Introduction
Lorlatinib is a highly effective third-generation
oral tyrosine kinase inhibitor against anaplastic lymphoma kinase
(ALK). In cell experiments, lorlatinib was shown to be more
effective than second-generation inhibitors, and had the broadest
coverage of ALK-resistant mutations (1). Despite the resistance of previous ALK
inhibitors (first generation, second generation or both),
lorlatinib exhibits strong antitumor activity (2), particularly in terms of intracranial
activity in patients with central nervous system metastases
(including leptomeningeal metastases) (3). Lorlatinib has been approved by Food
and Drug Administration for use in patients with locally advanced
or metastatic non-small cell lung cancer (NSCLC) who were
previously treated with at least one ALK inhibitor (4). First- and second-generation ALK
inhibitors are available in oral capsule form (5,6).
Lorlatinib is administered as an oral tablet and no intravenous
administration preparation is available (7). In patients with esophageal
obstruction, it is unclear whether administration through a gastric
tube affects the drug efficacy.
Hyperprogression was first described in case reports
and retrospective studies of patients using immunosuppressive
checkpoint inhibitors (such as Nivolumab, Pembrolizumab), when
certain patients with cancer appeared to show accelerated tumor
growth after beginning immunotherapy (8–10).
Retrospective observational studies revealed that in patients with
epidermal growth factor receptor (EGFR) and ALK mutations,
first-line immune checkpoint inhibitors have a limited benefit and
increase the risk of tumor hyperprogression (11,12).
However, it remains unclear whether the efficacy of the targeted
drug is affected if an immune checkpoint inhibitor is used in
patients with the mutation, and if hyperprogression occurs because
of an inadequate assessment of the initial status.
The present case report describes a patient with
advanced NSCLC who was first evaluated as negative for gene
mutations, but developed hyperprogression combined with external
esophageal pressure stenosis after first-line chemotherapy combined
with immune checkpoint inhibitors. The ALK gene fusion mutation was
detected during reevaluation of the patient, and second-line
treatment via gastric tube injection of lorlatinib was effective.
In the present case, dysphagia and dyspnea improved after
treatment.
Case report
In a 49-year-old male patient who underwent a
physical examination at the Western Theater General Hospital
(Chengdu, China), a lung shadow with a size of 3.4×4.3×5.2 cm was
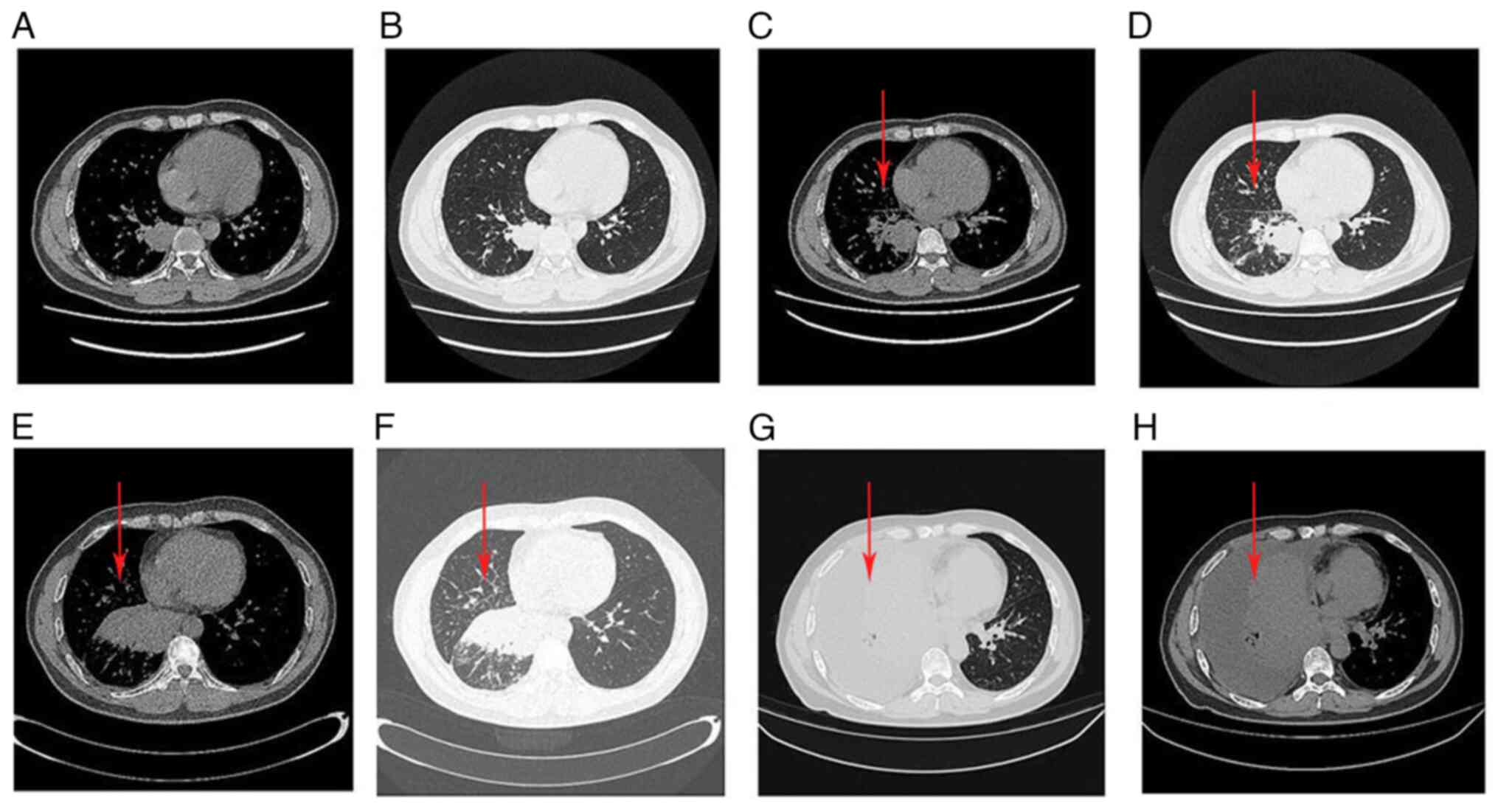
observed on computed tomography (CT) in September 2021 (Fig. 1A and B). However, the patient did
