Introduction
Penile metastasis from prostate cancer (PCa) is
exceptionally rare, reportedly occurring in <0.5% of PCa cases,
and is associated with a poor prognosis (1). While the bones and lymph nodes are
common metastatic sites, penile involvement can also arise by
mechanisms including retrograde venous spread due to anatomical
connections between the pelvic venous plexuses and penile dorsal
veins (2). The clinical
presentation of penile metastases is often non-specific (3), making differentiation from primary
penile lesions or other benign conditions difficult without
sophisticated imaging and histopathological confirmation. This
diagnostic ambiguity underscores the need for heightened clinical
suspicion in patients with advanced PCa presenting with new penile
abnormalities.
A key aspect in managing advanced PCa, including
rare metastatic presentations, is to understand the molecular
characteristics of the tumor. The mechanism by which CDK12
regulates homologous recombination repair (HRR) genes is linked to
intronic polyadenylation (IPA) (4).
IPA can lead to aberrant mRNA splicing and the emergence of mRNA
isoforms characterized by alterations in the 3′untranslated regions
and coding sequences, thereby affecting the stability, translation
efficiency and coding potential of HRR-related mRNAs (5).
The present case report describes a patient with
CDK12-mutated metastatic castration-resistant PCa (mCRPC)
who developed a rare penile metastasis after a prolonged period of
response to initial androgen deprivation therapy (ADT). The study
details the diagnostic workup, which utilized prostate-specific
membrane antigen (PSMA) positron emission tomography/computed
tomography (PET/CT), and the subsequent therapeutic strategy guided
by molecular profiling. This case underscores the clinical
challenge of penile metastasis, illustrates the pivotal role of
comprehensive genomic testing in guiding personalized therapy for
mCRPC, and presents a novel and sustained response to a combination
of enzalutamide and the poly(ADP-ribose) polymerase (PARP)
inhibitor olaparib in this specific genetic context.
Case report
A 65-year-old man was admitted to Nanfang Hospital
(Guangzhou, China) in July 2016 with progressive difficulty in
urinating. The patient had a history of type 2 diabetes. The
initial prostate-specific antigen (PSA) level was recorded as
>500 ng/ml (normal range, 0–4ng/ml). Magnetic resonance imaging
(MRI) suggested advanced metastatic PCa [8th American Joint
Committee On Cancer (AJCC) Staging, AJCC T4N1M1] (6) with invasion of the left seminal
vesicle, bladder and rectum, pelvic lymph node involvement, and
right iliac acetabular bone metastasis. Histological examination of
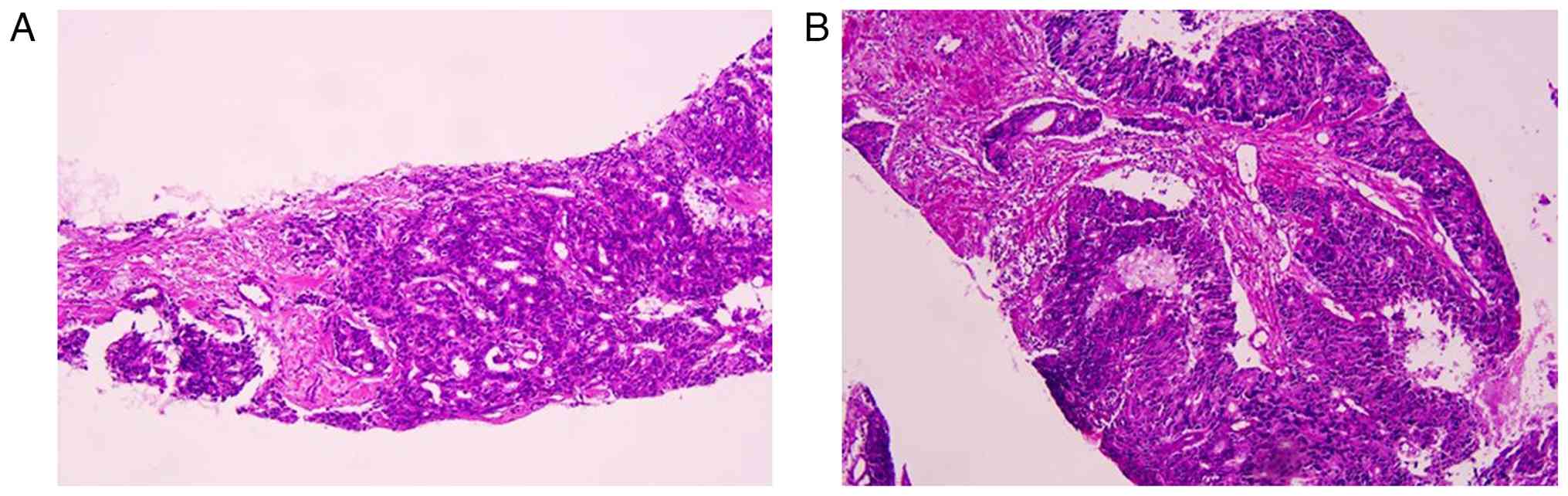
a biopsy tissue specimen confirmed acinar adenocarcinoma with a
Gleason score of 4+3=7 (Fig. 1).
