Introduction
Advances in diagnostics, treatment and patient
longevity have contributed to the increasing incidence of multiple
primary malignant tumors (MPMTs), defined as two or more
histologically distinct malignancies in a single individual
(1). MPMTs are classified as
synchronous or metachronous based on the timing of diagnosis, with
the latter often being associated with a poorer prognosis. First
described in the late 19th century, formal diagnostic criteria for
MPMTs were established by Warren and Gates (2) in 1932 and require each tumor to be: i)
Pathologically malignant, ii) histologically distinct and iii) not
representing metastasis of the other tumor. Malignancies diagnosed
within a 6-month interval are considered synchronous (2). The reported incidence of MPMTs ranges
from 0.52 to 11.7%, with a higher prevalence in men >50 years
old, and the most common combination is adenocarcinoma and squamous
cell carcinoma (3). Established
risk factors include smoking and heavy alcohol intake (4,5). In
addition, patients with MPMT may have a family history of cancer,
while other patients are sporadic and may possess DNA mismatch
repair gene mutations (6). The
diagnosis of MPMTs relies on pathological, histological and
immunohistochemical examinations, although no standardized
treatment guidelines are available at present. Due to the current
lack of standardized treatment guidelines, management is
individualized and based on tumor pathology, stage and patient
tolerance, ranging from radical to palliative intent.
MTSCC of the kidney is an extremely rare malignant
epithelial tumor of the kidney, accounting for <1% of all
primary renal tumors (7).
Recognized as a distinct entity in the 2004 World Health
Organization (WHO) classification (8), MTSCC predominantly affects middle-aged
and elderly patients (mean age, ~58 years) and exhibits a
significant female predominance (male:female ratio, ~1:3) (9). Characteristic histopathology involves
bland spindle cells and tubular structures within a mucinous
stroma, with a typical immunophenotype [positive for cytokeratin
(CK)7, α-methylacyl-CoA racemase and E-cadherin, and usually
negative for high-molecular-weight CKs] (9). Most cases follow an indolent course,
and nephron-sparing surgery is often curative (10). However, a minority of patients
exhibit aggressive features (such as sarcomatoid transformation and
metastasis), which are associated with complex cytogenetic
abnormalities (such as multiple chromosome losses) and specific
gene deletions (such as CDKN2A/2B) (11,12).
Comprehensive pathological evaluation supplemented by
immunohistochemistry and molecular analysis is therefore crucial
for an accurate diagnosis and prognosis.
To the best of our knowledge, the present case
represents the first documented occurrence of metachronous primary
lung adenocarcinoma and renal MTSCC. Although histologically
distinct, potentially shared pathophysiological mechanisms warrant
