Introduction
Breast cancer (BC) remains the foremost cause of
mortality in women worldwide (1).
Advanced BC encompasses both locally advanced BC that is inoperable
and BC that has metastasized to distant sites (mBC) (2). The majority of breast malignancies
arise from epithelial components, with ductal carcinoma [no special
type (NST)] being the most prevalent (3). Lobular carcinoma accounts for ~8% of
cases, whilst less common subtypes such as metaplastic (BC-Mp),
medullary, neuroendocrine, tubular and mucinous carcinomas make up
1–2% of cases (3). BC is classified
into several subtypes, including Luminal A, Luminal B, human
epidermal growth factor receptor 2 (HER2)+ and
basal-like (with triple-negative being the most common). These
classifications are determined by the expression of estrogen
receptor (ER), progesterone receptor (PrR), Ki-67 status and HER2
status (4).
BC-Mp, encompassing both epithelial and mixed
epithelial-mesenchymal tumours, are more likely to be
triple-negative and generally demonstrate a less favourable
prognosis when compared with triple-negative invasive ductal
carcinoma (5–7). BC-Mp has been reported to have low
chemosensitivity according to certain studies (8–10).
There is controversy, particularly regarding prognostic factors and
treatment guidelines, owing to its diverse nature and rarity. A
notable number of patients diagnosed with localized disease face
dissemination or local recurrences (8–10).
Data on outcomes of palliative treatment regimens used in this
setting are limited with the majority of publications concentrating
on the following: i) Clinicopathological characterisations with no
or insufficient data regarding palliative treatment (11–19);
ii) a small number of patients with mBC-Mp (20,21);
iii) radical treatment outcomes (22–24);
or iv) data from the general population without further details on
mBC-Mp (9,25,26).
The objective of the present study was to evaluate
overall survival and factors influencing it in patients with mBC-Mp
who received treatment at four Cancer Reference Centres/University
Hospitals located in Southern and Central Poland, and to establish
clinicopathological group characteristics.
Materials and methods
Patients and data extraction
Patients diagnosed with BC-Mp between 2012–2022 were
identified using the registry systems of four medical units: The
Maria Sklodowska-Curie National Research Institute of Oncology,
Branch in Warsaw, Krakow and Gliwice, Poland, and the Department of
Oncology at the University Hospital in Krakow, Poland. The imaging
results for patients with BC-Mp were reviewed to identify a subset
of individuals undergoing palliative treatment.
The inclusion criteria of the study encompassed
individuals with a confirmed diagnosis of BC-Mp in either
postsurgical or core biopsy pathology reports, along with evidence
of dissemination on imaging studies. The typical method for
determining the diagnosis of BC-Mp involved a combination of
morphological evaluation and immunohistochemical staining (27). There were no restrictions based on
the sex or age of the patients. Patients lacking an original
pathology report or those concurrently experiencing active
malignancies were excluded from the study.
Information pertaining to age and sex, as well as
clinical details such as tumour location and size, local lymph node
involvement, location of distant metastatic disease, dates and
types of palliative systemic treatment, the initial treatment
intention (palliative vs. radical) at the initial diagnosis of the
patients, survival status, dates of the last visit and
histopathological data (including histology, ER, PrR, HER2, Ki-67
status, presence of ductal carcinoma in situ, tumour grade
and presence of different BC-Mp components) were collected
retrospectively to ensure a comprehensive analysis.
Statistical analysis
All analyses were performed using R software,
version 4.3.2 (The R Foundation). P<0.05 was used to indicate a
statistically significance difference. The mean, standard deviation
(SD), median, quartiles and range of quantitative variables were
generated. For qualitative variables, the absolute and relative
frequencies (n and %) were reported. Univariate and multivariate
Cox regression (proportional hazards model) were used to model the
potential impact of predictors on a time to event. Hazard ratios
(HR) and 95% confidence intervals were generated. The choice of
independent variables was based on their significance in the
univariate analyses and so Events Per Variable was >10, or ≥5,
where 10 was not reachable. Multicollinearity was assessed using
the variance inflation factor (VIF). Predictors with VIF>5 were
removed from the model.
Results
Population clinicopathological
characteristics
The present research involved a cohort of 31 female
participants, with no male subjects included. The median age at the
time of mBC-Mp diagnosis was 61 years (quartiles, 50–69 years), and
the mean age ± SD was 59.7±14.2 years (range, 33–87 years). mBC-Mp
constituted <1% of the total breast cancer cases within each
institution and 25.4% of all BC-Mp cases identified in all
databases.
The median Ki-67 was 50% (quartiles, 35–70%) with a
mean ± SD of 51.5±23.1% (range, 10–90%; n=29). The median tumour
size was 70 mm (quartiles, 39.8–111.3 mm), and the mean ± SD was
73.4±35.8 mm (range, 20–130 mm; n=28). The most common site for
distant metastatic disease were lungs (61.3%). Further
clinicopathological data regarding patients are presented in
Table I. All patients presented
with distant metastases at the study entry and there were no
patients presenting with inoperable BC-Mp that were receiving
palliative treatment.
 | Table I.Clinical and pathological
characteristics of the patients (n=31). |
Table I.
Clinical and pathological
characteristics of the patients (n=31).
