Introduction
Neuroendocrine neoplasms (NENs) of the uterine
cervix account for approximately 1–2% of all cervical cancers,
wherein the vast majority are poorly differentiated neuroendocrine
carcinomas (NECs), such as small cell NEC (SCNEC) and large cell
NEC (LCNEC) (1). In contrast,
well-differentiated NETs (G1-G2) and mixed tumors with NET and
adenocarcinoma components are exceedingly rare, with reports
limited to individual case descriptions or small case series.
NETs of the gastrointestinal tract reportedly
exhibit increased proliferative activity during disease progression
or metastasis, and in some cases, morphological transformation from
NET to NEC-like features (2,3).
However, in gynecologic oncology, systematic evidence on changes in
proliferative activity in NETs remains scarce. This report
describes a rare case of cervical adenocarcinoma with a coexisting
NET G2 component, wherein postoperative bilateral pulmonary
metastases demonstrated increased proliferative activity in the NET
component alongside focal histological features suggesting NEC
transformation. This represents an unusual example of a gynecologic
NET exhibiting proliferative escalation in metastatic lesions
during disease progression.
Case report
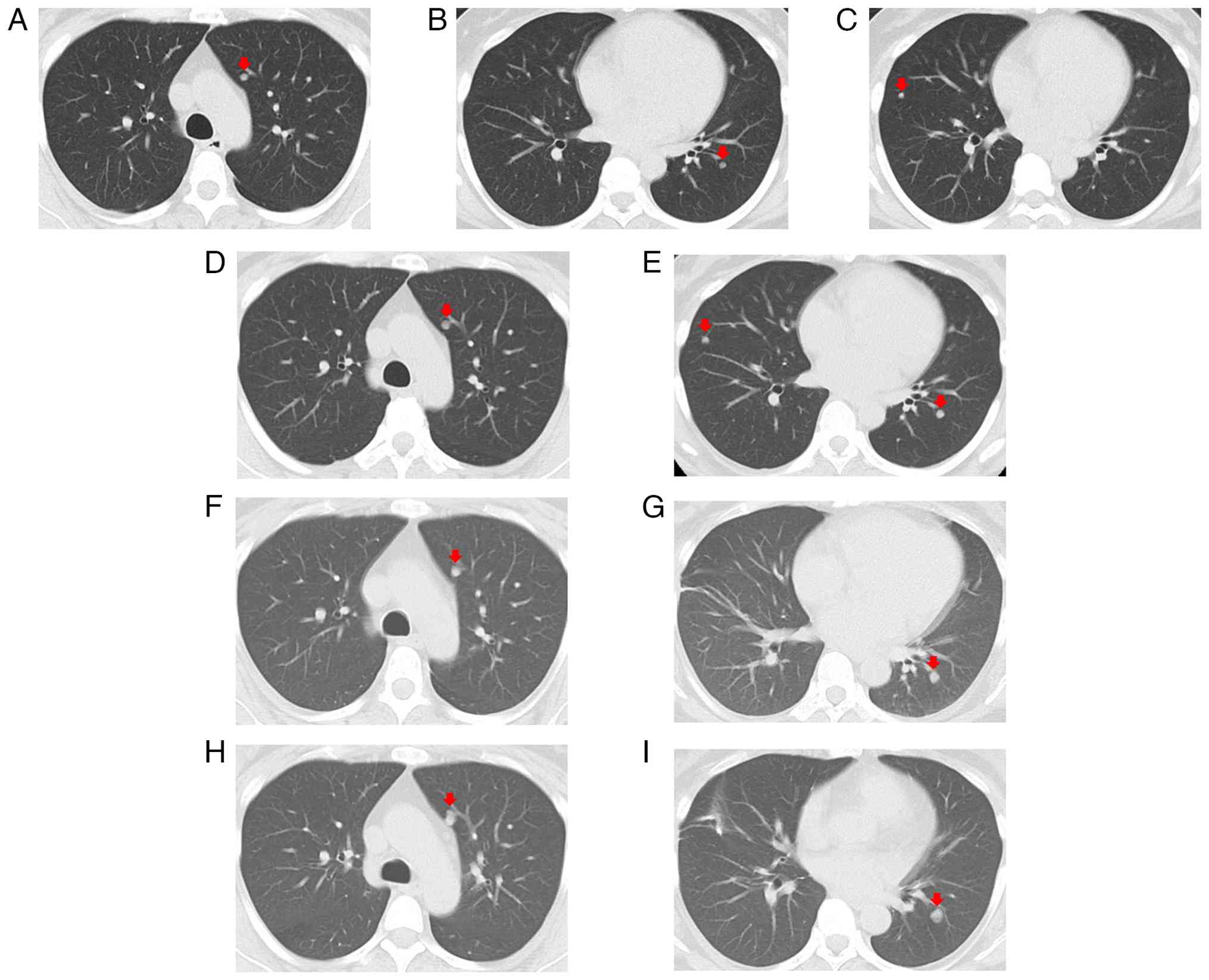
A 43-year-old woman presented at a respiratory
clinic with a chronic cough. Chest computed tomography (CT) in
March 2024 revealed 3 nodular lesions in the lung, each measuring
3–5 mm, located in the right middle, left upper, and left lower
lobes (Fig. 1A-C).
