Introduction
Malignant mesothelioma (MM) is a rare neoplasm
originating from mesothelial cells that line serosal surfaces,
including the pleura, peritoneum, pericardium and tunica vaginalis
testis (TVT). MM accounts for <1% of all malignancies (1) and is generally associated with a poor
prognosis (2). Although the pleura
is the most commonly affected site, MM primarily arising from the
TVT is exceedingly rare (3). Both
entities share an origin from the embryonic mesoderm (4) and exhibit three main histological
subtypes: Epithelioid, sarcomatoid and biphasic (5,6). As a
phenotypic variant of MM, MMTVT constitutes <1% of all
mesotheliomas (7,8) and represents 0.3–5% of MMs (9). Since its first description in 1957,
<300 cases have been reported globally (10), with only 41 cases documented in the
Chinese literature based on a search of the China National
Knowledge Infrastructure (https://www.cnki.net) and Wanfang (https://www.wanfangdata.com.cn) databases. The
epidemiological characteristics and risk factors of MMTVT remain
poorly defined. Although asbestos exposure is a well-established
risk factor for MM (11), its
association with the pathogenesis of MMTVT has not been
conclusively established (12).
Non-specific clinical presentations, such as hydrocele and scrotal
pain (13), along with the lack of
specific imaging features on computed tomography (CT) or magnetic
resonance imaging (MRI) (14,15),
often lead to diagnostic challenges and suboptimal surgical
management. Radical surgery remains the cornerstone of treatment
(16), yet the efficacy of adjuvant
chemotherapy or radiotherapy remains uncertain (13). Consequently, novel targeted agents
and immunotherapeutic strategies that have demonstrated efficacy in
malignant pleural mesothelioma may offer potential avenues for
optimizing the management of MMTVT.
Case report
A 71-year-old man presented to the General Surgery
Outpatient Clinic of Suzhou Municipal Hospital Affiliated to
Nanjing Medical University (Suzhou, China) in August 2024 with a
