Introduction
Lung cancer is currently one of the most common
malignancies in humans, and its incidence and mortality rates
ranking among the highest. The common symptoms of lung cancer are
cough, dyspnea, pain and weight loss. The main risk factors of lung
cancer include smoking, environmental exposure containing
carcinogens and chronic obstructive pulmonary disease (1). The epidermal growth factor receptor
(EGFR) gene is the most prevalent driver gene in patients
with non-small cell lung cancer (NSCLC). Among patients with NSCLC
harboring EGFR mutations, common mutations, such as EGFR
exon 19 deletion and exon 20 T790M missense mutation, account for
75–80% of all cases. Rare EGFR mutations, which have an
incidence rate of ~10%, are defined as mutations other than exon 19
deletions and exon 21 L858R substitutions (2).
Primary NSCLC metastasis to the breast is relatively
rare, accounting for <0.5% of cases of NSCLC (3). This type of metastasis typically
signifies disease progression and may be associated with drug
resistance. The T790M mutation in EGFR exon 20 was the first
resistance mechanism to tyrosine kinase inhibitors (TKIs)
identified in EGFR-mutant NSCLC (4). Currently, EGFR-TKIs are
considered the standard first-line treatment for patients with
locally advanced or metastatic NSCLC harboring sensitizing
EGFR mutations (5,6). The present case report mainly reports
a typical case of rare gene mutation lung cancer that eventually
metastasized to breast cancer, providing valuable clinical
management experience and treatment ideas.
Case report
In August 2023, a 36-year-old female patient
presented to a general physician in The People's Hospital of
