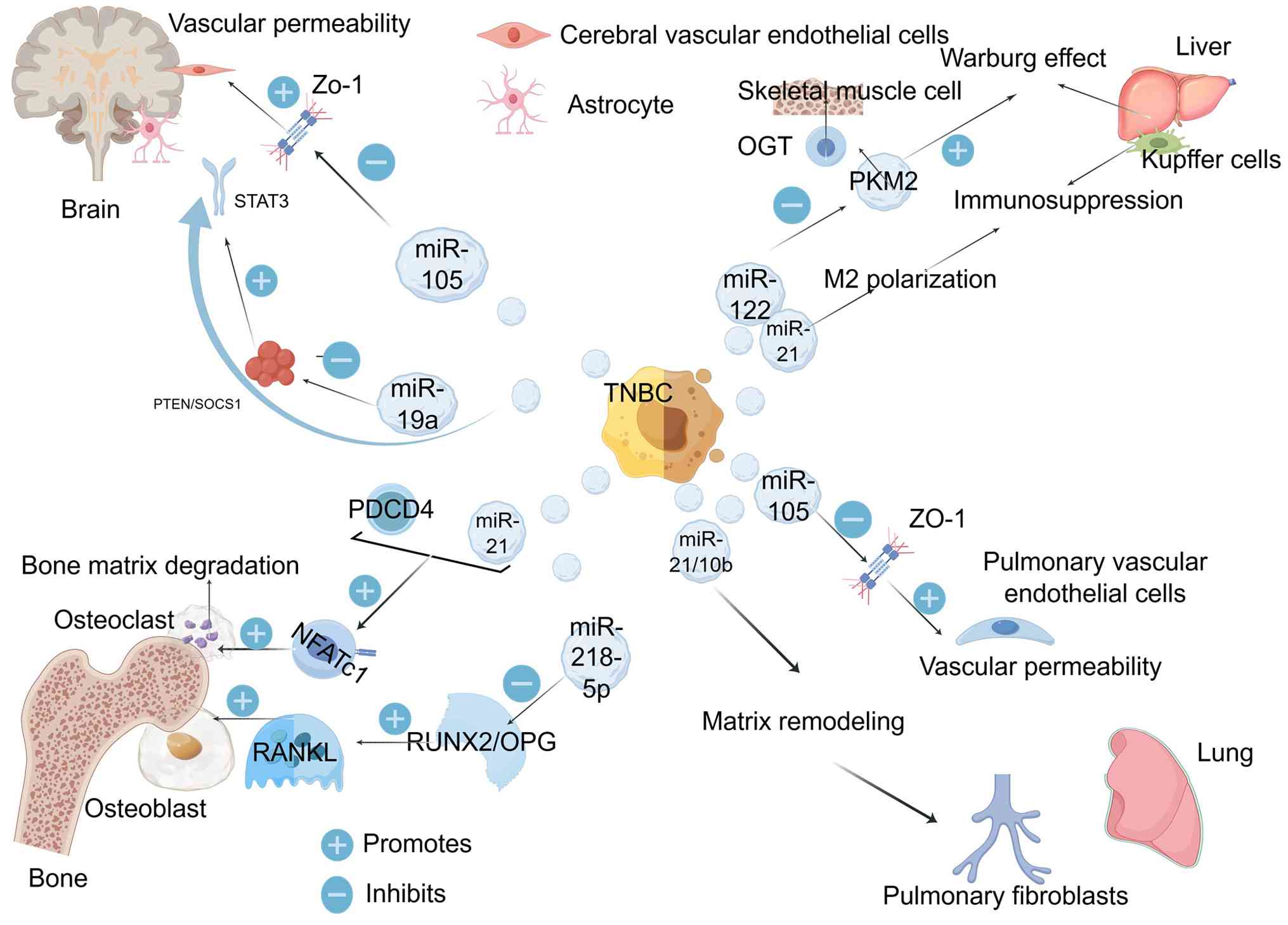
|
1
|
Wu SY, Wang H, Shao ZM and Jiang YZ:
Triple-negative breast cancer: New treatment strategies in the era
of precision medicine. Sci China Life Sci. 64:372–388. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Denkert C, Liedtke C, Tutt A and von
Minckwitz G: Molecular alterations in triple-negative breast
cancer-the road to new treatment strategies. Lancet. 389:2430–2442.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bianchini G, Balko JM, Mayer IA, Sanders
ME and Gianni L: Triple-negative breast cancer: Challenges and
opportunities of a heterogeneous disease. Nat Rev Clin Oncol.
