Introduction
Colorectal cancer (CRC) is one of the leading causes
of cancer-related mortality, accounting for 9.3% of all
cancer-related mortality worldwide (1). Patients with CRC are commonly
diagnosed at an advanced stage of the disease due to the subtle
nature of early-stage CRC (2).
Although targeted therapies and immunotherapies have been applied
to treat patients with advanced CRC, chemotherapy remains the
cornerstone of standard treatment regimens (3,4).
However, a notable proportion of patients with advanced CRC
gradually develop resistance to chemotherapy (5–7).
Therefore, it is necessary to identify novel therapeutic targets to
improve the management of CRC. In recent years, studies have
explored novel treatment modalities for CRC, such as glucagon-like
peptide 1 receptor agonists and the combination of autocrine
motility factor peptide and glycyrrhetinic acid (8,9).
Potential molecular targets of CRC have also been identified, such
as heat shock transcriptional factor 4, E3 ubiquitin ligase-related
genes and ribosomal protein L22-like 1 (10–12).
Curcumin, a compound derived from turmeric
(Curcuma longa), exhibits an extensive range of biological
activities, such as anti-inflammatory, anti-diabetic and antitumor
effects (13). Emerging evidence
has suggested that curcumin can serve as a promising agent for the
treatment of CRC. For example, Dal et al (14) demonstrated that curcumin could
inhibit the viability of CRC cells via activating
nucleotide-binding oligomerization domain-like receptor protein
3-mediated pyroptosis. Furthermore, Liu et al (15) demonstrated that curcumin increased
reactive oxidative species (ROS) in CRC cells, thus further
activating the Kelch-like ECH-associated protein 1/nuclear factor
erythroid 2-related factor 2/microRNA-34 axis and suppressing the
metastasis of CRC cells.
Ferroptosis is a type of programmed cell death
characterized by glutathione deprivation, accumulation of ferrous
iron (Fe2+) and enhanced oxidative stress (16,17).
Inducing ferroptosis has been considered as a potential strategy
for cancer treatment. For example, allicin, a garlic-derived
organosulfide, exhibits the potential to treat nasopharyngeal
cancer due to the ability of promoting ferroptosis (18). Progesterone is able to induce
ferroptosis, thus promoting the efficacy of niraparib in ovarian
cancer (19). Furthermore, it has
been extensively reported that curcumin can induce ferroptosis in
CRC (20–22).
Leucovorin is commonly incorporated into
chemotherapeutic regimens for CRC due to its effect on sensitizing
tumor cells to fluorouracil (23,24).
Considering this effect of leucovorin, and that curcumin inhibits
cell viability and induces ferroptosis in CRC cells, we made a
preliminary hypothesis that leucovorin could also sensitize
curcumin-mediated ferroptosis in CRC cells. In the present study,
CRC cells were treated with the combination of curcumin and
leucovorin to investigate the effect of this combination on cell
survival and ferroptosis.
Materials and methods
Cell culture
Caco-2 cells (Tongwei Co., Ltd.) and HCT116 cells
(iCell Gene Therapeutics, Inc.) were cultured in DMEM (cat. no.
4511; Wuhan Servicebio Technology Co., Ltd.) and RPMI1640 medium
(cat. no. G4531; Wuhan Servicebio Technology Co., Ltd.)
respectively, supplemented with 10% FBS (cat. no. 164210; Procell
Life Science & Technology Co., Ltd.). All cells were cultured
at 37°C in a humidified incubator with 5% CO2.
Curcumin and leucovorin
incubation
Initially, Caco-2 and HCT116 cells were cultured
with various concentrations of curcumin (0, 5, 10, 20, 50 and 100
µM; cat. no. HY-N0005; MedChemExpress) and leucovorin (0, 1, 2, 4,
8 and 16 µM; cat. no. HY-17556; MedChemExpress) for 24 h. Caco-2
and HCT116 cells were also incubated with combinations of 1, 2, 4,
8 and 16 µM leucovorin, with 50 and 100 µM curcumin in combination
for 24 h. Experimental groups established were as follows: Leu, 2
µM leucovorin; Cur, 50 µM curcumin; Leu + Cur, 2 and 50 µM
leucovorin and curcumin, respectively, in combination (24 h
incubation). Furthermore, Caco-2 and HCT116 cells were pre-treated
with 2 µM leucovorin for 24 h, followed by co-treatment with 2 µM
leucovorin and 50 µM curcumin (pre-Leu + Leu + Cur group) for an
additional 24 h. Caco-2 and HCT116 cells cultured under normal
conditions without any treatment served as the control group. The
duration of treatment was in accordance with previous studies
(25–27).
Ferrostatin-1 (Fer-1) incubation
The Caco-2 and HCT116 cells in Control, Leu, Cur,
Leu + Cur and pre-Leu + Leu + Cur groups were incubated with 10 µM
Fer-1 (cat. no. HY-100579, MedChemExpress) at 37°C for 24 h
(28). Subsequently, cell viability
and Fe2+ were analyzed.
Cell viability
Cell viability was evaluated utilizing a Cell
Counting Kit-8 assay (CCK-8; Wuhan Servicebio Technology Co.,
Ltd.). Briefly, Caco-2 and HCT116 cells were cultured in a 96-well
plate and treated with curcumin and leucovorin as aforementioned.
Following incubation, the cell medium (DMEM for Caco-2, and
RPMI-160 for HCT-116 cells) supplemented with CCK-8 reagent was
added in each well and cells were cultured in an incubator at 37°C
for 2 h. The optical density (OD) at a wavelength of 450 nm was
measured using a microplate reader (Nanjing Huadong Electronics
Group Co., Ltd.). The relative cell viability was calculated based
on the following formula: Cell viability rate=(OD of experimental
groups/OD of the corresponding control group) ×100%.
Glutathione (GSH) analysis
Cells were collected at 24 h after treatment with
curcumin and leucovorin as aforementioned. Subsequently, following
collection, the cells were lysed using repeated freeze-thaw cycles.
The cell lysates were centrifuged at 100 × g at 4°C for 10 min to
obtain the cell supernatant. GSH content in cell supernatant was
assessed utilizing the corresponding GSH Assay Kit (cat. no. S0053;
Beyotime Institute of Biotechnology), according to the
manufacturer's protocol. The OD was measured using a microplate
reader (Nanjing Huadong Electronics Group Co., Ltd.).