not recognize the severity of the disease. The patient was again
admitted to the General Hospital of Western Theater Command
(Chengdu, China) in January 2022 due to the presence of blood in
the sputum. Reexamination of the chest CT images of the patient
showed soft-tissue masses in the lower lobe of the right lung with
a size of 4.0×4.3×5.2 cm (Fig. 1C and
D). The pathological diagnosis of the patient was right lung
adenocarcinoma with mediastinum, right hilar lymph nodes, pleura
and bone metastases, stage IVB, based on CT-guided percutaneous
pulmonary aspiration. Blood sampling was performed for
next-generation sequencing (NGS) detection of lung cancer mutant
genes conducted externally at Accbio due to an insufficient amount
of biopsy tissue sample being available. Programmed death ligand 1
(PD-L1) expression was not detected, and mutations of EGFR, ALK,
ROS proto-oncogene 1 and MET were negative. A total of three cycles
(one cycle is 21 days) of chemotherapy consisting of pembrolizumab
200 mg, cisplatin 120 mg and pemetrexed disodium 800 mg injection
were administered in February, March and April 2022. A chest CT at
the end of May 2022 showed that the primary lesions were markedly
enlarged, new lung consolidation had occurred, and right lung
interstitial thickening with cancerous lymphangitis was more
evident than that in the previous CT scans (Fig. 1E and F). The symptoms of the
patient, such as cough and dyspnea, worsened, and hyperprogression
was considered. Accordingly, two cycles of the treatment regimen
consisting of cisplatin 120 mg, docetaxel 120 mg and bevacizumab
800 mg were administered. The patient's tumor continued to grow,
and considering that the rapid tumor growth may be caused by
immunosuppressive checkpoint inhibitors, pembrolizumab was
discontinued. The symptoms of the patient were not alleviated, and
the patient refused further treatment and did not return to the
clinic.
In October 18,2022, the patient visited the
physician again because of extreme dyspnea, complaining of nausea
and vomiting after drinking water and inability to eat. A chest CT
showed that the mass soft tissue density in the lower lobe of the
right lung had increased in the shadow range. In addition, right
lung interstitial thickening with carcinomatous lymphangitis was
more evident than previously and numerous pleural effusions were
detected on the right side (Fig. 1G and
H). The patient had a performance status score of 2 (13). Pleural effusion puncture and
drainage were performed, and adenocarcinoma cells were detected in
the pleural effusion. The pleural effusion was sent again for
genetic testing.