13:674–690. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Llévenes P, Chen A, Lawton M, Rondón-Ortiz
AN, Qiu Y, Seen M, Monti S and Denis GV: Plasma exosomes in insulin
resistant obesity exacerbate progression of triple negative breast
cancer. BMC Cancer. 25:10892025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Reddy SM, Barcenas CH, Sinha AK, Hsu L,
Moulder SL, Tripathy D, Hortobagyi GN and Valero V: Long-term
survival outcomes of triple-receptor negative breast cancer
survivors who are disease free at 5 years and relationship with low
hormone receptor positivity. Br J Cancer. 118:17–23. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ding L, Xu Y, Li C and Chen X: Clinical
characteristics, prognosis, and prognostic factors of patients with
second primary triple-negative breast cancer: A study based on
surveillance, epidemiology, and end results database. Eur J Cancer
Prev. 34:316–328. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Alzubi MA, Turner TH, Olex AL, Sohal SS,
Tobin NP, Recio SG, Bergh J, Hatschek T, Parker JS, Sartorius CA,
et al: Separation of breast cancer and organ microenvironment
transcriptomes in metastases. Breast Cancer Res. 21:362019.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang C, Fan P, Zhou Y, Ma M, Zhong H, Liu
L, Chen Q and Xu K: Heterogeneous tissue-specific macrophages
orchestrate metastatic organotropism of breast cancer: Implications
for promising therapeutics. J Transl Med. 23:6922025. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Qixiang MA, Xiaodan Z, Kaiwen HU and Chao
AN: New thoughts upon seed and soil hypothesis of tumor metastasis.
Cancer Res Prev Treat. 42:1049–1053. 2015.
|
|
10
|
Zhou W, Fong MY, Min Y, Somlo G, Liu L,
Palomares MR, Yu Y, Chow A, O'Connor STF, Chin AR, et al:
Cancer-secreted miR-105 destroys vascular endothelial barriers to
promote metastasis. Cancer Cell. 25:501–515. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Navarro G, Gómez-Autet M, Morales P,
Rebassa JB, Llinas Del Torrent C, Jagerovic N, Pardo L and Franco
R: Homodimerization of CB2 cannabinoid receptor
triggered by a bivalent ligand enhances cellular signaling.
Pharmacol Res. 208:1073632024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bai S, Wei Y, Liu R, Xu R, Xiang L and Du
J: Role of tumour-derived exosomes in metastasis. Biomed
Pharmacother. 147:1126572022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Peinado H, Zhang H, Matei IR, Costa-Silva
B, Hoshino A, Rodrigues G, Psaila B, Kaplan RN, Bromberg JF, Kang
Y, et al: Pre-metastatic niches: Organ-specific homes for
metastases. Nat Rev Cancer. 17:302–317. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ganesh K and Massagué J: Targeting
metastatic cancer. Nat Med. 27:34–44. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wu S, Lu J, Zhu H, Wu F, Mo Y, Xie L, Song
C, Liu L, Xie X, Li Y, et al: A novel axis of
circKIF4A-miR-637-STAT3 promotes brain metastasis in
triple-negative breast cancer. Cancer Lett. 581:2165082024.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li J, Liu M, Zeng B and Wang Z: Propofol
induces hepatocellular carcinoma cell apoptosis via regulating
miR-105/JAK2/STAT3 axis. Cytokine. 148:1556492021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Morad G, Carman CV, Hagedorn EJ, Perlin
JR, Zon LI, Mustafaoglu N, Park TE, Ingber DE, Daisy CC and Moses
MA: Tumor-derived extracellular vesicles breach the intact
blood-brain barrier via transcytosis. ACS Nano. 13:13853–13865.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lee JY, Park K, Lee E, Ahn T, Jung HH, Lim
SH, Hong M, Do IG, Cho EY, Kim DH, et al: Gene expression profiling