| Characteristic | n (%) |
|---|
| Lymph node
involvement |
|
|
Positive | 12 (38.7) |
|
Negative | 19 (61.3) |
| Distant metastases
location-first metastatic sitea |
|
|
Lung | 19 (61.3) |
| Distant
lymph node | 6 (19.4) |
|
Bone | 4 (12.9) |
|
Liver | 4 (12.9) |
| Central
nervous system | 4 (12.9) |
| Skin
and subcutaneous tissue | 3 (9.7) |
| Other
sites | 5 (16.1) |
| Distant metastases
location-all metastatic sitesa |
|
|
Lung | 19 (61.3) |
| Distant
lymph node | 7 (22.6) |
|
Bone | 5 (16.1) |
|
Liver | 4 (12.9) |
| Central
nervous system | 6 (19.4) |
| Skin
and subcutaneous tissue | 4 (12.9) |
| Other
sites | 5 (16.1) |
| Grade |
|
| 2 | 4 (12.9) |
| 3 | 25 (80.6) |
| No
data | 2 (6.5) |
| DCIS presence |
|
|
Yes | 7 (22.6) |
| No | 23 (74.2) |
|
Unknown | 1 (3.2) |
| ER status |
|
|
Positive | 2 (6.5) |
|
Negative | 29 (93.5) |
| PrR status |
|
|
Positive | 0 (0.0) |
|
Negative | 31 (100.0) |
| Subtype |
|
| Luminal
A | 0 (0.0) |
| Luminal
B | 2 (6.5) |
|
HER2+ | 4 (12.9) |
|
Triple-negative | 25 (80.6) |
| HER2 |
|
|
Positive | 4 (12.9) |
|
Negativeb | 27 (87.1) |
| Type of
componentc |
|
|
NST | 10 (32.3) |
|
Squamous | 16 (51.6) |
| Spindle
cell/pleomorphic/sarcomatid | 9 (29.0) |
|
Osseous/chondroid | 5 (16.1) |
|
Mesenchymal unspecified | 2 (6.4) |
|
Lipid-rich | 1 (3.2) |
| Menopausal
status |
|
|
Premenopausal | 11 (35.5) |
|
Postmenopausal | 19 (61.3) |
|
Unknown | 1 (3.2) |
| Comorbidities |
|
|
Yes | 13 (41.9) |
| No | 16 (51.6) |
|
Unknown | 2 (6.5) |
| Intention of
treatment at initial diagnosis |
|
|
Radical | 24 (77.4) |
|
Palliative | 7 (22.6) |
| Prior systemic
treatment received |
|
|
Yes | 19 (61.3) |
| No | 12 (38.7) |
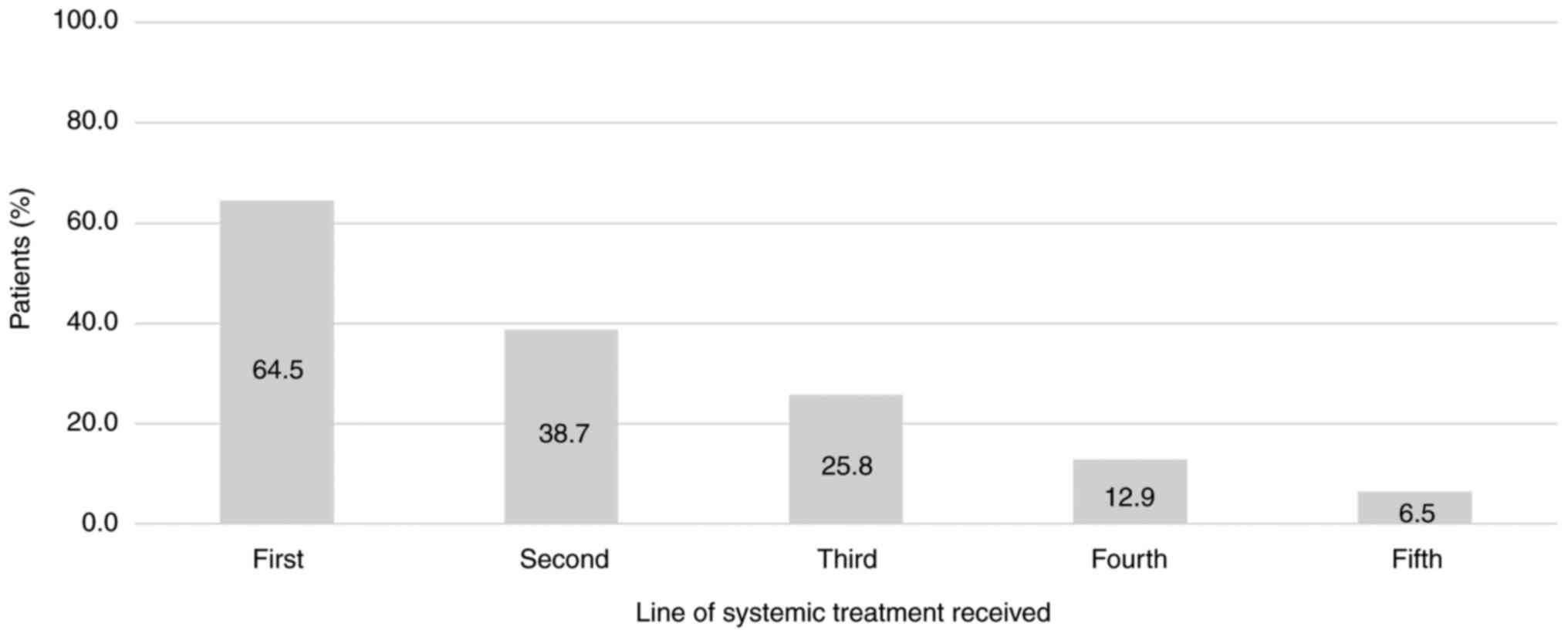
First-line systemic treatment
Overall, 20 patients (62.4%) received first-line
palliative systemic treatment. The types of treatments applied in
the first-line setting are presented in Table II. The proportion of patients that
received systemic treatment in the first- to fifth-line of
palliative therapy is presented in Fig.
1.
 | Table II.Systemic treatment regiments received
in a first-line setting (n=20). |
Table II.
Systemic treatment regiments received
in a first-line setting (n=20).
| First-line systemic
treatment typea | n (%) |
|---|
|
Anthracycline-based | 8 (40.0) |
| Taxan-based | 4 (20.0) |
| Platinum-based | 7 (35.0) |
|
Gemcitabine-based | 2 (10.0) |
|
Anti-HER2b | 2 (10.0) |
|
Anti-PD1c | 1 (5.0) |
| CMF scheme | 1 (5.0) |
| Hormonal
agentsd | 1 (5.0) |
| Single agent
therapy | 12 (60.0) |
| Combination
therapy | 8 (40.0) |
Median progression-free survival (PFS) in first-line
treatment was 15.8 weeks (range, 3–84 weeks) with 8 patients (40%)
responding to the treatment (at least stable disease). In
second-line treatment, PFS was 7.5 weeks (range, 3–30) with 3/12
patients (25%) responding to the treatment, including two patients
who were HER2+ and received lapatinib and capecitabine,
and one patient treated with a poly ADP-ribose polymerase inhibitor
(PARPi; within a clinical trial). In third- and fourth-line
treatments, 2/8 patients (with capecitabine, n=1; and with
cisplatin-gemcitabine regimens, n=1) and ¼ patients responded to
the treatment (with trastuzumab emtansine).
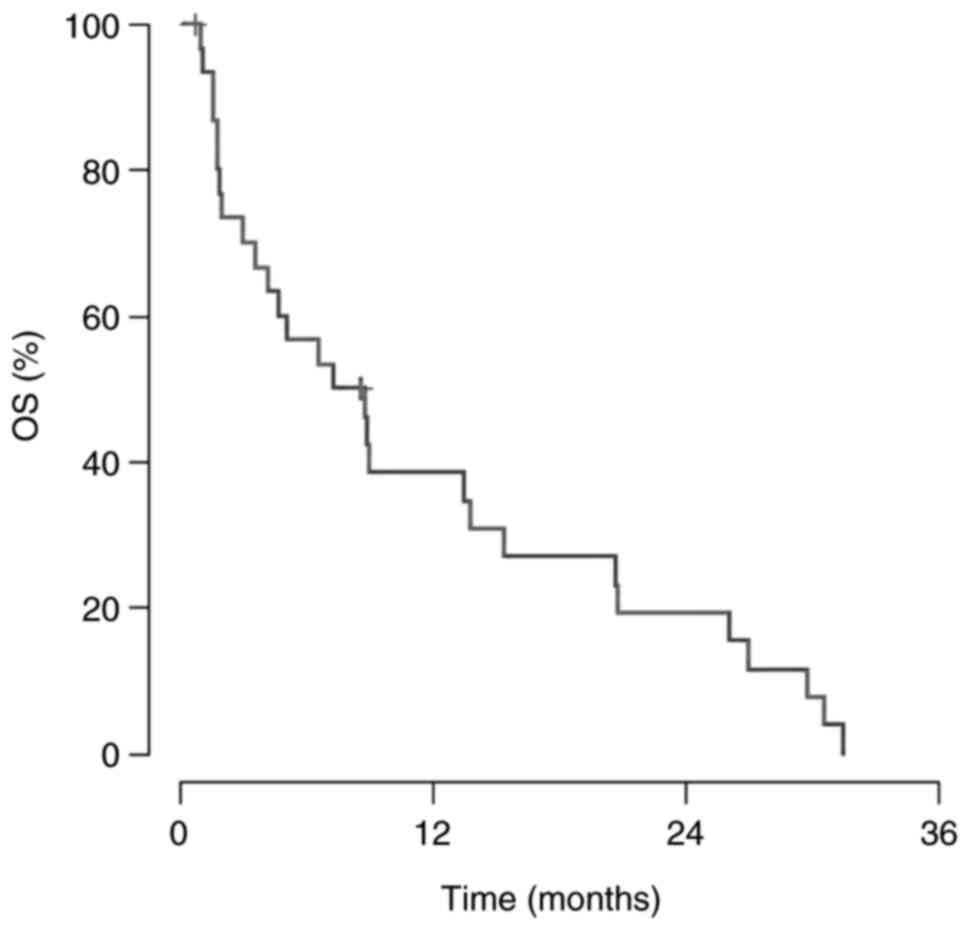
Overall survival
Up until July 2023, the median observation time was
7.4 months (range, 0.7–31.5 months). A total of 28/31 patients died
(90.32%). Table III and Fig. 2 present the overall survival (OS)
data.