The patient was started on endocrine therapy with goserelin (10.8
mg every 3 months) and bicalutamide (50 mg once daily) in July
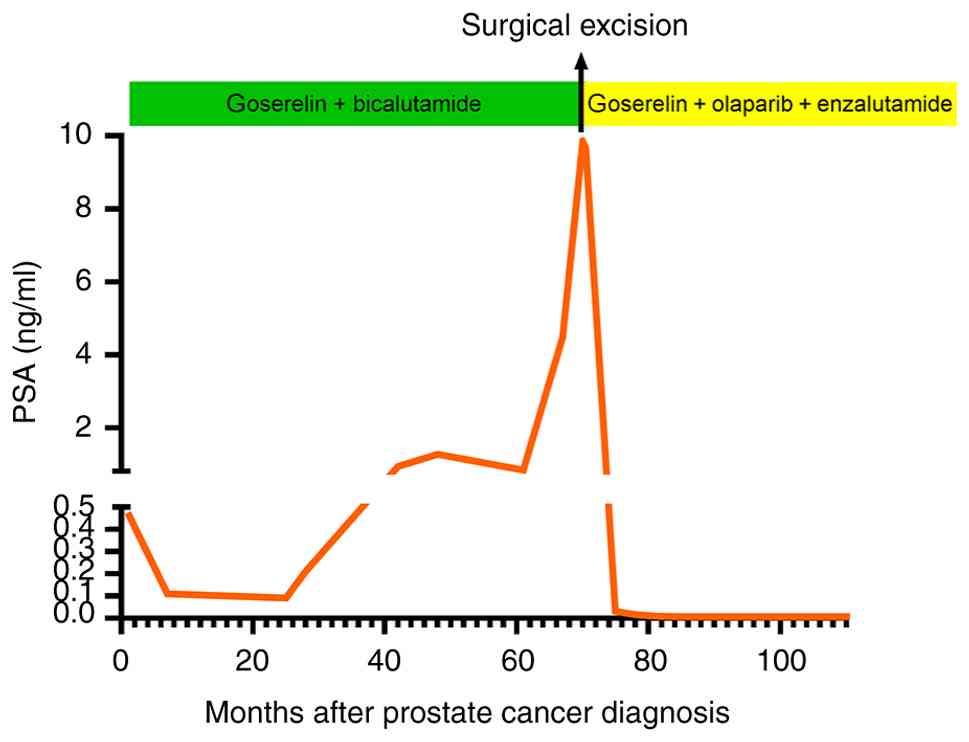
2016. The PSA levels subsequently decreased and reached a nadir of
0.09 ng/ml in July 2018 (Fig. 2),
accompanied by an improvement in urinary symptoms.
Beginning in January 2022, the PSA levels exhibited
a progressive increase, reaching 9.84 ng/ml in April 2022. During
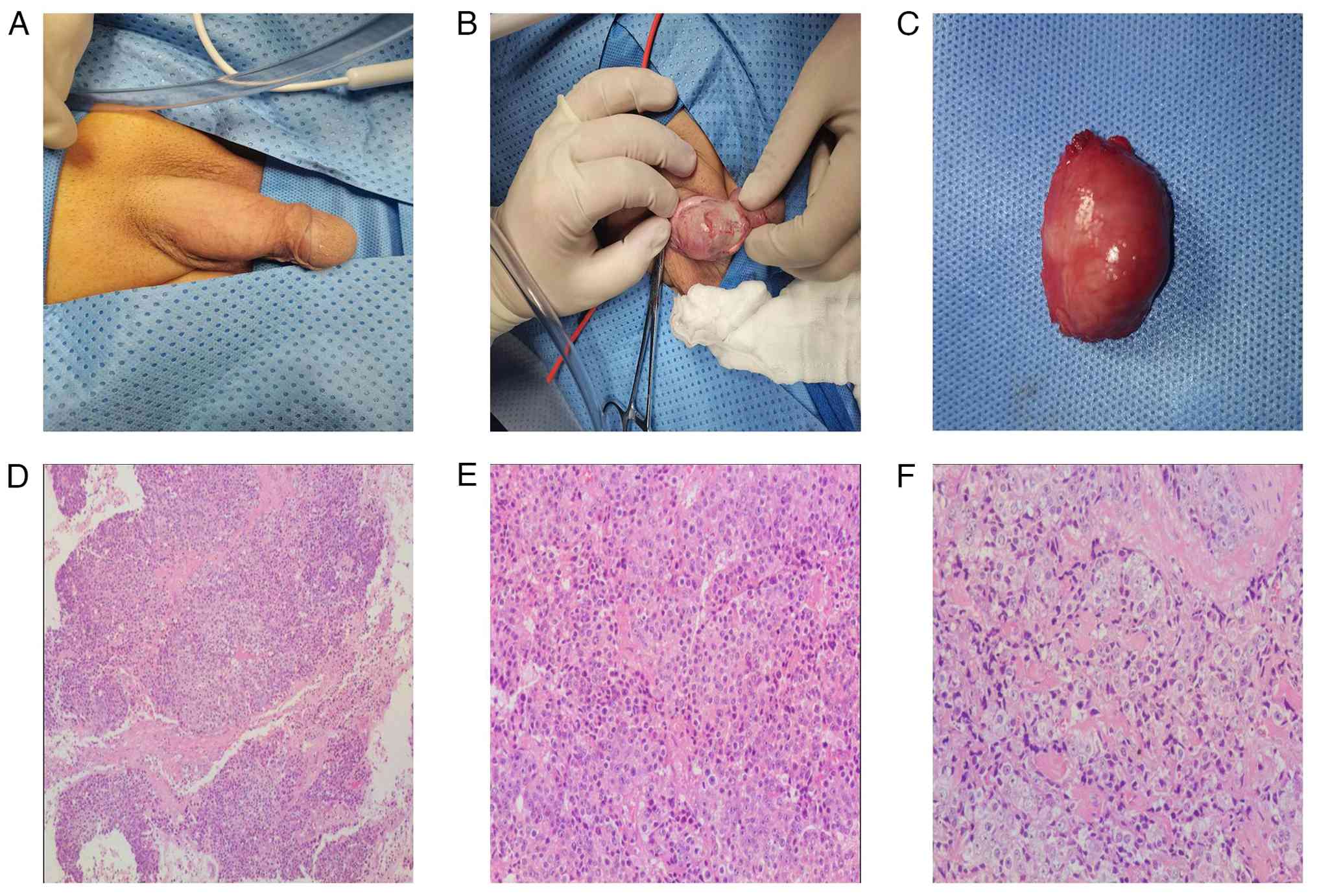
this period, a gradually enlarging mass was noted on the right side
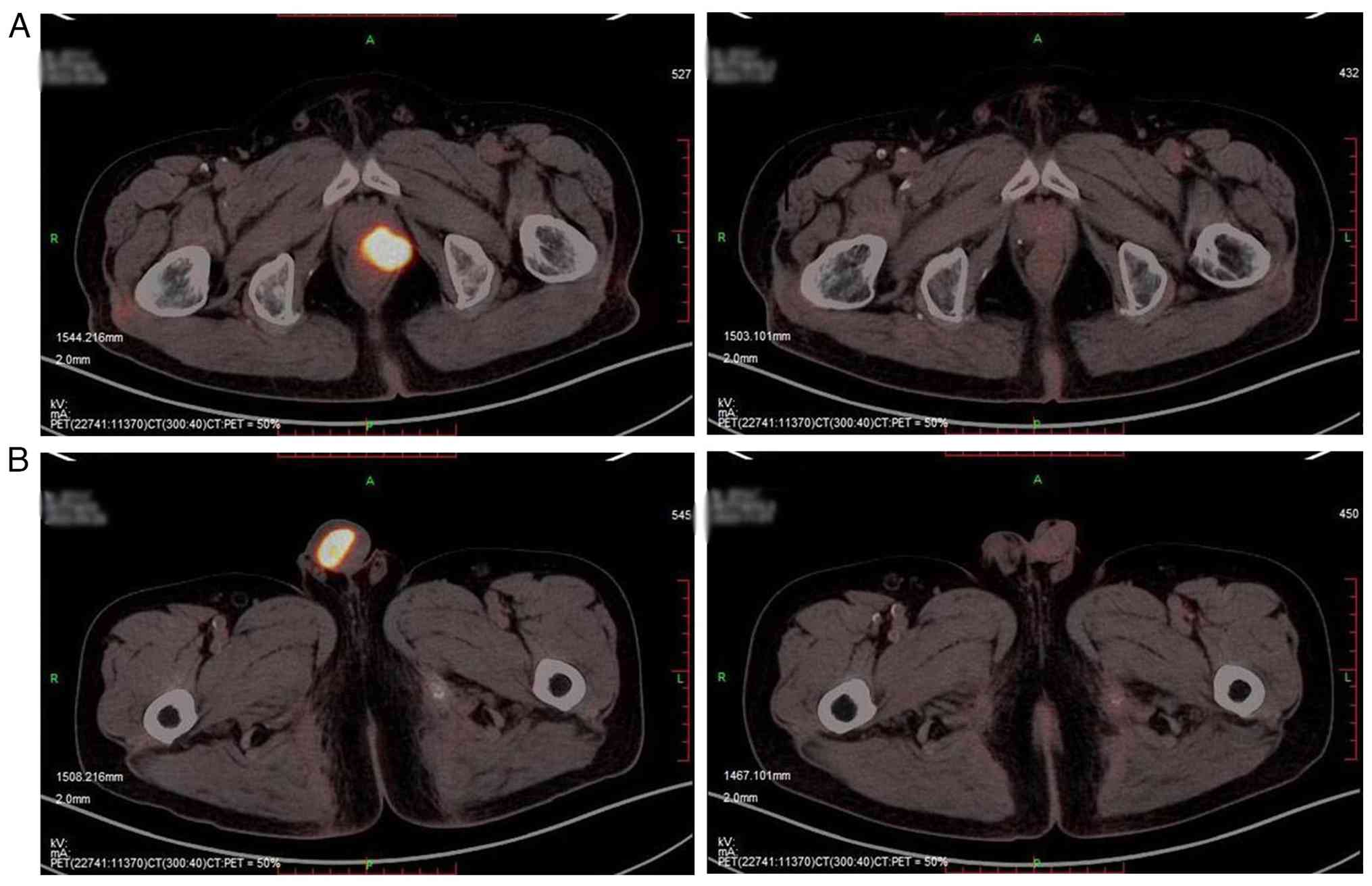
of the penis. PSMA PET/CT (Fig. 3A)
showed increased PSMA uptake in the left seminal vesicle and the
left anterior wall of the rectum, suggesting the PCa had invaded
these areas. Based on the PET/CT, there is increased PSMA uptake in
the penile mass on the right side, which is considered malignant.
No abnormal uptake was observed in previously involved lymph nodes,
and no other metastatic foci were identified. Therefore, the
patient was considered eligible for surgery to remove the penile
mass. In April 2022, the penile mass was removed under local
anesthesia.