of breast cancer brain metastasis. Sci Rep. 6:286232016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sato R, Nakano T, Hosonaga M, Sampetrean
O, Harigai R, Sasaki T, Koya I, Okano H, Kudoh J, Saya H and Arima
Y: RNA sequencing analysis reveals interactions between breast
cancer or melanoma cells and the tissue microenvironment during
brain metastasis. Biomed Res Int. 2017:80329102017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang B, Li X, Tang K, Xin Y, Hu G, Zheng
Y, Li K, Zhang C and Tan Y: Adhesion to the brain endothelium
selects breast cancer cells with brain metastasis potential. Int J
Mol Sci. 24:70872023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen X, Feng J, Chen W, Shao S, Chen L and
Wan H: Small extracellular vesicles: From promoting pre-metastatic
niche formation to therapeutic strategies in breast cancer. Cell
Commun Signal. 20:1412022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sharma R, Sharma R, Khaket TP, Dutta C,
Chakraborty B and Mukherjee TK: Breast cancer metastasis: Putative
therapeutic role of vascular cell adhesion molecule-1. Cell Oncol
(Dordr). 40:199–208. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang L, Zhang S, Yao J, Lowery FJ, Zhang
Q, Huang WC, Li P, Li M, Wang X, Zhang C, et al:
Microenvironment-induced PTEN loss by exosomal microRNA primes
brain metastasis outgrowth. Nature. 527:100–104. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Priego N, Zhu L, Monteiro C, Mulders M,
Wasilewski D, Bindeman W, Doglio L, Martínez L, Martínez-Saez E,
Ramón Y Cajal S, et al: STAT3 labels a subpopulation of reactive
astrocytes required for brain metastasis. Nat Med. 24:1024–1035.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Altea-Manzano P, Doglioni G, Liu Y,
Cuadros AM, Nolan E, Fernández-García J, Wu Q, Planque M, Laue KJ,
Cidre-Aranaz F, et al: A palmitate-rich metastatic niche enables
metastasis growth via p65 acetylation resulting in pro-metastatic
NF-κB signaling. Nat Cancer. 4:344–364. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wormald S, Zhang JG, Krebs DL, Mielke LA,
Silver J, Alexander WS, Speed TP, Nicola NA and Hilton DJ: The
comparative roles of suppressor of cytokine signaling-1 and −3 in
the inhibition and desensitization of cytokine signaling. J Biol
Chem. 281:11135–11143. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yamamoto T, Matsuda T, Muraguchi A,
Miyazono K and Kawabata M: Cross-talk between IL-6 and TGF-beta
signaling in hepatoma cells. FEBS Lett. 492:247–253. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cao M, Isaac R, Yan W, Ruan X, Jiang L,
Wan Y, Wang J, Wang E, Caron C, Neben S, et al:
Cancer-cell-secreted extracellular vesicles suppress insulin
secretion through miR-122 to impair systemic glucose homeostasis
and contribute to tumour growth. Nat Cell Biol. 24:954–967. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Fong MY, Zhou W, Liu L, Alontaga AY,
Chandra M, Ashby J, Chow A, O'Connor STF, Li S, Chin AR, et al:
Breast-cancer-secreted miR-122 reprograms glucose metabolism in
premetastatic niche to promote metastasis. Nat Cell Biol.
17:183–194. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Al Ageeli E: Dual roles of microRNA-122 in
hepatocellular carcinoma and breast cancer progression and
metastasis: A comprehensive review. Curr Issues Mol Biol.
46:11975–11992. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rathore D, Jain S, Patel S, Dharwal N,
Shukla N, Desai C, Shah J and Dave HV: MicroRNA-driven
organ-specific metastasis in triple-negative breast cancer:
Biogenesis, mechanisms, and therapeutic approaches. Med Oncol.