 | Table III.Overall survival data for patients
with metaplastic breast cancer with distant metastases. |
Table III.
Overall survival data for patients
with metaplastic breast cancer with distant metastases.
|
|
| Overall
survivala (%),
months |
|---|
|
|
|
|
|---|
| Patients, n | Events (deaths),
n | 6 | 12 | 24 | Median |
|---|
| 31 | 28 | 56.67 | 38.46 | 19.23 | 7.36 |
Factors influencing overall
survival
The univariate proportional hazards Cox models
demonstrated that the likelihood of death at any given time was
significantly reduced by 82.8% (HR=0.172) in individuals who
received at ≥1 line of palliative treatment. Additionally, de
novo diagnosis of disseminated disease significantly decreased
the probability of death at any given time by 72.2% (HR=0.278)
compared with patients previously treated with curative intent.
In the multivariate proportional hazards Cox model,
application of ≥1 line of palliative chemotherapy significantly
reduced the probability of death at any given time by 76.4%
(HR=0.236). Furthermore, de novo diagnosis of mBC-Mp
significantly decreased the likelihood of death at any given time
by 72.3% (HR=0.277) in comparison with patients with secondary
cancer dissemination. Factors such as comorbidities (requiring
pharmacological treatment), presence of specific epithelial or
mesenchymal components, location of metastases or chemotherapy
regimen type did not influence OS. Table IV presents the outcomes of the
univariate and multivariate proportional hazards Cox models.
 | Table IV.Outcomes of univariate and
multivariate proportional hazards Cox models. |
Table IV.
Outcomes of univariate and
multivariate proportional hazards Cox models.
|
|
|
| Overall
survival |
|---|
|
|
|
|
|
|---|
|
|
|
| Univariate
model | Multivariate
model |
|---|
|
|
|
|
|
|
|---|
| Variable | Total, n | Deaths, n | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Age | - | - | 1.032 | 0.997–1.067 | 0.072 | 1.029 | 0.994–1.065 | 0.106 |
| Menopause |
|
|
|
|
|
|
|
|
| No | 11 | 10 | 1 | ref. | - |
|
|
|
|
Yes | 19 | 17 | 1.078 | 0.486–2.388 | 0.854 |
|
|
|
| Histopathology
performed in a reference centre |
|
|
|
|
|
|
|
|
| No | 11 | 10 | 1 | Ref. | - |
|
|
|
|
Yes | 20 | 18 | 1.887 | 0.776–4.590 | 0.162 |
|
|
|
| Grade |
|
|
|
|
|
|
|
|
| 2 | 4 | 4 | 1 | ref. | - |
|
|
|
| 3 | 25 | 22 | 0.692 | 0.231–2.074 | 0.511 |
|
|
|
| ER+
status |
|
|
|
|
|
|
|
|
| No | 29 | 27 | 1 | ref. | - |
|
|
|
|
Yes | 2 | 1 | 5.859 | 0.680–50.449 | 0.108 |
|
|
|
| HER2+
status |
|
|
|
|
|
|
|
|
| No | 27 | 24 | 1 | ref. | - |
|
|
|
|
Yes | 4 | 4 | 0.633 | 0.214–1.871 | 0.408 |
|
|
|
| Ki-67 | - | - | 0.994 | 0.976–1.013 | 0.534 |
|
|
|
| Primary tumour
size | - | - | 0.999 | 0.988–1.011 | 0.904 |
|
|
|
| Lymph nodes
involved |
|
|
|
|
|
|
|
|
| No | 19 | 16 | 1 | ref. | - |
|
|
|
|
Yes | 12 | 12 | 0.656 | 0.293–1.468 | 0.305 |
|
|
|
| Primary
diagnosis |
|
|
|
|
|
|
|
|
|
Local | 24 | 21 | 1 | ref. | - | 1 | ref. | - |
|
Metastatic | 7 | 7 | 0.278 | 0.099–0.779 | 0.015a | 0.277 | 0.085–0.907 | 0.034a |
| Palliative systemic
treatment |
|
|
|
|
|
|
|
|
| No | 9 | 9 | 1 | ref. | - | 1 | ref. | - |
|
Yes | 20 | 18 | 0.172 | 0.069–0.428 |
<0.001a | 0.236 | 0.087–0.638 | 0.004a |
| First-line systemic
therapy |
|
|
|
|
|
|
|
|
|
Combination | 8 | 6 | 1 | ref. | - |
|
|
|
|
Monotherapy | 12 | 12 | 0.439 | 0.143–1.344 | 0.149 |
|
|
|
Discussion
The present study presented clinical data for one of
the largest cohorts of patients with mBC-Mp published and is second
most extensive study thus far with regards to publications
discussing treatment responses and the types of regimens used, to
the best of our knowledge (28).
The majority of patients presented as triple negative (TN)BC with
poorly differentiated tumours aligning with the general trend in
studies exploring BC-Mp clinicopathological data (Table V) (29). However, the population in the
present study had even lower rates of
ER+/PrR+ and HER2+ in comparison
with other cohorts, although there were certain studies reporting
100% or ≤100% of HER2− populations (11,30),
and a sparse occurrence of ER+ cases (22). The prognostic significance of
ER+/(PrR+) in a mBC-Mp population is
uncertain, given that only two patients in the present study were
ER+. Other studies have not reported such prognostic
relevance (31,32). In a cohort from Pakistan, the
hormone-positive status was demonstrated in >50% of the
population; however, looking at additional factors (such as the %
local lymph node positivity), the cohort presentation was more
reminiscent of NST-like BC (33).
 | Table V.Literature regarding patients with
metastatic metaplastic breast cancer receiving palliative systemic
treatment. |
Table V.