complaint of recurrent dull pain in the right lower abdomen for
half a month. The past medical history was unremarkable. A physical
examination revealed mild deep tenderness in the right lower
abdomen without rebound tenderness or guarding. Enlarged lymph
nodes were palpable bilaterally in the inguinal regions.
An ultrasound scan performed subsequently showed an
enlarged right testis with internal linear echoes and bilateral
inguinal lymphadenopathy, more prominent on the right side
(Fig. S1). Non-contrast and
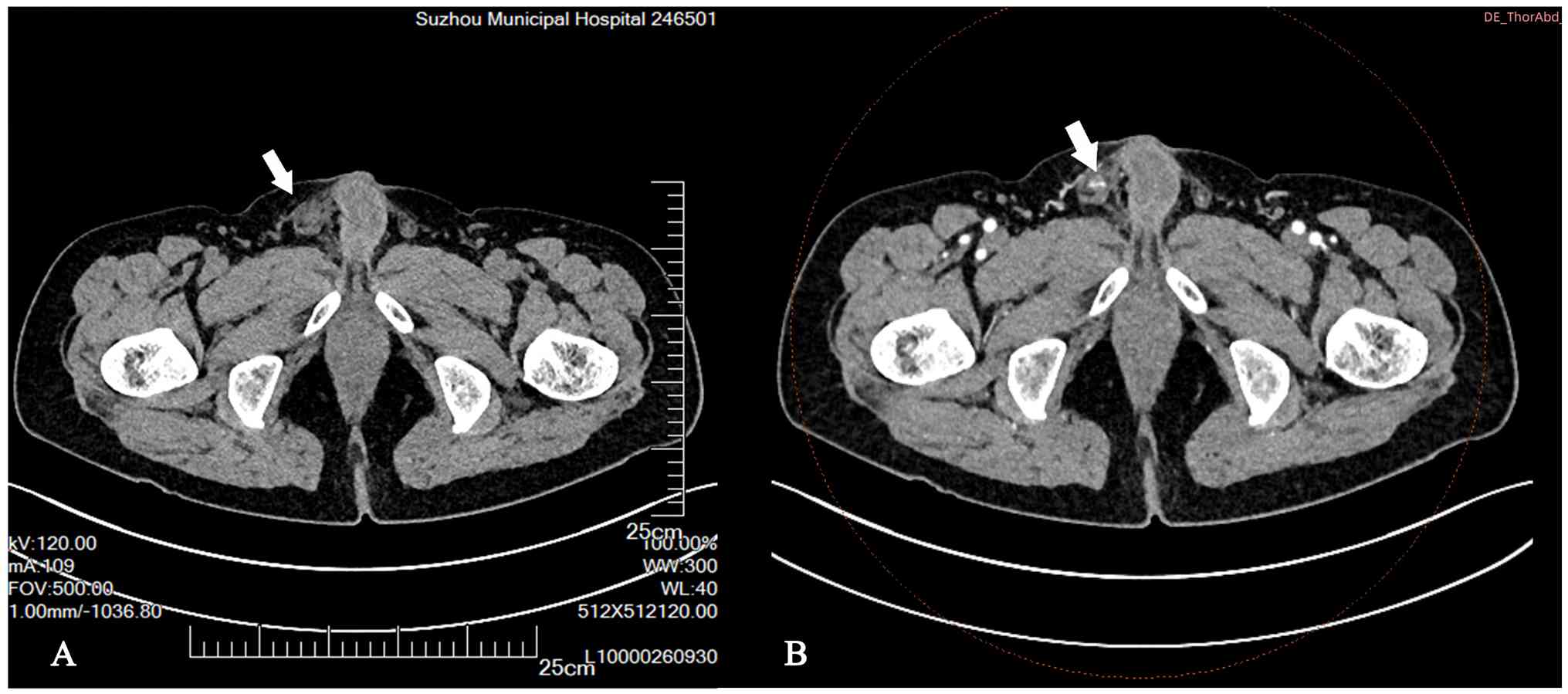
contrast-enhanced CT scans performed on the same day confirmed an
enlarged right testis with heterogeneous enhancement and cystic
changes, along with enlarged lymph nodes adjacent to the right
external iliac artery (Fig. 1A and
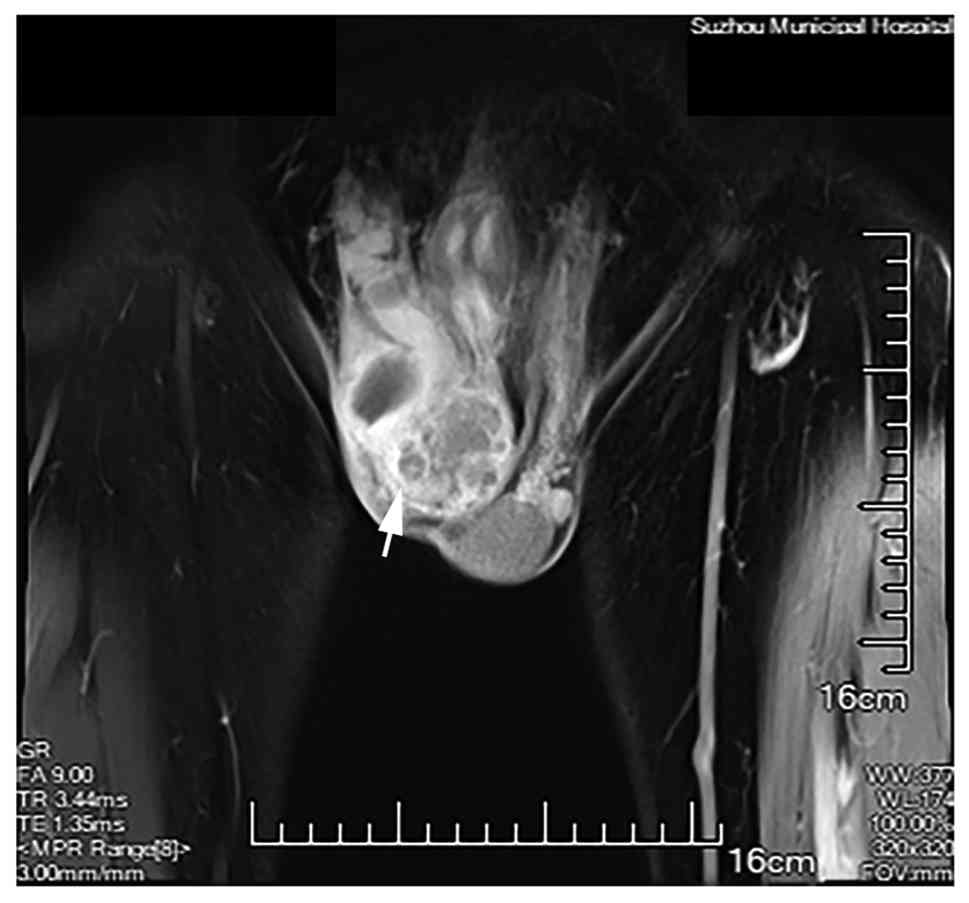
B). Enhanced scrotal MRI 3 days later revealed a lesion in the
right epididymis causing compression of the testis (Fig. 2).
The patient was subsequently transferred to the
Department of Urology of Suzhou Municipal Hospital Affiliated to
Nanjing Medical University. A physical examination showed redness
and swelling of the right scrotum with a slightly elevated local
temperature. The epididymis was enlarged and tender, with poorly
defined borders from the testis. No significant abnormalities were
noted on the left side. A preliminary diagnosis of right
epididymitis was made. A follow-up ultrasound scan indicated
thickening of the right scrotal wall, localized fluid collection
within the right tunica vaginalis cavity containing heterogeneous
internal echoes, and inflammatory changes in the epididymis and
surrounding tissues (Fig. S2).
In September 2024, the patient underwent a right
epididymo-orchidectomy under general anesthesia. Intraoperatively,
significant epididymal enlargement was noted, particularly in the
head of the epididymis, with severe adhesions to the testis. As an
epididymectomy alone was deemed insufficient for complete
resection, a combined epididymo-orchidectomy was performed.