Malondialdehyde (MDA) and
Fe2+assessment
Briefly, following treatment with the indicated
compounds for 24 h, cells were lysed with RIPA Buffer (Wuhan
Servicebio Technology Co., Ltd.) for 30 min. The protein
concentration in cell supernatant was quantified using a BCA Kit
(Wuhan Servicebio Technology Co., Ltd.). MDA and Fe2+
levels were assessed using the Lipid Peroxidation MDA Assay Kit
(cat. no. S0131S; Beyotime Institute of Biotechnology) and Ferrous
Ion Content Assay Kit (cat. no. BC5415; Beijing Solarbio Science
& Technology Co., Ltd.), respectively, according to the
manufacturer's protocol.
Reactive oxygen species staining
Following incubation for 24 h, ROS levels in CRC
cells were calculated utilizing a ROS Assay Kit (cat. no. S0033S;
Beyotime Institute of Biotechnology), according to the
manufacturer's instructions. Prior to analysis, the working probe
was freshly prepared. Cells were then incubated with the working
probe at 37°C for 10 min. Images were captured using an inverted
fluorescence microscope (Motic Incorporation, Ltd.).
Mitochondrial membrane potential (MMP)
detection
To assess MMP, CRC cells were incubated with MMP
Assay Kit with JC-1 (cat. no. C2006; Beyotime Institute of
Biotechnology) for 24 h. The JC-1 working solution was prepared
according to the manufacturer's instructions. The cells were
incubated with the JC-1 working solution for 20 min at 37°C. After
the working solution was removed, images of the stained cells were
captured using an inverted fluorescence microscope (Motic
Incorporation, Ltd.).
Western blot analysis
Following incubation with curcumin and leucovorin as
aforementioned for 24 h at 37°C, cells were lysed using a RIPA
buffer (Wuhan Servicebio Technology Co., Ltd.). To ensure complete
lysis, cells in RIPA buffer were scrapped and incubated on ice for
30 min. Following centrifugation at 12,000 × g for 5 min at 4°C,
the cell supernatant was obtained. The protein concentration was
measured utilizing a BCA kit (Wuhan Servicebio Technology Co.,
Ltd.), according to the manufacturer's instructions. After mixing
with loading buffer, the supernatant was denatured at 98°C for 5
min. Subsequently, the protein samples (10 µg) were separated using
a 10% Precast Gel (Willget) for 30 min, then transferred onto a
nitrocellulose membrane (MilliporeSigma) for 90 min. Following by
washing with Tris Buffered Saline with Tween 20 (TBST; cat. no.
G2150; Wuhan Servicebio Technology Co., Ltd.), the membrane was
immersed in 5% BSA (Beyotime Institute of Biotechnology) for 90 min
at 37°C. Subsequently, the membrane was incubated with primary
