|
1
|
Friedman AA, Letai A, Fisher DE and
Flaherty KT: Precision medicine for cancer with next-generation
functional diagnostics. Nat Rev Cancer. 15:747–756. 2015.
View Article : Google Scholar : PubMed/NCBI
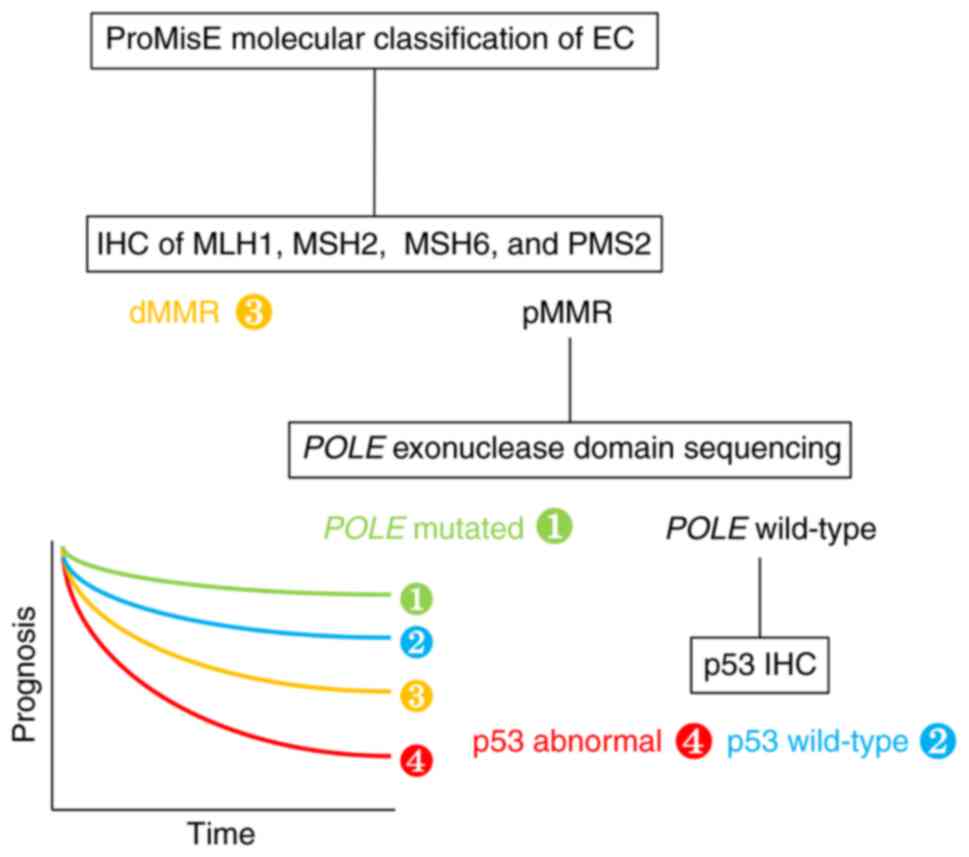
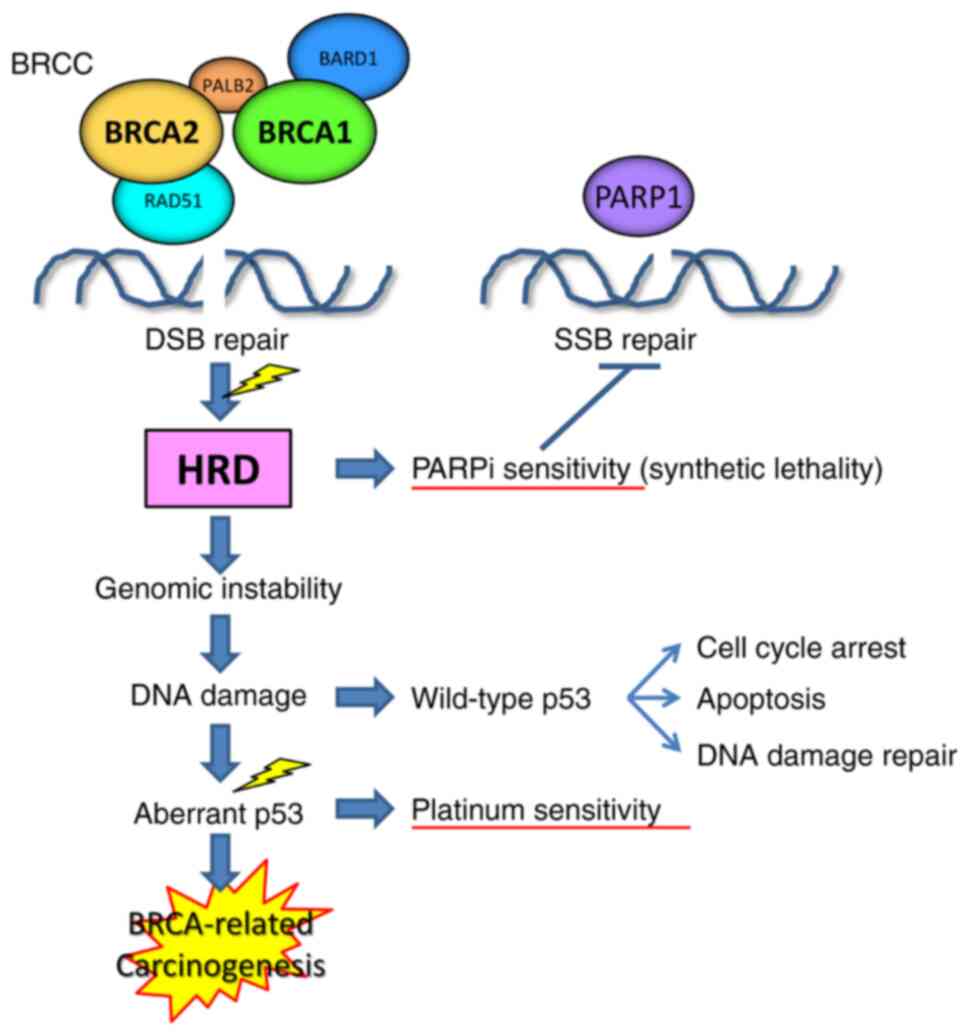
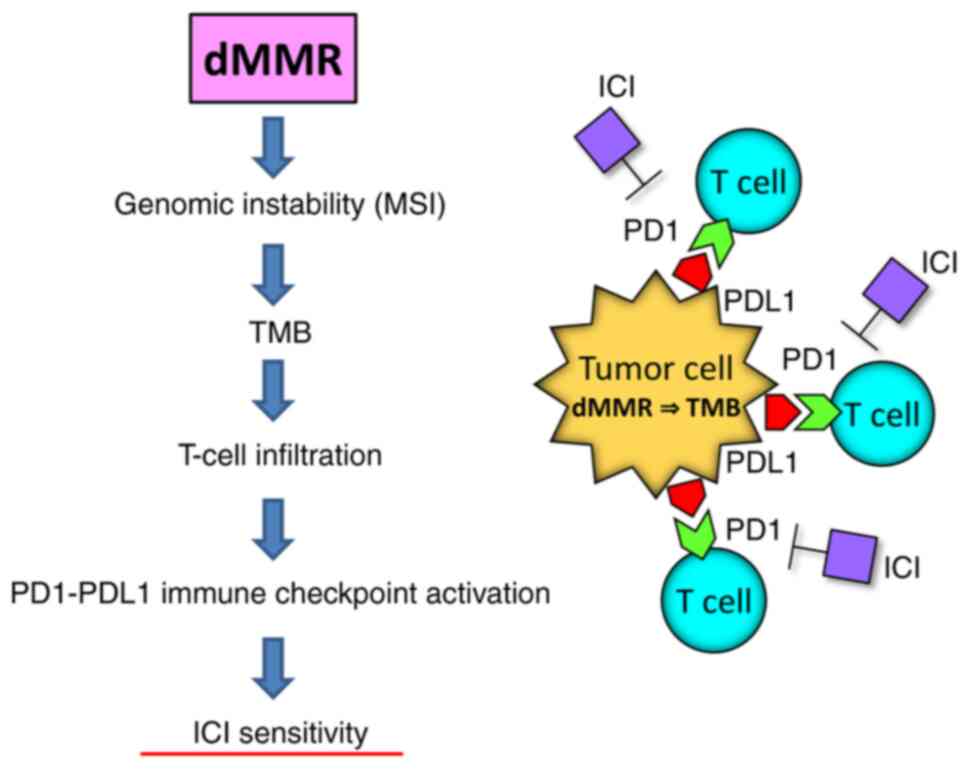
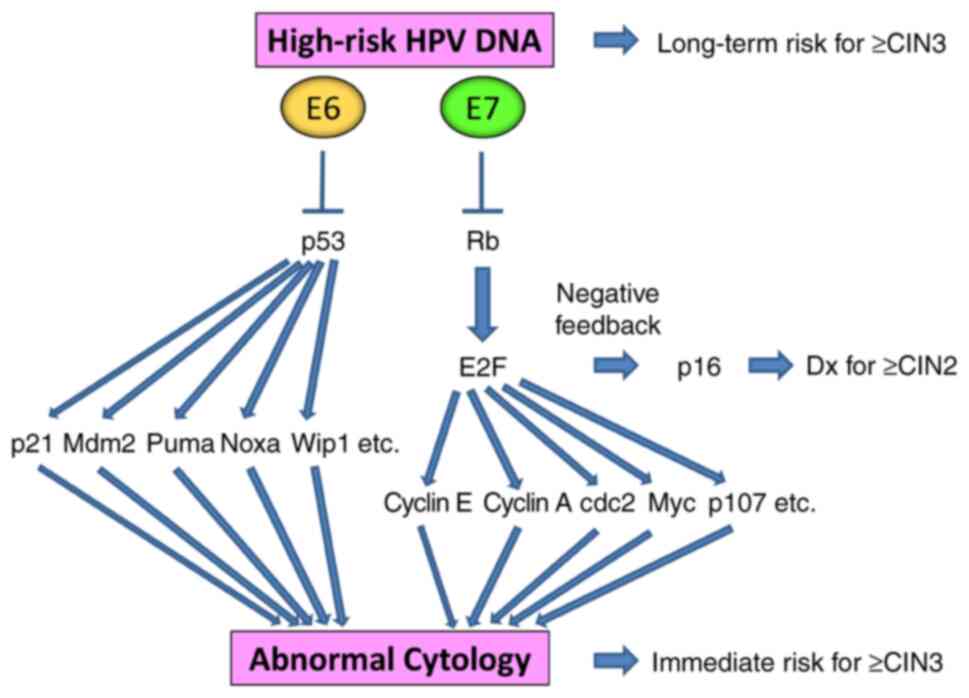
|
|
2
|
Yang W, Lei C, Song S, Jing W, Jin C, Gong
S, Tian H and Guo T: Immune checkpoint blockade in the treatment of
malignant tumor: Current statue and future strategies. Cancer Cell
Int. 21:5892021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Herzog TJ, Vergote I, Gomella LG,
Milenkova T, French T, Tonikian R, Poehlein C and Hussain M:
Testing for homologous recombination repair or homologous
recombination deficiency for poly (ADP-ribose) polymerase
inhibitors: A current perspective. Eur J Cancer. 179:136–146. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bokhman JV: Two pathogenetic types of
endometrial carcinoma. Gynecol Oncol. 15:10–17. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Murali R, Soslow RA and Weigelt B:
Classification of endometrial carcinoma: More than two types.
Lancet Oncol. 15:e268–e278. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cancer Genome Atlas Research Network, .
Kandoth C, Schultz N, Cherniack AD, Akbani R, Liu Y, Shen H,
Robertson AG, Pashtan I, Shen R, et al: Integrated genomic
characterization of endometrial carcinoma. Nature. 497:67–73. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Talhouk A, McConechy MK, Leung S, Li-Chang
HH, Kwon JS, Melnyk N, Yang W, Senz J, Boyd N, Karnezis AN, et al:
A clinically applicable molecular-based classification for
endometrial cancers. Br J Cancer. 113:299–310. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Talhouk A, McConechy MK, Leung S, Yang W,
Lum A, Senz J, Boyd N, Pike J, Anglesio M, Kwon JS, et al:
Confirmation of ProMisE: A simple, genomics-based clinical
classifier for endometrial cancer. Cancer. 123:802–813. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
PORTEC-4a. Molecular profile-based versus
standard adjuvant radiotherapy in endometrial cancer (PORTEC-4a).