further consideration and may include: i) Common environmental
carcinogen exposure (for example, tobacco smoke, a known risk
factor for both lung and some renal carcinomas) (13,14);
ii) shared genetic susceptibility, such as underlying germline
mutations in cancer predisposition genes or age-related genomic
instability, which could facilitate the development of independent
primaries (15); and iii) altered
immune surveillance in the context of aging and previous
oncological treatment (16,17). Positioning this case within the
broader literature relating to MPMT emphasizes the unique nature of
the present patient. Unlike more common combinations (such as dual
aerodigestive tract carcinomas) (18), the concurrence of a pulmonary
adenocarcinoma with a rare renal subtype such as MTSCC is
exceptional. This highlights the diverse spectrum of MPMTs,
reinforces the critical need for meticulous pathological workup to
exclude metastasis, and contributes to a more nuanced understanding
of the etiological and clinical landscape of multiple primary
cancers.
Case report
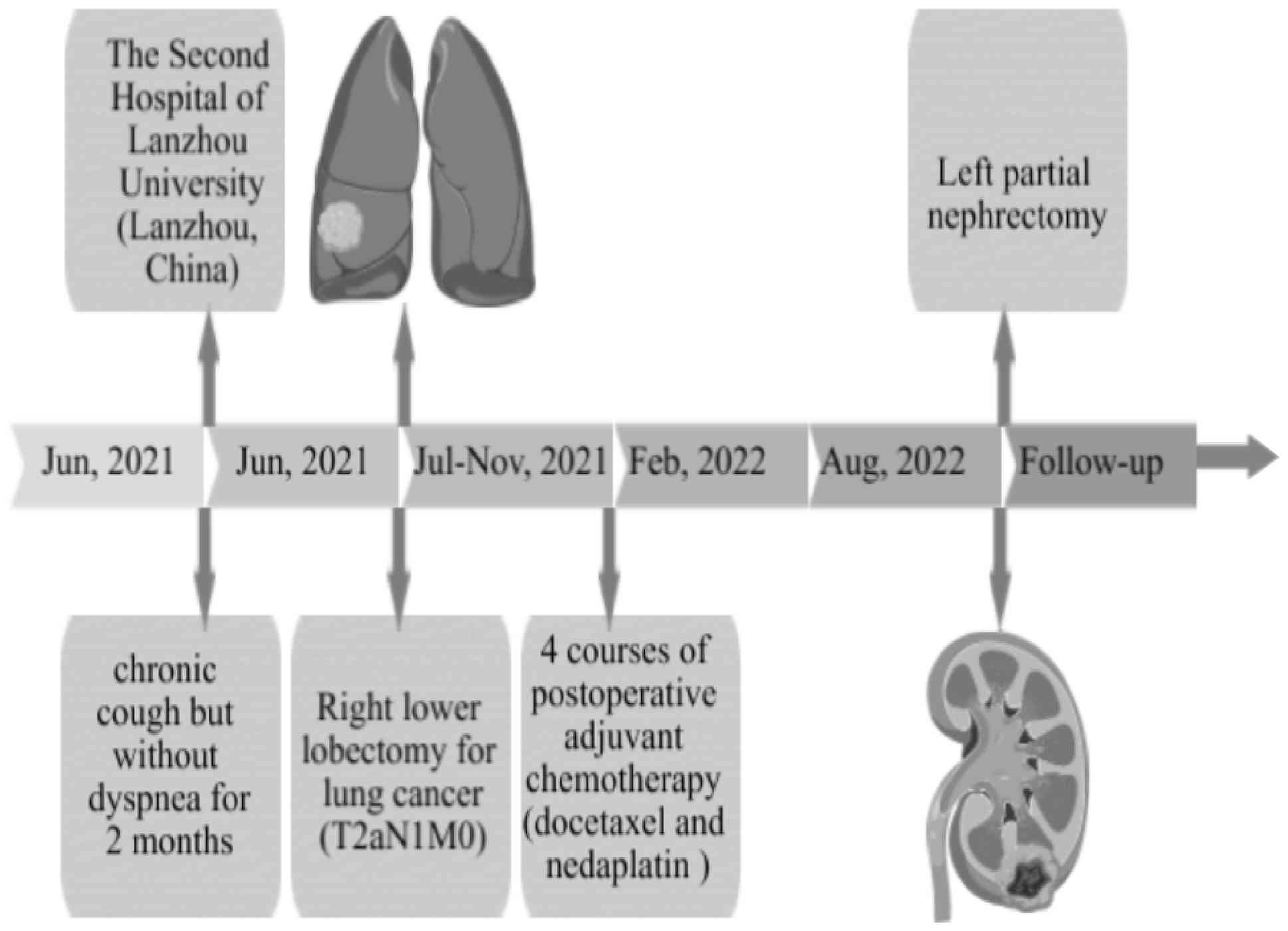
A 69-year-old man first presented to The Second
Hospital of Lanzhou University (Lanzhou, China) in June 2021 with a
chronic cough but without dyspnea for 2 months, and with no history
of pulmonary disease. The patient had smoked 15 cigarettes daily
for 43 years and had not experienced recent weight loss. Throughout
the 2-month period, the patient had maintained a good mental status
and a clear consciousness, with appetite being unaffected and sleep
being undisturbed. Normal bowel movements were noted, with no
symptoms of urinary frequency, urgency, pain or hematuria. A
physical examination indicated normal skin and conjunctiva, clear
lung breath sounds, no tenderness or rebound tenderness in the
abdomen, painless percussion over the kidney area and no lower limb
swelling. The patient had been diagnosed with depression 3 years
previously and had no other surgical or traumatic history. Computed
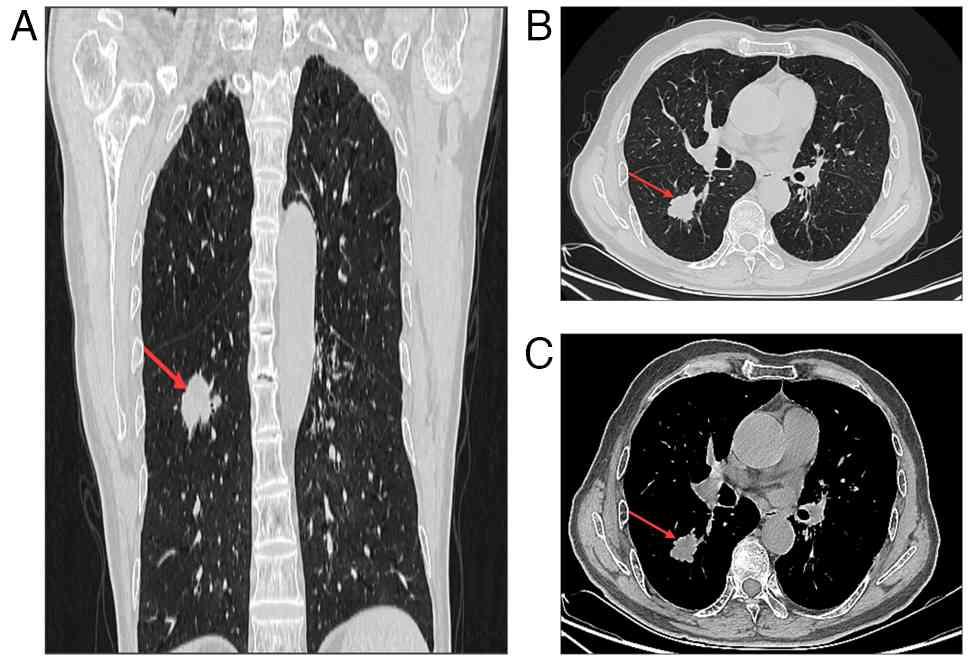
tomography (CT) revealed a mass in the dorsal segment of the lower
lobe of the right lung, measuring ~3.3×2.6×2.5 cm, along with
enlargement of the mediastinal and hilar lymph nodes (Fig. 1). Additional imaging, including
contrast-enhanced abdominal CT, magnetic resonance imaging (MRI) of
the brain and a bone scan, revealed no abnormalities. Based on
these findings, the patient was clinically staged as stage IB
(cT2aN0M0), according to the TNM classification system (19).
Analysis of tumor markers was indicative of primary
NSCLC. Tumor biomarker profiling demonstrated notable increases in
carcinoembryonic antigen (1,021.2 ng/ml; reference value, <4.7
ng/ml) levels, whereas routine hematological, coagulation and
metabolic panels were normal. Subsequently, the patient underwent a
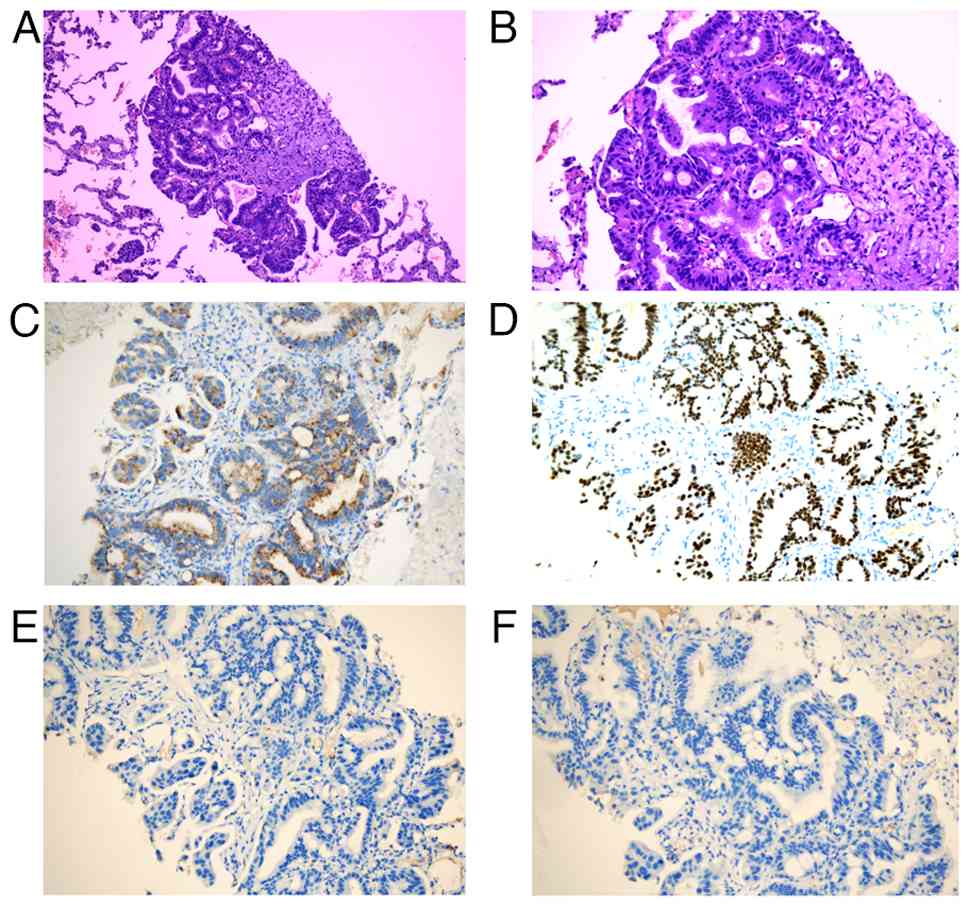
right lower lobectomy. Postoperative pathology identified the tumor
as lung invasive adenocarcinoma (Fig.