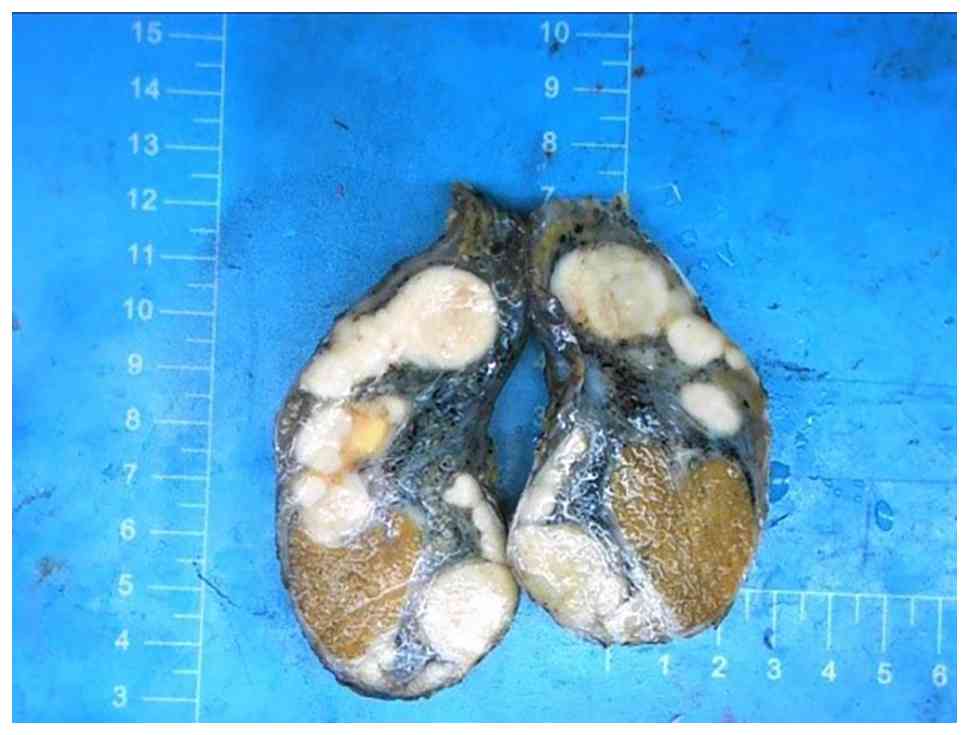
The pathological report described a gross specimen
measuring 9.5×6.0×4.7 cm. The cut surface was grayish-white, solid,
of medium consistency, and exhibited a multinodular and lobulated
appearance (Fig. 3). No tumor
involvement was identified at the surgical margins.
All specimens were fixed within 30 min
post-resection in 4% neutral buffered formalin at room temperature
for 24 h, followed by gross examination, routine dehydration,
clearing and paraffin embedding. From each paraffin block, 4-µm
thick sections were cut using a microtome for hematoxylin and eosin
(H&E) staining. After deparaffinization in xylene and
rehydration through a graded ethanol series, the sections were
stained with hematoxylin at room temperature for 5 min, rinsed in
running tap water, differentiated with 1% acid-alcohol,
counterstained with eosin at room temperature for 3 min, dehydrated
through ethanol, cleared in xylene and finally mounted with a
resinous mounting medium. Histological examination was performed
under an Olympus BX53 upright light microscope. H&E staining
revealed that the tumor was composed of cells with cuboidal,
polygonal or flattened morphology (Fig.
4A; scale bar, 200 µm; magnification, ×20). The cytoplasm was
abundant and predominantly eosinophilic, but occasionally
vacuolated. Nuclei were relatively regular, often featuring
discernible nucleoli. Various morphological patterns were observed,
including tubulopapillary, micropapillary, solid, clear cell,
deciduoid, signet-ring cell-like, rhabdoid and pleomorphic
variants.
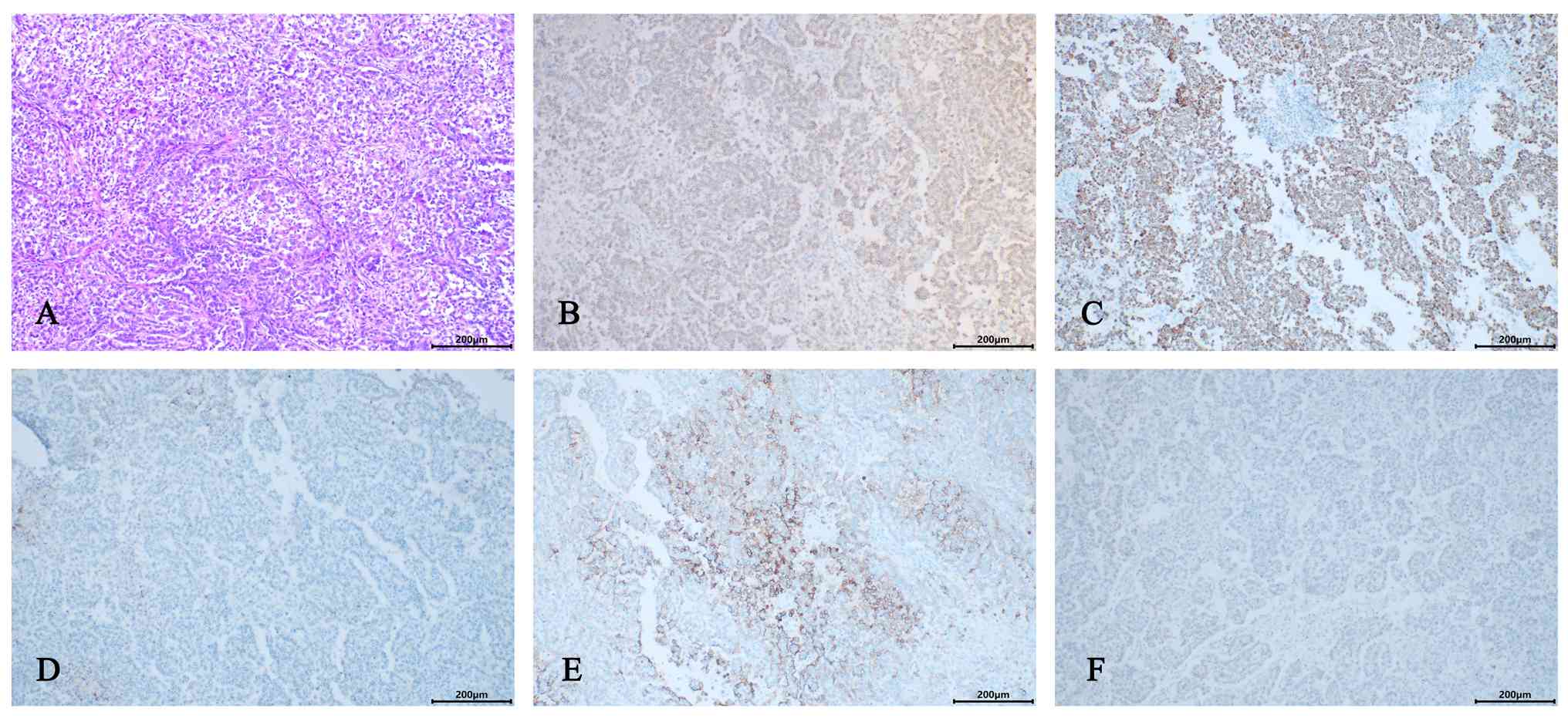 | Figure 4.(A) Tumor cells exhibit cuboidal,
polygonal or flattened morphology, with abundant cytoplasm that is
predominantly eosinophilic but may appear vacuolated. Nuclei show
regular contours and often contain visible nucleoli. Multiple
morphological variants are present, including tubulopapillary,
micropapillary, solid, clear cell, deciduoid, signet-ring cell-like
rhabdoid, and pleomorphic patterns (hematoxylin and eosin;
magnification, ×20). (B) Calretinin immunohistochemical staining of
tumor tissue (magnification, ×10). (C) Cytokeratin 5/6
immunohistochemical staining of tumor tissue (magnification, ×10).
(D) Podoplanin immunohistochemical staining of tumor tissue
(magnification, ×10). (E) Epithelial membrane antigen
immunohistochemical staining of tumor tissue (magnification, ×10).
(F) Wilms' tumor-1 immunohistochemical staining of tumor tissue