antibodies against solute carrier family 7 member 11 (SLC7A11;
1:1,000; cat. no. GB150180; Wuhan Servicebio Technology Co., Ltd.),
acyl-CoA synthetase long chain family member 4 (ACSL4; cat. no.
81196-1-RR; dilution, 1:1,000; Proteintech Group, Inc.) and GAPDH
(cat. no. GB15004; 1:5,000, Wuhan Servicebio Technology Co., Ltd.)
at 4°C overnight. After the primary antibodies were discarded, the
membrane was incubated with the corresponding secondary antibody
(cat. no. GB23303; 1:10,000; Wuhan Servicebio Technology Co., Ltd.)
for 90 min at 37°C. Subsequently, the membrane was washed using
TBST and immersed in ECL Reagent (cat. no. MA0186; Dalian Meilun
Biology Technology Co., Ltd.). Lastly, the protein bands were
visualized on X-ray films. The experiments were conducted in
triplicate. The intensity value was measured utilizing ImageJ
software (version 1.8.0; National Institutes of Health).
Target gene analysis
The target genes of curcumin and leucovorin were
analyzed from the PharmMapper database (https://www.lilab-ecust.cn/pharmmapper/index.html).
The common target genes of curcumin and leucovorin were displayed
using Cytoscape (version 3.10.3). The Gene Ontology (GO) and Kyoto
Encyclopedia of Genes and Genomes (KEGG) enrichment was performed
using the Database for Annotation, Visualization and Integrated
Discovery tool (https://david.ncifcrf.gov/). The significance of each
enrichment item was identified with the cut-off FDR <0.05.
Statistical analysis
All data were expressed as the mean ± SD. The
experiments were performed in triplicate. All analyses were
performed by GraphPad software (version 9.0; Dotmatics). The
differences among groups were compared using one-way ANOVA followed
by Dunnett's or Tukey's post hoc test. P<0.05 was considered to
indicate a statistically significant difference.
Results
Effect of curcumin and leucovorin on
cell viability
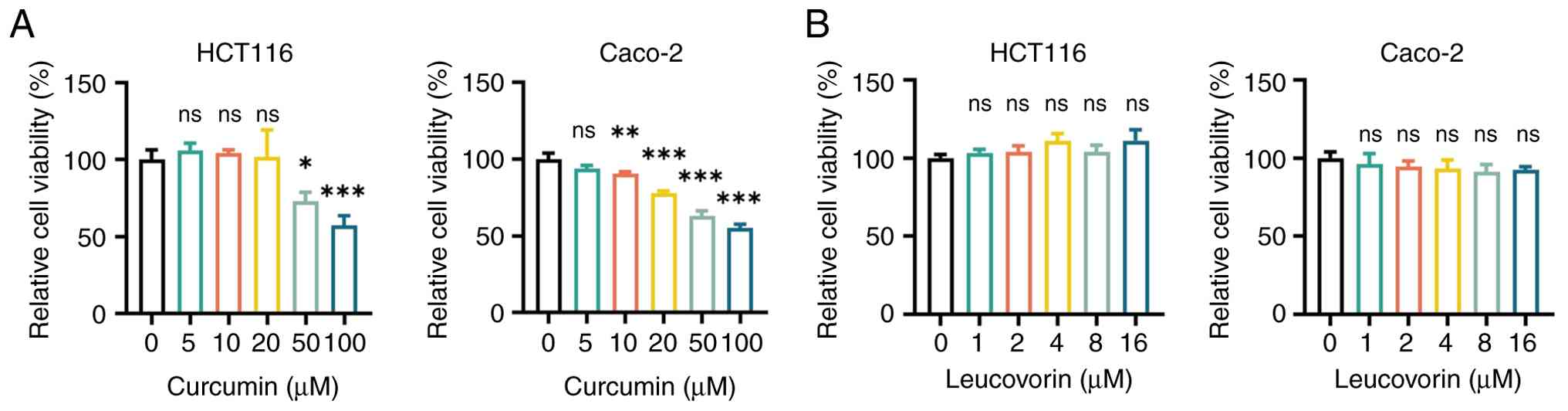
The results indicated that treatment of Caco-2 and
HCT116 cells with curcumin alone significantly suppressed their
viability in a dose-dependent manner. Statistically significant
differences were observed in Caco-2 cells treated with 10, 20, 50
and 100 µM curcumin, and in HCT116 cells treated with 50 and 100 µM
(all P<0.05; Fig. 1A). By
contrast, treatment with 1–16 µM leucovorin alone had no effect on
the viability of Caco-2 or HCT116 cells (all P>0.05; Fig. 1B). Considering that 50 µM curcumin
exhibited significant inhibitory effect in both cell lines, this
concentration was selected for the subsequent experiments, which
was also in accordance with a previous study (29). Furthermore, 2 µM leucovorin was