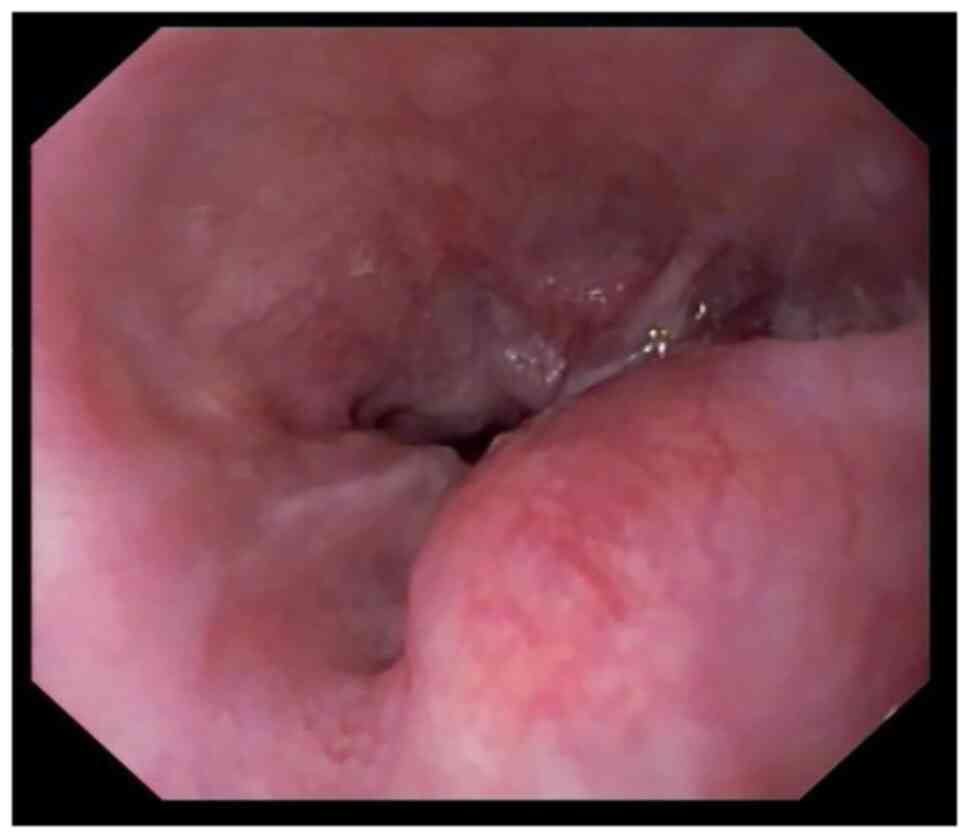
Gastroscopy revealed external pressure-associated
esophageal stenosis, which could not be passed by the gastroscope
(Fig. 2). A gastric tube was placed
under the guidance of a guide wire under gastroscopy and X-ray
imaging, and enteral nutrition support was provided through the
gastric tube. Next-generation sequencing of the posterior pleural
effusion showed that the cells were ALK (E13:A20) fusion
mutation-positive.
It was decided that ALK inhibitors were to be
administered, but the efficacy of administration through a gastric
tube was unknown. Both crizotinib, a first-generation ALK
inhibitor, and alectinib, a second-generation ALK inhibitor, are
available as capsules and cannot be dissolved in water for
injection through a gastric tube (5,6). The
third-generation inhibitor lorlatinib was administered to the
patient as a tablet in October 2022 with patient's consent. The
drug was dissolved in water at room temperature and injected
through the gastric tube as follows: The gastric tube was rinsed
with 20 ml warm water and the 100 mg (1 piece) tablets were placed
in a syringe containing 20 ml warm water. The syringe was inverted
for 5 min until the tablets had disintegrated (Fig. 3A), after which the solution was
immediately injected into the gastric tube. The gastric tube and
syringe were rinsed with warm water again to ensure that all of the
drug had entered the stomach (Fig.