42:2962025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Arya SB, Chen S, Jordan-Javed F and Parent
CA: Ceramide-rich microdomains facilitate nuclear envelope budding
for non-conventional exosome formation. Nat Cell Biol.
24:1019–1028. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kanchan RK, Siddiqui JA, Mahapatra S,
Batra SK and Nasser MW: microRNAs orchestrate pathophysiology of
breast cancer brain metastasis: Advances in therapy. Mol Cancer.
19:292020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Udagawa N, Koide M, Nakamura M, Nakamichi
Y, Yamashita T, Uehara S, Kobayashi Y, Furuya Y, Yasuda H, Fukuda C
and Tsuda E: Osteoclast differentiation by RANKL and OPG signaling
pathways. J Bone Miner Metab. 39:19–26. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xiong S, Hu M, Li C, Zhou X and Chen H:
Role of miR-34 in gastric cancer: From bench to bedside (Review).
Oncol Rep. 42:1635–1646. 2019.PubMed/NCBI
|
|
36
|
Uehara N, Shibusawa N, Mikami Y,
Kyumoto-Nakamura Y, Sonoda S, Kato H, Yamaza T and Kukita T: Bone
metastatic mammary tumor cell-derived extracellular vesicles
inhibit osteoblast maturation via JNK signaling. Arch Biochem
Biophys. 750:1098212023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Yuan X, Qian N, Ling S, Li Y, Sun W, Li J,
Du R, Zhong G, Liu C, Yu G, et al: Breast cancer exosomes
contribute to pre-metastatic niche formation and promote bone
metastasis of tumor cells. Theranostics. 11:1429–1445. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ell B and Kang Y: SnapShot: Bone
metastasis. Cell. 151:690–690.e1. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kiechl S, Werner P, Knoflach M, Furtner M,
Willeit J and Schett G: The osteoprotegerin/RANK/RANKL system: A
bone key to vascular disease. Expert Rev Cardiovasc Ther.
4:801–811. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Baud'huin M, Lamoureux F, Duplomb L,
Rédini F and Heymann D: RANKL, RANK, osteoprotegerin: Key partners
of osteoimmunology and vascular diseases. Cell Mol Life Sci.
64:2334–2350. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Takayanagi H: RANKL as the master
regulator of osteoclast differentiation. J Bone Miner Metab.
39:13–18. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yu T, Wang Z, You X, Zhou H, He W, Li B,
Xia J, Zhu H, Zhao Y, Yu G, et al: Resveratrol promotes
osteogenesis and alleviates osteoporosis by inhibiting p53. Aging
(Albany NY). 12:10359–10369. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wu L, Luo Z and Liu Y, Jia L, Jiang Y, Du
J, Guo L, Bai Y and Liu Y: Aspirin inhibits RANKL-induced
osteoclast differentiation in dendritic cells by suppressing NF-κB
and NFATc1 activation. Stem Cell Res Ther. 10:3752019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Wang FS, Wang CJ, Chen YJ, Huang YT, Huang
HC, Chang PR, Sun YC and Yang KD: Nitric oxide donor increases
osteoprotegerin production and osteoclastogenesis inhibitory
activity in bone marrow stromal cells from ovariectomized rats.
Endocrinology. 145:2148–2156. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Liu B, Tian Y, Li F, Zhao Z, Jiang X, Zhai
C, Han X and Zhang L: Tumor-suppressing roles of miR-214 and
miR-218 in breast cancer. Oncol Rep. 35:3178–3184. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Gao M, Zhang Z, Sun J, Li B and Li Y: The
roles of circRNA-miRNA-mRNA networks in the development and
treatment of osteoporosis. Front Endocrinol (Lausanne).