The patient had a history of cervical cancer (FIGO
2018 stage IB1) diagnosed in March 2022, treated with radical
hysterectomy, bilateral salpingectomy, and pelvic lymph node
dissection. Histopathological examination revealed a mixed tumor
consisting of adenocarcinoma and NET (Fig. 2). Specifically, the transitional
area had adenocarcinoma components proliferating in tubular,
papillary, and cribriform patterns. Meanwhile, uniform cells with
round nuclei formed solid nests, trabeculae, and rosettes,
consistent with neuroendocrine morphology. The neuroendocrine
component was positive for synaptophysin, chromogranin A, INSM1,
p16, and CK7, while it was negative for CK20, TTF-1, PAX8, and
YAP1. The tumor showed a p53 wild-type pattern with retained Rb
expression. The Ki-67 index was approximately 30% in hotspots
(overall range: 3–30%), and mitotic activity was 5–8 per 10
high-power fields (HPFs). Within the cervical tissue, the
neuroendocrine component was confined to a well-differentiated NET,
with no evidence of NEC. If classified using the gastrointestinal
system criteria, the neuroendocrine component would correspond to
NET G3, but the current classification of cervical NETs extends
only up to G2. Thus, the tumor was considered a mixed
adenocarcinoma and NET G2. According to the AJCC 9th edition, the
postoperative pathological stage was stage IB1 (pT1b1 N0 M0)
without distant metastasis.
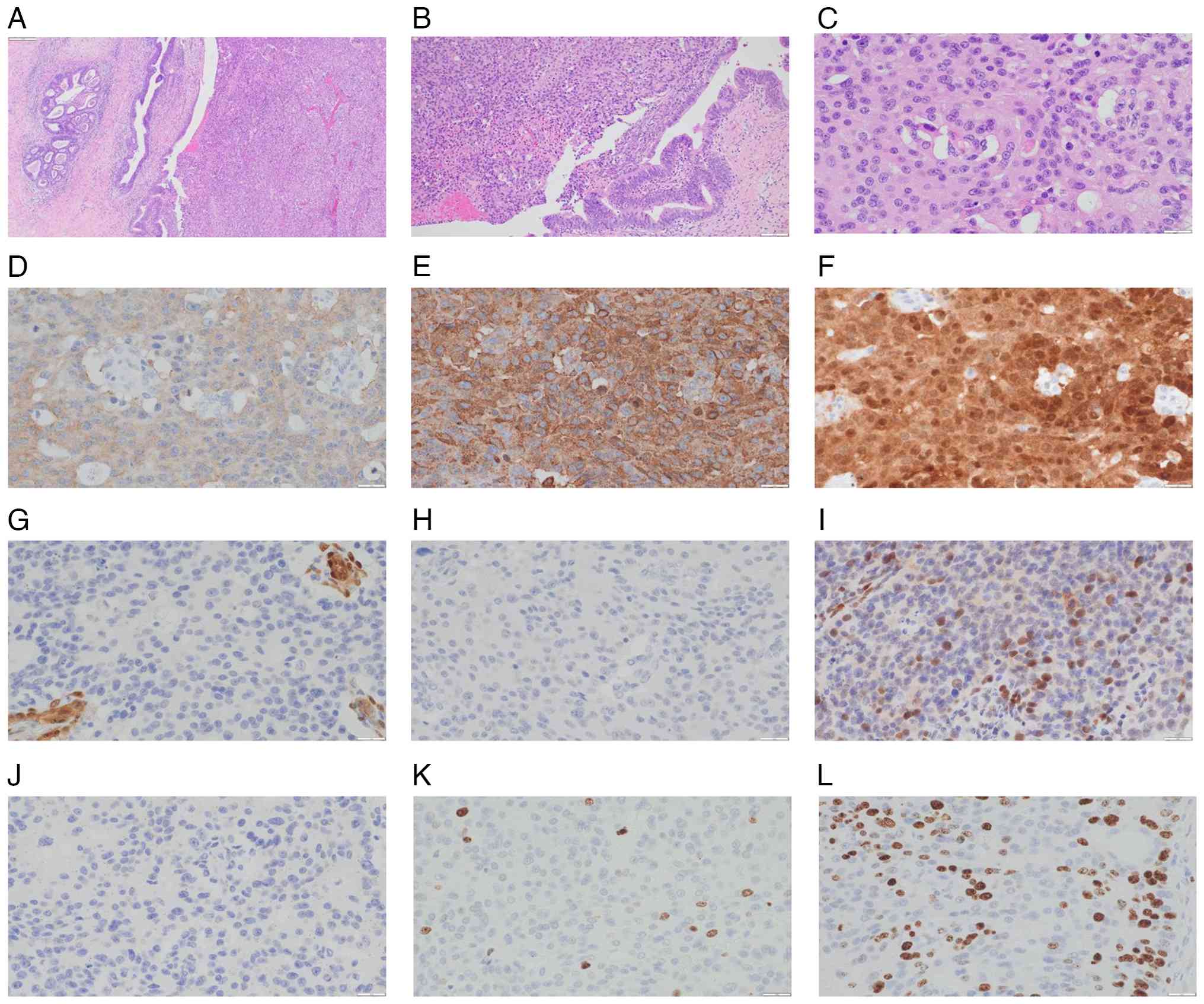 | Figure 2.Histopathological and
immunohistochemical findings of the cervical tumor. (A) H&E
staining (ocular, ×10; objective, ×4; final magnification, ×40)
showing mixed histology. (B) H&E staining (ocular, ×10;
objective, ×10; final magnification, ×100) revealing coexistence of