3B). A total of 6 days after this administration, the dyspnea
and dysphagia of the patient had markedly improved and there was no
increase in pleural effusion. After 18 days of lorlatinib
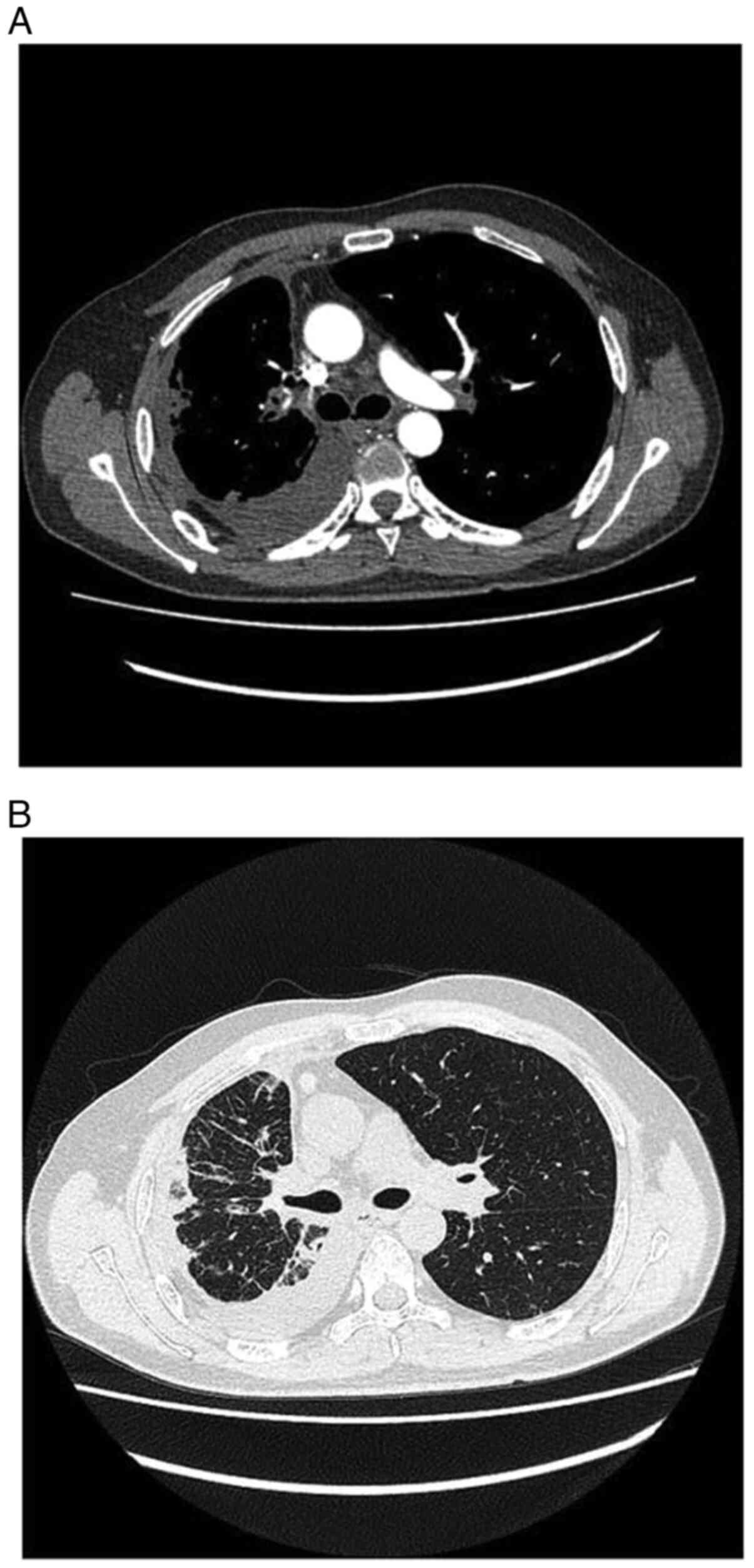
treatment, the chest CT lesions of the patient were notably smaller
than those observed previously (Fig. 4A
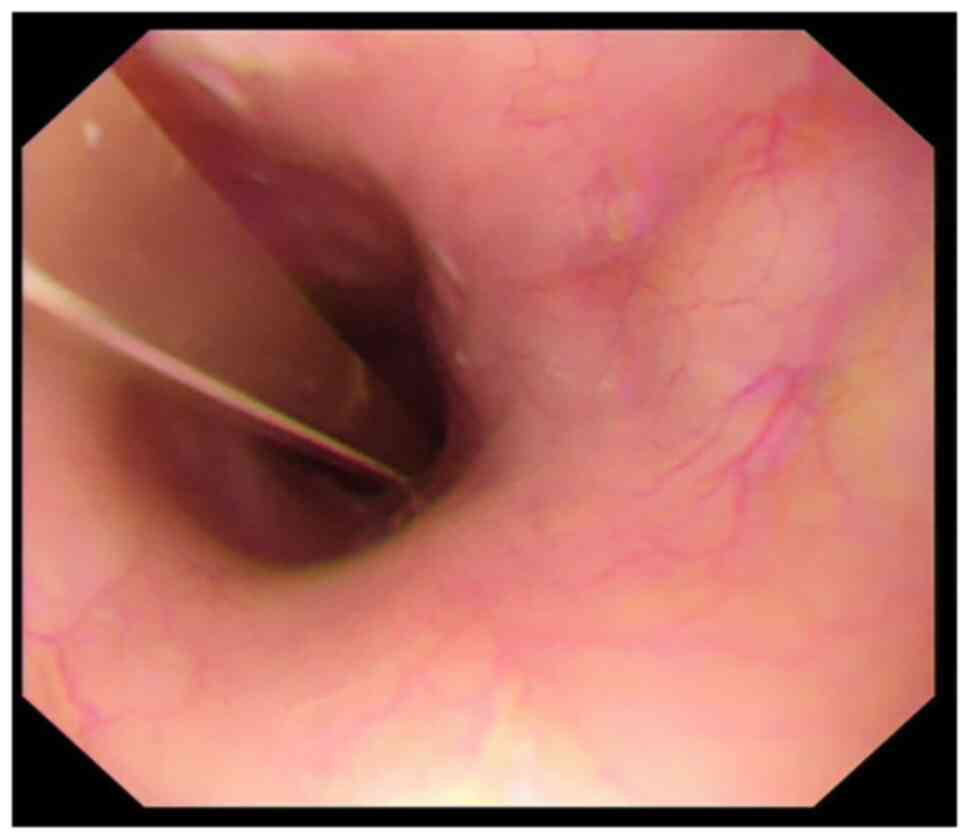
and B). Gastroscopy showed that anterior esophageal stenosis
was significantly relieved and the gastric mucosa was normal
(Fig. 5). Thus, the gastric tube
was removed and oral medication was taken autonomously.
Subsequently, the patient was given oral lorlatinib and the tumor
lesions continued to shrink.
After 18 days of taking lorlatinib, the patient was
examined for blood lipids and developed hyperlipidemia.
Atorvastatin 10 mg each time, once a day was given to reduce blood
lipids. After 1 month of taking lorlatinib and atorvastatin, the
patient's blood lipids returned to normal. Except for
hyperlipidemia, other adverse events, such as edema, weight gain,
diarrhea and constipation (7), were
not observed at follow-up.
Discussion
As a potent third-generation ALK inhibitor,
lorlatinib inhibits almost all known mutated ALK types, including
G1202R (14). G1202R is the most
common secondary resistant ALK mutant in patients post progression
with second generation ALK inhibitor (15). In the B7461001 study, second-line
treatment with lorlatinib in patients who previously received
crizotinib achieved an overall response rate of 72.9% and
progression-free survival of 11.1 months (2). In the CROWN clinical trial, first-line
use of lorlatinib exhibited superior efficacy compared with that of
crizotinib in head-to-head studies, with an overall response rate
of 77.2% and progression-free survival of at least 66 months
(16). There is a tendency to use
lorlatinib as a first-line treatment rather than as a second-line
therapy. The CROWN phase 3 clinical trial of lorlatinib did not
include patients with immunotherapy-associated hyperprogression who
could not take the drug orally, and therefore, there are few
studies on the administration of lorlatinib by gastric tube
(17).