Histopathological examination of the penile mass
revealed infiltrating poorly differentiated PCa (Fig. 4). Tissue specimens were fixed in 10%
formalin, embedded in paraffin and sectioned into 4-µm serial
slices. The sections were mounted on APES-coated slides (Beijing
Zhongshan Jinqiao Biotechnology Co., Ltd.), baked at 60°C for 1–2 h
and stored at 4°C for later use. Immunohistochemistry was performed
using the streptavidin-peroxidase (SP) method: After dewaxing,
rehydration and antigen retrieval with citrate buffer (pH 6.0) via
microwave heating, endogenous peroxidase activity was blocked with
3% H2O2 (Reagent A of Ultra-Sensitive™ S-P
kit; Fuzhou Maixin Biotechnology Development Co., Ltd.) at room
temperature for 10 min; non-specific binding was blocked with 10%
goat serum (Reagent B of the same kit) at room temperature for 10
min; the primary antibody against Cripto-1 (diluted 1:75) was
applied and incubated overnight at 4°C; this was followed by
sequential incubation with biotin-labeled goat anti-mouse/rabbit
IgG (Reagent C) and streptavidin-peroxidase solution (Reagent D),
each at room temperature for 10 min; color development was
performed using diaminobenzidine (DAB) chromogen solution (Beijing
Zhongshan Jinqiao Biotechnology Co., Ltd.) for 1–3 min; sections
were counterstained with hematoxylin for 5 min, then dehydrated,
cleared and mounted. All antibodies and kits were purchased from
Fuzhou Maixin Biotechnology Development Co., Ltd., DAB reagent and
APES-coated slides were obtained from Beijing Zhongshan Jinqiao and
the HRP-labeled secondary antibody was sourced from Wuhan Boster
Biological Technology, Ltd. Stained sections were examined under a
BH-2 light microscope (Olympus Corp.). Control experiments were
performed by replacing the primary antibody with PBS while keeping
all other steps identical. Immunohistochemical examination revealed
the mass was positive for cytokeratin (CK), PSA, α-methylacyl-CoA
racemase (P504S) and Ki-67 40%, and focally positive for CK20. In
addition, the mass was negative for CK7, p40, synaptophysin (Syn),
uroplakin and spalt-like transcription factor 4 (SALL4). CK
positivity indicates that the mass is of epithelial origin. PSA and
P504S supports a prostatic origin. P40 negativity confirms basal
cell loss and verifies malignancy. Meanwhile, CK7 negativity and
uroplakin negativity rule out urothelial carcinoma; Syn negativity
rules out neuroendocrine tumors; and SALL4 negativity rules out
germ cell tumors (7) (Fig. 5). Therefore, the diagnosis was
progression to mCRPC.
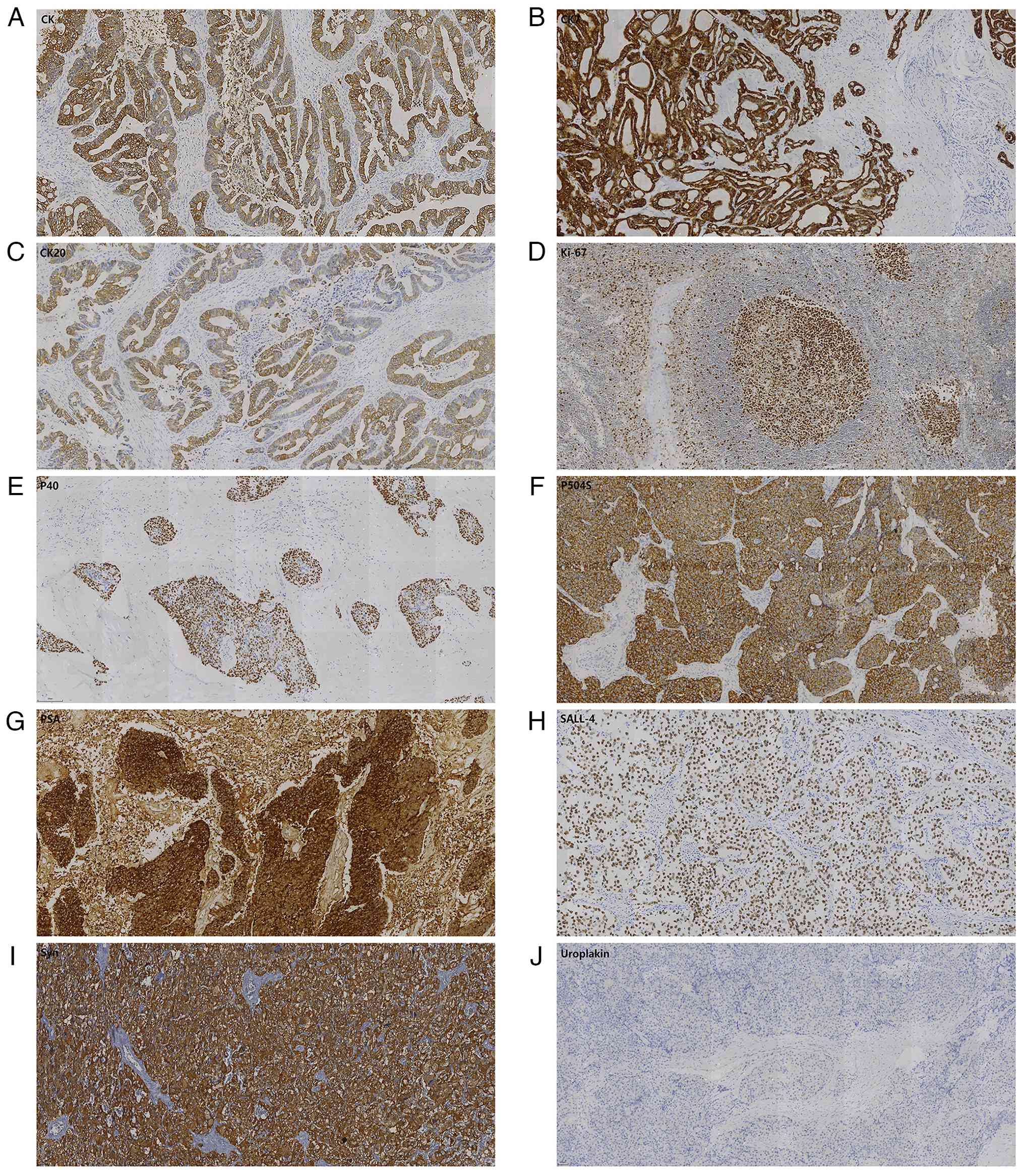 | Figure 5.Immunohistochemical staining of the
penile tumor. Staining results are (A) CK-positive, (B)
CK7-negative, (C) CK20-focally positive, (D) Ki-67-positive, (E)
p40-negative, (F) P504S-positive, (G) PSA-positive, (H)
SALL4-negative, (I) Syn-negative and (J) uroplakin-negative.