adenocarcinoma and neuroendocrine components. (C) H&E staining
of the neuroendocrine component (ocular, ×10; objective, ×40; final
magnification, ×400). (D-L) Immunohistochemical analysis of the
neuroendocrine component (ocular, ×10; objective, ×40; final
magnification, ×400) revealing the following: (D) Synaptophysin,
positive; (E) chromogranin A, positive; (F) p16, positive; (G)
yes-associated protein 1, negative; (H) thyroid transcription
factor-1, negative; (I) retinoblastoma protein, wild-type
expression; (J) paired box 8, negative; (K) Ki-67, low
proliferative activity; and (L) Ki-67, hotspot with high
proliferative activity. |
Follow-up chest CT in June 2024 revealed that the
pulmonary nodules increased in size compared to March 2024
(Fig. 1D and E). A right middle
lobe lesion was partially resected for diagnostic purposes,
yielding a small nodule measuring 5 mm in diameter. Histologically,
solid nests of columnar to short spindle-shaped cells with oval
nuclei were observed (Fig. 3).
Central necrosis was present within the nests, with nuclear
palisading at the periphery. More than 50 mitotic figures per 10
HPFs were counted. The Ki-67 index was 35% (hotspot). These
features are consistent with G3 disease according to the
gastrointestinal NET criteria. Immunohistochemically, the tumor
cells were positive for CK7, p16, INSM1, and chromogranin A, but
these were negative for CK20 and p40. These findings are
morphologically and immunophenotypically consistent with metastatic
NET originating from the uterine cervix. No areas lacking
expression of neuroendocrine markers were identified within the
specimen.
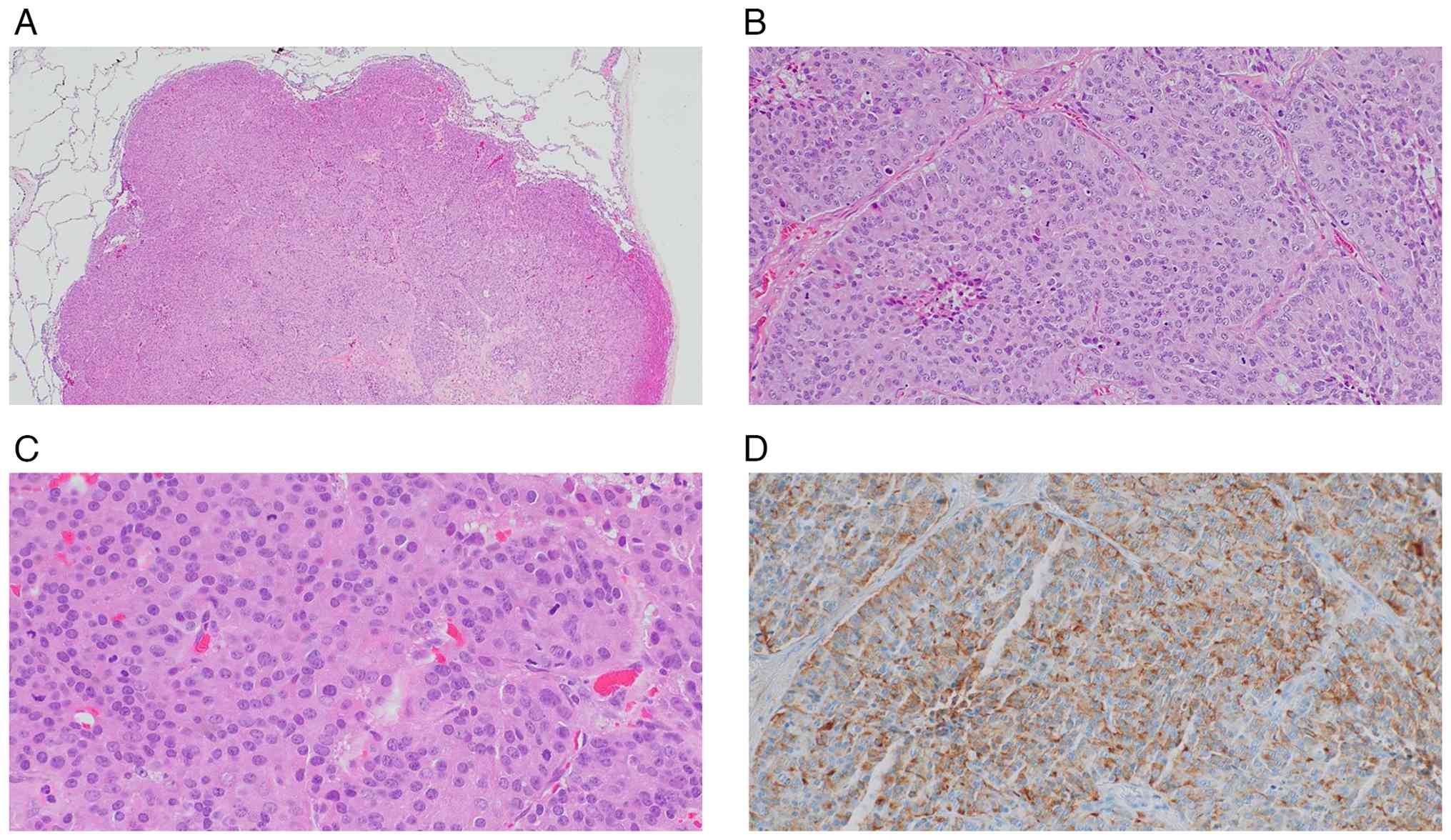 | Figure 3.Histopathological and
immunohistochemical findings of the right middle lobe lung tumor.