13:9453102022. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Nakashima K, Zhou X, Kunkel G, Zhang Z,
Deng JM, Behringer RR and de Crombrugghe B: The novel zinc
finger-containing transcription factor osterix is required for
osteoblast differentiation and bone formation. Cell. 108:17–29.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Tomasetti M, Monaco F, Rubini C, Rossato
M, De Quattro C, Beltrami C, Sollini G, Pasquini E, Amati M, Goteri
G, et al: AGO2-RIP-Seq reveals miR-34/miR-449 cluster targetome in
sinonasal cancers. PLoS One. 19:e02959972024. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Yang X, Yang J, Lei P and Wen T: LncRNA
MALAT1 shuttled by bone marrow-derived mesenchymal stem
cells-secreted exosomes alleviates osteoporosis through mediating
microRNA-34c/SATB2 axis. Aging (Albany NY). 11:8777–8791. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Xu M, Jin H, Xu CX, Bi WZ and Wang Y:
MiR-34c inhibits osteosarcoma metastasis and chemoresistance. Med
Oncol. 31:9722014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wu X, Xiang H, Cong W, Yang H, Zhang G,
Wang Y, Guo Z, Shen Y and Chen B: PLOD1, a target of miR-34c,
contributes to cell growth and metastasis via repressing LATS1
phosphorylation and inactivating Hippo pathway in osteosarcoma.
Biochem Biophys Res Commun. 527:29–36. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Wang S, Zhang S, Xu H, Zhang M, Liu X, Liu
S, Li H and Hu Y: Ano5 deficiency leads to abnormal bone formation
via miR-34c-5p/KLF4/β-catenin in gnathodiaphyseal dysplasia. Int J
Mol Sci. 26:52672025. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Padua D, Zhang XH, Wang Q, Nadal C, Gerald
WL, Gomis RR and Massagué J: TGFbeta primes breast tumors for lung
metastasis seeding through angiopoietin-like 4. Cell. 133:66–77.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Mohammad KS and Akhund SA: From tumor to
bone: Growth factor receptors as key players in cancer metastasis.
Front Biosci (Landmark Ed). 29:1842024. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Han M, Wang F, Gu Y, Pei X, Guo G, Yu C,
Li L, Zhu M, Xiong Y and Wang Y: MicroRNA-21 induces breast cancer
cell invasion and migration by suppressing smad7 via EGF and TGF-β
pathways. Oncol Rep. 35:73–80. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Liu Z, Xin B, Zhang N, An P, Shi Y, Yang
J, Wan Y, He Y and Hu X: LSD1 modulates the bone metastasis of
breast cancer cells through hnRNPA2B1-mediated sorting of exosomal
miRNAs. Cell Death Discov. 10:1152024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Faubert B, Solmonson A and DeBerardinis
RJ: Metabolic reprogramming and cancer progression. Science.
368:eaaw54732020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Shen J, Ma Z, Yang J, Qu T, Xia Y, Xu Y,
Zhou M and Liu W: CircPHGDH downregulation decreases papillary
thyroid cancer progression through miR-122-5p/PKM2 axis. BMC
Cancer. 24:5112024. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Yan W, Cao M, Ruan X, Jiang L, Lee S,
Lemanek A, Ghassemian M, Pizzo DP, Wan Y, Qiao Y, et al:
Cancer-cell-secreted miR-122 suppresses O-GlcNAcylation to promote
skeletal muscle proteolysis. Nat Cell Biol. 24:793–804. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Huang JH, Li YH, Hong JZ, Li RN, Wang R,
Chen ZQ, Li SY, Chi YL, Huang JY and Zhu Y: Bile acid accumulation
induced by miR-122 deficiency in liver parenchyma promotes cancer
cell growth in hepatocellular carcinoma. Mol Ther Nucleic Acids.