Magnification, ×200. (For negative markers, staining is limited to
non-neoplastic tissue components). CK, cytokeratin; P504S,
α-methylacyl-CoA racemase; PSA, prostate-specific antigen; SALL-4,
Spalt-like transcription factor 4; Syn, synaptophysin. |
As the disease progressed, bicalutamide was
discontinued and the patient was switched to a next-generation
antiandrogen. Meanwhile, considering the patient's history of
diabetes and the fact that abiraterone can affect blood glucose
levels, enzalutamide (160 mg once daily) was selected. A total of
99 genes (Xiangxian™-Prostate Cancer 99-Gene Panel; AcornMed)
associated with prostate cancer were tested, covering those related
to hereditary tumors, endocrine therapy, targeted therapy,
chemotherapy drugs and prognosis. Eventually, CDK12 p.L636fs
and CDK12 p.D1004fs were identified. Based on these
findings, olaparib (300 mg twice daily) was added to the treatment
regimen in May 2022. Following this combination therapy, PSA levels
decreased markedly, reaching 0.03 ng/ml in September 2022, which
was substantially lower than the nadir prior to the development of
penile metastasis. PSA levels have since remained stable and are
consistently at <0.006 ng/ml (Fig.
2). In October 2024, follow-up PET/CT (Fig. 3B) showed no increased PSMA uptake in
the left posterior lobe of the prostate, suggesting sustained tumor
suppression following treatment. No evidence of local recurrence
was observed at the site of the resected penile metastasis, and no
lymph node metastases were detected in the pelvic or
retroperitoneal regions. The patient has been followed up every
three months, and no new abnormal symptoms or discomfort have been
observed.
Discussion
Penile metastasis from PCa is rare, and a search for
relevant literature on PubMed (https://pubmed.ncbi.nlm.nih.gov/) using the keywords
‘prostate cancer’ AND (‘penile metastasis’ OR ‘penis metastasis’)
identified <100 reported cases. The condition has been reported
to have a mean survival time after diagnosis of ~9 months (8). The longest reported survival duration
to date is 62 months (9). By
comparison, the patient described in the present case report has
survived for 41 months since the diagnosis of penile metastasis.
Although only five other cases were analyzed in the present review,
they are highly representative in terms of both PSA level
distribution and treatment strategy coverage. These cases include
instances of both very low and very high PSA, demonstrating that
penile metastasis can occur across different tumor burdens. They
also encompass a range of treatment approaches, from traditional
androgen deprivation therapy to novel combination regimens. By
comparing prognostic differences among various strategies, this
study particularly highlights the breakthrough efficacy achieved
through genotype-guided precision combination therapy in key cases,
thereby supporting the critical role of molecular testing in rare
metastatic prostate cancer and the value of innovative combination
strategies (Table I).
 | Table I.Summary of selected reported cases of
penile metastasis from prostate cancer. |
Table I.
Summary of selected reported cases of
penile metastasis from prostate cancer.
| First author,
year | Age, years | Gleason Score | Initial PSA,
ng/ml | Treatment of penile
metastasis | Follow-up period,
months | Outcome | (Refs.) |
|---|
| Martz et al,
2021 | 61 | 4+4=8 | 1.57 | MHB | 6 | Alive | (1) |
| Fiaschetti et
al, 2016 | 84 | NA | 8.07 | ADT | 30 | Dead | (2) |
| Kamaleshwaran et
al, 2018 | 79 | 3+4=7 | >100 | ADT + RT | NA | NA | (8) |
| Fujita et
al, 2021 | 80 | 4+4=8 | 48 | ADT + RT | 62 | Alive | (9) |
| He et al,
2012 | 78 | 5+5=10 | 0.09 | ADT + surgery | 7 | Dead | (11) |
| Present case | 65 | 4+3=7 | 9.84 | ADT + enzalutamide
+ olaparib | 41 | Alive | - |
When PCa metastasizes to the penis, the metastatic
lesions typically occur in the penile root, penile shaft or glans
(10). Proposed mechanisms of
penile metastasis include direct invasion, previous device
implantation, retrograde venous flow, and arterial or lymphatic
dissemination (11). Among these,
retrograde venous flow is considered the most likely pathway for
penile metastasis from PCa (2). He
et al (11) reported tumor
thrombi within the veins of the penile corpus cavernosum,
supporting this mechanism.
Penile metastases can manifest as painless nodules,
surface nodules, ulcers, erythema, urinary retention, irritative
voiding symptoms, perineal pain, hematuria and/or priapism
(3). Due to their low incidence and
non-specific clinical manifestations, penile metastases must be
differentiated from other conditions such as idiopathic priapism,
sexually transmitted infections, tuberculosis, Peyronie's disease,
primary tumors and non-specific inflammatory lesions (2,12–14).
Various imaging techniques are valuable aids in the differential
diagnosis. Color Doppler ultrasonography can detect uneven nodules
in the penile corpora cavernosa, but lacks specificity for penile
metastasis (3). CT is helpful for
detecting secondary penile lesions (14), while MRI, with its high soft-tissue
contrast, can reliably differentiate and accurately stage penile
lesions (15). PSMA is an integral
membrane protein located on the membrane of prostate glandular
epithelial cells. In prostate cancer cells, including those from
both primary tumors and metastatic lesions, the expression level of
PSMA is significantly upregulated, leading to markedly increased
radiotracer uptake on PET/CT images. Thus, PSMA PET/CT is
particularly useful for diagnosing atypical penile metastases from
PCa (16,17).