(magnification, ×10). |
Immunohistochemical staining was performed on 4-µm
thick sections of formalin-fixed, paraffin-embedded tissue using a
fully automated immunostainer (Roche BenchMark ULTRA PLUS; Roche
Diagnostics GmbH). All staining procedures followed the
standardized protocols of the instrument. Briefly, after
preheating, dewaxing with EZ Prep solution and antigen retrieval
(incubation in Cell Conditioner #1 at 95°C for 8 min), endogenous
peroxidase activity was blocked using the instrument-matched
inhibitor (UV INHIBITOR). Subsequently, the sections were incubated
with specific primary antibodies at 37°C for 32 min. All primary
antibodies were applied at ready-to-use concentrations and
included: Calretinin, cytokeratin (CK)5/6, AE1/AE3, vimentin, CK7,
p53, Ki-67, β-catenin, epithelial membrane antigen (EMA),
podoplanin (D2-40), Wilms' tumor 1 (WT-1), spalt-like transcription
factor 4 (SALL4), placental alkaline phosphatase (PLAP), S-100,
α-inhibin, carcinoembryonic antigen (CEA), CD30, α-fetoprotein
(AFP), CD117, CD99, melanoma markers, P504s (AMACR), GATA binding
protein 3 (GATA3), CK20, EpCAM, programmed death-ligand 1 (PD-L1)
and VEGF (Table I). Following
primary antibody incubation, the sections were sequentially
incubated with a horseradish peroxidase (HRP)-labeled polymer
secondary antibody (UV HRP UNIV MULT) at 36°C for 8 min and the
chromogenic substrate (UV DAB and UV DAB
H2O2) at room temperature for 8 min. The
reaction product was enhanced with copper ions (UV COPPER) to
optimize the signal. Finally, the sections were counterstained with
hematoxylin (Hematoxylin II), blued (Bluing Reagent), washed,
dehydrated, cleared, and mounted with a synthetic resin.
Immunohistochemical results showed positivity for calretinin,
CK5/6, AE1/AE3, vimentin, CK7, p53 (wild-type pattern), Ki-67
(hotspot 35%) and β-catenin (membranous), partial positivity for
EMA, and negativity for D2-40, WT-1, SALL4, PLAP, S-100, α-inhibin,
CEA, CD30, AFP, CD117, CD99, melanoma markers, P504s, GATA3, CK20,
MOC31, PD-L1 (Fig. S3) and VEGF
(Fig. S4) (Figs. 4B-F and S5). Based on morphological features and
immunohistochemical results, a diagnosis of MM (epithelioid type)
was rendered. The patient received no further treatment
postoperatively.
 | Table I.Markers, catalog numbers, clones and
suppliers used in immunohistochemical analysis. |
Table I.
Markers, catalog numbers, clones and
suppliers used in immunohistochemical analysis.