Tiantai County (Taizhou, China) with a cough and asthma that had
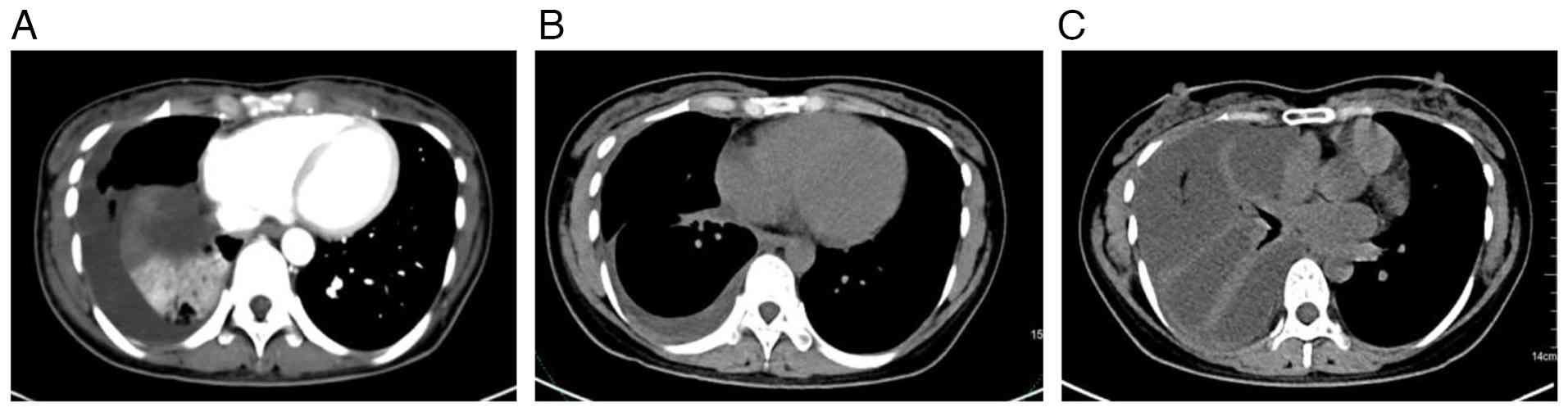
persisted for >1 month. A lung CT scan (Fig. 1A) revealed a mass near the hilum of
the right lung, suggesting a malignant tumor with partial
atelectasis of the right lower lung. A thoracic puncture and
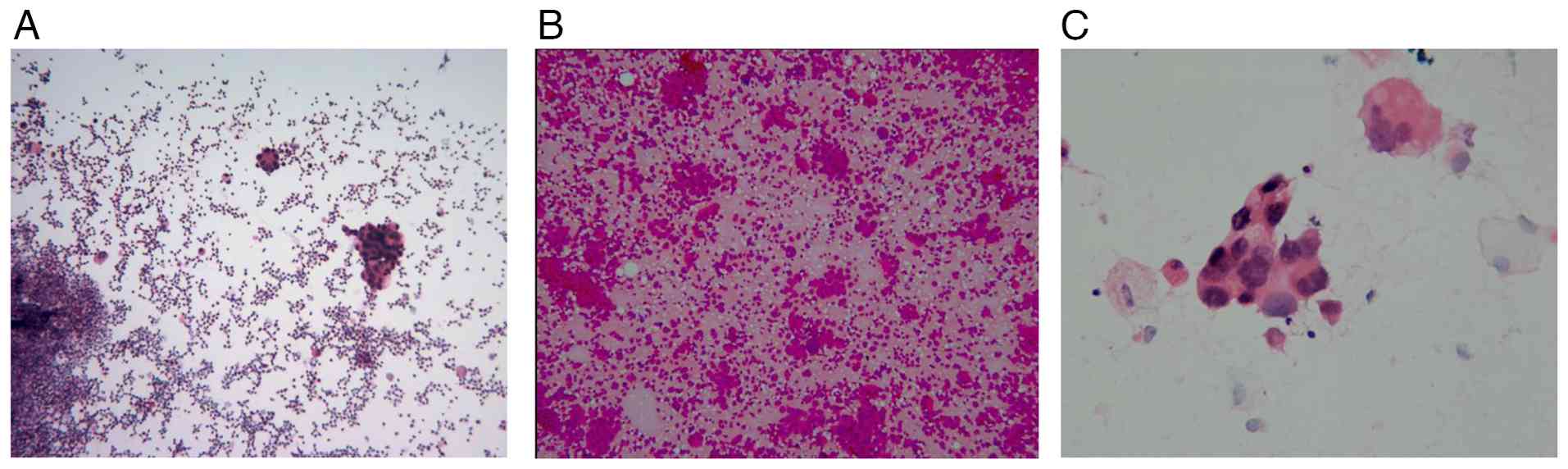
drainage were performed. Cytopathology of centrifugal smears from
the right pleural effusion and from bronchoalveolar lavage fluid-a
diagnostic technique that analyzes fluid retrieved from the
alveolar surface-revealed red blood cells, inflammatory cells and
heterogeneous cell clusters, findings diagnostic for adenocarcinoma
(Fig. 2). Genetic analysis (Data
S1) detected an EGFR exon 19 deletion
[c.22402254del; p.L747T751del; variant allele
frequency (VAF)=8.49%] and an exon 20 T790M mutation (c.2369C>T;
p.T790M; VAF=10.53%). Following the Chinese Society of Clinical
Oncology and National Comprehensive Cancer Network guidelines for
lung cancer (7,8), the patient was treated with
furmonertinib (80 mg once daily in the morning), a third-generation
targeted antitumor therapy.
After 8 months of furmonertinib treatment (April
2024), a CT scan of the upper abdomen and pelvic cavity (Fig. 1B) indicated that the mass near the
hilum of the right lung was markedly smaller compared with that at
baseline, and the enlarged lymph nodes in the mediastinum were no
longer detectable. Furthermore, the multiple small nodules in the
right lung were reduced in size and the inflammation in both lungs
had resolved. There was also notable improvement in the right lung
cancer lymphadenitis, right pleural thickening and pleural
effusion.