Literature regarding patients with
metastatic metaplastic breast cancer receiving palliative systemic
treatment.
| First author/s,
year | Participants | Recruitment
period | IHC | Pathology | Systemic palliative
treatment (including data about regimen type) | Outcomes | Conclusions | (Refs.) |
|---|
| Esbah et al,
2012 | 6 | 2005-2011 | ER−,
83.3%; PrR−, 83.3%; and HER2−, 83.3% | Adenosquamous
carcinoma, n=2; Squamous cell carcinoma, n=2; and carcinosarcoma,
n=2 | CTH, n=5; CED →PE',
n=1; DX, n=2; PG, n=1; PE', n=1; no data, n=1 | PD, n=4; SD, n=1;
and died of disease, n=4 | The study proposed
adding platinum to the treatment for squamous and triple negative
subgroups. In the sarcoma subgroup, high dose anthracyclines could
be used. The study presented data for both radically- and
palliatively-treated patients. | (59) |
| Kim et al,
2021 | 5 | Unknown | ER−,
80%; PrR−, 100%; and HER2−, 100% | Squamous cell
carcinoma, n=2; mixed metaplastic squamous carcinoma and
pleomorphic invasive lobular carcinoma, n=1; and metaplastic
carcinoma with heterologous mesenchymal differentiation
(chondroid), n=2 | Pembrolizumab + X →
RTH; pembrolizumab + X → eribulin → G → P → T → sacituzumab
govitecan; RTH → fulvestrant + Palbociclib → exemestane +
everolimus → nivolumab + bicalutamide → nivolumab in monotherapy;
pembrolizumab + X; and pembrolizumab + T → RTH → D + bevacizumab +
everolimus | PR, n=2; PD, n=2;
CR, n=1; and PFS, 5.3–8.0 months | Response to
immunotherapy in BC-Mp can be achieved in patients with modest
PD-L1 (CPS 1–10) expression. Palliative patients only. | (30) |
| Rayson et
al, 1999 | 14 | 1976-1997 | ER−,
70%; PR−, 70%; HER2 status, no data; and unknown,
15% | Spindle, n=8;
squamous, n=1; osseous, n=1; chondroid, n=1; mixed BC-Mp, n=5; and
unknown, n=11 | 14 patients with
metastatic disease: CTH, n=7 including: A, n=4; CF + prednisone,
n=2; P, n=2; MMC, n=2; CAF, n=1; C, n=1; CMF, n=1; M, n=1; Taxol,
n=1; F, n=1; actinomycin, n=1; vincristine, n=1; tamoxifen,n=4;
surgery, n=3; RTH, n=6; no treatment, n=3 | PR, n=1 (A); PD,
n=10; CR, n=2; SD, n=1; mOS after detection of metastasis, 8
months | Among the
chemotherapeutic agents used in this cohort, none reliably produced
responses in metastatic disease. The study presented data for both
radically- and palliatively-treated patients. | (41) |
| Takala et
al, 2019 | 28 | 2002-2016 | ER−,
88%; PrR−, 97%; and HER2−, 96% | Low-grade
adenosquamous, n=2; squamous, n=20; spindle, n=17; chondroid
differentiation, n=11; osseous differentiation, n=2; mixed BC-Mp,
n=9; and mixed type, n=17 | 14/28 patients with
metastatic disease received palliative systemic therapy and 1
patient received palliative endocrinetherapy. First-line: D/T, n=6;
X, n=2; PX, n=1; PN', n=1; DG, n=1; vincristine + MF, n=1; FEC,
n=1; and D + P, n=1. Second-line: FEC, n=5; TC', n=2; C'G, n=1; and
D/T, n=1. Third-line: TC', n=1; D, n=1; GP, n=1; X, n=1; and AC,
n=1. Fourth-line: XN', n=2 and D, n=1. Fifth-line: FEC, n=1.
Sixth-line: C'G, n=1. Seventh-line: weekly A, n=1 | PR, n=2; PD, n=22;
NE, n=4; SD, n=6; mOS, 3.4 months or 6.4 months if treatment was
administered; mOS, 1.1 month if treatment was not administered | The response to
palliative systemic therapy in metastatic BC-Mp was poor. The few
responses observed in the study were in those treated with
anthracyclines (FEC-regimen) and capecitabine. The study presented
data for both radically- and palliatively-treated patients. | (38) |
| Chen et al,
2011 | 18 | 1988-2009 | ER−,
89.1%; PrR−,78.2%; and HER2−, 95.7% | Spindle cell
carcinoma, n=8; squamous cell carcinoma, n=12; chondrosarcoma, n=1;
chondromyxoid, n=1; sarcomatous, n=5; mixed, n=18; and unknown,
n=1 | 18 patients
developed metastatic disease, of which 12 received CTH. First-line:
N'P, n=1; oral CE', n=1; CEF, n=1; N'FL, n=1; imatinib, n=1; T +
FL, n=1; CMF, n=2; oral uracil-tegafur, n=1; EC, n=1; N'X, n=1; HT,
n=1. Second-line: uracil-tegafur/P, n=1; bevacizumab + MMC, n=1;
bevacizumab + IE, n=1; HN'+ FL, CEF, n=1; D+P, n=1; T + FL, n=1; HT
+ FL, n=1. Third-line: Cetuximab, n=1; bevacizumab + M' + MMC, n=1;
CE, n=2; M' + PN', n=1. Fourth-line: H, n=1; G, n=1; TGH, n=1; N'P,
n=1. Fifth-line: X/lapatinib, n=1. Sixth-line: TGH, n=1.
Seventh-line: Oral E'M, n=1. Eighth-line: DH, n=1. Ninth-line: DPH,
n=1. Tenth-line: EH, n=1. Eleventh-line: Oral C, n=1. Twelfth-line:
VMH, n=1. Thirteenth-line: V, n=1. Fourteenth-line: H + CMF,
n=1 | PR, n=2. First
patient: paclitaxel and 24-h high-dose fluorouracil/leucovorin
treatment. Second patient: oral uracil-tegafur for 8 months before
disease progression. PD, n=10. Median time from diagnosis of
metastatic disease to death, 10.65 months if palliative CTH was
administered and 5.29 months if palliative CTH was not
administered. mTTP of first-line chemotherapy for metastatic BC-Mp
treated using taxane-based regimen was 1.55 months and
non-taxane-based was 0.73 months | The response of
patients with BC-Mp to systemic chemotherapy was poor. Taxane-or
doxorubicin-containing regimens may be the two major categories of
traditional chemotherapeutic agents worth recommending. The study
presented data for both radically- and palliatively-treated
patients. | (42) |
| Lee et al,
2012 | 30 | January
2001-December 2009 |
ER−/PrR−, 94% and
HER2−, 91% | Squamous cell
carcinoma, n=35; spindle cell carcinoma, n=9; chondroid
differentiation, n=7; osseous differentiation, n=2;
matrix-producing, n=6; squamous + spindle, n=1; adenosquamous +
spindle, n=1; squamous + chrondroid + spindle, n=1; squamous +
osseous, n=1; squamous + matrix-producing, n=1; chondroid +
matrix-producing, n=1 | 30 patients
developed recurrence or de novo stage IV disease. CTH, n=25:
Anthracycline-based, 17%; taxane-based, 35%; X-containing, 30%; and
other regimens, 17%. Mean number of CTH cycles=5.56 (range,
2–15) | ORR=38.9%;
CBR=50% | There was no
significant difference in response to preferred CTH between BC-Mp
and triple-negative invasive ductal carcinoma. | (35) |
| Basho et al,
2018 | 59 | April 2009-November
2014 | ER−,
100%; PrR−, 100%; HER2−, 98% | No data | Prior systemic
therapy for metastatic disease, n=30; prior anthracycline, n=46;
prior taxane n=48; prior bevacizumab, n=4; orior mTOR inhibitor,
n=4; liposomal A + bevacizumab + temsirolimus, n=38; and liposomal
A + bevacizumab + everolimus, n=21 | ORR=19%; CBR=36%;
median PFS, 4.8 months; and mOS, 10.0 months | Prior anthracycline
use was an independent predictor of worse OS. Prior taxane use was
an independent predictor of improved OS. Prior systemic therapy in
metastatic setting was associated with worse outcomes. BC-MP may be
more sensitive to mTOR inhibition due to hyperactivation of the
PI3K/Akt/mTOR pathway. | (28) |
| Youssef et
al, 2020 | 417 | 2004-2015 | ER−,
73%; PrR−, 78%; HER2−, 42%; and unknown,
54% | No data | Endocrine therapy,
n=43; CTH, n=156; no data regarding the regimen type or response to
the treatment. | mOS, 12 months if
CTH was administered and 8 months if CTH was not administered | The use of
chemotherapy was associated with improved survival. Palliative
patients only were included in the study. | (40) |
The majority of the patients in the present study
were diagnosed with squamous component, commonly with accompanying
NST BC (Table I). The data did not
reveal any prognostic significance associated with the histologic
subtype of BC-Mp, aligning with findings reported by certain
researchers (18,19) but in contrary to results published
by others (34,35). Emerging data indicate that the
expression of proteins serving as potential targets for novel
therapies is associated with histological subtypes (36). Therefore, this factor warrants
re-evaluation in the context of clinical trials or cohort studies
of patients treated recently.