In preclinical studies, advantageous pharmacokinetic
properties have been reported for lorlatinib, including a moderate
volume of distribution, good oral bioavailability and low plasma
clearance (15,18). Hibma et al (19) compared the bioavailability of a
single oral dose of 100 mg and an intravenous dose of 50 mg of
lorlatinib. The adjusted geometric mean (90% confidence interval)
for the absolute oral bioavailability was 80.78% (75.73–86.16%),
and the mean plasma elimination half-life was 25.5 h for oral
administration and 27.0 h for intravenous administration,
indicating the high bioavailability of oral lorlatinib. However,
current targeted drugs for ALK gene mutations are only available as
oral tablets, limiting the feasibility to administer these drugs to
patients with dysphagia (5,6). As described in the present case
report, lorlatinib was dissolved to obtain a suspension and
injected into the gastric tube of the patient, leading to a notable
curative effect. Thus, gastric injection may be used as a
therapeutic approach for patients with advanced lung cancer who
have dysphagia.
Immune hyperprogression is a serious condition
experienced by a small number of patients treated with immune
checkpoint inhibitors, the reported incidence of which varied
between 4 and 29% (20). The
mechanism and potential predictors of this process remain unclear.
The survival time of patients with immune hyperprogression is
shorter than that of other patients. Ferrara et al (21) showed that hyperprogression and
non-hyperprogression PFS (1.4 vs. 4.9 months, P<0.05). In
patients with lung cancer showing epidermal growth factor receptor
and ALK fusion, regardless of their PD-L1 expression, the overall
survival of cases treated with first- or second-line immunotherapy
followed by tyrosine kinase inhibitor treatment was significantly
shorter than that of patients who were negative for gene mutations
(22). This result may be related
to the immunosuppressive state of the tumor microenvironment
containing the gene mutation. The tumor immune microenvironment
associated with the ALK fusion mutation lacks CD8+ T
cells and activated memory CD4+ T cells; therefore,
tumor immunogenicity is low (23).
It has been shown that the co-existence of certain gene mutations
is significantly associated with adverse outcomes of immunotherapy
(24). Thus, further evaluation is
needed to examine gene mutations in patients with advanced NSCLC
before administering immune checkpoint inhibitor therapy. If a
negative result is obtained when blood samples are used for genetic
testing, the possibility of a false-negative result should be
considered, particularly if the patient shows a poor response to
immunotherapy, and repeated genetic testing should be
performed.
In the present case report, the patient responded
rapidly to treatment with lorlatinib, demonstrating another
potential advantage of this drug. This effect was also observed in
another patient with esophageal stenosis (25). In this previous study, the patient
with ALK+ NSCLC with esophageal metastasis showed rapid
improvement of dysphagia symptoms after 5 days of treatment with
lorlatinib without local therapy. In another study similar to the
present study, within three days, the patient's consciousness
improved (26). In the present
case, dysphagia and dyspnea improved after 6 days of treatment.
After 18 days of treatment, the patient showed significant
improvement in cardiac orifice stenosis and the gastric tube was
removed. The patient was able to eat normally and avoid the risk of
esophageal stenting or gastrostomy. The efficacy of lorlatinib,
given its rapid objective response, makes this therapeutic agent a
good treatment choice after treatment with two or more tyrosine
kinase inhibitors, and may be effective in patients with whom rapid
tumor shrinkage is required.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study are not
publicly available, as the patient has not agreed to the public
availability of the raw sequencing data, but may be requested from
the corresponding author.
Authors' contributions
HW and JZ conceptualized the present study. HW was
the principal person responsible for the study and wrote the
original manuscript. GS and ZW provided the case report details
described in the manuscript. GS and ZW contributed to the
acquisition, analysis and interpretation of data and performed
analysis and interpretation of CT imaging data. ZX performed a
critical literature review, contributed to analysis and
interpretation of data and contributed to the drafting of the
Introduction and Discussion sections. JZ reviewed and edited
manuscript. JZ and ZX confirm the authenticity of all the raw data.