| Protein | Catalog number | Clone | Supplier |
|---|
| Calretinin | GT200904 | Poly | Gene Tech Co.,
Ltd. |
| EMA | GM061302 | E29 | Gene Tech Co.,
Ltd. |
| D2-40 | GM361902 | D2-40 | Gene Tech Co.,
Ltd. |
| CK5/6 | GM723707 | EP24&EP67 | Gene Tech Co.,
Ltd. |
| WT1 | GM356104 | 6F-H2 | Gene Tech Co.,
Ltd. |
| AE1/3 | GM351502 | AE1/3 | Gene Tech Co.,
Ltd. |
| SALL4 | ZM-0393 | 6E3 | Beijing Zhongshan
Jinqiao |
|
|
|
| Biotechnology Co.,
Ltd. |
| Vimentin | GM072504 | V9 | Gene Tech Co.,
Ltd. |
| PLAP | GM719102 | 8A9 | Gene Tech Co.,
Ltd. |
| CK7 | GM701807 | OV-TL 12/30 | Gene Tech Co.,
Ltd. |
| S100 | GT224902 | 4C4.9 | Gene Tech Co.,
Ltd. |
| P53 | GM700107 | DO-7 | Gene Tech Co.,
Ltd. |
| α-inhibin | GT230202 | EP378 | Gene Tech Co.,
Ltd. |
| Ki67 | GT209407 | GM027 | Gene Tech Co.,
Ltd. |
| CEA | GT210802 | COL-1 | Gene Tech Co.,
Ltd. |
| β-catenin | GT211902 | EP35 | Gene Tech Co.,
Ltd. |
| CD30 | GT213902 | JCM182 | Gene Tech Co.,
Ltd. |
| AFP | GA000802 | Poly | Gene Tech Co.,
Ltd. |
| CD117 | ZA-0523 | EP10 | Beijing Zhongshan
Jinqiao |
|
|
|
| Biotechnology Co.,
Ltd. |
| CD79 | GT212302 | EP8 | Gene Tech Co.,
Ltd. |
| P504S | GT200102 | 13H4 | Gene Tech Co.,
Ltd. |
| GATA3 | ZA-0661 | OTIR4F2 | Beijing Zhongshan
Jinqiao |
|
|
|
| Biotechnology Co.,
Ltd. |
| CK20 | GT204202 | Ks208 | Gene Tech Co.,
Ltd. |
| EPCAM | GM080402 | MOC-31 | Gene Tech Co.,
Ltd. |
| PD-L1 | GT228004 | ZR3 | Gene Tech Co.,
Ltd. |
| VEGF | GT217004 | VG1 | Gene Tech Co.,
Ltd. |
| GPC-3 | GT206802 | 1G12 | Gene Tech Co.,
Ltd. |
| Oct3-4 | GT207202 | C10 | Gene Tech Co.,
Ltd. |
| SOX-2 | ZA-0571 | EP103 | Beijing Zhongshan
Jinqiao |
|
|
|
| Biotechnology Co.,
Ltd. |
| AR | GM356202 | EP120 | Gene Tech Co.,
Ltd. |
| P40 | GT233807 | GR006 | Gene Tech Co.,
Ltd. |
| Ber-Ep4 | ZM-0099 | Ber-Ep4 | Beijing Zhongshan
Jinqiao |
|
|
|
| Biotechnology Co.,
Ltd. |
| P16 | GT233002 | GM501 | Gene Tech Co.,
Ltd. |
| Syn | GT206504 | SP11 | Gene Tech Co.,
Ltd. |
| CgA | GT211404 | GM306 | Gene Tech Co.,
Ltd. |
| CD56 | GT200504 | 123C3 | Gene Tech Co.,
Ltd. |
The patient was readmitted in April 2025 due to the
presence of a right spermatic cord mass for 5 days, accompanied by
localized fever and progressive enlargement over the preceding day.
No abdominal pain, nausea, vomiting, hematuria, dysuria, urinary
frequency or urgency was reported. Laboratory investigations
revealed a mildly elevated cytokeratin fragment CYFRA 21-1 level of
7.53 ng/ml (normal range, <3.3 ng/ml), while other tumor
markers, including AFP (1.14 ng/ml; normal range, <20 ng/ml),
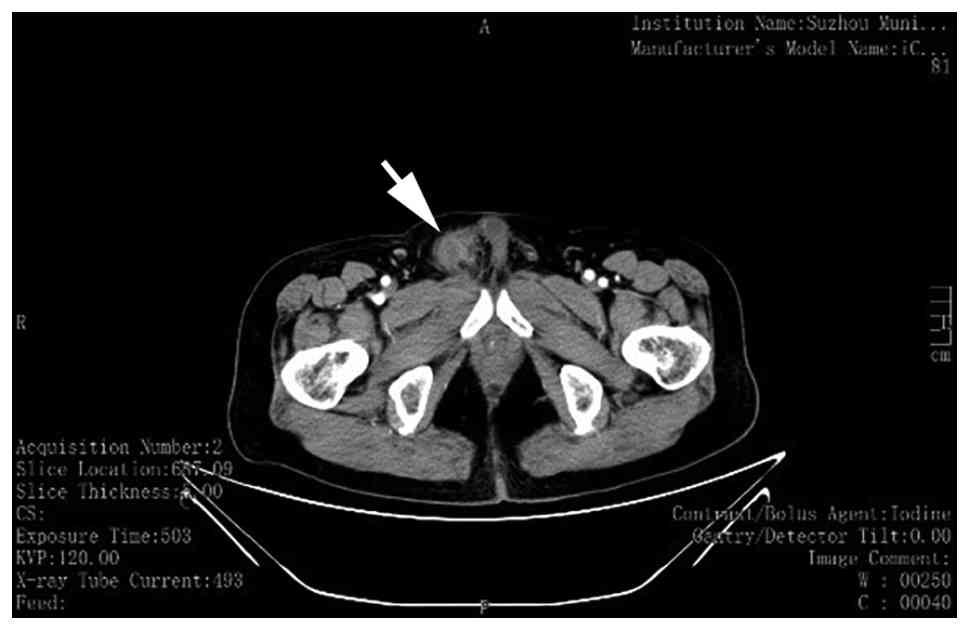
were within normal limits. An abdominal and pelvic non-contrast and
contrast-enhanced CT scan performed 3 days later showed
post-operative changes in the right testicular region and a
soft-tissue density within the right spermatic cord, suggestive of
recurrence. Additionally, dense opacities were noted in both iliac
bones (Fig. 5).