A right breast tumor was incidentally identified 1
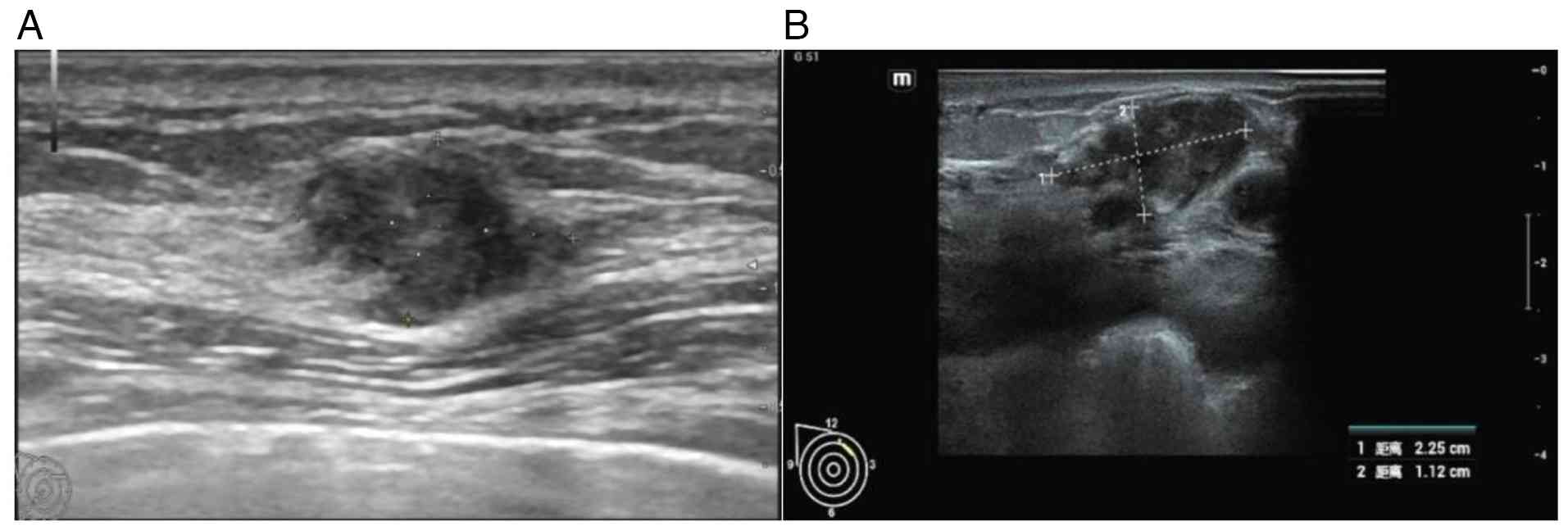
month after a CT scan (June 2024). A color Doppler ultrasonography
examination revealed a solid mass in the right breast, classified
as Breast Imaging Reporting and Data System (BI-RADS) category 4A
(Fig. 3A) (9). The right breast tumor was resected
under local anesthesia. Based on postoperative pathology,
immunohistochemistry (IHC) results and clinical history, the tumor
was diagnosed as breast cancer secondary to a primary lung
adenocarcinoma. An intravascular tumor thrombus was observed. IHC
results (Data S1) demonstrated that the patient was positive for
CK7, Ki-67 (~60% +), Napsin A and thyroid transcription factor 1
(TTF-1) and negative for estrogen receptor, progesterone receptor,
CK5/6, Sox10, GATA binding protein 3, mammaglobin and HER2
(Fig. 4).