No standardized treatment strategy exists for penile
metastases. Management should be individualized according to
symptom burden, the extent of the tumor and patient performance
status (3). Local treatment
approaches include surgery or radiotherapy (18); however, the latter carries a risk of
urethral stenosis or ulceration (19). ADT remains the cornerstone for the
management of metastatic PCa (20).
The addition of enzalutamide to ADT has been shown to improve
outcomes in metastatic hormone-sensitive PCa (21,22).
PARP inhibitors, including olaparib, have demonstrated efficacy in
tumors with HRR gene defects (23).
Preclinical and clinical studies suggest a synergistic effect may
be achieved using PARP inhibitors and androgen receptor-targeted
therapies (24). In particular,
clinical trials, including the PROpel (25), MAGNITUDE (26) and TALAPRO-2 (27) trials, have reported improved
outcomes with combination therapy among patients harboring HRR
mutations. It is worth noting that the updated TALAPRO-2 study
indicates that OS showed a statistically significant and clinically
meaningful improvement regardless of HRR gene mutation status.
In the present case, treatment with ADT combined
with enzalutamide and olaparib resulted in a marked decline in PSA
levels and sustained radiographic disease stability. At 41 months,
the survival time following a diagnosis of penile metastasis in the
present case exceeds the median overall survival time reported for
olaparib monotherapy (10.1 months) (28) and enzalutamide monotherapy (35.3
months) (29). Notably, this
survival duration also exceeds those described for multicatheter
interstitial high-dose rate brachytherapy-based regimens (6 months)
(1), ADT alone (30 months)
(2), or surgery combined with ADT
(7 months) (11) in men with PCa
metastatic to the penis (Table I).
Currently, the combination regimens involving novel endocrine drugs
and PARP inhibitors include olaparib plus abiraterone, niraparib
plus abiraterone, and enzalutamide combined with talazoparib.
However, the combination of enzalutamide and olaparib has not yet
been reported. Although limited by this being a single case, the
present outcome suggests that a combination of olaparib and
enzalutamide may be a viable option to promote the antitumor effect
of PARP inhibitors and novel endocrine drugs. The present case may
serve as a reference for the treatment of mCRPC with HRR
mutations.
CDK12 is a protein kinase that plays a crucial role
in cell cycle regulation and gene transcription (30). CDK12 mutations occur in ~7%
of PCa cases, and patients with CDK12 mutations have been
shown to have a significantly shorter overall survival time and a
shorter time to progression to castration-resistant disease
compared with those patients with wild-type CDK12 (31,32). A
multicenter study reported similar findings, demonstrating that
patients with CDK12 mutations exhibited a shorter
progression-free survival time and faster disease progression after
receiving ADT than those with wild-type CDK12, and only one
of seven patients achieved a reduction in PSA from a baseline of
>50% (a PSA50 response) (33).
In addition, patients with PCa harboring CDK12 mutations
have been shown to exhibit a shorter time to PSA progression
following first-line androgen receptor signaling inhibitor compared
with that exhibited by other patients (34). These findings suggest that
CDK12 mutations may result in PCa being intrinsically less
responsive to ADT.
Alternative systemic therapeutic approaches for this
molecular subtype are urgently needed and are currently being
investigated. Wu et al (4)
reported outcomes for four patients with mCRPC harboring CDK12
mutations treated with anti-programmed cell death protein-1
monotherapy, of whom 2 experienced marked reductions in PSA levels,
thus benefiting from the treatment. However, in another
retrospective study, only 2 out of 19 patients receiving immune
checkpoint inhibitor therapy achieved a PSA50 response (35). Whether CDK12 mutations can serve as
biomarkers to predict the potential response of PCa to ICIs remains
unclear and requires further evaluation. In addition, Zhu et
al (36) and Barata et
al (37) reported that
platinum-based chemotherapy can be effective for patients with
CDK12 mutations. However, evidence supporting this is limited to
only three case reports (36–38)
and a clinical study in 2020 that reported patients with CDK12
mutations achieved a PSA50 response after receiving platinum-based
chemotherapy, but specific data were not discussed (39).
In the present case, CDK12 p.L636fs and
CDK12 p.D1004fs mutations were detected. It has been
demonstrated that CDK12 loss drives prostate cancer
progression, and PARP inhibitors have some activity in patients
with prostate cancer with biallelic inactivating CDK12
alterations. In 2020, the U.S. Food and Drug Administration
approved olaparib for the treatment of patients with mCRPC
harboring HRR gene mutations. Based on these results, treatment
with olaparib was selected, achieving a sustained response.
In conclusion, the present case highlights the
diagnostic value of PSMA PET/CT in the identification of rare
penile metastases and underscores the importance of molecular
profiling in guiding therapy. The sustained response to
enzalutamide and olaparib in a patient with CDK12-mutated
mCRPC suggests that this combination may be a valuable option in
similar genetic contexts. However, further studies are necessary to
validate this approach.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
QX and YW analyzed data, and wrote and edited the
manuscript. QX and QD designed the study protocol. QD was the
primary care physician of the patient and developed and implemented
the treatment plan. QX, YW and BY performed the literature review
and analyzed and interpreted the data in the paper. BY and HT
obtained patient data and performed the histological examination of
the tumor. HT and QD reviewed the manuscript. HT performed the
analysis and interpretation of images. QX, YW, BY, HT and QD
confirm the authenticity of all the raw data. All authors have read
and approved the final version of the manuscript.