After another 2 days, the patient underwent a
laparoscopic right retroperitoneal lymph node dissection combined
with a resection of a right inguinal mass under general anesthesia.
Intraoperatively, a hard mass ~3 cm in diameter was identified at
the distal stump of the right spermatic cord. The proximal
spermatic cord was transected near the peritoneum, and the mass was
dissected along its margins. Lymphatic dissection commenced over
the surface of the right common iliac artery, extending superiorly
to the origin of the right renal artery from the abdominal aorta.
The lymphatic tissues overlying the sheath of the common iliac vein
were opened and dissected towards the inferior vena cava (IVC). A
thorough lymphadenectomy was performed in the interaortocaval
region and between the right ureter and the IVC, with ligation of
the internal spermatic vein. Finally, lymph nodes surrounding the
right external iliac vessels were dissected. Pathology results
indicated paratesticular mesothelioma at the right spermatic cord
stump, with areas of necrotic change. The resection margins were
free of tumor involvement. Metastatic tumor was identified in the
lymph nodes as follows: Right spermatic cord stump adipose tissue
(0/10), obturator (0/9), interaortocaval (1/6), right internal
inguinal ring (2/2) and paracaval (1/6). Immunohistochemical
analysis of the specimens was performed using the previously
described protocol. Details regarding each marker, including clone
numbers and suppliers are summarized in Table I. The results demonstrated
positivity for AE1/AE3, EMA, calretinin, D2-40, CK7, CK5/6, WT1
(focal) and β-catenin (membranous), along with a Ki-67
proliferation index of 40%. Stains for inhibin-α, CD117, CD30,
glypican-3, octamer binding transcription factor 3–4, SALL-4, SOX2,
PLAP, androgen receptor, p40, Ber-Ep4, p16, synaptophysin,
chromogranin A and CD56 were negative, while desmin showed focal
positivity (Fig. S6).
The patient subsequently received six cycles of
first-line AP regimen chemotherapy (600 mg/m2 pemetrexed
on day 1 + 30 mg/m2 cisplatin on days 1–3), administered
every 21 days between May and September 2025. Prophylactic
administration of varying doses of efbemalenograstim was
implemented following each cycle to maintain neutrophil counts, and
the patient was discharged after each session without reporting any
notable treatment-related discomfort. In June 2025, the patient was
re-admitted due to the emergence of an inguinal mass. Surgical
excision was performed the following day, during which the mass was
noted to be soft in consistency and well-circumscribed.
Histopathological examination of the resected specimen revealed
fibroconnective tissue with features of microvascular
proliferation, inflammatory cell infiltration, fat necrosis and a
multinucleated giant cell reaction, consistent with a
fibroinflammatory process rather than tumor recurrence. Restaging
CT scans performed after the 2nd (Fig.
S7) and 4th cycles of chemotherapy demonstrated stable disease,
indicating no evidence of progression. A follow-up
contrast-enhanced MRI study conducted in October 2025 confirmed the
absence of local recurrence or distant metastasis.
The patient commenced adjuvant radiotherapy in
October 2025. The target volume encompassed the pelvic regional
lymphatic drainage basin plus the right epididymis and spermatic
cord region, planned for a total dose of 45 Gy in 25 fractions.
However, after receiving 15 fractions (by November 2025), the
treatment course was interrupted due to the development of
pancytopenia on routine blood testing, accompanied by
patient-reported epigastric discomfort and erythema in the right
inguinal region. Radiotherapy was consequently suspended, and
appropriate supportive care was initiated. The patient has been
scheduled for regular follow-up visits every 3 months thereafter.
At the most recent assessment, the patient remains well with no
complaints of discomfort.
Discussion
Testicular mesothelioma typically occurs in men aged
55 to 75 years (17). The median
age at diagnosis is 62 years [interquartile range (IQR), 44–73
years] (13) and incidence
increases with age, being 18.6 times higher in men >80 years
compared with those <50 years (7). While asbestos exposure is strongly
associated with malignant pleural mesothelioma, accounting for up
to 80% of cases (2), its role in
the pathogenesis of testicular mesothelioma remains unclear
(18). In a review of 223 cases,
Bisceglia et al (12) found
that only 30–40% were associated with asbestos exposure. Similarly,
the patient in the present report, who worked in wool carpet
production, had no history of asbestos exposure.
Patients with testicular mesothelioma have a
generally poor prognosis that strongly correlates with the presence
of metastasis. Huang et al (19) reported a median overall survival
(OS) time of 71.5 months for 4 patients without metastasis. By
contrast, Grogg et al (13)
found that among 84 patients with metastatic disease, the median
survival time was only 18 months (IQR, 7–43 months), underscoring
the critical importance of early detection and treatment.
Furthermore, the overall recurrence rate (including both local
recurrence and widespread metastasis) is 52.5%, with >60% of
recurrences occurring within the first 2 years of follow-up
(20).
The majority of patients with testicular
mesothelioma are incidentally identified during hydrocelectomy
(21). The majority of patients
present with a painless testicular mass/swelling or hydrocele
(244/259; 94%), followed by scrotal pain (40/259; 15%), which may
be accompanied by orchitis or epididymitis (13,22).