All authors have read and approved the final version of the
manuscript. All authors have checked and confirmed the authenticity
of the raw data generated during the study.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient provided written informed consent for
the publication of any data and/or accompanying images.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ALK
|
anaplastic lymphoma kinase
|
|
CT
|
computed tomography
|
|
NSCLC
|
non-small cell lung cancer
|
References
|
1
|
Horn L, Whisenant JG, Wakelee H, Reckamp
KL, Qiao H, Leal TA, Du L, Hernandez J, Huang V, Blumenschein GR,
et al: Monitoring therapeutic response and resistance: Analysis of
circulating tumor DNA in patients with ALK+ lung cancer. J Thorac
Oncol. 14:1901–1911. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Solomon BJ, Besse B, Bauer TM, Felip E,
Soo RA, Camidge DR, Chiari R, Bearz A, Lin CC, Gadgeel SM, et al:
Lorlatinib in patients with ALK-positive non-small-cell lung
cancer: Results from a global phase 2 study. Lancet Oncol.
19:1654–1667. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bauer TM, Shaw AT, Johnson ML, Navarro A,
Gainor JF, Thurm H, Pithavala YK, Abbattista A, Peltz G and Felip
E: Brain penetration of lorlatinib: Cumulative incidences of CNS
and non-CNS progression with lorlatinib in patients with previously
treated ALK-positive non-small-cell lung cancer. Target Oncol.
15:55–65. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
US FDA, . FDA approves lorlatinib for
metastatic ALK-positive NSCLC. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-lorlatinib-metastatic-alk-positive-nsclcSeptember
29–2023
|
|
5
|
US FDA, . Crizotinib prescribing
information. www.accessdata.fda.gov/drugsatfda_docs/label/2016/202570s016lbl.pdfSeptember
29–2023
|
|
6
|
US FDA, . Alectinib prescribing
information. www.accessdata.fda.gov/drugsatfda_docs/label/2017/208434s003lbl.pdfSeptember
29–2023
|
|
7
|
US FDA, . Lorlatinib prescribing
information. www.accessdata.fda.gov/drugsatfda_docs/label/2018/210868s000lbl.pdfSeptember
29–2023
|
|
8
|
Chubachi S, Yasuda H, Irie H, Fukunaga K,
Naoki K, Soejima K and Betsuyaku T: A case of non-small cell lung
cancer with possible ‘disease flare’ on nivolumab treatment. Case
Rep Oncol Med. 2016:10756412016.PubMed/NCBI
|
|
9
|
Kazemi NY, Langstraat C and John Weroha S:
Non-gestational choriocarcinoma with hyperprogression on
pembrolizumab: A case report and review of the literature. Gynecol
Oncol Rep. 39:1009232022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Champiat S, Dercle L, Ammari S, Massard C,
Hollebecque A, Postel-Vinay S, Chaput N, Eggermont A, Marabelle A,
Soria JC and Ferté C: Hyperprogressive disease is a new pattern of
progression in cancer patients treated by anti-PD-1/PD-L1. Clin
Cancer Res. 23:1920–1928. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kato S, Goodman A, Walavalkar V,
Barkauskas DA, Sharabi A and Kurzrock R: Hyperprogressors after
immunotherapy: Analysis of genomic alterations associated with
accelerated growth rate. Clin Cancer Res. 23:4242–4250. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang L, Bai L, Liu X, Liu Y, Li S, Liu J,
Zhang S, Yang C, Ren X and Cheng Y: Factors related to rapid
progression of non-small cell lung cancer in Chinese patients
treated using single-agent immune checkpoint inhibitor treatment.
Thorac Cancer. 11:1170–1179. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Azam F, Latif MF, Farooq A, Tirmazy SH,
AlShahrani S, Bashir S and Bukhari N: Performance status assessment
by using ECOG (eastern cooperative oncology group) score for cancer
patients by oncology healthcare professionals. Case Rep Oncol.
12:728–736. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nagasaka M, Ge Y, Sukari A, Kukreja G and
Ou SI: A user's guide to lorlatinib. Crit Rev Oncol Hematol.
151:1029692020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zou HY, Li Q, Engstrom LD, West M,
Appleman V, Wong KA, McTigue M, Deng YL, Liu W, Brooun A, et al:
PF-06463922 is a potent and selective next-generation ROS1/ALK
inhibitor capable of blocking crizotinib-resistant ROS1 mutations.