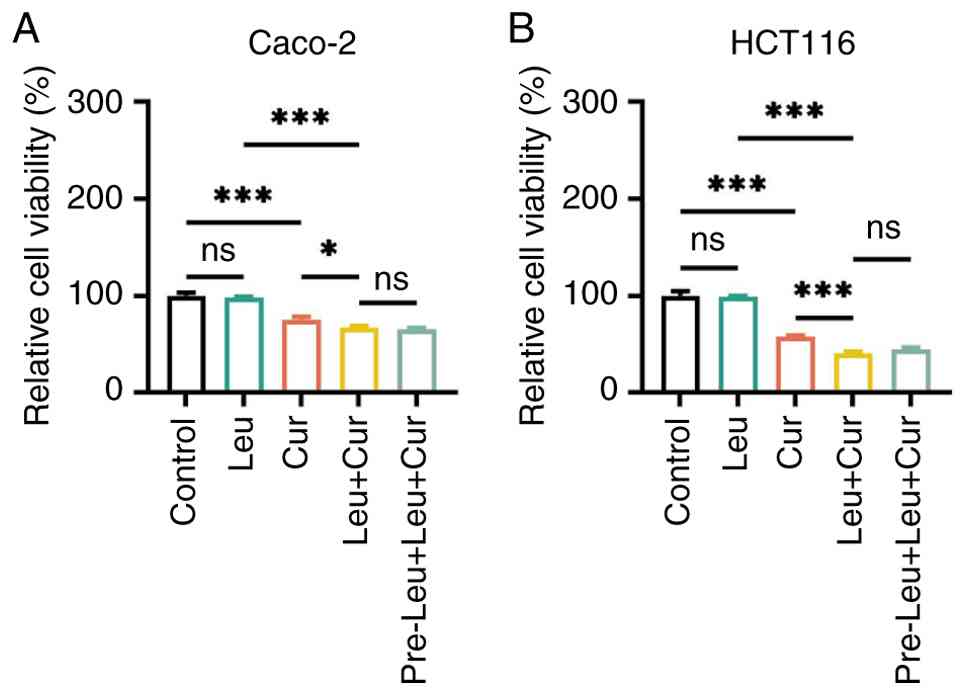
chosen for further experiments, as previously described (30). Co-treatment in the Leu + Cur group
resulted in significantly reduced cell viability compared with the
Cur group in both Caco-2 and HCT116 cells (both P<0.05; Fig. 2A and B). Furthermore, the pre-Leu +
Leu + Cur group displayed similar inhibitory effect on the
viability of Caco-2 and HCT116 cells compared with the simultaneous
Leu + Cur group (both P>0.05; Fig.
2A and B). The dose-dependent effects in the Leu + Cur group
were further investigated with varying concentrations of curcumin
and leucovorin (Figs. S1A-D and
S2A-D).
Oxidative stress and Fe2+
levels in CRC cells treated with curcumin/leucovorin
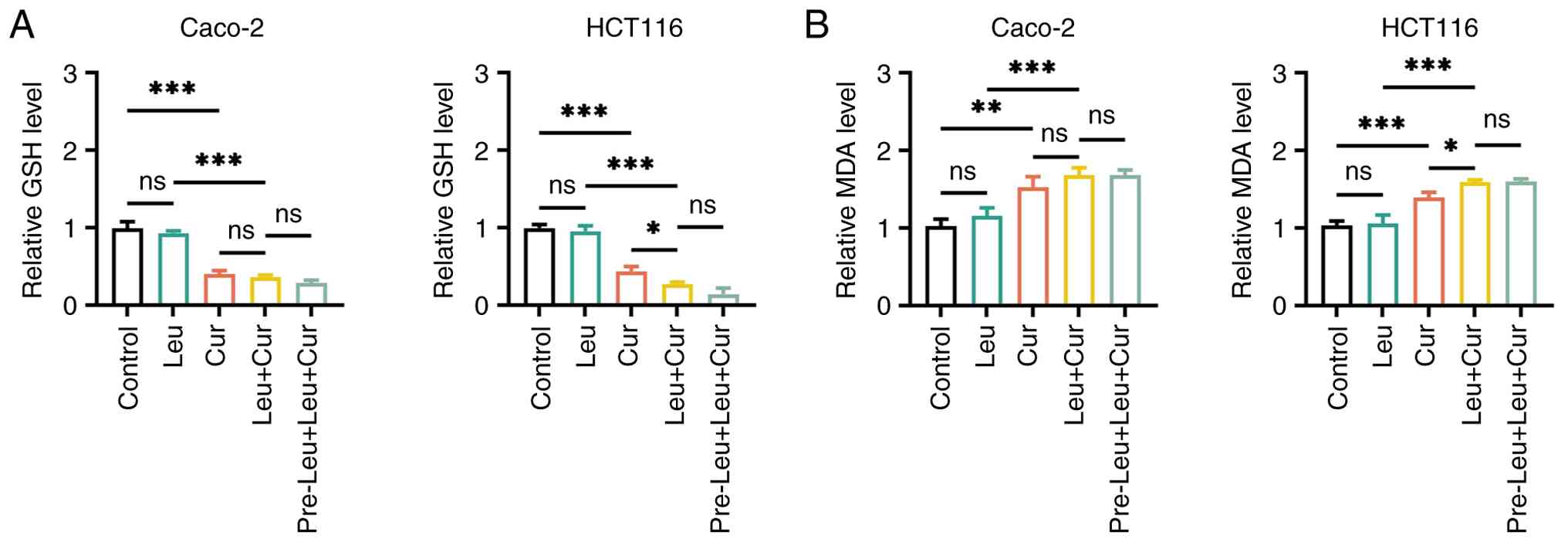
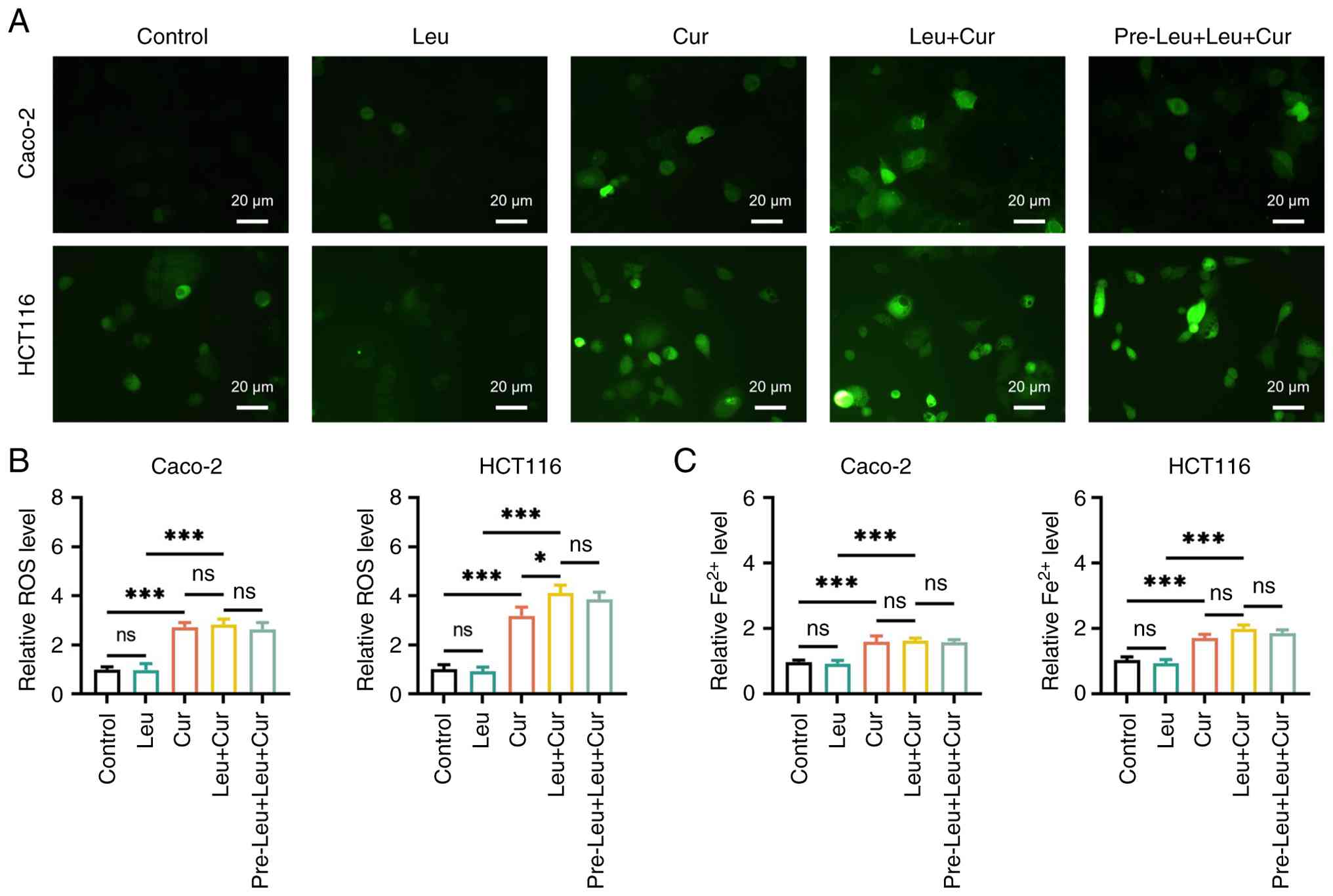
In both Caco-2 and HCT116 cells, treatment with
curcumin alone in the Cur group significantly reduced the relative
GSH levels (both P<0.001; Fig.
3A) and enhanced those of relative MDA (both P<0.01;
Fig. 3B) and ROS (both P<0.001;
Fig. 4A and B) compared with the
control group. Curcumin alone in the Cur group also elevated
relative intracellular Fe2+ levels in both cell lines
compared with the control group (both P<0.001; Fig. 4C). However, CRC cell treatment with
leucovorin alone in the Leu group had no effect on GSH, MDA, ROS or
Fe2+ levels compared with the control group (all
P>0.05). In addition, co-treatment in the Leu + Cur group
significantly further reduced the relative GSH levels and
significantly increased those of MDA and ROS compared with Cur
group in HCT116 cells (all P<0.05). However, this effect was not
observed in Caco-2 cells (all P>0.05). Although in HCT116 cells,
the Leu + Cur group showed notable increases in relative
Fe2+ levels compared with that of the Cur group, a
statistically significant difference was not reached (P>0.05).
Consistently, no significant difference in Fe2+ levels
was detected between the Cur and Leu + Cur groups in Caco-2 cells
(P>0.05; Fig 4C). The pre-Leu +
Leu + Cur cells did not demonstrate a more predominant effect on
GSH, MDA, ROS and Fe2+ levels compared with that of the
Leu + Cur group in both CRC cell lines. However, the relative GSH,
ROS and Fe2+ levels in HCT116 cells were markedly lower
in the pre-Leu + Leu + Cur group (all P>0.05). Furthermore,
ferroptosis inhibitor, Fer-1, was used for verification. In both
Caco-2 and HCT116 cells, the addition of Fer-1 [Fer-1 (+) cells]
improved cell viability and reduced Fe2+ level in the
Cur, Leu + Cur and pre-Leu + Leu + Cur groups compared with Fer-1
(−) cells, confirming ferroptosis (Fig. S3A and B).