Due to these non-specific manifestations, most cases are not
correctly diagnosed preoperatively (23). Common clinical misdiagnoses include
hydrocele, testicular tumors and epididymitis (24). Additionally, routine serum tumor
markers (e.g., AFP, β-human chorionic gonadotropin and lactate
dehydrogenase) are typically normal. However, positive PD-L1
expression has been reported (19),
suggesting a potential role for immunotherapy. By contrast, in the
present case, immunohistochemistry of the primary tumor showed
negative staining for PD-L1, indicating that immune checkpoint
blockade may not be a suitable therapeutic option for this specific
patient.
A preoperative diagnosis of MMTVT based on imaging
findings is challenging. Ultrasound can be used for the initial
screening. In the epithelioid subtype, ultrasonography may reveal a
solid lesion in the head of the epididymis and hypoechoic fluid
collection within the scrotum, containing fine floating internal
echoes (25). The tunica vaginalis
appears irregularly thickened, with multiple nodular protrusions of
varying sizes demonstrating high or slightly hyperechoic signals on
its wall, corresponding to intra-tunical deposits (26,27).
Doppler evaluation may show scant (28) or abundant (23) internal blood flow, which may be
associated with the papillary nodules on the parietal layer of the
tunica vaginalis seen in pathological specimens. CT offers low
specificity for diagnosing this condition (15), but is valuable for assessing
metastasis and staging (29). MRI
findings are also non-specific (14), typically demonstrating a
heterogeneous intratesticular mass that may contain partially
calcified tumor tissue (30). On
contrast-enhanced scans, the tumor often shows marked heterogeneous
enhancement. The presence of a hydrocele with enhancing wall
nodules, particularly in a hypervascular lesion, should raise
suspicion for MMTVT (31).
Mesothelial tumors are categorized into benign or
potentially premalignant lesions and malignant mesotheliomas. The
former group includes adenomatoid tumors, well-differentiated
papillary mesothelial tumors and mesothelioma in situ. MM is
further subdivided into three principal subtypes: Epithelioid,
sarcomatoid and biphasic (32). In
a review of 275 cases of MTVT reported up to January 2019 (13), 227 out of 275 (83%) cases were
classified as MM. The histopathological distribution among these
malignant cases was as follows: Epithelioid (130/227; 57%),
sarcomatoid (4/227; 2%), biphasic (53/227; 23%) and unspecified
subtype (40/227; 18%).
The immunohistochemical profile of MMTVT is similar
to that of other mesotheliomas (33). Tumor cells typically show positive
reactivity for calretinin, EMA, thrombomodulin, CK7, WT-1 and
D2-40, while expression of CK5/6 can be variable (34). Given that the morphological features
of epithelioid mesothelioma on H&E staining closely resemble
those of adenocarcinoma, immunohistochemical studies are essential
for the differential diagnosis. Among the various markers,
calretinin is considered the most reliable for distinguishing
mesothelioma from adenocarcinoma, as it is consistently negative in
adenocarcinomas. This marker demonstrates both high sensitivity and
specificity for mesothelioma (21).
Notably, D2-40 and WT1 constitute pivotal diagnostic markers
(35,36), with D2-40 demonstrating superior
sensitivity in confirming epithelioid mesothelioma (37). In the present patient,
immunohistochemical analysis of the primary tumor specimen showed
negativity for both D2-40 and WT-1. It is noteworthy that
additional sampling and re-testing from different areas of the same
primary specimen consistently yielded negative results for these
markers (Figs. S8 and 9). By contrast, the recurrent lesion
exhibited a divergent immunoprofile, with positive staining for
D2-40 and focal positivity for WT1. This discrepancy, wherein the
primary tumor cells lacked expression of D2-40 and WT-1 while the
recurrence acquired it, may be attributable to one or a combination
of the following factors: i) Intratumoral heterogeneity in the
expression of D2-40 and WT-1 within this malignant mesothelioma;
ii) selective survival and proliferation of residual tumor cells
post-surgery that were double-positive for D2-40 and WT-1; iii)
phenotypic transformation of tumor cells during the recurrence
process; or iv) technical artifacts such as sampling variation or
other unforeseen factors. This phenomenon finds support in the
literature, as Wang and Liu (38)
similarly reported a case of epithelioid testicular MM that was
D2-40 negative, and Yang et al (39) documented a patient with epithelioid
testicular MM exhibiting WT-1 negativity.
In MMTVT, electron microscopic examination reveals
that tumor cells are interconnected by desmosomes and junctional
complexes, forming luminal spaces lined by long, slender microvilli
with a characteristic length-to-diameter ratio typically >10
(9). Yuan et al (40) observed similar ultrastructural
features in MMTVT tumor cells and noted abundant cytoplasmic
intermediate filaments, including tonofilaments, along with a
moderate amount of glycogen within the cytoplasm. The presence of
these electron microscopic features serves as a definitive
diagnostic criterion, playing a pivotal role in the pathological
distinction of MMTVT from histological mimics, such as
adenocarcinoma.
For patients with MMTVT, the most common surgical
indications are a rapidly enlarging hydrocele or suspicion of
testicular tumor (20). For
localized MMTVT, radical inguinal orchidectomy is the treatment of
choice, while inguinal lymph node dissection is warranted when
signs of lymph node metastasis are present (41). For advanced or recurrent disease,
radical resection combined with adjuvant chemotherapy and/or
radiotherapy is recommended (23).