2) (20,21); the tumor was predominantly solid and
cribriform, with components of tumor thrombus, micropapillary type
and acinar structures. In addition, >90% of the tumor was
high-grade. The size of the tumor was revised to 3.5×3.0×2.8 cm,
with no invasion of the visceral pleura, or the bronchial and
vascular stumps. There was no lymph node metastasis, with the
exception of node number 12. Immunohistochemistry was positive for
thyroid transcription factor-1 (TTF-1) and NapsinA, but negative
for p40 and CK5/6. The Ki-67 index was 40%. The final postoperative
stage was classified as T2aN1M0, stage IIB.
Tumor tissue was sent for next-generation
sequencing-based targeted genomic profiling (OrigiMed; Zhiben
Medical Technology Co., Ltd.) and revealed a missense mutation in
TP53 exon 6, negative PD-L1 expression (tumor proportion score
<1%) and a tumor mutational burden of 8.15 Mut/MB. The diagnosis
was duly confirmed as adenocarcinoma. According to the 2022 Chinese
Society of Clinical Oncology Guidelines (22), The patient was initiated on
combination chemotherapy with docetaxel (75 mg/m2) and
nedaplatin (100 mg/m2) administered on day 1 of a 21-day
cycle. The patient completed four cycles and tolerated treatment
well.
The plan post-chemotherapy was to perform enhanced
chest and abdominal CT scans every 3 months for 1 year to assess
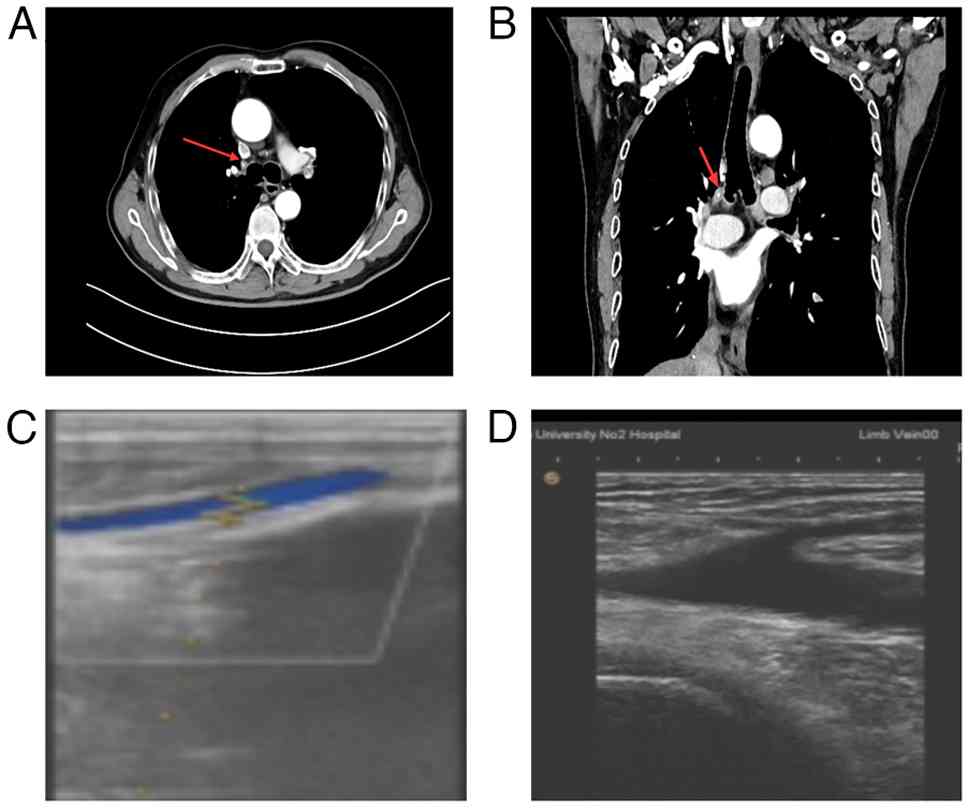
the patient's condition. A CT scan at 3 months post-chemotherapy
revealed a pulmonary artery embolism in the right upper lobe, and
color Doppler ultrasound of the lower extremities revealed a
thrombus in the right calf muscle vein (Fig. 3). Oral rivaroxaban (20 mg once
daily) was administered for anticoagulant therapy. After 3 months
of anticoagulation therapy, the pulmonary embolism had
resolved.