(A-C) H&E staining of the resected specimen showing exclusively
neuroendocrine tumor components. (A) Ocular, ×10; objective, ×4;
final magnification, ×40. (B) Ocular, ×10; objective, ×10; final
magnification, ×100. (C) Ocular, ×10; objective, ×40; final
magnification, ×400. (D) Chromogranin A immunostaining (ocular,
×10; objective, ×40; final magnification, ×400) demonstrating
positive expression in tumor cells. |
The patient was referred to our oncology department
in August 2024 for further evaluation and treatment.
Gastrointestinal endoscopy revealed no evidence of a primary tumor,
with no abnormal uptake on octreotide scintigraphy. Treatment with
everolimus was initiated in October 2024, and CT in December 2024
showed stable disease (Fig. 1F and
G). However, in April 2025, the residual pulmonary lesions
mildly increased in size, but with no new lesions (Fig. 1H and I). The patient declined
continuation of everolimus, opting instead for a local treatment
strategy.
Radiofrequency ablation (RFA) of a left lower lobe
lesion was performed in May 2025, followed by partial resection due
to its close proximity to the aorta in June 2025. Histopathological
examination of the resected left upper lobe specimen revealed
predominantly NET morphology with a focal area showing features
suspicious for small cell carcinoma (Fig. 4). The tumor was composed of atypical
cells proliferating in solid nests with rosette- and palisade-like
arrangements, accompanied by foci of necrosis. The tumor cells had
relatively abundant eosinophilic cytoplasm and enlarged, uniform,
round to oval nuclei without marked nuclear atypia.
Immunohistochemical analysis demonstrated positivity for
synaptophysin, chromogranin A, and INSM1, negativity for p40 and
YAP1, partial positivity for TTF-1, a p53 wild-type pattern, and no
loss of Rb expression. The Ki-67 index reached 93% in hotspots,
while the mitotic count was approximately 50 per 10 HPFs. These
findings supported a diagnosis of NEN. Although most of the lesion
exhibited morphology consistent with NET, there were focal areas
demonstrating a markedly high Ki-67 labeling index, partial TTF-1
positivity, increased chromatin, and a higher
nuclear-to-cytoplasmic ratio, thus raising concern for small cell
carcinoma. While these findings suggested a metastatic cervical
origin, the unusual morphological overlap warranted further
investigation for small cell carcinoma. Throughout the
perioperative course and subsequent follow-up, regular tumor marker
assessments showed no elevation of NSE or ProGRP. Postoperative
chemotherapy with a platinum-etoposide regimen was recommended, but
the patient declined. The patient has remained recurrence-free to
date, with radiologic evaluation at 6 months after resection of the
last tumor showing no evidence of recurrence under close
follow-up.