https://beta.clinicaltrials.gov/study/NCT03469674?cond=NCT03469674&rank=1February
25–2023
|
|
10
|
Tailored Adjuvant Therapy in POLE-mutated
and p53-wildtype Early Stage Endometrial Cancer (TAPER). https://beta.clinicaltrials.gov/study/NCT04705649?cond=NCT04705649&rank=1February
25–2023
|
|
11
|
Ghezelayagh TS, Pennington KP, Norquist
BM, Khasnavis N, Radke MR, Kilgore MR, Garcia RL, Lee M, Katz R,
Leslie KK, et al: Characterizing TP53 mutations in ovarian
carcinomas with and without concurrent BRCA1 or BRCA2 mutations.
Gynecol Oncol. 160:786–792. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fong PC, Boss DS, Yap TA, Tutt A, Wu P,
Mergui-Roelvink M, Mortimer P, Swaisland H, Lau A, O'Connor MJ, et
al: Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA
mutation carriers. N Engl J Med. 361:123–134. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fong PC, Yap TA, Boss DS, Carden CP,
Mergui-Roelvink M, Gourley C, De Greve J, Lubinski J, Shanley S,
Messiou C, et al: Poly(ADP)-ribose polymerase inhibition: Frequent
durable responses in BRCA carrier ovarian cancer correlating with
platinum-free interval. J Clin Oncol. 28:2512–2519. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ray-Coquard I, Pautier P, Pignata S, Pérol
D, González-Martín A, Berger R, Fujiwara K, Vergote I, Colombo N,
Mäenpää J, et al: Olaparib plus Bevacizumab as first-line
maintenance in ovarian cancer. N Engl J Med. 381:2416–2428. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Moore KN, Secord AA, Geller MA, Miller DS,
Cloven N, Fleming GF, Hendrickson AEW, Azodi M, DiSilvestro P, Oza
AM, et al: Niraparib monotherapy for late-line treatment of ovarian
cancer (QUADRA): A multicentre, open-label, single-arm, phase 2
trial. Lancet Oncol. 20:636–648. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
de Boer SM, Powell ME, Mileshkin L,
Katsaros D, Bessette P, Haie-Meder C, Ottevanger PB, Ledermann JA,
Khaw P, Colombo A, et al: Adjuvant chemoradiotherapy versus
radiotherapy alone for women with high-risk endometrial cancer
(PORTEC-3): Final results of an international, open-label,
multicentre, randomised, phase 3 trial. Lancet Oncol. 19:295–309.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Leon-Castillo A, de Boer SM, Powell ME,
Mileshkin LR, Mackay HJ, Leary A, Nijman HW, Singh N, Pollock PM,
Bessette P, et al: Molecular classification of the PORTEC-3 trial
for high-risk endometrial cancer: Impact on prognosis and benefit
from adjuvant therapy. J Clin Oncol. 38:3388–3397. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ott PA, Bang YJ, Piha-Paul SA, Razak ARA,
Bennouna J, Soria JC, Rugo HS, Cohen RB, O'Neil BH, Mehnert JM, et
al: T-cell-inflamed gene-expression profile, programmed death
ligand 1 expression, and tumor mutational burden predict efficacy
in patients treated with pembrolizumab across 20 cancers:
KEYNOTE-028. J Clin Oncol. 37:318–327. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ayers M, Lunceford J, Nebozhyn M, Murphy
E, Loboda A, Kaufman DR, Albright A, Cheng JD, Kang SP, Shankaran
V, et al: IFN-γ-related mRNA profile predicts clinical response to
PD-1 blockade. J Clin Invest. 127:2930–2940. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Marabelle A, Fakih M, Lopez J, Shah M,
Shapira-Frommer R, Nakagawa K, Chung HC, Kindler HL, Lopez-Martin
JA, Miller WH Jr, et al: Association of tumour mutational burden
with outcomes in patients with advanced solid tumours treated with
pembrolizumab: Prospective biomarker analysis of the multicohort,
open-label, phase 2 KEYNOTE-158 study. Lancet Oncol. 21:1353–1365.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Makker V, Colombo N, Herraez AC, Santin
AD, Colomba E, Miller DS, Fujiwara K, Pignata S, Baron-Hay S,
Ray-Coquard I, et al: Lenvatinib plus Pembrolizumab for advanced
endometrial cancer. N Engl J Med. 386:437–448. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Colombo N, Dubot C, Lorusso D, Caceres MV,
Hasegawa K, Shapira-Frommer R, Tewari KS, Salman P, Usta EH, Yañez
E, et al: Pembrolizumab for persistent, recurrent, or metastatic
cervical cancer. N Engl J Med. 385:1856–1867. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Schiffman M, Kinney WK, Cheung LC, Gage
JC, Fetterman B, Poitras NE, Lorey TS, Wentzensen N, Befano B,
Schussler J, et al: Relative performance of HPV and cytology
components of cotesting in cervical screening. J Natl Cancer Inst.