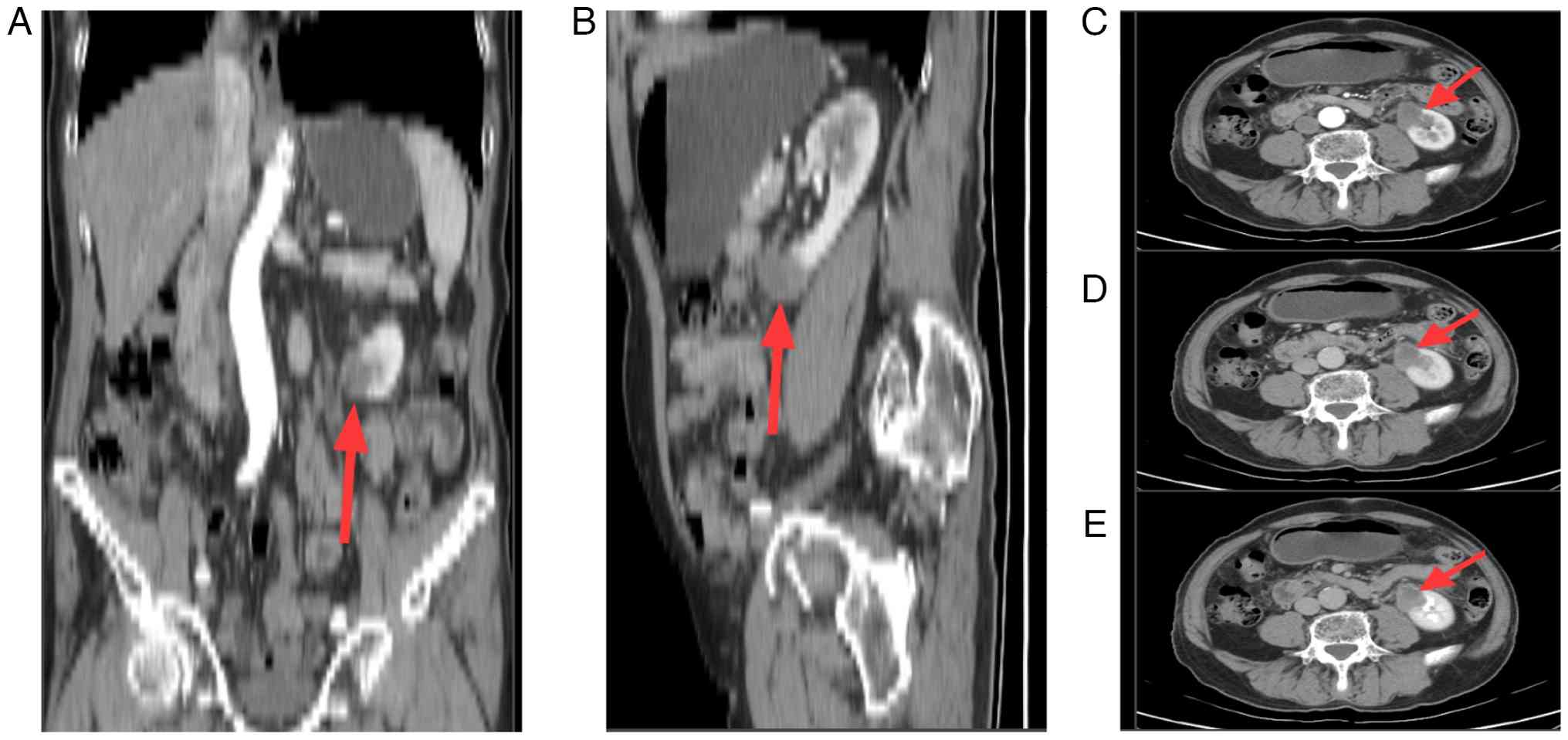
After a disease-free interval of ~9 months, enhanced
abdominal CT scans revealed a round isodense lesion in the lower
pole of the left kidney, mostly protruding beyond the renal
contour, measuring ~2.5×3.0×2.6 cm, with progressive enhancement on
the contrast scan (Fig. 4).
Enhanced chest CT and enhanced brain MRI scans did not reveal any
further metastases. Bone scans were negative. The patient was
asymptomatic, without abdominal discomfort, hematuria or other
urinary system symptoms. It was difficult to distinguish between
incidental renal cell carcinoma and renal metastasis based on
imaging alone, and the patient refused a biopsy of the renal
lesions. Therefore, after multidisciplinary discussion, in a
meeting involving urologic oncologists, medical oncologists,
radiologists and pathologists, surgical treatment was
recommended.
The patient underwent a laparoscopic transperitoneal
partial nephrectomy. The postoperative course was uneventful, and
the patient was discharged 2 days after surgery. Pathological
analysis revealed two pieces of gray-yellow to gray-brown tissue,
measuring 5.5×3.0×2 cm, with a gray-yellow cut surface and a small
amount of attached fat. Microscopically, the tumor was composed of
tightly arranged, small and elongated tubules. Some tumor cells
were spindle shaped, with a lightly stained mucinous stroma. The
tumor cells forming the tubules were small, cuboidal or oval in
shape. No nerves, vascular invasion or intravascular tumor thrombi
were detected. Tumor invasion was not detected in the ipsilateral
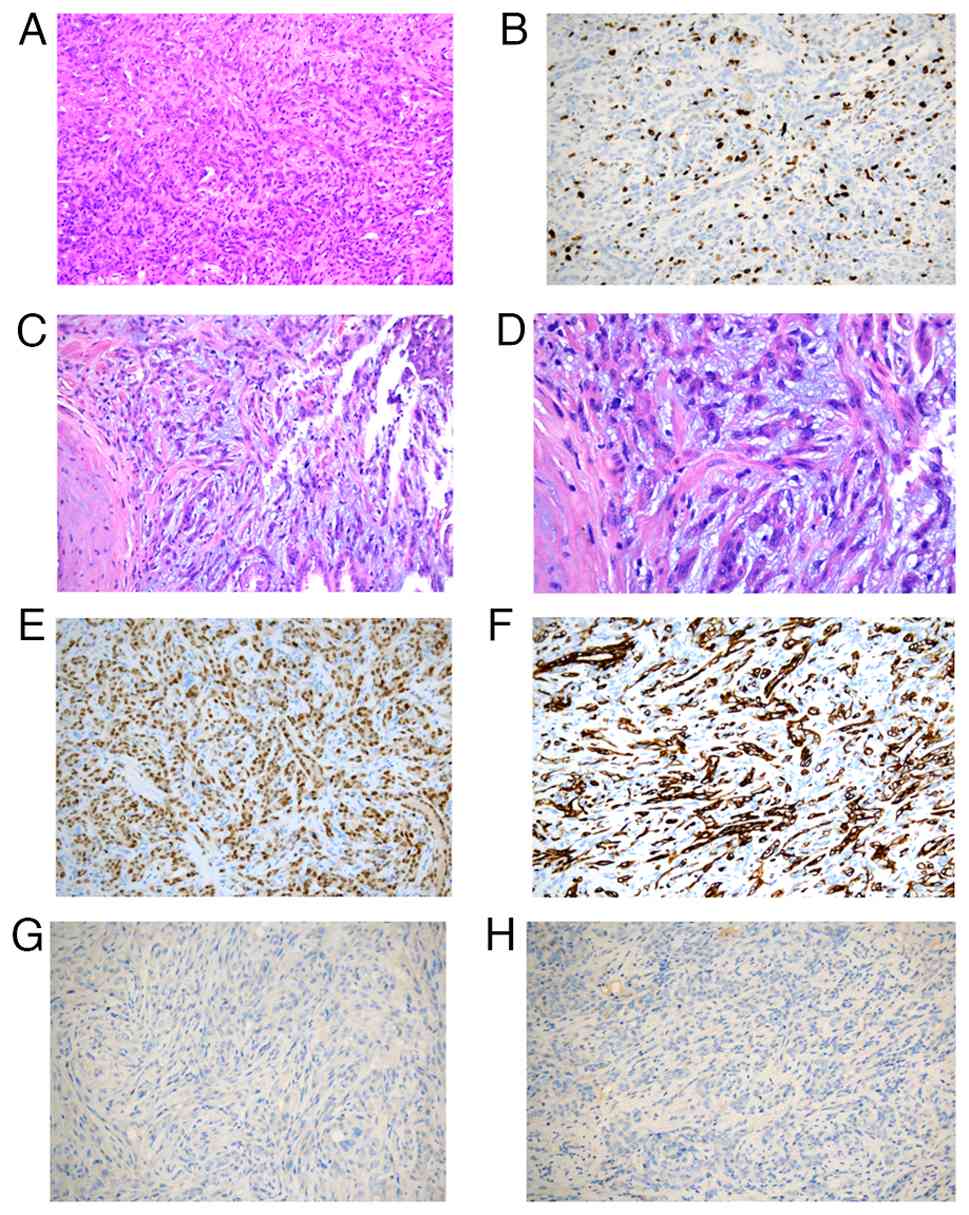
ureter or vascular margins. Immunohistochemistry results revealed
the following results: A Ki-67 labeling index of ~20%, paired box
protein Pax-8(+), CD117(−), CK7(+), napsin A(−) and TTF-1(−). Based
on morphology and immunohistochemistry, the patient was finally
diagnosed with high-grade MTSCC (Fig.
5). The patient declined genetic testing due to financial
reasons. The tumor was classified as T1N0M0, WHO/International
Society of Urological Pathology Grade 3 (23), but without sarcomatoid
transformation. According to the American (UCLA Integrated Staging
System) prognostic system (24,25),
this patient was classified as low progression risk [TNM stage,
T1N0M0; Fuhrman grade (26), 2;
Eastern Cooperative Oncology Group performance status (27), 0] (28). Therefore, the postoperative
monitoring plan involved clinical examinations (physical
examination, tumor markers, creatinine, uric acid and urinalysis)
and chest and abdominal CT scans every 6 months for 3 years, then
once a year for 2 years, and thereafter once every 5 years. After 3
years of follow-up, the patient remains in good condition, with no
local or contralateral recurrence, and no secondary lesions. The
clinical timeline for this patient is summarized in Fig. 6. Written informed consent was