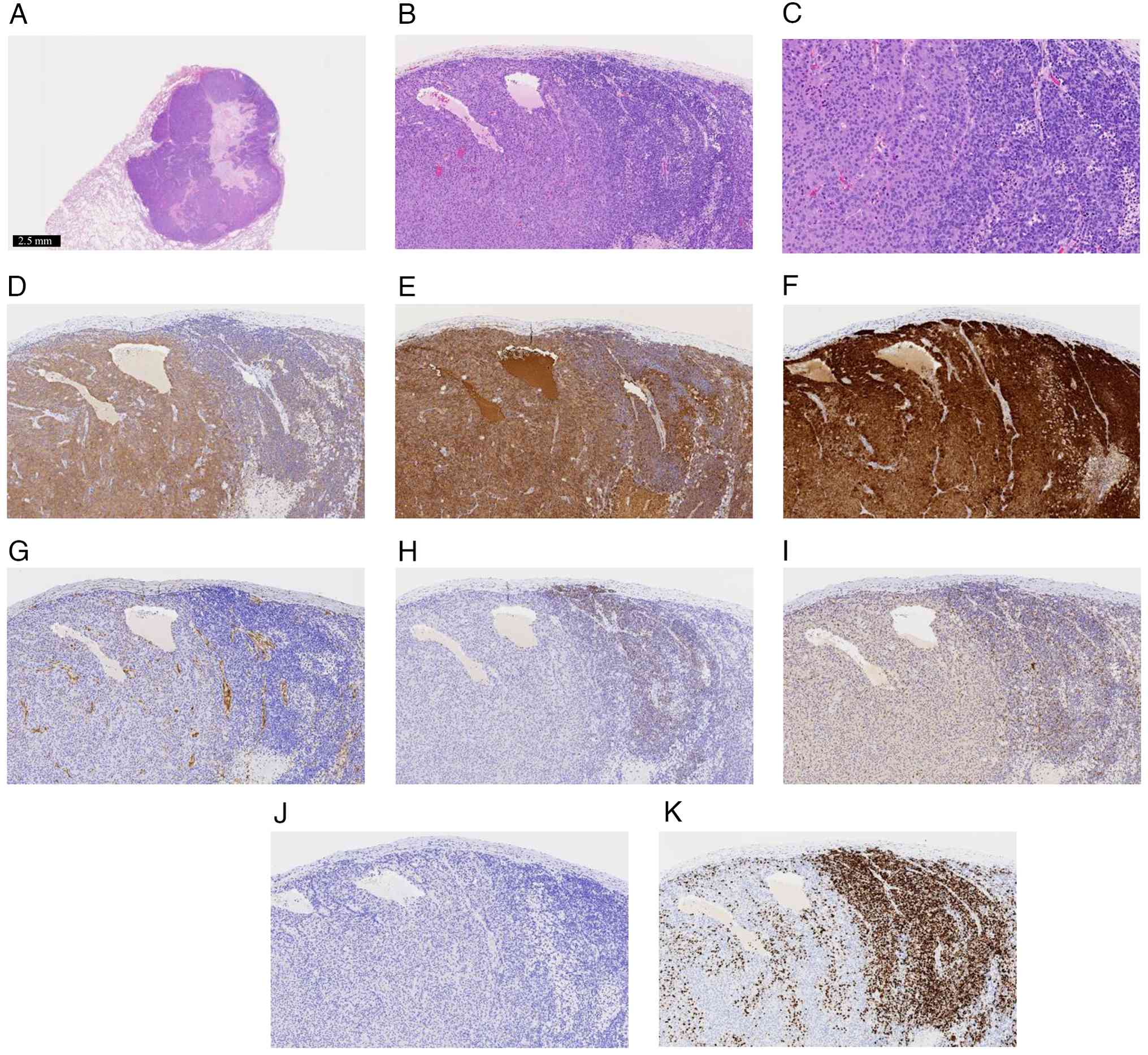 | Figure 4.Histopathological and
immunohistochemical findings of the left upper lobe lung tumor. (A)
Low-power view of H&E staining (scale bar, 2.5 mm). (B) H&E
staining (ocular, ×10; objective, ×10; final magnification, ×100).
(C) H&E staining (ocular, ×10; objective, ×20; final
magnification, ×200). (A-C) The tumor consisted predominantly of
NET components but with areas of increased chromatin and a higher
nuclear-to-cytoplasmic ratio, suggesting the presence of NEC
components. (D-K) Immunohistochemical analysis (ocular, ×10;
objective, ×10; final magnification, ×100) revealed the following:
(D) Synaptophysin, positive in NET areas but weaker in suspected
NEC areas; (E) chromogranin A, positive; (F) p16, positive; (G)
yes-associated protein 1, negative; (H) thyroid transcription
factor-1, negative in NET areas but focally positive in suspected
NEC areas; (I) retinoblastoma protein, wild-type expression; (J)
paired box 8, negative; and (K) Ki-67, high proliferative index in
suspected NEC areas. NEC, neuroendocrine carcinoma; NET,
neuroendocrine tumor. |
Immunohistochemical analyses for the immunostaining
images shown in the figures were performed on formalin-fixed,
paraffin-embedded (FFPE) tissue sections. The primary antibodies
used for the immunostaining shown in Figs. 2 and 4 were as follows: synaptophysin (catalog
no. 413831, clone 27G12; Nichirei Biosciences), chromogranin A
(catalog no. M086901-2, clone DAK-A3; DAKO), p16 (catalog no.
550834, clone G175-405; BD Biosciences), YAP1 (catalog no. ab52771,
clone EPR1674Y; Abcam), TTF-1 (catalog no. NCL-L-TTF-1, clone
SPT24; NOVO), retinoblastoma protein (Rb; catalog no. 554136, clone
G3-245; BD Biosciences), PAX8 (catalog no. ab53490, clone PAX8R1;
abcam), and Ki-67 (catalog no. M7240, clone MIB-1; DAKO). Antibody
dilutions were 1:1 for synaptophysin, 1:200 for chromogranin A,
1:20 for p16, 1:100 for YAP1, 1:100 for TTF-1, 1:400 for Rb, 1:20
for PAX8, and 1:2 for Ki-67. For the immunostaining shown in
Fig. 3D, chromogranin A (catalog
no. 412751; Nichirei) was used at a dilution of 1:1. Appropriate
positive and negative controls were included in each staining run.
Immunoreactivity was evaluated independently by experienced
pathologists.