A total of >60% of patients in the present study
presented with lung metastases, which aligns with data from other
studies (18,25,37,38).
In all cases, lung metastases were simultaneously the initial site
of distant metastatic disease. Thapa et al (25) identified metastases to the lungs as
a poor prognostic factor. A total of 87% of all the patients in the
present study had visceral metastases confirmed at a point in the
course of their disease (data not shown), contributing to
unfavourable outcomes.
The present study demonstrated a poor OS in the
mBC-Mp population, a finding generally supported by other studies
indicating several-month OS rates (Table V) (39). Although BC-Mp is commonly claimed to
be chemoresistant (8,18,40),
the results of the present study revealed that receiving ≥1 line of
palliative treatment significantly reduced the likelihood of death
at any given time, which is consistent with data from other studies
(40).
Currently, there are no established protocols or
recommendations for the management of mBC-Mp, to the best of our
knowledge. This can be attributed to the rarity of the condition
and the incomplete understanding of its natural course. In total,
64.5% of patients received first-line systemic treatment. This was
lower in previously published cohorts (38,40,41) or
had a comparable proportion (38,41,42).
The proportion of patients who received treatment in second and
later treatment lines rapidly decreased in the present study and
other studies (38,42). As highlighted by Youssef et
al (40) in a retrospective
analysis of the U.S. national registry, patients with mBC-Mp
underwent palliative chemotherapy more frequently when managed in
academic centres. The data from the present study were compiled
from patients treated in three Reference Cancer Centres and one
University Hospital. A substantial proportion of patients did not
undergo treatment due to a rapid disease progression associated
with poor performance status and inadequate blood test results.
Certain patients experienced deterioration during the diagnostic
process, such as whilst awaiting biopsy appointments with the
intent of assessing HER2 and ER/PrR status in metastatic sites. It
appears that expedited initiation of treatment is imperative in
this population whenever feasible.
Several systemic therapy regimens were administered
across different lines of treatment, revealing discernible trends
within the four Units in the present study. Overall, no regimen
demonstrated superiority in first-line treatment, and general
responses to treatment were predominantly poor, except in cases
where targeted treatments were used (anti-HER2 agents in two
HER2+ patients in first- and second-line treatments;
aromatase inhibitor in one ER+ patient in a first-line
treatment; anti-programmed cell death protein 1 monoclonal antibody
within a clinical trial in a first-line treatment; and PARPi within
a clinical trial in a second-line treatment). Whilst there are
suggestions regarding potentially improved responses to certain
agents, these conclusions are often drawn from small case series
and case reports, or extrapolated from (neo)adjuvant results,
including: Taxanes (42,43), capecitabine (38), anthracyclines (38) and cisplatin (13). Responses to the treatment in the
cohort in the present study were 40 and 25% in first- and
second-line treatments, respectively. Similar results for
first-line treatments were reported by Youssef et al
(40) In other studies, lower rates
were reported (38,42).
It is suggested that the concurrent activation of
numerous signalling pathways within a tumour could elucidate the
lower clinical response rates observed when using single agent
targeted therapeutic strategies, and combining multiple agents may
offer a potential solution to surmount this therapeutic barrier
(28). However, in the present
study, no differences were observed between single-agent and
multimodal therapies.
Pembrolizumab, when used in combination with
chemotherapy, received approval from Food and Drug Administration
for treating metastatic TNBC displaying positive programmed death
ligand 1 (PD-L1) expression (combined positive score ≥10) as shown
in the KEYNOTE-355 study (44,45).
Numerous reports have emphasized the notable expression of PD-L1 in
metaplastic breast carcinomas, observed in 40–50% of cases
(36,46). This suggests a potential for
enhanced effectiveness of immunotherapy-based treatments within
this subgroup. Case reports have illustrated positive responses and
clinical benefits with immunotherapy in patients diagnosed with
BC-Mp (47–50). In the present study, one patient
received pembrolizumab with chemotherapy within a clinical trial,
achieving a PFS of 20 months (the longest in the cohort) and an OS
of 21 months. Furthermore, the SWOG1609 trial has revealed notable
outcomes with the combination of the cytotoxic T lymphocyte antigen
4 inhibitor ipilimumab and programmed cell death protein 1
inhibitor nivolumab in mBC-Mp (51). Nevertheless, the application of
ipilimumab or nivolumab in this population is currently restricted
to a clinical trial (trial registration no. NCT02834013; http://clinicaltrials.gov/).
The study by Corso et al (52) revealed that breast cancer gene
1 (BRCA1) mutation was the most common germline
pathogenic variant within BC-Mp. Interestingly, among the 15
patients with genetic alterations, no patient was reported to have
a BRCA2 mutation. In the present study, there were 3
patients with BRCA1 mutation (15/31 tested) and one with a
checkpoint kinase 2 mutation (with unknown number of tested
individuals). Only one patient (reported in the database as unknown
BRCA status) received PARPi within a clinical trial as a
second-line treatment with a PFS of 25.2 weeks. Furthermore, it has
been documented that 6/31 patients had a history of other
malignancies treated radically in the past. These included the
following: Three cases of breast cancer, all NST and
ER+, treated >5 years before the diagnosis of BC-Mp;
two haematological malignancies; and one thyroid cancer. Only
limited data exists about synchronous and metachronous malignancies
in the BC-Mp population (53).
Patients diagnosed with mBC with a molecular profile
indicating TNBC or ER+/PrR+ BC may undergo
treatment with sacituzumab govitecan in a second or later line of
treatment, according to the ASCENT or TROPICS-02 trials. This
therapeutic agent is an antibody-drug conjugate featuring an
irinotecan derivative attached to a monoclonal antibody that
specifically targets trophoblast cell surface antigen-2 (TROP-2)
(54–56). In these trials, TROP-2 expression
did not exhibit predictive value within the patient populations,
which was likely attributable to the consistently elevated
expression levels observed. Furthermore, it has been reported that
patients with mBC also exhibit heightened TROP-2 expression
(36).