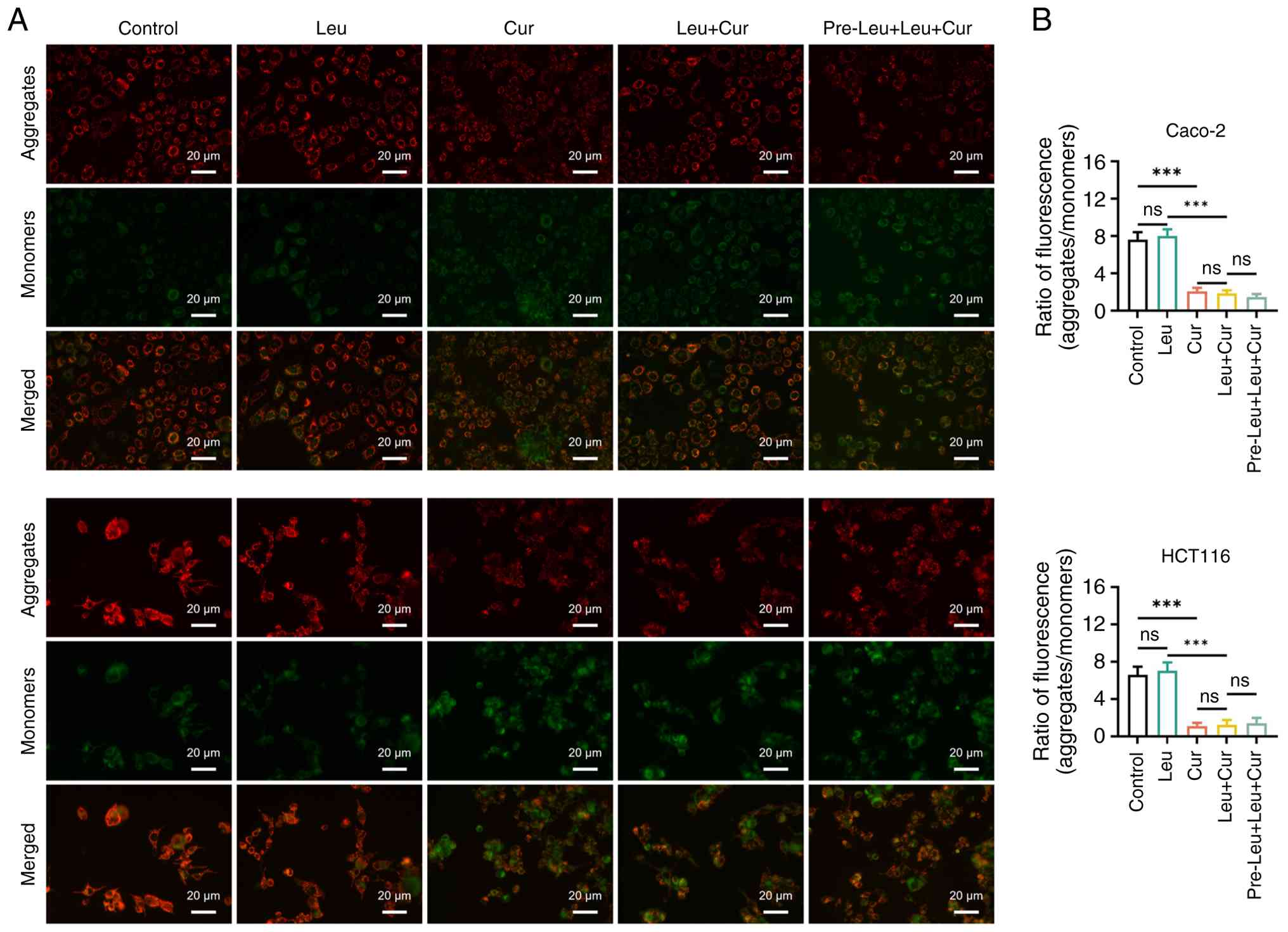
MMP in curcumin/leucovorin-treated CRC
cell lines
MMP, which was assessed by the ratio of
aggregates/monomers, was notably decreased in the Cur group in both
Caco-2 and HCT116 cells compared with the control group (both
P<0.001). However, treatment with leucovorin in the Leu group
had no effect on MMP in either cell line compared with the control
group (both P>0.05). The combination treatment in the Leu + Cur
group exhibited minor effects on MMP levels compared with Cur group
in both Caco-2 and HCT116 cells (both P>0.05). Furthermore, the
pre-Leu + Leu + Cur group did not show significantly altered MMP
levels compared with Leu + Cur group in HCT116 cells. In Caco-2
cells in pre-Leu + Leu + Cur group a slight, but not statistically
significant, decrease in MMP levels were observed compared with the
Leu + Cur group (both P>0.05; Fig.
5A and B).
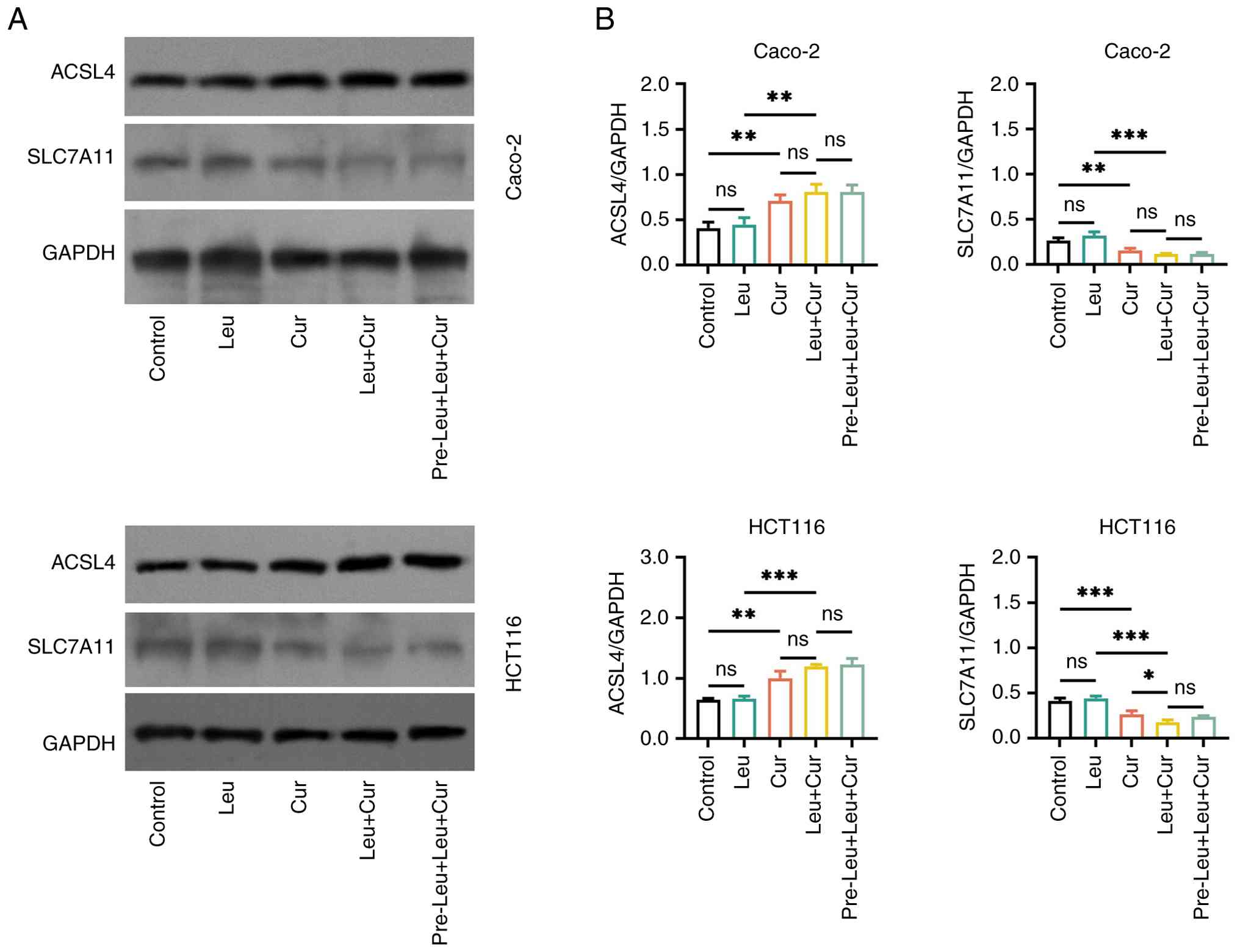
Expression of ferroptosis-related
markers in curcumin/leucovorin-treated CRC cell lines
In both Caco-2 and HCT116 cells, treatment with
curcumin alone in the Cur group upregulated ACSL4 and downregulated
SLC7A11 compared with the control group (all P<0.01). However,
leucovorin alone treatment in the Leu group had no significant
effect on the expression levels of both proteins compared with the
control group (all P>0.05). Furthermore, the Leu + Cur group
showed numerically increased ACSL4 levels compared with Cur group
in Caco-2 and HCT116 cells; however, a statistically significant
difference was not reached (both P>0.05). The Leu + Cur group
also presented significantly attenuated the expression levels of
SLC7A11 compared with Cur group in HCT116 cells (P<0.05), while
the reduction observed in Caco-2 cells did not reach statistical
significance (P>0.05). Furthermore, the pre-Leu + Leu + Cur
group displayed a similar effect on ACSL4 and SLC7A11 expression
compared with the Leu + Cur group in both cell lines (all
P>0.05; Fig. 6A and B).
Potential mechanisms of the
interaction between curcumin and leucovorin
A total of 218 co-target genes of curcumin and
leucovorin were identified from the PharmMapper database. GO
analyses indicated that the co-target genes of curcumin and
leucovorin were mainly enriched in biological processes of ‘signal
transduction’ and ‘peptidyl-serine phosphorylation’, cellular
component of ‘cytosol’ and ‘extracellular exosome’, and molecular
function of ‘enzyme binding’ and ‘nuclear receptor activity’. KEGG
analyses indicated that these genes were mainly enriched in
metabolic pathways and pathways in cancer (Fig. S4A-C).
Discussion
In the present study, two well-established CRC cell
lines, namely HCT116 and Caco-2, were utilized to perform the in
vitro experiments to assess the effect of curcumin and
leucovorin in CRC (31–33). The CCK-8 assay revealed that
curcumin inhibited the viability of HCT116 and Caco-2 cells in a
dose-dependent manner, thus supporting the anti-CRC capacity of
curcumin. These findings were consistent with those reported in
previous studies (20,25,34).
Other studies indicated that curcumin exerted its antitumor