110:501–508. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Perkins RB, Guido RS, Castle PE, Chelmow
D, Einstein MH, Garcia F, Huh WK, Kim JJ, Moscicki AB, Nayar R, et
al: 2019 ASCCP risk-based management consensus guidelines for
abnormal cervical cancer screening tests and cancer precursors. J
Low Genit Tract Dis. 24:102–131. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Serrano M, Hannon GJ and Beach D: A new
regulatory motif in cell-cycle control causing specific inhibition
of cyclin D/CDK4. Nature. 366:704–707. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Galgano MT, Castle PE, Atkins KA, Brix WK,
Nassau SR and Stoler MH: Using biomarkers as objective standards in
the diagnosis of cervical biopsies. Am J Surg Pathol. 34:1077–1087.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bergeron C, Ordi J, Schmidt D, Trunk MJ,
Keller T and Ridder R; European CINtec Histology Study Group, :
Conjunctive p16INK4a testing significantly increases accuracy in
diagnosing high-grade cervical intraepithelial neoplasia. Am J Clin
Pathol. 133:395–406. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kuchenbaecker KB, Hopper JL, Barnes DR,
Phillips KA, Mooij TM, Roos-Blom MJ, Jervis S, van Leeuwen FE,
Milne RL, Andrieu N, et al: Risks of breast, ovarian, and
contralateral breast cancer for BRCA1 and BRCA2 mutation carriers.
JAMA. 317:2402–2416. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Antoniou A, Pharoah PD, Narod S, Risch HA,
Eyfjord JE, Hopper JL, Loman N, Olsson H, Johannsson O, Borg A, et
al: Average risks of breast and ovarian cancer associated with
BRCA1 or BRCA2 mutations detected in case series unselected for
family history: A combined analysis of 22 studies. Am J Hum Genet.
72:1117–1130. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chen S and Parmigiani G: Meta-analysis of
BRCA1 and BRCA2 penetrance. J Clin Oncol. 25:1329–1333. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen J, Bae E, Zhang L, Hughes K,
Parmigiani G, Braun D and Rebbeck TR: Penetrance of breast and
ovarian cancer in women who carry a BRCA1/2 mutation and do not use
risk-reducing salpingo-oophorectomy: An updated meta-analysis. JNCI
Cancer Spectr. 4:pkaa0292020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pinsky PF, Yu K, Kramer BS, Black A, Buys
SS, Partridge E, Gohagan J, Berg CD and Prorok PC: Extended
mortality results for ovarian cancer screening in the PLCO trial
with median 15years follow-up. Gynecol Oncol. 143:270–275. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jacobs IJ, Menon U, Ryan A, Gentry-Maharaj
A, Burnell M, Kalsi JK, Amso NN, Apostolidou S, Benjamin E,
Cruickshank D, et al: Ovarian cancer screening and mortality in the
UK collaborative trial of ovarian cancer screening (UKCTOCS): A
randomised controlled trial. Lancet. 387:945–956. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Marchetti C, De Felice F, Perniola G,
Lecce F, Vertechy L, Monti M, Musio D, Muzii L, Tombolini V and
Panici PB: Screening program in ovarian cancer: A logical step in
clinical management? A meta-analysis. Curr Probl Cancer.