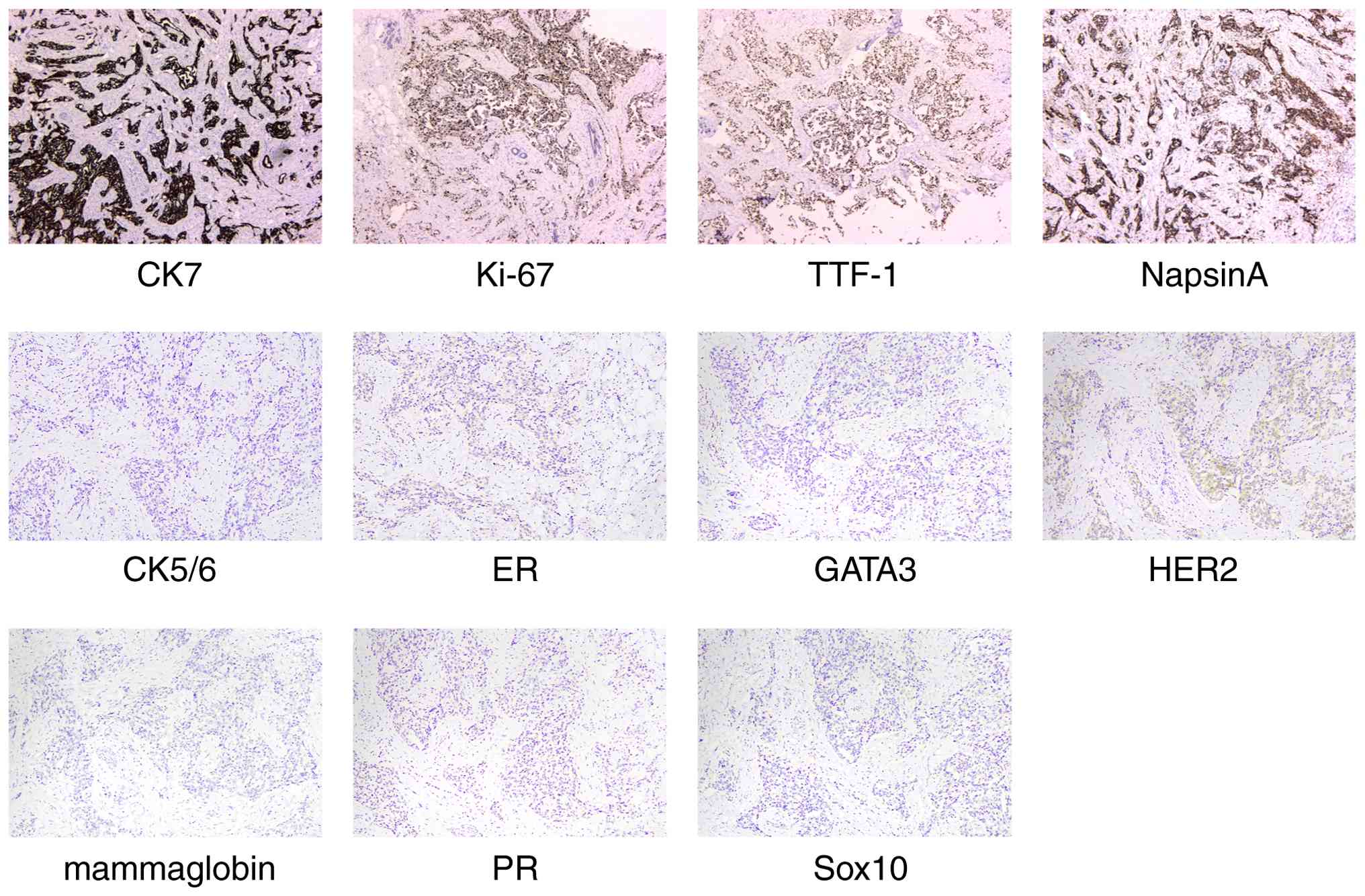 | Figure 4.Immunohistochemical results: Positive
for CK7, Ki-67 (~60%+), Napsin A and TTF-1 and negative for ER, PR,
CK5/6, Sox10, GATA3, mammaglobin and HER2 (magnification, ×200).
ER, estrogen receptor; PR, progesterone receptor; GATA3, GATA
binding protein 3; TTF-1, thyroid transcription factor 1. |
Asthma symptoms recurred 1 year after the first
onset of illness (August 2024). Thin-layer CT and three-dimensional
lung imaging (Fig. 1C) detected
progressive atelectasis and a considerable right chest effusion.