activity in CRC by inducing ferroptosis (20–22).
Therefore, the present study further verified this mechanism. The
results demonstrated that treatment of HCT116 and Caco-2 cells with
curcumin significantly increased oxidative stress and
Fe2+ levels, and the expression levels of the
ferroptosis-related marker ACSL4, thus supporting the notion that
curcumin could induce ferroptosis in CRC cells.
Leucovorin, a clinically notable medication adjunct
in CRC chemotherapy, is known to augment the efficacy of
fluorouracil via inhibition of thymidylate synthase (35), thus enhancing the cytotoxicity of
fluorouracil in CRC (36,37). However, whether leucovorin can
affect the antitumor effect of curcumin in CRC has not been
previously investigated. In the present study, the results
demonstrated that leucovorin alone had no effect on CRC cell
viability. However, the combination of curcumin and leucovorin more
effectively suppressed the viability of CRC cells compared with
curcumin alone. These findings suggested that leucovorin could
potentiate the antitumor effect of curcumin on CRC. Furthermore,
the present study revealed that leucovorin had a modest effect on
promoting curcumin-induced ferroptosis. However, this effect was
less pronounced compared with that observed on cell viability. It
was hypothesized that this discrepancy could be due to the fact
that apart from ferroptosis, curcumin could inhibit the viability
of CRC cells via additional mechanisms, including apoptosis and
pyroptosis (14,38). Notably, the results also
demonstrated that the effect of leucovorin on enhancing
curcumin-induced inhibition of cell viability and ferroptosis was
more evident in HCT116 cells compared with Caco-2 cells.
Furthermore, curcumin exerted a stronger effect on inhibiting the
viability of HCT116 cells compared with Caco-2 cells. This finding
could be attributed to intrinsic differences between the two cell
lines used. For instance, HCT116 cells exhibit microsatellite
instability and mutations in TGF-β (39), a known target of curcumin in CRC
(40). Meanwhile, the p53 status,
differentiation and ferroptosis sensitivity also differ markedly
between these two cell lines. Therefore, curcumin and curcumin
combined with leucovorin displayed enhanced efficacy in HCT116
cells. However, further studies are warranted to verify this
hypothesis.
Potentiating the efficacy of one therapeutic
modality with pre-treatment of another one is a common strategy
used in studies on treatments for cancer. For example,
pre-treatment with inhibitors of EGFR, c-Jun N-terminal kinase or
protein kinase C improved the efficacy of hypericin photodynamic
therapy in cancer cells (41).
Another previous study reported that the cytotoxicity of cisplatin
in resistant ovarian cancer cells could be promoted by
pre-treatment of quercetin (42).
Furthermore, it has been reported that pre-treatment of colon
cancer cells with leucovorin enhances the antitumor capacity of
fluorouracil (43). Therefore, the
present study further explored whether pre-treatment with
leucovorin could further potentiate the effect of curcumin on CRC.