42:235–240. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jacobs IJ, Skates SJ, MacDonald N, Menon
U, Rosenthal AN, Davies AP, Woolas R, Jeyarajah AR, Sibley K, Lowe
DG and Oram DH: Screening for ovarian cancer: A pilot randomised
controlled trial. Lancet. 353:1207–1210. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Henderson JT, Webber EM and Sawaya GF:
Screening for ovarian cancer: Updated evidence report and
systematic review for the US preventive services task force. JAMA.
319:595–606. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Finch AP, Lubinski J, Møller P, Singer CF,
Karlan B, Senter L, Rosen B, Maehle L, Ghadirian P, Cybulski C, et
al: Impact of oophorectomy on cancer incidence and mortality in
women with a BRCA1 or BRCA2 mutation. J Clin Oncol. 32:1547–1553.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Domchek SM, Friebel TM, Singer CF, Evans
DG, Lynch HT, Isaacs C, Garber JE, Neuhausen SL, Matloff E, Eeles
R, et al: Association of risk-reducing surgery in BRCA1 or BRCA2
mutation carriers with cancer risk and mortality. JAMA.
304:967–975. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
NCCN Guidelines Genetic/Familial High-risk
Assessment, . Breast, Ovarian, and Pancreatic. NCCN Clinical
Practice Guidelines in Oncology. 2023.https://www.nccn.org/guidelines/guidelines-detail?category=2&id=1503February
25–2023
|
|
40
|
Callahan MJ, Crum CP, Medeiros F,
Kindelberger DW, Elvin JA, Garber JE, Feltmate CM, Berkowitz RS and
Muto MG: Primary fallopian tube malignancies in BRCA-positive women
undergoing surgery for ovarian cancer risk reduction. J Clin Oncol.
25:3985–3990. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Powell CB, Kenley E, Chen LM, Crawford B,
McLennan J, Zaloudek C, Komaromy M, Beattie M and Ziegler J:
Risk-reducing salpingo-oophorectomy in BRCA mutation carriers: Role
of serial sectioning in the detection of occult malignancy. J Clin
Oncol. 23:127–132. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shaw PA, Rouzbahman M, Pizer ES, Pintilie
M and Begley H: Candidate serous cancer precursors in fallopian
tube epithelium of BRCA1/2 mutation carriers. Mod Pathol.
22:1133–1138. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Vang R and Shih IM: Serous tubal
intra-epithelial carcinoma: What do we really know at this point?
Histopathology. 81:542–555. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lopez F, Belloc F, Lacombe F, Dumain P,
Reiffers J, Bernard P and Boisseau MR: Modalities of synthesis of
Ki67 antigen during the stimulation of lymphocytes. Cytometry.
12:42–49. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Latham A, Srinivasan P, Kemel Y, Shia J,
Bandlamudi C, Mandelker D, Middha S, Hechtman J, Zehir A,
Dubard-Gault M, et al: Microsatellite instability is associated
with the presence of lynch syndrome pan-cancer. J Clin Oncol.
37:286–295. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Hendriks YM, de Jong AE, Morreau H, Tops
CM, Vasen HF, Wijnen JT, Breuning MH and Bröcker-Vriends AH:
Diagnostic approach and management of Lynch syndrome (hereditary
nonpolyposis colorectal carcinoma): A guide for clinicians. CA
Cancer J Clin. 56:213–225. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Crosbie EJ, Ryan NAJ, Arends MJ, Bosse T,
Burn J, Cornes JM, Crawford R, Eccles D, Frayling IM, Ghaem-Maghami
S, et al: The Manchester international consensus group
recommendations for the management of gynecological cancers in
Lynch syndrome. Genet Med. 21:2390–2400. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Giardiello FM, Allen JI, Axilbund JE,
Boland CR, Burke CA, Burt RW, Church JM, Dominitz JA, Johnson DA,
Kaltenbach T, et al: Guidelines on genetic evaluation and
management of Lynch syndrome: A consensus statement by the US
multi-society task force on colorectal cancer. Gastroenterology.
147:502–526. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Stoffel E, Mukherjee B, Raymond VM, Tayob
N, Kastrinos F, Sparr J, Wang F, Bandipalliam P, Syngal S and
Gruber SB: Calculation of risk of colorectal and endometrial cancer
among patients with Lynch syndrome. Gastroenterology.