BC-Mp may also exhibit molecular aberrations
associated with epithelial-to-mesenchymal transition,
phosphoinositide 3-kinase (PI3K), nitric oxide or Wnt/β-catenin
signalling (46,57,58).
Several of these potential targets for personalized treatment have
been evaluated in clinical trials. PI3K catalytic subunit
alpha (PI3KCA) mutations and PTEN) loss may
contribute to heightened susceptibility to mTOR) inhibitors. In a
phase 1 study by Basho et al (28), the safety and efficacy of combining
mTOR inhibitors (temsirolimus or everolimus) with vascular
endothelial growth factor inhibitor bevacizumab and liposomal
doxorubicin in 52 patients with mBC-Mp was assessed (28). Genetic testing revealed 74% of
tumours had a PIK3CA hotspot mutation. Patients with these
mutations presented with an improved objective response rate (ORR).
In an ongoing phase 2 study (trial registration no. NCT05660083;
http://clinicaltrials.gov/) it is
hypothesised that the inclusion of the PI3K inhibitor, alpelisib,
in combination with a pan-nitric oxide synthase inhibitor and
nab-paclitaxel may increase the ORR in patients with
HER2− mBC-Mp during the first and second lines of
systemic therapy. The SABINA clinical trial, a multicentre,
two-cohort, non-comparative, open-label, phase II study (trial
registration no. NCT05810870; http://clinicaltrials.gov/) is aimed at evaluating the
safety and effectiveness of MEN1611 (an oral PI3K inhibitor), both
as a standalone treatment and in combination with eribulin. The
trial specifically targets patients with HER2− mBC-Mp
with alterations in PIK3CA and PTEN. There are also a single report
of successful applications of combined anti-angiogenic agent and
immune check-point inhibitor (50).
A limitation of the present study is its
retrospective design. The low incidence of the studied malignancy
poses a challenge for prospective observation. Furthermore, the
study acknowledges another constraint related to the size of the
population. Nevertheless, it is noteworthy that the cohort of
patients ranks among the largest published cohorts concerning
patients with mBC-Mp, to the best of our knowledge. Secondly,
receiving systemic treatment in the metastatic setting vs. not
receiving it influenced OS; however, since poor performance status
was not an exclusion criterion for study entry, it is possible that
more advanced patients with worse performance status were the ones
not receiving systemic treatment. For patients who experienced
progression during follow-up, reassessment of ER/PrR, HER2 and
Ki-67 was typically not performed. This implies that the data were
derived from the primary tumour assessment. Given the rapid
progression and the limited likelihood of acquiring ER/PrR
positivity, this practice can be justified.
The data from the present study that demonstrated
worse survival in patients who progressed during follow up in
comparison with patients with de novo metastasis are
interesting, but difficult to explain. One explanation may be the
small patient sample size; another hypothesis may be the limitation
in the available therapeutic interventions for progressing
patients. The impact of secondary mutations or genetic alterations
acquired during disease progression and contribution of
treatment-induced resistance mechanisms in the progressing group
could also serve a role.
In conclusion, the present study demonstrated the
real-world multi-centre data of one of the largest populations of
patients with mBC-Mp. The study underscores the challenging
prognosis of mBC-Mp, with 1- and 2-year OS rates of 38.5 and 19.2%,
respectively. Notably, receiving ≥1 line of palliative treatment
was associated with significantly improved OS. However, other
factors such as age, Ki-67, molecular subtypes, metastatic site and
chemotherapy regimen did not demonstrate a significant impact on
survival. A highly negative ER/PrR and HER2 status restricted the
available treatment options in most of patients with mBC-Mp. The
findings highlight the need for dedicated clinical trials in
mBC-Mp, emphasizing the importance of tailored therapeutic
strategies and continued research to enhance outcomes in this
patient population.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
MP, with the support of AMM, JM, AR and RPM
conceived and MP, KŚ, AK, JS, AGW, AR, JD, RPM, AMM, MK, JM, MJ and
MZ were involved in designing the study. MP, JWM, KŚ, AK, MK, RPM
and JS performed searches within the Cancer Centre/Hospital
registry system. Organizing the database and gathering patient data
were undertaken by MP, KŚ, AK, MK, AR and JD, with MP handling the
statistical analysis. MP, KŚ, AK and JD confirm the authenticity of
all the raw data. The first draft of the manuscript was penned by
MP, whilst AR and RPM contributed to specific sections. All authors
were involved in revising the manuscript and have read and approved
the final manuscript.
Ethics approval and consent to
participate
The Maria Sklodowska-Curie National Research
Institute of Oncology Branch Krakow Ethical Committee (Krakow,
Poland) granted ethical approval for the present study (approval
no. 3/2023). Given the retrospective nature of the study, written
informed consent was not obtained from patients, in line with the
decision of the Ethical Committee.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
BRCA1
|
breast cancer gene 1
|
|
ER
|
estrogen receptor
|
|
HER2
|
human epidermal growth factor receptor
2
|
|
HR
|
hazard ratio
|
|
BC
|
breast cancer
|
|
mBC
|
BC with distant metastasis
|
|
BC-Mp
|
metaplastic BC
|
|
mTOR
|
mammalian target of rapamycin
|
|
OS
|
overall survival
|
|
PFS
|
progression free survival
|
|
NST
|
no special type
|
|
ORR
|
objective response rate
|
|
PARPi
|
poly ADP-ribose polymerase
inhibitor
|
|
PD-L1
|
programmed death ligand 1
|
|
PI3K
|
phosphoinositide 3-kinase
|
|
PrR
|
progesterone receptor
|
|
SD
|
standard deviation
|
|
TROP-2
|
trophoblast cell surface antigen-2
|
|
VIF
|
variance inflation factor
|
References
|
1
|
Siegel RL, Miller KD, Fuchs HE and Jemal
A: Cancer statistics, 2022. CA Cancer J Clin. 72:7–33. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cardoso F, Paluch-Shimon S, Senkus E,
Curigliano G, Aapro MS, André F, Barrios CH, Bergh J, Bhattacharyya
GS, Biganzoli L, et al: 5th ESO-ESMO international consensus
guidelines for advanced breast cancer (ABC 5). Ann Oncol.
31:1623–1649. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li CI, Uribe DJ and Daling JR: Clinical
characteristics of different histologic types of breast cancer. Br
J Cancer. 93:1046–1052. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Burstein HJ, Curigliano G, Thürlimann B,
Weber WP, Poortmans P, Regan MM, Senn HJ, Winer EP and Gnant M;
Panelists of the St Gallen Consensus Conference, : Customizing
local and systemic therapies for women with early breast cancer:
The St. gallen international consensus guidelines for treatment of
early breast cancer 2021. Ann Oncol. 32:1216–1235. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rechsteiner A, Dietrich D and Varga Z:
Prognostic relevance of mixed histological subtypes in invasive
breast carcinoma: A retrospective analysis. J Cancer Res Clin
Oncol. 149:4967–4978. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sherwani M, Vohra L, Ali D, Soomro R,
Adnan S and Idrees R: Clinicopathological features and survival
outcomes of metaplastic breast carcinoma-an observational
multi-centric study. Breast Cancer (Dove Med Press). 15:237–250.