Discussion
Cervical NENs are rare, accounting for approximately
1–2% of all malignant cervical tumors, among which the vast
majority are high-grade NECs (1).
Low- to intermediate-grade (G1-G2) NETs are extremely uncommon, and
cases with an admixed adenocarcinoma component are exceedingly
rare. The 2020 WHO classification categorizes cervical NENs as
either NET G1, NET G2, or NEC, but some reports have described
tumors corresponding to NET G3 (4).
At present, a formal category corresponding to NET G3 has not been
established in the current classification of cervical
neuroendocrine neoplasms. However, as described in this report, the
existence of cervical NENs showing well-differentiated morphology
despite a high Ki-67 proliferation index has been suggested in the
literature, albeit mainly at the level of individual case reports.
By analogy with pancreatic and gastrointestinal NET G3, such tumors
of the cervix may exhibit biological behavior and treatment
responsiveness distinct from those of morphologically defined
neuroendocrine carcinoma (NEC). In particular, they may be less
sensitive to platinum-based chemotherapy compared with classic NEC,
although robust evidence to support this assumption is currently
lacking. Currently, available data are insufficient to justify a
distinct therapeutic strategy for these tumors, and therefore no
change in clinical management can be recommended. Nevertheless,
recognition of a potential NET G3-like subgroup among cervical NENs
may have important clinical implications in the future,
particularly with respect to treatment selection and clinical trial
design. Further accumulation of cases and comprehensive
clinicopathological and molecular analyses will be essential to
clarify the biological and clinical significance of this
entity.
The primary tumor in this case had Ki-67 labeling
index of 30% at the hotspot, which corresponds to G3 according to
the pancreatic NEN grading system. Cervical NENs are considered
more prone to metastasis than squamous cell carcinomas (5,6). In
our patient, while the primary lesion had adenocarcinoma and NET
components, nearly all the metastatic lesions were composed solely
of NET. This observation raises the possibility that a NEN clone
present within the primary tumor possessed superior metastatic
potential compared with other tumor components and was therefore
selectively able to disseminate and expand at metastatic sites.
Furthermore, during the clinical course, the NET component had
increased proliferative activity (as reflected by Ki-67 index and
mitotic count), and the resected metastatic lesion ultimately
contained foci that required differentiation from small cell
carcinoma. In particular, the focal lesion exhibiting a Ki-67 index
of 93% may represent a further selected subclone that acquired
enhanced proliferative capacity during the metastatic process
and/or under therapeutic pressure. These findings suggest the
possibility that NEC may arise from preexisting NET and may provide
a possible explanation for the phenomenon of grade progression of
NET over the course of treatment. At the molecular level, the
acquisition of TP53 and RB1 mutations is considered a key event in
the progression toward NEC-like morphology, as reported in
pancreatic and gastrointestinal NENs (3,7).
However, beyond these alterations, there is a lack of large-scale
molecular analyses in cervical NENs, and the precise mechanisms
driving such transformation need to be further elucidated. Previous
reports found that cervical NECs harbor non-silent TP53 and RB1
mutations at a much lower frequency compared to small cell
carcinomas of the lung and bladder (8). Consistent with these observations,
even the metastatic lesions showing markedly increased
proliferative activity and NEC-like morphology demonstrated
retained Rb expression and a wild-type p53 immunophenotype. These
findings suggest that, at least in a subset of cervical
neuroendocrine neoplasms, progression toward NEC-like features may
occur through TP53/RB1-independent mechanisms. In the present case,
as shown in Fig. 4H, focal TTF-1
positivity was identified in areas exhibiting morphological
features suggestive of NEC. This spatial correlation between TTF-1
expression and NEC-like morphology suggests that the tumor may have
acquired a pulmonary neuroendocrine-like phenotype during disease
progression, making a nonspecific or artifactual finding less
likely. With respect to the pathological distinction between
high-grade NET and NEC arising from NET, no single diagnostic
parameter is sufficient. Rather, an integrated assessment of
multiple pathological features is required, including the degree of
cytologic atypia, the presence and pattern of necrosis, and the
Ki-67 proliferation index. These features should be interpreted in
conjunction with immunohistochemical findings, such as Rb
expression status and p53 expression patterns. While the
immunophenotype provides important supportive information, it
should be regarded as an adjunct rather than an independent
determinant of the final diagnosis. It should be noted that a
limitation of the present study is that next-generation sequencing
(NGS) was not performed on either the primary tumor or the
metastatic lesions. As a result, specific acquired genetic
alterations that may have driven the increased proliferative
activity or the emergence of NEC-like features could not be
identified, and any discussion of molecular drivers of
transformation in this case remains speculative. Thus, future
studies need to clarify the mechanisms underlying the morphological
transformation from cervical NET to NEC. In addition, diffuse p16
positivity was observed in both the primary and metastatic lesions,
including the NEC-like components, suggesting possible involvement
of high-risk HPV infection. Although previous analytical studies
have reported an association between cervical neuroendocrine
neoplasms and high-risk HPV infection (9), HPV PCR testing was not performed for
the individual tumor components in the present case; therefore, a
common origin from a single HPV-transformed progenitor cell could
not be definitively demonstrated.