The right lung was not fully inflated and new inflammation was
noted. Pleural fluid smears indicated cancer cells. Genetic
analysis of the pleural effusion exhibited an EGFR exon 19
deletion and the exon 20 T790M missense mutation. The patient
returned for a follow-up breast examination two months later, in
August 2024. A breast color Doppler ultrasonography (August 2024)
detected a solid mass of 2.25×1.12 cm in the upper quadrant of the
right breast. The mass had blurred edges and was classified as
BI-RADS category 5 (Fig. 3B). A
physical examination indicated that the mass was hard and had poor
mobility. After the patient's first diagnosis, follow-up was
conducted every 2–3 months, with the last follow-up in February
2024. No other abnormalities were found during the follow-up.
Therefore, based on the patient's medical history, recurrence of
metastatic cancer after local resection is considered.
Discussion
The EGFR exon 20 T790M mutation was the first
resistance mechanism to TKIs identified in EGFR-mutant
NSCLC. In 2005, Kobayashi et al (4) reported a 71-year-old male patient with
NSCLC treated with an EGFR-TKI. The patient achieved
complete remission after gefitinib treatment but relapsed 2 years
later. The researchers examined new biopsy samples and identified
that, in addition to the EGFR exon deletion 19, the patient
had developed an EGFR exon 20 T790M mutation, a C-T
transformation in codon 790 in which threonine is replaced with
methionine. Pao et al (10)
analyzed samples from six patients with disease progression after
EGFR-TKI treatment and detected the exon 20 T790M mutation
in three of them. The researchers further compared the drug
resistance of tumor cells with both L858R and T790M gene mutations
with those with the L858R gene mutation alone. This comparison
confirmed that the T790M mutation increased drug resistance by
100-fold. Among the various EGFR mutation types, the most
common are in-frame deletions in exon 19 (19 del; ~44%), which
encompass mutations such as L747 to E749 and the L858R point
mutation in exon 21 (~41%) (11).
Notably, tyrosine kinases exhibit reduced affinity to ATP in the
presence of EGFR exon 19 del and L858R mutations. However,
they have a relatively high affinity to EGFR-TKIs and,
therefore, generate an antitumor effect (12). In the present case, the patient had
both an EGFR exon 19 deletion and the exon 20 T790M missense
mutation at baseline, which meant that both sensitive and
drug-resistant mutations were present. A cohort study conducted by
Hong et al (13) reported
that the coexistence of these mutations negatively affected the
efficacy of EGFR-TKIs. Their study enrolled 58 patients with
advanced NSCLC and EGFR mutations. The mutation status in
the circulating tumor DNA before TKI treatment was detected using
next-generation sequencing technology. The associations between
coexisting mutations and clinicopathological features, curative
effects and survival time were analyzed. Their results demonstrated
that the objective remission rate (44 vs. 77%), median
progression-free survival (6.20 vs. 18.77 months) and median
overall survival (22.70 months vs. not reached) of patients with
coexisting mutations were markedly worse compared with those of
patients without coexisting mutations. Coexisting mutations
remained an independent prognostic predictor after multivariate
analysis corrected for EGFR mutation subtypes and
clinicopathological features (13).
The rarity and mechanism of breast metastasis
secondary to a primary lung tumor have been reported in relevant
literature. The incidence of breast metastasis from primary tumors
outside the breast is 0.5–6.0% (14). A previous study of 6,668 patients
with various malignant tumors, including lung cancer, liver cancer
and kidney cancer, reported that the incidence of breast metastasis
from solid malignant tumors was 0.76% (15). Of these cases, 16% were metastases
from primary lung tumors. Another previous study identified 45
cases of secondary breast malignant tumor (0.56%) among 6,334
patients with breast cancer. The most common primary tumor source
was lung cancer (33.3%) (16). The
metastasis of cancer cells from the lungs to the breast can occur
through the blood and lymph pathways (17). Huang et al (18) analyzed the path of lung cancer
metastasis to the breast through the lymphatic system. They
proposed that lung cancer cells spread onto the pleura, transferred
to the axillary lymph nodes through the lymphatic system and then
retrogradely transferred to the ipsilateral breast through the
lymphatic vessels. Based on this hypothesis, patients may exhibit
ipsilateral pleural thickening with pleural effusion, breast
metastasis and axillary lymph node enlargement. This hypothesis was
consistent with the characteristics of the present case. Ding et
al (19) analyzed a case of
lung cancer with contralateral breast metastasis. In such cases,
tumor cells may have entered the lymphatic circulation or, through
a vein, into the systemic circulation, delivering the tumor cells
through the thoracic duct or blood circulation to the contralateral
breast.