obtained from the patient for the publication of this case report
and any accompanying images. At initial diagnosis, the patient
presented with an elevated CEA level, which normalized after
surgery. The detailed laboratory monitoring during treatment and
follow-up, including tumor marker, uric acid, and creatinine
levels, is summarized in Tables SI
and SII.
Pathological analysis
All postoperative specimens were fixed in 4% neutral
formaldehyde, routinely dehydrated and embedded in paraffin.
Continuous sections 4-µm thick were prepared and stained with
hematoxylin and eosin (HE). Immunohistochemistry employed the SP
method. Primary rat anti-human monoclonal antibodies for CK5/6
(cat. no. ZM-0313; dilution, 1:120), p40 (cat. no. ZM-0472;
dilution, 1:60), Napsin A (cat. no. ZM-0473; dilution, 1:100) and
TTF-1 (cat. no. ZM-0270; dilution, 1:100) were purchased from
Beijing Zhongshan Jinqiao Biotechnology Co., Ltd.
Immunohistochemical procedures strictly followed kit protocols. PBS
served as the primary antibody negative control. TTF-1 and Ki-67
was localized to the nucleus, with positive expression appearing as
yellow-brown granular deposits in the nucleus. NapsinA positive
staining was located in the cytoplasm, appearing as punctate or
granular staining. p40 positive staining was predominantly
cytoplasmic, appearing as yellow or brownish-yellow granules. p63
staining was considered positive only when diffusely strongly
expressed in the nucleus. Cells were graded by percentage as
follows: No positive cells (−); <30% positive cells (+); 30–50%
positive cells (++); and >50% positive cells (+++).
Discussion
The patient in the present case was diagnosed with
metachronous NSCLC and MTSCC. For cancer survivors, detecting a new
mass necessitates differentiating between metastasis and a second
primary malignancy, which directly dictates the strategy to be used
for clinical management. To the best of our knowledge, no previous
cases of these concurrent tumors have been reported in the
literature.
NSCLC is the leading cause of cancer-related death
and represents ~85% of all lung cancer cases (29). MTSCC constitutes <1% of all renal
cell carcinoma (RCC) cases (30).
Although initially classified as a low-grade neoplasm, the
potential of MTSCC for aggressive behavior has now been recognized,
and the term ‘indolent renal carcinoma’ was removed from the 2016
WHO classification (31–34). Histopathological and
immunohistochemical analysis remains the cornerstone for a
definitive diagnosis (14). As
illustrated in Table I, the
systematic application of clinical context, imaging features,
distinctive morphology and lineage-specific immunohistochemistry
profiles is essential to accurately distinguish MTSCC from renal
metastasis and other RCC subtypes.
 | Table I.Comparison between MTSCC and other
relevant differential diagnoses. |
Table I.