2023.PubMed/NCBI
|
|
7
|
McCart Reed AE, Kalaw EM and Lakhani SR:
An update on the molecular pathology of metaplastic breast cancer.
Breast Cancer (Dove Med Press). 13:161–170. 2021.PubMed/NCBI
|
|
8
|
Wong W, Brogi E, Reis-Filho JS, Plitas G,
Robson M, Norton L, Morrow M and Wen HY: Poor response to
neoadjuvant chemotherapy in metaplastic breast carcinoma. NPJ
Breast Cancer. 7:962021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ismail Y, Kamal A, Allam R and Zakaria AS:
The conundrum of metaplastic breast cancer: A single Egyptian
institution retrospective 10-year experience (2011–2020). J Egypt
Natl Canc Inst. 35:162023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lan T, Lu Y, Zheng R, Shao X, Luo H, He J,
Yang H, Xu H, Wang X and Hu Z: The role of adjuvant chemotherapy in
metaplastic breast carcinoma: A competing risk analysis of the SEER
database. Front Oncol. 11:5722302021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Barnes PJ, Boutilier R, Chiasson D and
Rayson D: Metaplastic breast carcinoma: Clinical-pathologic
characteristics and HER2/neu expression. Breast Cancer Res Treat.
91:173–178. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Papatheodoridi A, Papamattheou E,
Marinopoulos S, Ntanasis-Stathopoulos I, Dimitrakakis C, Giannos A,
Kaparelou M, Liontos M, Dimopoulos MA and Zagouri F: Metaplastic
carcinoma of the breast: Case series of a single institute and
review of the literature. Med Sci (Basel). 11:352023.PubMed/NCBI
|
|
13
|
Nowara E, Drosik A, Samborska-Plewicka M,
Nowara EM and Stanek-Widera A: Metaplastic breast
carcinomas-analysis of prognostic factors in a case series. Contemp
Oncol (Pozn). 18:116–119. 2014.PubMed/NCBI
|
|
14
|
Xu D and Hou L: Clinicopathologic
characteristics of mixed epithelial/mesenchymal metaplastic breast
carcinoma (carcinosarcoma): A meta-analysis of Chinese patients.
Pol J Pathol. 70:174–182. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lai HW, Tseng LM, Chang TW, Kuo YL, Hsieh
CM, Chen ST, Kuo SJ, Su CC and Chen DR: The prognostic significance
of metaplastic carcinoma of the breast (MCB)-a case controlled
comparison study with infiltrating ductal carcinoma. Breast.
22:968–973. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cimino-Mathews A, Verma S,
Figueroa-Magalhaes MC, Jeter SC, Zhang Z, Argani P, Stearns V and
Connolly RM: A clinicopathologic analysis of 45 patients with
metaplastic breast carcinoma. Am J Clin Pathol. 145:365–372. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chaudhary D, Balhara K, Mandal S, Mallya
V, Tomar R, Khurana N and Bains L: Metaplastic breast carcinoma:
Analysis of clinical and pathologic features, a five-year study. J
Cancer Res Ther. 19:1226–1230. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang Y, Lv F, Yang Y, Qian X, Lang R, Fan
Y, Liu F, Li Y, Li S, Shen B, et al: Clinicopathological features
and prognosis of metaplastic breast carcinoma: Experience of a
major Chinese cancer center. PLoS One. 10:e01314092015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Leyrer CM, Berriochoa CA, Agrawal S,
Donaldson A, Calhoun BC, Shah C, Stewart R, Moore HCF and Tendulkar
RD: Predictive factors on outcomes in metaplastic breast cancer.
Breast Cancer Res Treat. 165:499–504. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ishizuka Y, Horimoto Y, Yanagisawa N,
Arakawa A, Nakai K and Saito M: Clinicopathological examination of
metaplastic spindle cell carcinoma of the breast: Case series.
Breast Cancer (Auckl). 15:117822342110394332021.PubMed/NCBI
|
|
21
|
Ghosh M, Muneer A, Trivedi V, Mandal K and
Shubham S: Metaplastic carcinoma breast: A clinical analysis of
nine cases. J Clin Diagn Res. 11:XR01–XR03. 2017.PubMed/NCBI
|
|
22
|
Salimoğlu S, Sert İ, Emiroğlu M, Karaali
C, Kuzukıran D, Kırmızı YA, Diniz G and Aydın C: Metaplastic breast
carcinoma: Analysis of clinical and pathologic characteristics-a
case series. J Breast Health. 12:63–66. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rakha EA, Tan PH, Varga Z, Tse GM, Shaaban
AM, Climent F, van Deurzen CH, Purnell D, Dodwell D, Chan T and
Ellis IO: Prognostic factors in metaplastic carcinoma of the
breast: A multi-institutional study. Br J Cancer. 112:283–289.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Xiao M, Yang Z, Tang X, Mu L, Cao X and
Wang X: Clinicopathological characteristics and prognosis of
metaplastic carcinoma of the breast. Oncol Lett. 14:1971–1978.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Thapa B, Arobelidze S, Clark BA, Xuefei J,
Daw H, Cheng YC, Patel M, Spiro TP and Haddad A: Metaplastic breast
cancer: Characteristics and survival outcomes. Cureus.
14:e285512022.PubMed/NCBI
|
|
26
|
Damera VV, Chowdhury Z, Tripathi M, Singh
R, Verma RK and Jain M: Clinicopathologic features of metaplastic
breast carcinoma: Experience from a tertiary cancer center of North
India. Cureus. 14:e289782022.PubMed/NCBI
|
|
27
|
McMullen ER, Zoumberos NA and Kleer CG:
Metaplastic breast carcinoma: Update on histopathology and
molecular alterations. Arch Pathol Lab Med. 143:1492–1496. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Basho RK, Yam C, Gilcrease M, Murthy RK,
Helgason T, Karp DD, Meric-Bernstam F, Hess KR, Valero V,
Albarracin C, et al: Comparative effectiveness of an mTOR-based
systemic therapy regimen in advanced, metaplastic and
nonmetaplastic triple-negative breast cancer. Oncologist.
23:1300–1309. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rakha EA, Coimbra NDM, Hodi Z, Juneinah E,
Ellis IO and Lee AHS: Immunoprofile of metaplastic carcinomas of
the breast. Histopathology. 70:975–985. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kim I, Rajamanickam V, Bernard B, Chun B,
Wu Y, Martel M, Sun Z, Redmond WL, Sanchez K, Basho R, et al: A
case series of metastatic metaplastic breast carcinoma treated with
anti-PD-1 therapy. Front Oncol. 11:6352372021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Abada E, Kim S, Dozier K, Fehmi O, Jang H,
Fehmi Z and Bandyopadhyay S: Estrogen receptor status has no
prognostic relevance in metaplastic breast carcinoma. Cancer Treat
Res Commun. 33:1006302022. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang S, Hu J, Zhang Y, Shen J, Dong F,
Zhang X, Lu C and Shang D: Presentation and survival by hormonal
receptor status in metaplastic breast cancer: A propensity
score-matched analysis. Breast. 60:168–176. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Samoon Z, Beg M, Idress R and Jabbar AA:
Survival and treatment outcomes of metaplastic breast carcinoma:
Single tertiary care center experience in Pakistan. Indian J
Cancer. 56:124–129. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hu J, Lang R, Zhao W, Jia Y, Tong Z and
Shi Y: The mixed subtype has a worse prognosis than other
histological subtypes: A retrospective analysis of 217 patients
with metaplastic breast cancer. Breast Cancer Res Treat. 200:23–36.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lee H, Jung SY, Ro JY, Kwon Y, Sohn JH,
Park IH, Lee KS, Lee S, Kim SW, Kang HS, et al: Metaplastic breast
cancer: Clinicopathological features and its prognosis. J Clin
Pathol. 65:441–446. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chartier S, Brochard C, Martinat C, Coussy
F, Feron JG, Kirova Y, Cottu P, Marchiò C and Vincent-Salomon A:
TROP2, androgen receptor, and PD-L1 status in histological subtypes
of high-grade metaplastic breast carcinomas. Histopathology.