Ethics approval and consent to
participate
This study was approved by the Ethics Committee of
Nanfang Hospital (Guangzhou, China; approval no.
NFEC-202511-K26).
Patient consent for publication
Written informed consent for the publication of
clinical details and images was obtained from the patient.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Martz N, Benziane-Ouaritini N, Gautier M,
Brenot-Rossi I, Montagne L, Salem N, Bodokh Y and Hannoun-Levi JM:
Brachytherapy for oligometastatic prostate cancer to the penis. J
Contemp Brachytherapy. 13:593–597. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fiaschetti V, Liberto V, Claroni G, Loreni
G, Formica V, Roselli M, Mauriello A and Floris R: Relevance of
computed tomography and magnetic resonance imaging for penile
metastasis after prostatectomy: uncommon case report and brief
review of the literature. Radiol Case Rep. 11:255–259. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang Z, Xu M, Shang M, Liu Z, Yang L and
Yu D: Case report: Malignant priapism: Penile metastasis from
prostate cancer with low serum PSA level. Front Oncol.
14:13953012025. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wu YM, Cieślik M, Lonigro RJ, Vats P,
Reimers MA, Cao X, Ning Y, Wang L, Kunju LP, de Sarkar N, et al:
Inactivation of CDK12 delineates a distinct immunogenic class of
advanced prostate cancer. Cell. 173:1770–1782.e14. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jiang C, Hong Z, Liu S, Hong Z and Dai B:
Roles of CDK12 mutations in PCa development and treatment. Biochim
Biophys Acta Rev Cancer. 1880:1892472025. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Amin MB, Greene FL, Edge SB, Compton CC,
Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR and
Winchester DP: The eighth edition AJCC cancer staging manual:
Continuing to build a bridge from a population-based to a more
‘personalized’ approach to cancer staging. CA Cancer J Clin.
67:93–99. 2017.PubMed/NCBI
|
|
7
|
Montironi R, Cheng L, Scarpelli M and
Lopez-Beltran A: Pathology and genetics: Tumours of the urinary
system and male genital system: clinical implications of the 4th
Edition of the WHO Classification and Beyond. Eur Urol. 70:120–123.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kamaleshwaran KK, Balasundararaj BKP, Jose
R and Shinto AS: Penile metastasis from prostate cancer presenting
as malignant priapism detected using gallium-68 prostate-specific
membrane antigen positron emission tomography/computed tomography.
Indian J Nucl Med. 33:57–58. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fujita N, Kurokawa R, Kaneshima R, Machida
M, Kawai G, Wada T, Takahashi M, Nakaya M, Sakamoto N, Cho S, et
al: Patient with penile metastasis from prostate cancer and
survival over 5 years: A case report with longitudinal evaluation
using computed tomography and magnetic resonance imaging. Radiol
Case Rep. 16:1255–1258. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cocci A, Hakenberg OW, Cai T, Nesi G, Livi
L, Detti B, Minervini A, Morelli G, Carini M, Serni S and Gacci M:
Prognosis of men with penile metastasis and malignant priapism: A
systematic review. Oncotarget. 9:2923–2930. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
He D, Zeng J, Li X, Wu K, Wu D, He H, Song
W and Li L: Priapism as the initial manifestation of a penile and
lower limb cutaneous metastasis of prostate adenocarcinoma with low
serum PSA level. J Androl. 33:1160–1164. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cardoso Guimarães G, Rodrigues De Souza R,
Paiva Gadêlha Guimarães A, Filho WD, Valeschka De Matos Granja N,
Karan Kalil R, Augusto Soares F and Lopes A: Penile metastasis of
chondrosarcoma of the jaw. Urology. 61:8372003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lin YH, Kim JJ, Stein NB and Khera M:
Malignant Priapism Secondary to Metastatic Prostate Cancer: A case
report and review of literature. Rev Urol. 13:90–94.
2011.PubMed/NCBI
|
|
14
|
Dai Y, Shi BL, Zhang J, Liu SN and Jia YT:
Penile metastasis from prostate cancer misdiagnosed as Peyronie
disease: A case report. Sex Med. 11:qfac0112023. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rocher L, Glas L, Cluzel G, Ifergan J and
Bellin MF: Imaging tumours of the penis. Diagn Interv Imaging.
93:319–328. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tatkovic A, McBean R, Schoeman J and Wong
D: Prostate penile metastasis: Incidence and imaging pattern on 68
Ga-PSMA PET/CT. J Med Imaging Radiat Oncol. 64:499–504. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dhull VS, Kshirsagar P, Chowhan M and
Patil SC: Solitary penile metastasis from prostate cancer on
18F-Prostate-specific membrane antigen positron emission
tomography/computed tomography. Indian J Nucl Med. 37:402–403.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Landen L, Devos G, Joniau S and Albersen
M: Penile metastasis in prostate cancer patients: Two case reports,
surgical excision technique, and literature review. Curr Urol.
17:165–172. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Atag E, Semiz HS, Kazaz SN, Tuna EB,
Ozdogan O, Bozkurt O, Demir O and Karaoglu A: Response to
cabazitaxel beyond 20 cycles in a patient with penile metastasis of
prostate cancer: A case report. Urol J. 14:2985–2988.
2017.PubMed/NCBI
|
|
20
|
Huggins C: Effect of orchiectomy and
irradiation on cancer of the prostate. Ann Surg. 115:1192–1200.
1942. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Armstrong AJ, Szmulewitz RZ, Petrylak DP,
Holzbeierlein J, Villers A, Azad A, Alcaraz A, Alekseev B, Iguchi
T, Shore ND, et al: ARCHES: A Randomized, phase III study of
androgen deprivation therapy with enzalutamide or placebo in men
with metastatic hormone-sensitive prostate cancer. J Clin Oncol.