36:1025602025. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Yi M, Xu L, Jiao Y, Luo S, Li A and Wu K:
The role of cancer-derived microRNAs in cancer immune escape. J
Hematol Oncol. 13:252020. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Wang G, Li J, Bojmar L, Chen H, Li Z,
Tobias GC, Hu M, Homan EA, Lucotti S, Zhao F, et al: Tumour
extracellular vesicles and particles induce liver metabolic
dysfunction. Nature. 618:374–382. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Bautista J and López-Cortés A: Deciphering
organotropism reveals therapeutic targets in metastasis. Front Cell
Dev Biol. 13:16774812025. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Ma L, Teruya-Feldstein J and Weinberg RA:
Tumour invasion and metastasis initiated by microRNA-10b in breast
cancer. Nature. 449:682–688. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Frankel LB, Christoffersen NR, Jacobsen A,
Lindow M, Krogh A and Lund AH: Programmed cell death 4 (PDCD4) is
an important functional target of the microRNA miR-21 in breast
cancer cells. J Biol Chem. 283:1026–1033. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Galardi A, Fogazzi V, Tottone C, Giussani
M, Pupa SM, Cosentino G and Iorio MV: ‘Small extracellular
vesicles: Messengers at the service of breast cancer agenda in the
primary and distant microenvironments’. J Exp Clin Cancer Res.
44:2162025. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Camps C, Saini HK, Mole DR, Choudhry H,
Reczko M, Guerra-Assunção JA, Tian YM, Buffa FM, Harris AL,
Hatzigeorgiou AG, et al: Integrated analysis of microRNA and mRNA
expression and association with HIF binding reveals the complexity
of microRNA expression regulation under hypoxia. Mol Cancer.
13:282014. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Kundu ST, Byers LA, Peng DH, Roybal JD,
Diao L, Wang J, Tong P, Creighton CJ and Gibbons DL: The miR-200
family and the miR-183~96~182 cluster target Foxf2 to inhibit
invasion and metastasis in lung cancers. Oncogene. 35:173–186.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Korpal M, Ell BJ, Buffa FM, Ibrahim T,
Blanco MA, Celià-Terrassa T, Mercatali L, Khan Z, Goodarzi H, Hua
Y, et al: Direct targeting of Sec23a by miR-200s influences cancer
cell secretome and promotes metastatic colonization. Nat Med.
17:1101–1108. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Cuiffo BG, Campagne A, Bell GW, Lembo A,
Orso F, Lien EC, Bhasin MK, Raimo M, Hanson SE, Marusyk A, et al:
MSC-regulated microRNAs converge on the transcription factor FOXP2
and promote breast cancer metastasis. Cell Stem Cell. 15:762–774.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Lefebvre CC, Giowachini P, Derrien J,
Naour M, Corre I, Thirouard L, Douillard E, Chiron D, Guillonneau
F, Treps L, et al: MCL-1 as a molecular switch between
myofibroblastic and pro-angiogenic features of breast
cancer-associated fibroblasts. Cell Death Dis. 16:6032025.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Ma L: Role of miR-10b in breast cancer
metastasis. Breast Cancer Res. 12:2102010. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Li B, Lu Y, Yu L, Han X, Wang H, Mao J,
Shen J, Wang B, Tang J, Li C and Song B: miR-221/222 promote cancer
stem-like cell properties and tumor growth of breast cancer via
targeting PTEN and sustained Akt/NF-κB/COX-2 activation. Chem Biol
Interact. 277:33–42. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zhuang G, Wu X, Jiang Z, Kasman I, Yao J,
Guan Y, Oeh J, Modrusan Z, Bais C, Sampath D and Ferrara N:
Tumour-secreted miR-9 promotes endothelial cell migration and
angiogenesis by activating the JAK-STAT pathway. EMBO J.
31:3513–3523. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Bautista-Sánchez D, Arriaga-Canon C,
Pedroza-Torres A, De La Rosa-Velázquez IA, González-Barrios R,
Contreras-Espinosa L, Montiel-Manríquez R, Castro-Hernández C,
Fragoso-Ontiveros V, Álvarez-Gómez RM and Herrera LA: The promising
role of miR-21 as a cancer biomarker and its importance in
RNA-based therapeutics. Mol Ther Nucleic Acids. 20:409–420. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Dykxhoorn DM, Wu Y, Xie H, Yu F, Lal A,
Petrocca F, Martinvalet D, Song E, Lim B and Lieberman J: miR-200
enhances mouse breast cancer cell colonization to form distant
metastases. PLoS One. 4:e71812009. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Debeb BG, Lacerda L, Anfossi S,
Diagaradjane P, Chu K, Bambhroliya A, Huo L, Wei C, Larson RA,
Wolfe AR, et al: miR-141-mediated regulation of brain metastasis
from breast cancer. J Natl Cancer Inst. 108:djw0262016. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Duhachek-Muggy S and Zolkiewska A:
ADAM12-L is a direct target of the miR-29 and miR-200 families in
breast cancer. BMC Cancer. 15:932015. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Mock K, Preca BT, Brummer T, Brabletz S,
Stemmler MP and Brabletz T: The EMT-activator ZEB1 induces bone
metastasis associated genes including BMP-inhibitors. Oncotarget.