However, pre-treatment of CRC cells with leucovorin followed by
curcumin + leucovorin treatment had no effect on cell viability and
ferroptosis compared with co-treatment with curcumin + leucovorin.
These findings suggested that the capacity of leucovorin in
potentiating curcumin in CRC could not be further improved by
pre-treatment. A potential explanation for these findings is that
the co-treatment strategy with leucovorin already fully promoted
the effects of curcumin. However, this hypothesis requires
verification.
The present study demonstrated that leucovorin could
augment the antitumor efficacy of curcumin in CRC, thus suggesting
that their combination could serve as a potential therapeutic
regimen for patients with CRC. The detailed molecular mechanisms of
their interaction were not explored in the present study.
Nevertheless, the present study identified 218 co-target genes of
leucovorin and curcumin, which were mainly enriched in ‘signal
transduction’, ‘peptidyl-serine phosphorylation’, ‘enzyme binding’
and ‘nuclear receptor activity’, as well as ‘metabolic pathways’
and ‘pathways in cancer’. Based on these findings, further studies
could explore the detailed mechanisms by which leucovorin enhanced
the effects of curcumin in CRC. Furthermore, the present study
findings suggested that leucovorin promoted curcumin-induced
ferroptosis in CRC. Therefore, an assumption could be made that
leucovorin might affect ferroptosis-related mechanisms in CRC, such
as redox metabolism, iron metabolism and ferroptosis regulators,
thus promoting the effect of curcumin in CRC. Nevertheless, this
assumption should be further verified. However, in vivo
studies are warranted to verify the findings of the present study.
In addition, further clinical research is necessary before this
regimen can be considered for clinical application; the findings of
the present study should be verified in other cell lines of CRC.
Additionally, the lack of enhanced effect with leucovorin
pre-treatment should be interpreted with caution, as timing and
pharmacodynamics may differ between experimental models and
clinical settings.
In conclusion, the present study indicated that
leucovorin could enhance curcumin-induced suppression of cell
viability and ferroptosis in CRC cells, demonstrating the potential
of curcumin combined with leucovorin as a therapeutic regimen for
treatment of CRC in the future.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
HX contributed to the study conception and design.
Material preparation, data collection and analysis were performed
by RM and QX. Data interpretation was performed by XW. The first
draft of the manuscript was written by XW and all authors commented
on previous versions of the manuscript. HX and RM confirm the
authenticity of all the raw data. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
2
|
Almeida-Lousada H, Mestre A, Ramalhete S,
Price AJ, de Mello RA, Marreiros AD, Neves RPD and Castelo-Branco
P: Screening for colorectal cancer leading into a new decade: The
‘Roaring '20s’ for epigenetic biomarkers? Curr Oncol. 28:4874–4893.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Biller LH and Schrag D: Diagnosis and
treatment of metastatic colorectal cancer: A review. JAMA.
325:669–685. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shin AE, Giancotti FG and Rustgi AK:
Metastatic colorectal cancer: Mechanisms and emerging therapeutics.
Trends Pharmacol Sci. 44:222–236. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Singh U, Kokkanti RR and Patnaik S: Beyond
chemotherapy: Exploring 5-FU resistance and stemness in colorectal
cancer. Eur J Pharmacol. 991:1772942025. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu M, Li TZ and Xu C: The role of
tumor-associated fibroblast-derived exosomes in chemotherapy
resistance of colorectal cancer and its application prospect.
Biochim Biophys Acta Gen Subj. 1869:1307962025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Luo S, Yue M, Wang D, Lu Y, Wu Q and Jiang
J: Breaking the barrier: Epigenetic strategies to combat platinum
resistance in colorectal cancer. Drug Resist Updat. 77:1011522024.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tong G, Peng T, Chen Y, Sha L, Dai H,
Xiang Y, Zou Z, He H and Wang S: Effects of GLP-1 receptor agonists
on biological behavior of colorectal cancer cells by regulating
PI3K/AKT/mTOR signaling pathway. Front Pharmacol. 13:9015592022.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kim SG, Duong TV, Lee S, Ryu KJ, Kwon HK
and Park HS: Synergistic inhibition of colorectal cancer cells by
autocrine motility factor peptide and glycyrrhetinic acid. Discov
Med. 36:2063–2070. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang K, Ning S, Zhang S, Jiang M, Huang Y,
Pei H, Li M and Tan F: Extracellular matrix stiffness regulates
colorectal cancer progression via HSF4. J Exp Clin Cancer Res.
44:302025. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liang L, Liang X, Yu X and Xiang W:
Bioinformatic analyses and integrated machine learning to predict
prognosis and therapeutic response based on E3 ligase-related genes
in colon cancer. J Cancer. 15:5376–5395. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li C, Du X, Zhang H and Liu S: Knockdown
of ribosomal protein L22-like 1 arrests the cell cycle and promotes
apoptosis in colorectal cancer. Cytojournal. 21:452024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang W, Li M, Wang L, Chen L and Goh BC:
Curcumin in cancer therapy: Exploring molecular mechanisms and
overcoming clinical challenges. Cancer Lett. 570:2163322023.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dal Z and Aru B: The role of curcumin on
apoptosis and NLRP3 inflammasome-dependent pyroptosis on colorectal
cancer in vitro. Turk J Med Sci. 53:883–893. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu C, Rokavec M, Huang Z and Hermeking H:
Curcumin activates a ROS/KEAP1/NRF2/miR-34a/b/c cascade to suppress
colorectal cancer metastasis. Cell Death Differ. 30:1771–1785.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jiang X, Stockwell BR and Conrad M:
Ferroptosis: Mechanisms, biology and role in disease. Nat Rev Mol
Cell Biol. 22:266–282. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li J, Cao F, Yin HL, Huang ZJ, Lin ZT, Mao
N, Sun B and Wang G: Ferroptosis: Past, present and future. Cell
Death Dis. 11:882020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li X, Luo JQ, Liao XQ, Zhang S, Yang LF,
Wu T, Wang L, Xu Q, He BS and Guo Z: Allicin inhibits the growth of
HONE-1 and HNE1 human nasopharyngeal carcinoma cells by inducing
ferroptosis. Neoplasma. 71:243–254. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wu N, Zhang X, Fang C, Zhu M, Wang Z, Jian
L, Tan W, Wang Y, Li H, Xu X, et al: Progesterone enhances
niraparib efficacy in ovarian cancer by promoting
palmitoleic-acid-mediated ferroptosis. Research (Wash D C).