Comparison between MTSCC and other
relevant differential diagnoses.
| Feature | Renal metastasis | Primary renal
MTSCC | Primary renal
PRCC |
|---|
| Incidence | Relatively uncommon,
accounting for 2–12% of renal malignancies. Common primaries
include lung, breast and gastric cancers, and melanoma. | Extremely rare,
constituting <1% of RCCs. With <100 cases reported in the
literature. Female predominance (M:F=1:4). | Relatively common,
representing 10–15% of RCCs. The second most common RCC subtype.
Slight male predominance. |
| Key clinical
features | i) Often a history of
a primary malignancy elsewhere; ii) frequently discovered in the
setting of widespread metastatic disease; and iii) may be
asymptomatic or present with hematuria/flank pain. | i) Most are
incidental findings on imaging; and ii) can present with flank pain
or hematuria. | i) Vast majority are
incidental findings; and ii) may present with hematuria, flank pain
or an abdominal mass. |
| Imaging
characteristics (CT) | Non-specific. Common
features include: i) Multiple, bilateral, small (<3 cm)
parenchymal masses; ii) mild to moderate, heterogeneous enhancement
on contrast; and iii) a wedge-shaped, endophytic growth
pattern. | Characteristic
‘progressive enhancement’: i) Non-contrast: Iso- or hypodense; ii)
postcontrast: Mild enhancement in the corticomedullary phase, with
persistent, progressive enhancement in the nephrographic/excretory
phases (‘slow wash-in, slow wash-out’), and less than normal
parenchyma. | Also exhibits
‘progressive enhancement’, often indistinguishable from MTSCC on
imaging. i) Non-contrast may be slightly hyperdense (due to
hemosiderin); and ii) more prone to cystic change and
calcification. |
| Definitive
pathological diagnosis | Morphology identical
to the primary tumor. IHC: Pax-8-negative, positive for primary
site markers (such as TTF-1 for the lungs). | Pathognomonic:
Microscopy shows a triphasic pattern of tightly packed, elongated
tubules + spindle cells + pale myxoid stroma, often blending. IHC
profile: Pax-8+, CK7+, vimentin+,
CAIX−, CD10 (variable; + in the present case). The
present case exhibited all of the aforementioned features, with a
high Ki-67 (~20%), diagnosing high-grade MTSCC. | Pathognomonic: True
papillary structures with fibrovascular cores. Lacks myxoid stroma.
IHC profile: Pax-8+, CK7+, CD10 (typically
strong/diffuse +) and AMACR+. |
| Molecular
features | Genomic profile
matches the primary carcinoma. | Characteristic
multiple chromosomal losses (for example, 1, 4, 6, 8, 9, 13, 14, 15
and 22). Absence of gains in chromosomes 7/17. | Characteristic
trisomy/multisomy of chromosomes 7 and 17, often with loss of
chromosome Y. |
| Primary
treatment | Systemic therapy is
the mainstay (chemotherapy, targeted therapy and immunotherapy),
based on primary tumor type. Surgical resection or SBRT may be
considered for isolated metastases. | Radical/partial
nephrectomy is the only curative modality. For high-grade, locally
advanced disease (pT2bN1 in the present case), multidisciplinary
discussion for adjuvant targeted therapy (such as sunitinib) is
warranted post-operatively. | Radical/partial
nephrectomy. For advanced/metastatic disease, targeted therapies
(such as cabozantinib and sunitinib) or immunocombinations are
used. |
| Prognosis | Generally poor,
dictated by the biology of the primary tumor and metastatic
burden. | Bimodal
distribution: i) Typical/low-grade MTSCC: indolent, excellent
prognosis, near 100% 5-year survival; and ii)
high-grade/sarcomatoid MTSCC: Aggressive, significantly worse
prognosis, prone to metastasis/recurrence. | Depends on subtype
and grade: i) Type I PRCC: Favorable prognosis; and ii) type II or
high-grade PRCC: Poorer prognosis, with higher metastatic potential
than typical MTSCC. |
Beyond morphology, elucidating the molecular
mechanisms underlying such rare metachronous malignancies holds
marked promise. Genomic sequencing could reveal whether these
independent primary cancers share common susceptibility factors
(for example, germline mutations or signatures of environmental
carcinogen exposure) or possess entirely distinct driver profiles.
While such analyses were not pursued in the present case due to
patient preference and resource constraints, a common challenge in
real-world clinical practice, this highlights a critical gap and an
important direction for future research. Establishing collaborative
frameworks and funding pathways to support the molecular
characterization of rare tumor combinations is essential to advance
our understanding of their biology and to identify potential
therapeutic targets.
The rise in MPMT incidence is likely due to the
growing number of cancer survivors, the long-term side effects of
chemotherapy and radiotherapy, and the influences from genetic,
environmental and endocrine factors (35,36).
Tobacco and alcohol use may heighten the risk of multiple
independent malignant foci in mucosal epithelial cells (36). Radiotherapy is also implicated in
the development of MPMTs (37). In
the context of molecular mechanisms, a potential commonality
between lung adenocarcinoma and MTSCC may lie in the dysregulation
of the Hippo signaling pathway. In lung adenocarcinoma, functional
inactivation of the upstream kinases mammalian STE20-like protein
kinase 1/2 is associated with tumor progression and can lead to
aberrant activation of the downstream oncogenic effectors
yes-associated protein (YAP)/transcriptional coactivator with
PDZ-binding motif (38). Notably,
MTSCC is characterized by a distinct molecular alteration, namely,
the frequent occurrence of biallelic loss of core Hippo pathway
tumor suppressor genes, such as protein tyrosine phosphatase
non-receptor type 14, neurofibromin-2 and salvador homolog 1. This
genetic loss similarly results in persistent nuclear accumulation
and activation of YAP1 (39,40).
Therefore, the inactivation of the Hippo pathway and the consequent
dysregulation of its key downstream effectors may serve as a
plausible molecular explanation for the co-occurrence of these two
metachronous malignancies. This observation also provides a
rationale for future exploration of therapeutic strategies
targeting this pathway.