Several reports have described metastatic
gastrointestinal NETs undergoing grade progression and acquiring
NEC-like morphology (2,3). In contrast, cases of primary cervical
NET with direct documentation of sequential changes in metastatic
lesions (e.g., increased proliferative capacity and morphological
transformation toward NEC) are extremely rare. Given that the
present report describes a single case, it is difficult to draw
robust conclusions regarding the frequency or generalizability of
this evolutionary pattern. Nevertheless, careful documentation and
reporting of such cases may incrementally contribute to a better
collective understanding of the biological behavior of cervical
neuroendocrine neoplasms. From a future research perspective,
further studies are needed to validate the evolutionary model
suggested by this case. In particular, accumulation of additional
cases with detailed longitudinal pathological evaluation of both
primary and metastatic lesions will be essential to determine the
frequency and reproducibility of grade progression from NET to NEC
in cervical neuroendocrine neoplasms. Comprehensive molecular
profiling, ideally using next-generation sequencing of paired
primary and metastatic samples, may help identify genetic
alterations associated with increased proliferative activity and
morphological transformation. In addition, prospective collection
of fresh or appropriately preserved tumor tissue may enable
functional studies, such as patient-derived organoid or other
experimental models, to further elucidate tumor evolution and
therapeutic vulnerabilities. Although such approaches were beyond
the scope of the present case report, they represent important
directions for future investigation.
Currently, there is no established standard
treatment for cervical NENs, although in cases of NEC, adjuvant
chemotherapy with platinum agents and etoposide is often
administered alongside surgery and/or radiotherapy (1). In gastroenteropancreatic NET G3,
sensitivity to platinum-based chemotherapy with etoposide has been
reported to be lower than that observed in NEC (10,11).
Based on this consideration, everolimus was selected and initiated
in the present case. Everolimus was administered for approximately
6 months for the treatment pulmonary metastases, during which no
new lesions appeared. Considering that the pulmonary metastatic
tumors were initially regarded as neuroendocrine tumors, that no
new metastatic lesions developed over a six-month period, and that
the patient did not wish to undergo intravenous cytotoxic
chemotherapy, we considered that reducing the overall tumor burden
through local interventions might contribute to prolongation of
disease control and potentially improve prognosis. This enabled
local surgical resection and histopathological examination, which
revealed areas requiring differential diagnosis from small cell
carcinoma, strongly suggesting progression from NET to NEC. Marked
spatial heterogeneity of the Ki-67 proliferation index may reduce
the representativeness of small biopsy specimens and complicate
prognostic assessment and evaluation of treatment response. From a
prognostic and therapeutic standpoint, when substantial
intrapatient heterogeneity is present, it is reasonable to consider
that the clinical behavior of the disease is driven by the most
aggressive tumor component. Accordingly, particularly when
high-Ki-67 areas are associated with aggressive growth patterns or
morphological features suggestive of progression toward NEC,
treatment decisions should be guided by the highest-grade lesion.
In the present case, NEC-like morphology was identified in the
additionally resected pulmonary tumor tissue; therefore, adjuvant
chemotherapy was recommended postoperatively. However, as the
patient declined further systemic therapy, close clinical follow-up
was subsequently adopted.
This case highlights three important clinical
implications. First, even in cases diagnosed as NET, tumors with a
high Ki-67 proliferation index warrant close follow-up because of
the potential for morphological transition toward NEC. In
particular, for cervical neuroendocrine tumors with an initial
Ki-67 index greater than 20%, closer radiologic surveillance should
be considered, even in patients with low-stage disease who have
undergone apparently curative resection. The present case suggests
that tumors with high proliferative activity may harbor an
increased risk of early recurrence or progression that may not be
adequately captured by standard follow-up protocols. Second, the
pathological re-evaluation of metastatic lesions is crucial in
guiding therapeutic decision-making. When the primary tumor
demonstrates mixed histology, the emergence of new lesions during
follow-up should prompt histopathological confirmation whenever
feasible. In such situations, repeat biopsy is preferable to
radiologic surveillance alone, as reassessment of tumor histology
and grade may reveal phenotypic evolution with direct implications
for treatment selection. Third, given the rarity, biological
heterogeneity, and potential for rapid progression of cervical
neuroendocrine tumors, multidisciplinary management is essential.