6:14399–14412. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Pan Y, Li J, Zhang Y, Wang N, Liang H, Liu
Y, Zhang CY, Zen K and Gu H: Slug-upregulated miR-221 promotes
breast cancer progression through suppressing E-cadherin
expression. Sci Rep. 6:257982016. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Matsas S, Ruiz Simões A, Nazareth Aguiar P
Jr, Abdou Y, Krontiras H and Del Giglio A: Prognostic role of
miR-190, miR-221, and miR-381 in breast cancer: A systematic review
and meta-analysis. Eur Rev Med Pharmacol Sci. 29:301–312.
2025.PubMed/NCBI
|
|
82
|
Bayat M and Sadri Nahand J: Exosomal
miRNAs: The tumor's trojan horse in selective metastasis. Mol
Cancer. 23:1672024. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Deng Z, Rong Y, Teng Y, Zhuang X,
Samykutty A, Mu J, Zhang L, Cao P, Yan J, Miller D and Zhang HG:
Exosomes miR-126a released from MDSC induced by DOX treatment
promotes lung metastasis. Oncogene. 36:639–651. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Li J, Huang L, He Z, Chen M, Ding Y, Yao
Y, Duan Y, Zixuan L, Qi C, Zheng L, et al: Andrographolide
suppresses the growth and metastasis of luminal-like breast cancer
by inhibiting the NF-κB/miR-21-5p/PDCD4 signaling pathway. Front
Cell Dev Biol. 9:6435252021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Uen Y, Wang JW, Wang C, Jhang Y, Chung JY,
Tseng T, Sheu M and Lee S: Mining of potential microRNAs with
clinical correlation-regulation of syndecan-1 expression by
miR-122-5p altered mobility of breast cancer cells and possible
correlation with liver injury. Oncotarget. 9:28165–28175. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Guo X, Yang F, Yu L, Wen R, Zhang X and
Lin H: MiR-598-5p inhibits breast cancer tumor growth and lung
metastasis by targeting PPAPDC1A. Chin J Physiol. 66:103–110. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Jiang Z, Pei L, Xie Y, Ye Q, Liang X, Ye Y
and Liu S: Ruyiping formula inhibits metastasis via the
microRNA-134-SLUG axis in breast cancer. BMC Complement Med Ther.
21:1912021. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Liu Y, Tang J, Qiu X, Teng LA, Sriwastva
MK, Han X, Li Z, Liu M, Liu S, Da D, et al: Rab1A-mediated exosomal
sorting of miR-200c enhances breast cancer lung metastasis. Breast
Cancer (Dove Med Press). 15:403–419. 2023.PubMed/NCBI
|
|
89
|
Li C, Li R, Hu X, Zhou G and Jiang G:
Tumor-promoting mechanisms of macrophage-derived extracellular
vesicles-enclosed microRNA-660 in breast cancer progression. Breast
Cancer Res Treat. 192:353–368. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Edmonds MD, Hurst DR, Vaidya KS, Stafford
LJ, Chen D and Welch DR: Breast cancer metastasis suppressor 1
coordinately regulates metastasis-associated microRNA expression.
