and state of the art. Breast. 48 (Suppl 1):S7–S20. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Waks AG and Winer EP: Breast cancer
treatment: A review. JAMA. 321:288–300. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Fisher B, Brown A, Mamounas E, Wieand S,
Robidoux A, Margolese RG, Cruz AB Jr, Fisher ER, Wickerham DL,
Wolmark N, et al: Effect of preoperative chemotherapy on
local-regional disease in women with operable breast cancer:
Findings from national surgical adjuvant breast and bowel project
B-18. J Clin Oncol. 15:2483–2493. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Moreno-Aspitia A: Neoadjuvant therapy in
early-stage breast cancer. Crit Rev Oncol Hematol. 82:187–199.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Early Breast Cancer Trialists'
Collaborative Group (EBCTCG), . Long-term outcomes for neoadjuvant
versus adjuvant chemotherapy in early breast cancer: Meta-analysis
of individual patient data from ten randomised trials. Lancet
Oncol. 19:27–39. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Masuda N, Lee SJ, Ohtani S, Im YH, Lee ES,
Yokota I, Kuroi K, Im SA, Park BW, Kim SB, et al: Adjuvant
capecitabine for breast cancer after preoperative chemotherapy. N
Engl J Med. 376:2147–2159. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Murphy BL, Day CN, Hoskin TL, Habermann EB
and Boughey JC: Neoadjuvant chemotherapy use in breast cancer is
greatest in excellent responders: Triple-negative and HER2+
subtypes. Ann Surg Oncol. 25:2241–2248. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
von Minckwitz G, Huang CS, Mano MS, Loibl
S, Mamounas EP, Untch M, Wolmark N, Rastogi P, Schneeweiss A,
Redondo A, et al: Trastuzumab emtansine for residual invasive
HER2-positive breast cancer. N Engl J Med. 380:617–628. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Vaidya JS, Massarut S, Vaidya HJ,
Alexander EC, Richards T, Caris JA, Sirohi B and Tobias JS:
Rethinking neoadjuvant chemotherapy for breast cancer. BMJ.
360:j59132018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Reyal F, Hamy AS and Piccart MJ:
Neoadjuvant treatment: The future of patients with breast cancer.
ESMO Open. 3:e0003712018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fisher B, Bryant J, Wolmark N, Mamounas E,
Brown A, Fisher ER, Wickerham DL, Begovic M, DeCillis A, Robidoux
A, et al: Effect of preoperative chemotherapy on the outcome of
women with operable breast cancer. J Clin Oncol. 16:2672–2685.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rastogi P, Anderson SJ, Bear HD, Geyer CE,
Kahlenberg MS, Robidoux A, Margolese RG, Hoehn JL, Vogel VG, Dakhil
SR, et al: Preoperative chemotherapy: updates of national surgical
adjuvant breast and bowel project protocols B-18 and B-27. J Clin
Oncol. 26:778–785. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mauri D, Pavlidis N and Ioannidis JP:
Neoadjuvant versus adjuvant systemic treatment in breast cancer: A
meta-analysis. J Natl Cancer Inst. 97:188–194. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang J, Xiu BQ, Guo R, Yang BL, Zhang Q,
Su YH, Li L, Ji WR, Zhang YY, Cao AY, et al: Current trend of
breast cancer neoadjuvant treatment in China: A cross-sectional
study. Zhonghua Zhong Liu Za Zhi. 42:931–936. 2020.(In Chinese).
PubMed/NCBI
|
|
16
|
Edge SB and Compton CC: The American joint
committee on cancer: the 7th edition of the AJCC cancer staging
manual and the future of TNM. Ann Surg Oncol. 17:1471–1474. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Senkus E, Kyriakides S, Ohno S,
Penault-Llorca F, Poortmans P, Rutgers E, Zackrisson S and Cardoso
F; ESMO Guidelines Committee, . Primary breast cancer: ESMO
clinical practice guidelines for diagnosis, treatment and
follow-up. Ann Oncol. 26 (Suppl 5):v8–v30. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
National Comprehensive Cancer Network
(NCCN), . Clinical Practice Guideline in Oncology. Breast Cancer
version 1.2021. https://www.nccn.org/professionals/physician_gls/pdf/breast.pdfMarch
20–2021
|
|
19
|
Denduluri N, Chavez-MacGregor M, Telli ML,
Eisen A, Graff SL, Hassett MJ, Holloway JN, Hurria A, King TA,
Lyman GH, et al: Selection of optimal adjuvant chemotherapy and
targeted therapy for early breast cancer: ASCO clinical practice
guideline focused update. J Clin Oncol. 36:2433–2443. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Poggio F, Bruzzone M, Ceppi M, Pondé NF,
La Valle G, Del Mastro L, de Azambuja E and Lambertini M:
Platinum-based neoadjuvant chemotherapy in triple-negative breast
cancer: A systematic review and meta-analysis. Ann Oncol.
29:1497–1508. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Association of Breast Surgery at Baso
2009, . Surgical guidelines for the management of breast cancer.
Eur J Surg Oncol. 35 (Suppl 1):S1–S22. 2009. View Article : Google Scholar
|
|
22
|
Early Breast Cancer Trialists'
Collaborative Group (EBCTCG), . Effects of chemotherapy and
hormonal therapy for early breast cancer on recurrence and 15-year
survival: An overview of the randomised trials. Lancet.
365:1687–1717. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Berry DA, Cirrincione C, Henderson IC,
Citron ML, Budman DR, Goldstein LJ, Martino S, Perez EA, Muss HB,
Norton L, et al: Estrogen-receptor status and outcomes of modern
chemotherapy for patients with node-positive breast cancer. JAMA.
295:1658–1667. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Pondé NF, Zardavas D and Piccart M:
Progress in adjuvant systemic therapy for breast cancer. Nat Rev
Clin Oncol. 16:27–44. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Early Breast Cancer Trialists'
Collaborative Group (EBCTCG), . Increasing the dose intensity of
chemotherapy by more frequent administration or sequential
scheduling: A patient-level meta-analysis of 37 298 women with
early breast cancer in 26 randomised trials. Lancet. 393:1440–1452.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Harris L, Fritsche H, Mennel R, Norton L,
Ravdin P, Taube S, Somerfield MR, Hayes DF and Bast RC Jr; American
Society of Clinical Oncology, . American society of clinical
oncology 2007 update of recommendations for the use of tumor
markers in breast cancer. J Clin Oncol. 25:5287–5312. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sparano JA and Paik S: Development of the
21-gene assay and its application in clinical practice and clinical
trials. J Clin Oncol. 26:721–728. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sparano JA, Gray RJ, Makower DF, Pritchard
KI, Albain KS, Hayes DF, Geyer CE Jr, Dees EC, Perez EA, Olson JA
Jr, et al: Prospective validation of a 21-gene expression assay in
breast cancer. N Engl J Med. 373:2005–2014. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cooke T, Reeves J, Lanigan A and Stanton
P: HER2 as a prognostic and predictive marker for breast cancer.
Ann Oncol. 12 (Suppl 1):S23–S28. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Caparica R, Lambertini M, Pondé N,
Fumagalli D, de Azambuja E and Piccart M: Post-neoadjuvant
treatment and the management of residual disease in breast cancer:
State of the art and perspectives. Ther Adv Med Oncol.
11:17588359198277142019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bagegni NA, Tao Y and Ademuyiwa FO:
Clinical outcomes with neoadjuvant versus adjuvant chemotherapy for
triple negative breast cancer: A report from the national cancer
database. PLoS One. 14:e02223582019. View Article : Google Scholar : PubMed/NCBI
|