The present case highlights key clinical
implications. First, an integrated diagnostic approach combining
clinical features, imaging and definitive pathology is essential.
Second, the case emphasizes the need for structured, long-term,
cross-organ surveillance in cancer survivors, particularly those
with persistent risk factors. Finally, management should adopt a
multidisciplinary team framework, prioritizing curative surgery
when possible, with adjuvant therapy guided by specific pathology
and the stage of each primary tumor.
In summary, enhancing awareness of multiple primary
malignancies is crucial to avoid misdiagnosis. Distinguishing
between metastasis and an independent primary tumor directly guides
therapeutic strategy and prognosis. Regular follow-up and lifestyle
modifications, such as smoking cessation, remain integral to
comprehensive survivorship care.
In conclusion, the present study reports a rare case
of metachronous primary NSCLC and MTSCC. The case highlights a
critical clinical principle in that, for all cancer survivors, a
new mass should be investigated as a potential second primary
malignancy and not merely presumed as representing metastatic
disease. Accurate differentiation relies upon definitive
histopathological and immunohistochemical evaluation. Finally, the
case emphasizes the necessity of maintaining a high index of
diagnostic suspicion and implementing structured and long-term
surveillance to optimize outcomes in patients with multiple primary
tumors.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by the Natural Science
Foundation of Gansu Province (grant no. 24JRRA1090).
Availability of data and materials
All data generated in the present study are included
in the figures and/or tables of this article.
Authors' contributions
JXZ conceived the study, acquired, analyzed and
interpreted the data, and wrote the manuscript. PFS contributed to
the clinical interpretation and differential diagnosis, and
critically revised the manuscript. WZ performed pathological
evaluations and revised the manuscript as a pathologist. JXZ, PFS,
JBP and WZ contributed to clinical investigation and data
acquisition (dialysis parameters, laboratory and imaging data),
participated in diagnostic discussions, conducted the literature
review, revised the Discussion and critically revised the
manuscript. JXZ and PFS confirm the authenticity of all raw data.
All authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
This case report was approved by the Ethics
Committee of the Second Hospital of Lanzhou University (Lanzhou,
China; approval no. 2024A-803).
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of this case report, including any
potentially identifiable images or data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Matzkin H and Braf Z: Multiple primary
malignant neoplasms in the genitourinary tract: Occurrence and
etiology. J Urol. 142:1–12. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Warren S and Gates O: Multiple primary
malignant tumors. A survey of the literature and statistical study.
Am J Cancer. 16:1358–1414. 1932.
|
|
3
|
Lv M, Zhang X, Shen Y, Wang F and Yang J,
Wang B, Chen Z, Li P, Zhang X, Li S and Yang J: Clinical analysis
and prognosis of synchronous and metachronous multiple primary
malignant tumors. Medicine (Baltimore). 96:e67992017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Vogt A, Schmid S, Heinimann K, Frick H,
Herrmann C, Cerny T and Omlin A: Multiple primary tumours:
Challenges and approaches, a review. ESMO Open. 2:e0001722017.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Irelli A, Sirufo MM, D'Ugo C, Ginaldi L
and De Martinis M: Sex and gender influences on cancer
immunotherapy response. Biomedicines. 8:2322020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kong P, Wu R, Lan Y, He W, Yang C, Yin C,
Yang Q, Jiang C, Xu D and Xia L: Association between
Mismatch-repair genetic variation and the risk of multiple primary
cancers: A meta-analysis. J Cancer. 8:3296–3308. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ordóñez NG and Mackay B: Renal cell
carcinoma with unusual differentiation. Ultrastruct Pathol.
20:27–30. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lopez-Beltran A, Scarpelli M, Montironi R
and Kirkali Z: 2004 WHO classification of the renal tumors of the
adults. Eur Urol. 49:798–805. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ged Y, Chen YB, Knezevic A, Donoghue MTA,
Carlo MI, Lee MI, Feldman MI, Patil MI, Hakimi MI, Russo P, et al:
Mucinous tubular and spindle-cell carcinoma of the kidney: Clinical
features, genomic profiles, and treatment outcomes. Clin Genitourin
Cancer. 17:268–274.e1. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ivey JA III, Cortese C, Baird BA, Thiel DD
and Lyon TD: Mucinous tubular and spindle cell carcinoma of the
kidney with nodal metastasis managed with surgical resection. Eur
Urol Open Sci. 29:10–14. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang C, Cimera RS, Aryeequaye R,
Jayakumaran G, Sarungbam J, Al-Ahmadie HA, Gopalan A, Sirintrapun
SJ, Fine SW, Tickoo SK, et al: Adverse histology, homozygous loss
of CDKN2A/B, and complex genomic alterations in locally
advanced/metastatic renal mucinous tubular and spindle cell
carcinoma. Mod Pathol. 34:445–456. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Trpkov K, Hes O, Williamson SR, Adeniran
AJ, Agaimy A, Alaghehbandan R, Amin MB, Argani P, Chen YB, Cheng L,
et al: New developments in existing WHO entities and evolving
molecular concepts: The Genitourinary Pathology Society (GUPS)
update on renal neoplasia. Mod Pathol. 34:1392–1424. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
He H, He MM, Wang H, Qiu W, Liu L, Long L,
Shen Q, Zhang S, Qin S, Lu Z, et al: In utero and
childhood/adolescence exposure to tobacco smoke, genetic risk, and
lung cancer incidence and mortality in adulthood. Am J Respir Crit
Care Med. 207:173–182. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hou Y, Zhang Q and Ma B: Kidney, prostate,
and bladder cancer Burden attributable to tobacco smoke exposure in
BRICS Countries from 1990 to 2021: A systematic analysis based on
the global Burden of disease study. Healthcare (Basel).
13:30822025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zou M, Deng R, Liu H, Qiu J, Tian P, Shang
J, Zhou J, Li X, Cai L, Wang Y and Gong K: Risk-based screening and
prognostic analysis for second primary malignancies in kidney
cancer patients: A retrospective cohort study based on large-scale
population and Mendelian randomization analysis. Int J Med Sci.