Regarding the differentiation between primary and
secondary breast tumors, primary breast cancer often has fibrous
connective tissue hyperplasia in the tumor area, while metastatic
lesions lack this proliferative reaction. As a result, their size
recorded during clinical examination is often similar to that
observed in breast radiographs or ultrasonography (20). Metastatic tumors are more likely to
occur in superficial positions than primary breast lesions and
often do not cause clinical manifestations such as skin depression,
possibly because metastatic tumors are often located outside the
ductal system (21). These tumors
often manifest as a palpable, painless, rapidly-growth mass with a
clear boundary, mostly in the upper outer quadrant (22).
Radiologically, secondary breast metastasis lacks
the structural distortion, burr and microcalcification observed in
primary breast tumors. It also lacks the smooth outline of benign
masses (for example, fibroadenoma or cyst) (23). The absence of carcinoma in
situ is another characteristic of metastatic lesions (24). Histologically, almost all
neuroendocrine tumors that have metastasized to the breast are
strongly and diffusely positive for the neuroendocrine markers
synaptophysin and chromaffin (25).
Neuroendocrine metastases to the breast are often positive for
TTF-1, while primary breast cancer is negative for TTF-1 (26). However, certain studies have
reported TTF-1+ primary breast cancer. For example,
previous studies summarizing the expression of TTF-1 in breast
metastases of primary lung cancer identified 22 positive and four
negative cases (27,28). These findings demonstrated that
TTF-1 is a notable biomarker of lung cancer metastasis to the
breast. In previous studies, increasing evidence has demonstrated
that even metastatic carcinomas derived from the same primary tumor
may exhibit divergent immunohistochemical profiles, largely
attributable to tissue heterogeneity and potential alterations in
receptor expression during metastatic progression (29–31).
In the present case, the positivity for TTF-1 and Napsin A, both
highly specific markers of pulmonary origin, along with the absence
of breast-specific markers, markedly supports a lung origin for the
breast lesions (32). Therefore,
TTF-1 immunoreactivity should not be ignored in the pathological
examination of breast lesions from patients with a known history of
lung cancer.
In conclusion, although breast metastasis of lung
cancer is rare, accurate diagnosis, clear staging and a correct
treatment plan are of notable clinical significance. If breast
nodules are identified in patients with malignant lung tumors,
screening is suggested to avoid misdiagnosis. It is particularly
key to identify the source of tumors by combining medical imaging
and IHC results.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data related to gene sequencing can be found in
the Mendeley Data database (doi: 10.17632/wpw7mstpp8.1) at the
following URL: https://data.mendeley.com/datasets/wpw7mstpp8/1. The
rest of the data generated in the present study may be requested
from the corresponding author.
Authors' contributions
FW conceptualized the present case report. XL
devised the methodology. GC performed the follow-up collection of
the patient data and validated the data. TY conducted the formal
analysis. YX conducted the patient follow-up and wrote the original
draft. All authors read and approved the final manuscript. TY and
FW confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
The present study was approved by Ethics Committee
of Tiantai Hospital [approval no. Ethical Review 2025 Other No.
(014); Taizhou, China]. The patient in the present case report
agreed to participate in the present study.
Patient consent for publication
The patient in the present case report provided
written informed consent for publication of the present study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Leiter A, Veluswamy RR and Wisnivesky JP:
The global burden of lung cancer: Current status and future trends.