We believe that such cases should be routinely discussed in a
multidisciplinary tumor board involving gynecologic oncologists,
pathologists, radiologists, and medical oncologists with expertise
in neuroendocrine neoplasms. A multidisciplinary treatment strategy
that integrates systemic therapy and local interventions may help
optimize patient management and improve prognosis. Another
limitation of this study relates to radiologic assessment. All
metastatic lesions were small and did not meet RECIST criteria for
measurable target lesions; therefore, radiologic evaluation was
qualitative rather than quantitative. In addition, 18F-FDG PET/CT
was not performed, precluding assessment of metabolic activity and
correlation between SUVmax and pathological features such as the
Ki-67 proliferation index or NEC-like morphology. Although such
radiologic-pathologic correlations may strengthen the link between
imaging phenotype and biological aggressiveness, these data were
not available in the present case. Future studies incorporating
standardized quantitative imaging and metabolic evaluation are
warranted. Taken together, clinical decision-making in gynecologic
oncology should consider not only the initial histologic
classification but also proliferative activity, histologic
heterogeneity, and dynamic changes over time, including the
potential progression from NET to NEC-like morphology that has been
described in the gastrointestinal field.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
NH conceptualized the manuscript. Data acquisition
and interpretation were performed by NH, HK, RS, YK, YY, MO, SO,
YN, TU, RM, MH, AY and MT. The original draft was written by NH,
while the final manuscript was written, reviewed and edited by NH
and MT. NH and MT confirm the authenticity of all the raw data. All
authors agree to be accountable for all aspects of the research in
ensuring that the accuracy or integrity of any part of the work are
appropriately investigated and resolved. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient for publication of this case report and the accompanying
images. The patient was informed that all identifying information
would be removed to ensure anonymity.
Competing interests
MT received honoraria from Chugai Pharmaceutical,
AstraZeneca K.K., Bristol-Myers Squibb Company, Novartis Pharma
K.K. and Ono Pharmaceutical. The other authors declare that they
have no competing interests.
Glossary
Abbreviations
Abbreviations:
|
NET
|
neuroendocrine tumor
|
|
NEC
|
neuroendocrine carcinoma
|
|
HPF
|
high-power field
|
|
NEN
|
neuroendocrine neoplasm
|
|
RFA
|
radiofrequency ablation
|
References
|
1
|
Tempfer CB, Tischoff I, Dogan A, Hilal Z,
Schultheis B, Kern P and Rezniczek GA: Neuroendocrine carcinoma of
the cervix: A systematic review of the literature. BMC Cancer.
18:5302018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Singh S, Hallet J, Rowsell C and Law CHL:
Variability of Ki67 labeling index in multiple neuroendocrine tumor
specimens over the course of the disease. Eur J Surg Oncol.
40:1517–1522. 2014. View Article : Google Scholar
|
|
3
|
Joseph NM, Umetsu SE, Kim GE, Terry M,
Perry A, Bergsland E and Kakar S: Progression of low-grade
neuroendocrine tumors to high-grade neoplasms harboring the
NEC-like co-alteration of RB1 and TP53. Endocr Pathol. 35:325–337.
2024. View Article : Google Scholar
|
|
4
|
Katafuchi T, Kawakami F, Iwagoi Y, Saito F
and Mikami Y: ‘Neuroendocrine tumor grade 3 (NET G3)’ of the
uterine cervix: A report of 2 cases. Int J Gynecol Pathol.
41:470–475. 2022. View Article : Google Scholar
|
|
5
|
Margolis B, Tergas AI, Chen L, Hou JY,
Burke WM, Hu JC, Ananth CV, Neugut AI, Hershman DL and Wright JD:
Natural history and outcome of neuroendocrine carcinoma of the
cervix. Gynecol Oncol. 141:247–254. 2016. View Article : Google Scholar
|
|
6
|
McCusker ME, Coté TR, Clegg LX and
Tavassoli FJ: Endocrine tumors of the uterine cervix: Incidence,
demographics, and survival with comparison to squamous cell
carcinoma. Gynecol Oncol. 88:333–339. 2003. View Article : Google Scholar
|
|
7
|
Kasajima A, Pfarr N, Mayr EM, Ura A, Moser
E, von Werder A, Agaimy A, Pavel M and Klöppel G: Rapid evolution
of metastases in patients with treated G3 neuroendocrine tumors
associated with NEC-like transformation and TP53 mutation. Endocr
Pathol. 35:313–324. 2024. View Article : Google Scholar
|
|
8
|
Hillman RT, Cardnell R, Fujimoto J, Lee
WC, Zhang J, Byers LA, Ramalingam P, Leitao M, Swisher E, Futreal
PA and Frumovitz M: Comparative genomics of high grade
neuroendocrine carcinoma of the cervix. PLoS One. 15:e02345052020.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Alejo M, Alemany L, Clavero O, Quiros B,
Vighi S, Seoud M, Cheng-Yang C, Garland SM, Juanpere N, Lloreta J,
et al: Contribution of human papillomavirus in neuroendocrine
tumors from a series of 10,575 invasive cervical cancer cases.
Papillomavirus Res. 5:134–142. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sorbye H, Hjortland GO, Vestermark LW,
Ladekarl M, Svensson J, Sundlöv A, Tiensuu Janson E, Garresori H,
Hofsli E, Kersten C, et al: Characteristics and treatment outcome
in a prospective cohort of 639 advanced high-grade digestive
neuroendocrine neoplasms (NET G3 and NEC): The NORDIC NEC 2 study.
Br J Cancer. 133:316–324. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Heetfeld M, Chougnet CN, Olsen IH, Rinke
A, Borbath I, Crespo G, Barriuso J, Pavel M, O'Toole D and Walter
T; Knowledge Network members, : Characteristics and treatment of
patients with G3 gastroenteropancreatic neuroendocrine neoplasms.
Endocr Relat Cancer. 22:657–664. 2015. View Article : Google Scholar : PubMed/NCBI
|