82:664–671. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Yigit S, Pehlivan FS, Evcim G and Etit D:
Clinicopathologic features of the mixed epithelial and mesenchymal
type metaplastic breast carcinoma with myoepithelial
differentiation in a subset of six cases. Pathol Res Pract.
208:147–150. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Takala S, Heikkilä P, Nevanlinna H,
Blomqvist C and Mattson J: Metaplastic carcinoma of the breast:
Prognosis and response to systemic treatment in metastatic disease.
Breast J. 25:418–424. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Moreno AC, Lin YH, Bedrosian I, Shen Y,
Babiera GV and Shaitelman SF: Outcomes after treatment of
metaplastic versus other breast cancer subtypes. J Cancer.
11:1341–1350. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Youssef I, Lee A, Kukunoor S, Taiwo E,
Luhrs CA and Schreiber D: Patterns of care and survival of
metastatic metaplastic breast cancer patients. Cureus.
12:e103392020.PubMed/NCBI
|
|
41
|
Rayson D, Adjei AA, Suman VJ, Wold LE and
Ingle JN: Metaplastic breast cancer: Prognosis and response to
systemic therapy. Ann Oncol. 10:413–419. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chen IC, Lin CH, Huang CS, Lien HC, Hsu C,
Kuo WH, Lu YS and Cheng AL: Lack of efficacy to systemic
chemotherapy for treatment of metaplastic carcinoma of the breast
in the modern era. Breast Cancer Res Treat. 130:345–351. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Noro A, Ishitobi M, Hanamura N, Kashikura
Y, Yamashita M, Kozuka Y, Saito K, Mizuno T and Ogawa T: A case of
metaplastic squamous cell carcinoma of the breast that showed a
pathological complete response after neoadjuvant chemotherapy with
weekly paclitaxel. Am J Case Rep. 23:e9350352022.PubMed/NCBI
|
|
44
|
Cortes J, Cescon DW, Rugo HS, Nowecki Z,
Im SA, Yusof MM, Gallardo C, Lipatov O, Barrios CH, Holgado E, et
al: Pembrolizumab plus chemotherapy versus placebo plus
chemotherapy for previously untreated locally recurrent inoperable
or metastatic triple-negative breast cancer (KEYNOTE-355): A
randomised, placebo-controlled, double-blind, phase 3 clinical
trial. Lancet. 396:1817–1828. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
FDA, . FDA grants accelerated approval to
pembrolizumab for locally recurrent unresectable or metastatic
triple negative breast cancer. 2020.
|
|
46
|
Joneja U, Vranic S, Swensen J, Feldman R,
Chen W, Kimbrough J, Xiao N, Reddy S, Palazzo J and Gatalica Z:
Comprehensive profiling of metaplastic breast carcinomas reveals
frequent overexpression of programmed death-ligand 1. J Clin
Pathol. 70:255–259. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ladwa A, Elghawy O, Schroen A, Abernathy
K, Schlefman J and Dillon P: Complete response of triple-negative
metaplastic carcinoma of the breast using pembrolizumab. Case Rep
Oncol. 16:1129–1135. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Gul A, Alberty-Oller JJ, Sandhu J,
Ayala-Bustamante E and Adams S: A case of pathologic complete
response to neoadjuvant chemotherapy and pembrolizumab in
metaplastic breast cancer. JCO Precis Oncol. 7:e22005062023.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Gorshein E, Matsuda K, Riedlinger G, Sokol
L, Rodriguez-Rodriguez L, Eladoumikdachi F, Grandhi M, Ganesan S,
Toppmeyer DL, Potdevin L, et al: Durable response to PD1 inhibitor
pembrolizumab in a metastatic, metaplastic breast cancer. Case Rep
Oncol. 14:931–937. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Fu Y, Liu J and Jiang Y: Partial response
after toripalimab plus anlotinib for advanced metaplastic breast
carcinoma: A case report. Front Endocrinol (Lausanne).
13:8107472022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Adams S, Othus M, Patel SP, Miller KD,
Chugh R, Schuetze SM, Chamberlin MD, Haley BJ, Storniolo AMV, Reddy
MP, et al: A Multicenter phase II trial of ipilimumab and nivolumab
in unresectable or metastatic metaplastic breast cancer: Cohort 36
of dual anti-CTLA-4 and anti-PD-1 blockade in rare tumors (DART,
SWOG S1609). Clin Cancer Res. 28:271–278. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Corso G, Marabelli M, Calvello M, Gandini
S, Risti M, Feroce I, Mannucci S, Girardi A, De Scalzi AM, Magnoni
F, et al: Germline pathogenic variants in metaplastic breast cancer
patients and the emerging role of the BRCA1 gene. Eur J Hum Genet.
31:1275–1282. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Lane R, Yan F, Higgins D and Agarwal G:
Synchronous metaplastic breast carcinoma and lung adenocarcinoma: A
rare case and review of the literature. BMJ Case Rep.
13:e2324212020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Rugo HS, Bardia A, Tolaney SM, Arteaga C,
Cortes J, Sohn J, Marmé F, Hong Q, Delaney RJ, Hafeez A, et al:
TROPiCS-02: A phase III study investigating sacituzumab govitecan
in the treatment of HR+/HER2-metastatic breast cancer. Future
Oncol. 16:705–715. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Bardia A, Hurvitz SA, Tolaney SM, Loirat
D, Punie K, Oliveira M, Brufsky A, Sardesai SD, Kalinsky K, Zelnak
AB, et al: Sacituzumab govitecan in metastatic triple-negative
breast cancer. N Engl J Med. 384:1529–1541. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Püsküllüoğlu M, Rudzińska A and
Pacholczak-Madej R: Antibody-drug conjugates in HER-2 negative
breast cancers with poor prognosis. Biochim Biophys Acta Rev
Cancer. 1878:1889912023. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Reddy TP, Rosato RR, Li X, Moulder S,
Piwnica-Worms H and Chang JC: A comprehensive overview of
metaplastic breast cancer: Clinical features and molecular
aberrations. Breast Cancer Res. 22:1212020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Hayes MJ, Thomas D, Emmons A, Giordano TJ
and Kleer CG: Genetic changes of Wnt pathway genes are common
events in metaplastic carcinomas of the breast. Clin Cancer Res.
14:4038–4044. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Esbah O, Turkoz FP, Turker I, Durnali A,
Ekinci AS, Bal O, Sonmez OU, Budakoglu B, Arslan UY and Oksuzoglu
B: Metaplastic breast carcinoma: Case series and review of the
literature. Asian Pac J Cancer Prev. 13:4645–4649. 2012. View Article : Google Scholar : PubMed/NCBI
|