Nat Rev Clin Oncol. 20:624–639. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ke EE and Wu YL: Afatinib in the
first-line treatment of Epidermal-growth-factor-receptor
mutation-positive non-small cell lung cancer: A review of the
clinical evidence. Ther Adv Respir Dis. 10:256–264. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ramar K, Pervez H, Potti A and Mehdi S:
Breast metastasis from non-small-cell lung carcinoma. Med Oncol.
20:181–184. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kobayashi S, Boggon TJ, Dayaram T, Jänne
PA, Kocher O, Meyerson M, Johnson BE, Eck MJ, Tenen DG and Halmos
B: EGFR mutation and resistance of non-small-cell lung cancer to
gefitinib. N Engl J Med. 352:786–792. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ettinger DS, Wood DE, Aisner DL, Akerley
W, Bauman JR, Bharat A, Bruno DS, Chang JY, Chirieac LR, D'Amico
TA, et al: Non-Small cell lung cancer, version 3.2022, NCCN
clinical practice guidelines in oncology. J Natl Compr Canc Netw.
20:497–530. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Reck M, Popat S, Reinmuth N, De Ruysscher
D, Kerr KM and Peters S; ESMO Guidelines Working Group, :
Metastatic non-small-cell lung cancer (NSCLC): ESMO Clinical
Practice Guidelines for diagnosis, treatment and follow-up. Ann
Oncol. 25 (Suppl 3):iii27–iii39. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Langer CJ: Epidermal growth factor
receptor inhibition in mutation-positive non-small-cell lung
cancer: Is afatinib better or simply newer? J Clin Oncol.
31:3303–3306. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cheng Y, He Y, Li W, Zhang HL, Zhou Q,
Wang B, Liu C, Walding A, Saggese M, Huang X, et al: Osimertinib
versus comparator EGFR TKI as First-line treatment for EGFR-mutated
advanced NSCLC: FLAURA China, A randomized study. Target Oncol.
16:165–176. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Amin MB, Greene FL, Edge SB, Compton CC,
Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR and
Winchester DP: The eighth edition AJCC Cancer Staging Manual:
Continuing to build a bridge from a population-based to a more
‘Personalized’ approach to cancer staging. CA Cancer J Clin.
67:93–99. 2017.PubMed/NCBI
|
|
10
|
Pao W, Miller VA, Politi KA, Riely GJ,
Somwar R, Zakowski MF, Kris MG and Varmus H: Acquired resistance of
lung adenocarcinomas to gefitinib or erlotinib is associated with a
second mutation in the EGFR kinase domain. PLoS Med. 2:e732005.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gazdar AF: Activating and resistance
mutations of EGFR in non-small-cell lung cancer: Role in clinical
response to EGFR tyrosine kinase inhibitors. Oncogene. 28 (Suppl
1):S24–S31. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Carey KD, Garton AJ, Romero MS, Kahler J,
Thomson S, Ross S, Park F, Haley JD, Gibson N and Sliwkowski MX:
Kinetic analysis of epidermal growth factor receptor somatic mutant
proteins shows increased sensitivity to the epidermal growth factor
receptor tyrosine kinase inhibitor, erlotinib. Cancer Res.
66:8163–8171. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hong S, Gao F, Fu S, Wang Y, Fang W, Huang
Y and Zhang L: Concomitant genetic alterations with response to
treatment and epidermal growth factor receptor tyrosine kinase
inhibitors in patients with EGFR-mutant advanced non-small cell
lung cancer. JAMA Oncol. 4:739–742. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yeh CN, Lin CH and Chen MF: Clinical and
ultrasonographic characteristics of breast metastases from
extramammary malignancies. Am Surg. 70:287–290. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Surov A, Fiedler E, Holzhausen HJ, Ruschke
K, Schmoll HJ and Spielmann RP: Metastases to the breast from
non-mammary malignancies: Primary tumors, prevalence, clinical
signs, and radiological features. Acad Radiol. 18:565–574. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sauer T: Fine-needle aspiration cytology
of extra mammary metastatic lesions in the breast: A retrospective
study of 36 cases diagnosed during 18 years. Cytojournal. 7:102010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mun SH, Ko EY, Han BK, Shin JH, Kim SJ and
Cho EY: Breast metastases from extramammary malignancies: Typical
and atypical ultrasound features. Korean J Radiol. 15:20–28. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Huang HC, Hang JF, Wu MH, Chou TY and Chiu
CH: Lung adenocarcinoma with ipsilateral breast metastasis: A
simple coincidence? J Thorac Oncol. 8:974–979. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ding Y, Zhou J and Shan H: One case report
of male left lung cancer patients with contralateral breast
metastasis. Zhongguo Fei Ai Za Zhi. 13:1082–1084. 2010.(In
Chinese). PubMed/NCBI
|
|
20
|
Bitencourt AGV, Gama RRM, Graziano L,
Negrão EMS, Sabino SMPS, Watanabe AHU, Guatelli CS, Souza JA, Mauad
EC and Marques EF: Breast metastases from extramammary
malignancies: Multimodality imaging aspects. Br J Radiol.