37:2974–2986. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Davis ID, Martin AJ, Stockler MR, Begbie
S, Chi KN, Chowdhury S, Coskinas X, Frydenberg M, Hague WE, Horvath
LG, et al: Enzalutamide with standard first-line therapy in
metastatic prostate cancer. N Engl J Med. 381:121–131. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fong PC, Boss DS, Yap TA, Tutt A, Wu P,
Mergui-Roelvink M, Mortimer P, Swaisland H, Lau A, O'Connor MJ, et
al: Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA
mutation carriers. N Engl J Med. 361:123–134. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Asim M, Tarish F, Zecchini HI, Sanjiv K,
Gelali E, Massie CE, Baridi A, Warren AY, Zhao W, Ogris C, et al:
Synthetic lethality between androgen receptor signalling and the
PARP pathway in prostate cancer. Nat Commun. 8:3742017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Saad F, Clarke NW, Oya M, Shore N,
Procopio G, Guedes JD, Arslan C, Mehra N, Parnis F, Brown E, et al:
Olaparib plus abiraterone versus placebo plus abiraterone in
metastatic castration-resistant prostate cancer (PROpel): Final
prespecified overall survival results of a randomised,
double-blind, phase 3 trial. Lancet Oncol. 24:1094–1108. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Chi KN, Sandhu S, Smith MR, Attard G, Saad
M, Olmos D, Castro E, Roubaud G, Pereira de Santana Gomes AJ, Small
EJ, et al: Niraparib plus abiraterone acetate with prednisone in
patients with metastatic castration-resistant prostate cancer and
homologous recombination repair gene alterations: Second interim
analysis of the randomized phase III MAGNITUDE trial. Ann Oncol.
34:772–782. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Agarwal N, Azad AA, Carles J, Fay AP,
Matsubara N, Heinrich D, Szczylik C, De Giorgi U, Young Joung J,
Fong PCC, et al: Talazoparib plus enzalutamide in men with
first-line metastatic castration-resistant prostate cancer
(TALAPRO-2): A randomised, placebo-controlled, phase 3 trial.
Lancet. 402:291–303. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mateo J, Carreira S, Sandhu S, Miranda S,
Mossop H, Perez-Lopez R, Nava Rodrigues D, Robinson D, Omlin A,
Tunariu N, et al: DNA-Repair defects and olaparib in metastatic
prostate cancer. N Engl J Med. 373:1697–1708. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Beer TM, Armstrong AJ, Rathkopf D, Loriot
Y, Sternberg CN, Higano CS, Iversen P, Evans CP, Kim CS, Kimura G,
et al: Enzalutamide in men with chemotherapy-naïve metastatic
castration-resistant prostate cancer: Extended analysis of the
phase 3 PREVAIL Study. Eur Urol. 71:151–154. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Greenleaf AL: Human CDK12 and CDK13,
multi-tasking CTD kinases for the new millenium. Transcription.
10:91–110. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang W, Zhou L and Di J: Prognostic and
clinicopathological value of CDK12 mutation in prostate cancer: A
meta-analysis. Expert Rev Anticancer Ther. 23:207–216. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Nguyen B, Mota JM, Nandakumar S, Stopsack
KH, Weg E, Rathkopf D, Morris MJ, Scher HI, Kantoff PW, Gopalan A,
et al: Pan-cancer analysis of CDK12 alterations identifies a subset
of prostate cancers with distinct genomic and clinical
characteristics. Eur Urol. 78:671–679. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dong B, Fan L, Yang B, Chen W, Li Y, Wu K,
Zhang F, Dong H, Cheng H, Pan J, et al: Use of circulating tumor
DNA for the clinical management of metastatic castration-resistant
prostate cancer: A multicenter, real-world study. J Natl Compr Canc
Netw. 19:905–914. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Reimers MA, Yip SM, Zhang L, Cieslik M,
Dhawan M, Montgomery B, Wyatt AW, Chi KN, Small EJ, Chinnaiyan AM,
et al: Clinical outcomes in cyclin-dependent kinase 12 mutant
advanced prostate cancer. Eur Urol. 77:333–341. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Schweizer MT, Ha G, Gulati R, Brown LC,
McKay RR, Dorff T, Hoge ACH, Reichel J, Vats P, Kilari D, et al:
CDK12-mutated prostate cancer: Clinical outcomes with standard
therapies and immune checkpoint blockade. JCO Precis Oncol.
4:382–392. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhu S, Bao Y, Zheng L, Zhao J, Chen Y,
Huang R, Sun G, Zhao F, Zhang X, Liang J, et al: Chronological
liquid biopsy reveals the impact of platinum-based chemotherapy on
a prostate cancer patient's CDK12 mutation: A case report. Onco
Targets Ther. 15:947–952. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Barata P, Ledet E, Manogue C, Cotogno P,
Harris K, Lewis B, Layton J and Sartor O: Long-term disease control
using taxane/platinum-based chemotherapy in CDK12-mutated advanced
prostate cancer. Oncologist. 25:e1421–e1422. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Murata Y, Kosaka T, Nakamura K, Baba Y,
Arai E, Yasumizu Y, Matsumoto K, Nishihara H and Oya M: A case of
testicular dysgenesis syndrome with squamous cell carcinoma of the
prostate harboring a CDK12 mutation. IJU Case Rep. 8:125–128. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mota JM, Barnett E, Nauseef JT, Nguyen B,
Stopsack KH, Wibmer A, Flynn JR, Heller G, Danila DC, Rathkopf D,
et al: Platinum-based chemotherapy in metastatic prostate cancer
with DNA repair gene alterations. JCO Precis Oncol. 4:355–366.
2020. View Article : Google Scholar : PubMed/NCBI
|