22:4432–4450. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kejamurthy P and Devi KTR: Immune
checkpoint inhibitors and cancer immunotherapy by aptamers: An
overview. Med Oncol. 41:402023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tang C, Hartley GP, Couillault C, Yuan Y,
Lin H, Nicholas C, Srinivasamani A, Dai J, Dumbrava EEI, Fu S, et
al: Preclinical study and parallel phase II trial evaluating
antisense STAT3 oligonucleotide and checkpoint blockade for
advanced pancreatic, non-small cell lung cancer and mismatch
repair-deficient colorectal cancer. BMJ Oncol. 3:e0001332024.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yang XB, Zhang LH, Xue JN, Wang YC, Yang
X, Zhang N, Liu D, Wang YY, Xun ZY, Li YR, et al: High incidence
combination of multiple primary malignant tumors of the digestive
system. World J Gastroenterol. 28:5982–5992. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lim W, Ridge CA, Nicholson AG and
Mirsadraee S: The 8(th) lung cancer TNM classification and clinical
staging system: Review of the changes and clinical implications.
Quant Imaging Med Surg. 8:709–718. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang A, Sun Y, Xu S, He H and Cao D: The
role of a panel of immunohistochemical markers in differential
diagnosis between prhnary lung squamous cell carcinoma and
adenocarcinoma in biopsies. J Diag Pathol. 23:70–73. 2016.(In
Chinese).
|
|
21
|
Mu X and Gao W: Application
immunohistochemical markers such as TTF-1 in the diagnosis of
non-small cell lung adenocarcinoma and squamous cell carcinoma in
different ethnic groups. Chin J Surg Oncol. 17:69–74. 2025.(In
Chinese).
|
|
22
|
Chen P, Liu Y, Wen Y and Zhou C: Non-small
cell lung cancer in China. Cancer Commun (Lond). 42:937–970. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Perrino CM, Cramer HM, Chen S, Idrees MT
and Wu HH: World Health Organization (WHO)/International Society of
Urological Pathology (ISUP) grading in fine-needle aspiration
biopsies of renal masses. Diagn Cytopathol. 46:895–900. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Patard JJ, Kim HL, Lam JS, Dorey FJ,
Pantuck AJ, Zisman A, Ficarra V, Han KR, Cindolo L, De La Taille A,
et al: Use of the University of California Los Angeles integrated
staging system to predict survival in renal cell carcinoma: an
international multicenter study. J Clin Oncol. 22:3316–3322. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zisman A, Pantuck AJ, Dorey F, Said JW,
Shvarts O, Quintana D, Gitlitz BJ, deKernion JB, Figlin RA and
Belldegrun AS: Improved prognostication of renal cell carcinoma
using an integrated staging system. J Clin Oncol. 19:1649–1657.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gudbjartsson T, Hardarson S, Petursdottir
V, Thoroddsen A, Magnusson J and Einarsson GV: Histological
subtyping and nuclear grading of renal cell carcinoma and their
implications for survival: A retrospective nation-wide study of 629
patients. Eur Urol. 48:593–600. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern Cooperative Oncology Group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ljungberg B, Albiges L, Abu-Ghanem Y,
Bensalah K, Dabestani S, Fernández-Pello S, Giles RH, Hofmann F,
Hora M, Kuczyk MA, et al: European association of urology
guidelines on renal cell carcinoma: The 2019 update. Eur Urol.
75:799–810. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Aramini B, Masciale V, Samarelli AV,
Dubini A, Gaudio M, Stella F, Morandi U, Dominici M, De Biasi S,
Gibellini L and Cossarizza A: Phenotypic, functional, and metabolic
heterogeneity of immune cells infiltrating non-small cell lung
cancer. Front Immunol. 13:9591142022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Moch H, Amin MB, Berney DM, Compérat EM,
Gill AJ, Hartmann A, Menon S, Raspollini MR, Rubin MA, Srigley JR,
et al: The 2022 World Health Organization classification of tumours
of the urinary system and male genital organs-part A: Renal,
penile, and testicular tumours. Eur Urol. 82:458–468. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang H, Xie J, Lu C, Zhang D and Jiang J:
Renal mucinous tubular and spindle cell carcinoma: Report of four
cases and literature review. Int J Clin Exp Pathol. 8:3122–3126.
2015.PubMed/NCBI
|
|
32
|
Isono M, Seguchi K, Yamanaka M, Miyai K,
Okubo K and Ito K: Rapid progression of mucinous tubular and
spindle cell carcinoma of the kidney without sarcomatoid changes: A
case report. Urol Case Rep. 31:1011622020.PubMed/NCBI
|
|
33
|
Rakozy C, Schmahl GE, Bogner S and Störkel
S: Low-grade tubular-mucinous renal neoplasms: Morphologic,
immunohistochemical, and genetic features. Mod Pathol.
15:1162–1171. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Geramizadeh B, Salehipour M and Moradi A:
Mucinous tubular and spindle cell carcinoma of kidney: A rare case
report and review of the literature. Indian J Pathol Microbiol.
52:514–516. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018.PubMed/NCBI
|
|
36
|
Oeffinger KC, Baxi SS, Novetsky Friedman D
and Moskowitz CS: Solid tumor second primary neoplasms: Who is at
risk, what can we do? Semin Oncol. 40:676–689. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Berrington de Gonzalez A, Curtis RE, Kry
SF, Gilbert E, Lamart S, Berg CD, Stovall M and Ron E: Proportion
of second cancers attributable to radiotherapy treatment in adults:
A cohort study in the US SEER cancer registries. Lancet Oncol.
12:353–360. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yang S, Xie H, Lin Q, Zhou L, Liu J, Fang
Z, Tang Z, Yuan R, Su J, Li S, et al: EM2, a natural product MST1/2
kinase activator, suppresses non-small cell lung cancer via hippo
pathway activation. Adv Sci (Weinh). 13:e105082026. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ren Q, Wang L, Al-Ahmadie HA, Fine SW,
Gopalan A, Sirintrapun SJ, Tickoo SK, Reuter VE and Chen YB:
Distinct genomic copy number alterations distinguish mucinous
tubular and spindle cell carcinoma of the kidney from papillary
renal cell carcinoma with overlapping histologic features. Am J
Surg Pathol. 42:767–777. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yu FX, Zhao B and Guan KL: Hippo pathway
in organ size control, tissue homeostasis, and cancer. Cell.
163:811–828. 2015. View Article : Google Scholar : PubMed/NCBI
|