137:1621–1627. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Dunlop MG, Farrington SM, Carothers AD,
Wyllie AH, Sharp L, Burn J, Liu B, Kinzler KW and Vogelstein B:
Cancer risk associated with germline DNA mismatch repair gene
mutations. Hum Mol Genet. 6:105–110. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Barrow E, Robinson L, Alduaij W, Shenton
A, Clancy T, Lalloo F, Hill J and Evans DG: Cumulative lifetime
incidence of extracolonic cancers in Lynch syndrome: A report of
121 families with proven mutations. Clin Genet. 75:141–149. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hampel H, Stephens JA, Pukkala E, Sankila
R, Aaltonen LA, Mecklin JP and de la Chapelle A: Cancer risk in
hereditary nonpolyposis colorectal cancer syndrome: Later age of
onset. Gastroenterology. 129:415–421. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Aarnio M, Sankila R, Pukkala E, Salovaara
R, Aaltonen LA, de la Chapelle A, Peltomäki P, Mecklin JP and
Järvinen HJ: Cancer risk in mutation carriers of
DNA-mismatch-repair genes. Int J Cancer. 81:214–218. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Schmeler KM, Lynch HT, Chen LM, Munsell
MF, Soliman PT, Clark MB, Daniels MS, White KG, Boyd-Rogers SG,
Conrad PG, et al: Prophylactic surgery to reduce the risk of
gynecologic cancers in the Lynch syndrome. N Engl J Med.
354:261–269. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
NCCN Guidelines, . Genetic/Familial
High-risk Assessment: Colorectal. NCCN Clinical Practice Guidelines
in Oncology 2023. https://www.nccn.org/guidelines/guidelines-detail?category=2&id=1436February
25–2023
|
|
56
|
Dong Y, Hakimi MA, Chen X, Kumaraswamy E,
Cooch NS, Godwin AK and Shiekhattar R: Regulation of BRCC, a
holoenzyme complex containing BRCA1 and BRCA2, by a
signalosome-like subunit and its role in DNA repair. Mol Cell.
12:1087–1099. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Christou CM and Kyriacou K: BRCA1 and its
network of interacting partners. Biology. 2:40–63. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Brugarolas J and Jacks T: Double
indemnity: p53, BRCA and cancer. p53 mutation partially rescues
developmental arrest in Brca1 and Brca2 null mice, suggesting a
role for familial breast cancer genes in DNA damage repair. Nat
Med. 3:721–722. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Patel KJ, Yu VP, Lee H, Corcoran A,
Thistlethwaite FC, Evans MJ, Colledge WH, Friedman LS, Ponder BA
and Venkitaraman AR: Involvement of Brca2 in DNA repair. Mol Cell.
1:347–357. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Xu X, Weaver Z, Linke SP, Li C, Gotay J,
Wang XW, Harris CC, Ried T and Deng CX: Centrosome amplification
and a defective G2-M cell cycle checkpoint induce genetic
instability in BRCA1 exon 11 isoform-deficient cells. Mol Cell.
3:389–395. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Kindelberger DW, Lee Y, Miron A, Hirsch
MS, Feltmate C, Medeiros F, Callahan MJ, Garner EO, Gordon RW,
Birch C, et al: Intraepithelial carcinoma of the fimbria and pelvic
serous carcinoma: Evidence for a causal relationship. Am J Surg
Pathol. 31:161–169. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Ahmed AA, Etemadmoghadam D, Temple J,
Lynch AG, Riad M, Sharma R, Stewart C, Fereday S, Caldas C, Defazio
A, et al: Driver mutations in TP53 are ubiquitous in high grade
serous carcinoma of the ovary. J Pathol. 221:49–56. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Dudley JC, Lin MT, Le DT and Eshleman JR:
microsatellite instability as a biomarker for PD-1 blockade. Clin
Cancer Res. 22:813–820. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Refining Adjuvant Treatment IN Endometrial
Cancer Based On Molecular Features (RAINBO). https://beta.clinicaltrials.gov/study/NCT05255653?cond=NCT05255653&rank=1February
25–2023
|