90:201701972017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ji FF, Gao P, Wang JG, Zhao J and Zhao P:
Contralateral breast metastasis from pulmonary adenocarcinoma: Two
cases report and literature review. J Thorac Dis. 4:384–389.
2012.PubMed/NCBI
|
|
22
|
McIntosh IH, Hooper AA, Millis RR and
Greening WP: Metastatic carcinoma within the breast. Clin Oncol.
2:393–401. 1976.PubMed/NCBI
|
|
23
|
Lee AH: The histological diagnosis of
metastases to the breast from extramammary malignancies. J Clin
Pathol. 60:1333–1341. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Klingen TA, Klaasen H, Aas H, Chen Y and
Akslen LA: Secondary breast cancer: A 5-year population-based study
with review of the literature. APMIS. 117:762–767. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mohanty SK, Kim SA, DeLair DF, Bose S,
Laury AR, Chopra S, Mertens RB and Dhall D: Comparison of
metastatic neuroendocrine neoplasms to the breast and primary
invasive mammary carcinomas with neuroendocrine differentiation.
Mod Pathol. 29:788–798. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Chong CR, Wirth LJ, Nishino M, Chen AB,
Sholl LM, Kulke MH, McNamee CJ, Jänne PA and Johnson BE:
Chemotherapy for locally advanced and metastatic pulmonary
carcinoid tumors. Lung Cancer. 86:241–246. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Klingen TA, Chen Y, Gundersen MD, Aas H,
Westre B and Sauer T: Thyroid transcription factor-1 positive
primary breast cancer: A case report with review of the literature.
Diagn Pathol. 5:372010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ordonez NG: Value of thyroid transcription
factor-1 immunostaining in tumor diagnosis: A review and update.
Appl Immunohistochem Mol Morphol. 20:429–444. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Testa U, Castelli G and Pelosi E: Breast
Cancer: A molecularly heterogenous disease needing Subtype-specific
treatments. Med Sci (Basel). 8:182020.PubMed/NCBI
|
|
30
|
Song SG, Kim S, Koh J, Yim J, Han B, Kim
YA, Jeon YK and Chung DH: Comparative analysis of the tumor
immune-microenvironment of primary and brain metastases of
non-small-cell lung cancer reveals organ-specific and EGFR
mutation-dependent unique immune landscape. Cancer Immunol
Immunother. 70:2035–2048. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Vidarsdottir H, Tran L, Nodin B, Jirström
K, Planck M, Jönsson P, Mattsson JSM, Botling J, Micke P and
Brunnström H: Immunohistochemical profiles in primary lung cancers
and epithelial pulmonary metastases. Hum Pathol. 84:221–230. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ye J, Findeis-Hosey JJ, Yang Q, McMahon
LA, Yao JL, Li F and Xu H: Combination of napsin A and TTF-1
immunohistochemistry helps in differentiating primary lung
adenocarcinoma from metastatic carcinoma in the lung. Appl
Immunohistochem Mol Morphol. 19:313–317. 2011. View Article : Google Scholar : PubMed/NCBI
|