7:03712024.PubMed/NCBI
|
|
20
|
Chen M, Tan AH and Li J: Curcumin
represses colorectal cancer cell proliferation by triggering
ferroptosis via PI3K/Akt/mTOR signaling. Nutr Cancer. 75:726–733.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ming T, Lei J, Peng Y, Wang M, Liang Y,
Tang S, Tao Q, Wang M, Tang X, He Z, et al: Curcumin suppresses
colorectal cancer by induction of ferroptosis via regulation of p53
and solute carrier family 7 member 11/glutathione/glutathione
peroxidase 4 signaling axis. Phytother Res. 38:3954–3972. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Miyazaki K, Xu C, Shimada M and Goel A:
Curcumin and andrographis exhibit anti-tumor effects in colorectal
cancer via activation of ferroptosis and dual suppression of
glutathione Peroxidase-4 and ferroptosis suppressor Protein-1.
Pharmaceuticals (Basel). 16:3832023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gustavsson B, Carlsson G, Machover D,
Petrelli N, Roth A, Schmoll HJ, Tveit KM and Gibson F: A review of
the evolution of systemic chemotherapy in the management of
colorectal cancer. Clin Colorectal Cancer. 14:1–10. 2015.PubMed/NCBI
|
|
24
|
Saad ED and Hoff PM: UFT and oral
leucovorin as radiation sensitizers in rectal and other
gastrointestinal malignancies. Cancer Invest. 21:624–629. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mao X, Zhang X, Zheng X, Chen Y, Xuan Z
and Huang P: Curcumin suppresses LGR5(+) colorectal cancer stem
cells by inducing autophagy and via repressing TFAP2A-mediated ECM
pathway. J Nat Med. 75:590–601. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yu H, Xie Y, Zhou Z, Wu Z, Dai X and Xu B:
Curcumin regulates the progression of colorectal cancer via LncRNA
NBR2/AMPK pathway. Technol Cancer Res Treat.
18:15330338198707812019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Güllü N, Smith J, Herrmann P and Stein U:
MACC1-dependent antitumor effect of curcumin in colorectal cancer.
Nutrients. 14:47922022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zheng S, Yin J, Wang B, Ye Q, Huang J,
Liang X, Wu J, Yue H and Zhang T: Polydatin protects against
DSS-induced ulcerative colitis via Nrf2/Slc7a11/Gpx4-dependent
inhibition of ferroptosis signalling activation. Front Pharmacol.
15:15130202025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kumar P, Malhotra P, Ma K, Singla A,
Hedroug O, Saksena S, Dudeja PK, Gill RK and Alrefai WA: SREBP2
mediates the modulation of intestinal NPC1L1 expression by
curcumin. Am J Physiol Gastrointest Liver Physiol. 301:G148–G155.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Guler Y and Ovey IS: Synergic and
comparative effect of 5-fluorouracil and leucoverin on breast and
colon cancer cells through TRPM2 channels. Bratisl Lek Listy.
119:692–700. 2018.PubMed/NCBI
|
|
31
|
Pan P, Zhang Z, Xu Y, Li F, Yang Q and
Liang B: Sarsasapogenin inhibits HCT116 and Caco-2 cell malignancy
and tumor growth in a xenograft mouse model of colorectal cancer by
inactivating MAPK signaling. J Biochem Mol Toxicol. 39:e701892025.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Barrera LN, Johnson IT, Bao Y, Cassidy A
and Belshaw NJ: Colorectal cancer cells Caco-2 and HCT116 resist
epigenetic effects of isothiocyanates and selenium in vitro. Eur J
Nutr. 52:1327–1341. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ferrante M, Grasso A, Salemi R, Libra M,
Tomasello B, Fiore M and Copat C: DNA damage and apoptosis as
in-vitro effect biomarkers of titanium dioxide nanoparticles
(TiO(2)-NPs) and the food additive E171 toxicity in colon cancer
cells: HCT-116 and Caco-2. Int J Environ Res Public Health.
20:20022023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Guo LD, Chen XJ, Hu YH, Yu ZJ, Wang D and
Liu JZ: Curcumin inhibits proliferation and induces apoptosis of
human colorectal cancer cells by activating the mitochondria
apoptotic pathway. Phytother Res. 27:422–430. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Cheng AL, Li J, Vaid AK, Ma BB, Teh C, Ahn
JB, Bello M, Charoentum C, Chen LT, de Lima Lopes G Jr, et al:
Adaptation of international guidelines for metastatic colorectal
cancer: An asian consensus. Clin Colorectal Cancer. 13:145–155.
2014.PubMed/NCBI
|
|
36
|
Grogan L, Sotos GA and Allegra CJ:
Leucovorin modulation of fluorouracil. Oncology (Williston Park).
7:63–72; discussion 75–6. 1993.PubMed/NCBI
|
|
37
|
Morgan RG: Leucovorin enhancement of the
effects of the fluoropyrimidines on thymidylate synthase. Cancer.
63:1008–1012. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ismail NI, Othman I, Abas F, H Lajis N and
Naidu R: Mechanism of apoptosis induced by curcumin in colorectal
cancer. Int J Mol Sci. 20:24542019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Carethers JM and Pham TT: Mutations of
transforming growth factor beta 1 type II receptor, BAX, and
insulin-like growth factor II receptor genes in microsatellite
unstable cell lines. In Vivo. 14:13–20. 2000.PubMed/NCBI
|
|
40
|
Yin J, Wang L, Wang Y, Shen H, Wang X and
Wu L: Curcumin reverses oxaliplatin resistance in human colorectal
cancer via regulation of TGF-β/Smad2/3 signaling pathway. Onco
Targets Ther. 12:3893–3903. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Romanovová M, Jendželovská Z, Barčáková I,
Majerník M, Jendželovský R and Fedoročko P: Pretreatment of cancer
cells with inhibitors of PKCδ, EGFR, and JNK increased
intracellular hypericin content and enhanced the effectiveness of
photodynamic therapy. J Photochem Photobiol B. 268:1131832025.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Hasan AA, Kalinina E, Nuzhina J, Volodina
Y, Shtil A and Tatarskiy V: Potentiation of cisplatin cytotoxicity
in resistant ovarian cancer SKOV3/cisplatin cells by quercetin
pre-treatment. Int J Mol Sci. 24:109602023. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Nadal JC, van Groeningen CJ, Pinedo HM and
Peters GJ: Schedule-dependency of in vivo modulation of
5-fluorouracil by leucovorin and uridine in murine colon carcinoma.
Invest New Drugs. 7:163–172. 1989. View Article : Google Scholar : PubMed/NCBI
|