Int J Cancer. 125:1778–1785. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Zuo J, Yu Y, Zhu M, Jing W, Yu M, Chai H,
Liang C and Tu J: Inhibition of miR-155, a therapeutic target for
breast cancer, prevented in cancer stem cell formation. Cancer
Biomark. 21:383–392. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Sun H, Ding C, Zhang H and Gao J: Let-7
miRNAs sensitize breast cancer stem cells to radiation-induced
repression through inhibition of the cyclin D1/Akt1/Wnt1 signaling
pathway. Mol Med Rep. 14:3285–3292. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Lee JW, Guan W, Han S, Hong DK, Kim LS and
Kim H: MicroRNA-708-3p mediates metastasis and chemoresistance
through inhibition of epithelial-to-mesenchymal transition in
breast cancer. Cancer Sci. 109:1404–1413. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Yu YZ, Mu Q, Ren Q, Xie LJ, Wang QT and
Wang CP: miR-381-3p suppresses breast cancer progression by
inhibition of epithelial-mesenchymal transition. World J Surg
Oncol. 19:2302021. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Yang T, Martin P, Fogarty B, Brown A,
Schurman K, Phipps R, Yin VP, Lockman P and Bai S: Exosome
delivered anticancer drugs across the blood-brain barrier for brain
cancer therapy in Danio rerio. Pharm Res. 32:2003–2014. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Kamerkar S, LeBleu VS, Sugimoto H, Yang S,
Ruivo CF, Melo SA, Lee JJ and Kalluri R: Exosomes facilitate
therapeutic targeting of oncogenic KRAS in pancreatic cancer.
Nature. 546:498–503. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Abdelsalam M, Ahmed M, Osaid Z, Hamoudi R
and Harati R: Insights into exosome transport through the
blood-brain barrier and the potential therapeutical applications in
brain diseases. Pharmaceuticals (Basel). 16:5712023. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Li Y, Liu R and Zhao Z: Targeting brain
drug delivery with macromolecules through receptor-mediated
transcytosis. Pharmaceutics. 17:1092025. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Tao B, Du R, Zhang X, Jia B, Gao Y, Zhao Y
and Liu Y: Engineering CAR-NK cell derived exosome disguised
nano-bombs for enhanced HER2 positive breast cancer brain
metastasis therapy. J Control Release. 363:692–706. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Zhao M, Li Q, Chai Y, Rong R, He L, Zhang
Y, Cui H, Xu H, Zhang X, Wang Z, et al: An anti-CD19-exosome
delivery system navigates the blood-brain barrier for targeting of
central nervous system lymphoma. J Nanobiotechnology. 23:1732025.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Schapira M, Calabrese MF, Bullock AN and
Crews CM: Targeted protein degradation: Expanding the toolbox. Nat
Rev Drug Discov. 18:949–963. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Yang C, Kim SH, Bianco NR and Robbins PD:
Tumor-derived exosomes confer antigen-specific immunosuppression in
a murine delayed-type hypersensitivity model. PLoS One.
6:e225172011. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Ma L, Reinhardt F, Pan E, Soutschek J,
Bhat B, Marcusson EG, Teruya-Feldstein J, Bell GW and Weinberg RA:
Therapeutic silencing of miR-10b inhibits metastasis in a mouse
mammary tumor model. Nat Biotechnol. 28:341–347. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Dai L, Yu P, Fan H, Xia W, Zhao Y, Zhang
P, Zhang JZH, Zhang H and Chen Y: Identification and validation of
new DNA-PKcs inhibitors through high-throughput virtual screening
and experimental verification. Int J Mol Sci. 25:79822024.
View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Jiang Y, Zhang W, Liu L, Wu Y, Li W, Liang
J, Shen H, Fang S, Huang X, Chu Z, et al: Gelatin methacryloyl
xerogel puncture implants loaded with
Cu0.5Mn2.5O4 nanoparticles
synergizes cuproptosis and STING activation for enhanced breast
cancer immunotherapy. ACS Nano. 19:27902–27918. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Lawson DA, Bhakta NR, Kessenbrock K,
Prummel KD, Yu Y, Takai K, Zhou A, Eyob H, Balakrishnan S, Wang CY,
et al: Single-cell analysis reveals a stem-cell program in human
metastatic breast cancer cells. Nature. 526:131–135. 2015.
View Article : Google Scholar : PubMed/NCBI
|