Proc Natl Acad Sci USA. 112:3493349–8. 2015. View Article : Google Scholar
|
|
16
|
Solomon BJ, Bauer TM, Ignatius Ou SH, Liu
G, Hayashi H, Bearz A, Penkov K, Wu YL, Arrieta O, Jassem J, et al:
Post hoc analysis of lorlatinib intracranial efficacy and safety in
patients with ALK-positive advanced non-small-cell lung cancer from
the phase III CROWN study. J Clin Oncol. 40:3593–3602. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shaw AT, Bauer TM, de Marinis F, Felip E,
Goto Y, Liu G, Mazieres J, Kim DW, Mok T, Polli A, et al:
First-line lorlatinib or crizotinib in advanced ALK-positive lung
cancer. N Engl J Med. 383:2018–2029. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Johnson TW, Richardson PF, Bailey S,
Brooun A, Burke BJ, Collins MR, Cui JJ, Deal JG, Deng YL, Dinh D,
et al: Discovery of
(10R)-7-amino-12-fluoro-2,10,16-trimethyl-15-oxo-10,15,16,17-tetrahydro-2H-8,4-(metheno)pyrazolo[4,3-h][2,5,11]-benzoxadiazacyclotetradecine-3-carbonitrile
(PF-06463922), a macrocyclic inhibitor of anaplastic lymphoma
kinase (ALK) and c-ros oncogene 1 (ROS1) with preclinical brain
exposure and broad-spectrum potency against ALK-resistant
mutations. J Med Chem. 57:4720–4744. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hibma JE, O'Gorman M, Nepal S, Pawlak S,
Ginman K and Pithavala YK: Evaluation of the absolute oral
bioavailability of the anaplastic lymphoma kinase/c-ROS oncogene 1
kinase inhibitor lorlatinib in healthy participants. Cancer
Chemother Pharmacol. 89:71–81. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Frelaut M, Le Tourneau C and Borcoman E:
Hyperprogression under Immunotherapy. Int J Mol Sci. 20:26742019.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ferrara R, Caramella C, Texier M, Audigier
Valette C, Tessonnier L, Mezquita L, Lahmar J, Mazieres J, Zalcman
G, Brosseau S, et al: Hyperprogressive disease (HPD) is frequent in
non-small cell lung cancer (NSCLC) patients (pts) treated with anti
PD1/PD-L1 monoclonal antibodies (IO). Ann Oncol. 28 (Suppl
5):v464–v465. 2017. View Article : Google Scholar
|
|
22
|
Peters S, Gettinger S, Johnson ML, Jänne
PA, Garassino MC, Christoph D, Toh CK, Rizvi NA, Chaft JE,
Carcereny Costa E, et al: Phase II trial of atezolizumab as
first-line or subsequent therapy for patients with programmed
death-ligand 1-selected advanced non-small-cell lung cancer
(BIRCH). J Clin Oncol. 35:2781–2789. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu SY, Dong ZY, Wu SP, Xie Z, Yan LX, Li
YF, Yan HH, Su J, Yang JJ, Zhou Q, et al: Clinical relevance of
PD-L1 expression and CD8+ T cells infiltration in patients with
EGFR-mutated and ALK-rearranged lung cancer. Lung Cancer.
125:86–92. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kang YK, Reck M, Nghiem P, Feng Y, Plautz
G, Kim HR, Owonikoko TK, Boku N, Chen LT, Lei M, et al: Assessment
of hyperprogression versus the natural course of disease
development with nivolumab with or without ipilimumab versus
placebo in phase III, randomized, controlled trials. J Immunother
Cancer. 10:e0042732022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Longo V, Catino A, Montrone M, Pizzutilo
P, Ugenti I, Lacalamita R, Del Bene G, Pesola F, Marech I and
Galetta D: Esophageal stricture caused by ALK-positive NSCLC
esophageal metastasis resolved after a few days of lorlatinib
therapy without stent placement. JTO Clin Res Rep.
1:1000442020.PubMed/NCBI
|
|
26
|
Sasaki K, Yokota Y, Isojima T, Fujii M,
Hasui K, Chen Y, Saito K, Takahata T, Kindaichi S and Sato A:
Enteral lorlatinib after alectinib as a treatment option in
anaplastic lymphoma kinase-positive non-small cell lung cancer with
triple problems: Carcinomatous meningitis, poor performance status,
and dysphagia-a case report. Respirol Case Rep. 9:e007962021.
View Article : Google Scholar : PubMed/NCBI
|