Primary radical inguinal orchidectomy with partial scrotal
resection and retroperitoneal lymph node dissection has also been
proposed (13,23). It is noteworthy that the inguinal
approach is preferred as it allows early control of the testicular
vascular and lymphatic supply, effectively reducing the risk of
tumor dissemination via the blood and lymphatics during surgery,
thereby potentially lowering the local recurrence rate (42). Furthermore, cisplatin and
doxorubicin have been suggested for chemotherapy (43). In a previous study focusing on MMTVT
patients who underwent lymph node dissection, of the 9 patients who
received adjuvant chemotherapy or chemoradiotherapy, 5 (56%)
developed local and/or metastatic recurrence. Similarly, among 11
patients who underwent radiotherapy targeting the scrotum, as well
as the retroperitoneal and inguinal lymph nodes (maximum dose
55–60.5 Gy), 6 (55%) experienced recurrence, suggesting that the
efficacy achieved with radiotherapy may be comparable to that of
chemotherapy (13).
Since 2004, platinum-pemetrexed chemotherapy has
remained the first-line standard for unresectable mesothelioma,
without approved second- or third-line regimens (44). Bevacizumab targeting vascular
endothelial growth factor combined with cisplatin-pemetrexed
demonstrated significant median overall survival time improvements
in the phase III MAPS trial for malignant pleural mesothelioma when
compared with platinum-pemetrexed alone (45). Leveraging a shared molecular
pathogenesis, pleural mesothelioma therapies may offer clinical
utility for MMTVT. A phase II trial evaluating amatuximab
(anti-mesothelin monoclonal antibody) with cisplatin-pemetrexed in
unresectable malignant pleural mesothelioma achieved a median OS
time of 14.8 months, surpassing historical controls (13.3 months)
(46). The phase III CONFIRM trial
established the superiority of nivolumab over a placebo in patients
with recurrent mesothelioma, demonstrating improved
progression-free survival (median PFS, 3.0 vs. 1.8 months; HR,
0.67; P=0.0012) and OS (median, 10.2 vs. 6.9 months; HR, 0.69;
P=0.0090) times (47). Furthermore,
the CheckMate 743 trial demonstrated that nivolumab plus
ipilimumab, compared with platinum-pemetrexed chemotherapy,
significantly prolonged overall survival time (median OS time, 18.1
vs. 14.1 months) and resulted in a higher 2-year OS rate (41 vs.
27%), subsequently leading to approvals by the US Food and Drug
Administration and the European Medicines Agency (48). Mishra et al (49) reported a case of metastatic
testicular mesothelioma in which the patient underwent a left
orchiectomy followed by immunotherapy with ipilimumab and
nivolumab. After 6 months of treatment, the patient exhibited a
partial response, with reduction in size of known pleural nodules
and effusion. In the present case, however, immunohistochemistry of
the primary tumor showed negative staining for both PD-L1 and VEGF,
suggesting a potentially lower likelihood of clinical benefit from
corresponding targeted antibodies. Notably, in rare tumors,
clinical trials have reported efficacy of immunotherapy in certain
subtypes, including biliary tract cancer, neuroendocrine tumors,
carcinoma of unknown primary and pancreatic acinar carcinoma
(50–54). Collectively, these findings indicate
that immunotherapy holds promise in the treatment of malignant
testicular mesothelioma.
The present report shares the inherent limitations
common to single-case descriptions: i) The small sample size
precludes statistical analysis and limits the ability to draw
definitive conclusions regarding treatment efficacy or prognostic
factors. ii) The surgical, chemotherapeutic and radiotherapy
regimens were tailored to this specific patient and may not
represent an optimized or standardized strategy. iii) The follow-up
period in this case is relatively short, and long-term outcomes
remain to be observed. Furthermore, this study has the following
additional shortcomings: i) The proposed treatment suggestions lack
supporting mechanistic data, comparative genomic evidence or
specific treatment response information from this patient. ii)
There is an absence of further ultrastructural studies. iii) The
study lacks key ancillary tests that are now crucial in
mesothelioma diagnosis, such as BAP1 immunohistochemistry (55), assessment of methylthioadenosine
phosphorylase deletion (56),
cyclin dependent kinase inhibitor 2A fluorescence in situ
hybridization (57) or
next-generation sequencing (58).
This, to some extent, limits the referential value of this case
within the modern diagnostic and classificatory framework for
mesothelioma. Future studies could address these limitations as
potential research directions.
In conclusion, MMTVT is an extremely rare and highly
aggressive neoplasm characterized by non-specific clinical
presentations, with a definitive diagnosis relying on a
pathological examination. In suspected cases, a comprehensive
imaging evaluation is essential for the differential diagnosis and
the detection of metastasis. The disease is prone to local invasion
and distant spread, and patients generally have a short overall
survival time. Therefore, an early diagnosis and radical surgery
via an inguinal approach are critical. When lymph node metastasis
is identified, lymph node dissection should be performed, and
postoperative adjuvant chemotherapy or radiotherapy should be
considered to improve outcomes. Meanwhile, novel targeted
therapeutic strategies may hold potential benefit. Furthermore,
long-term and regular follow-up surveillance is necessary for these
patients.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
Funding was provided by Suzhou Clinical Medicine Center for
Urological Diseases (grant no. Szlcyxzx202106).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YCL designed the study, drafted the initial
manuscript, collated research data, created original figures and
flowcharts, and revised key academic content. JJM assisted in
editing the manuscript and contributed to the examination and
analysis of pathology slides. ZRF and LH were responsible for the
selection, critical evaluation and analytical annotation of
clinical imaging data. ZZ played a key role in the examination and
analysis of pathology slides. JPD and JJX provided administrative
support and contributed to the study design. YCQ, XHS, and JQH
analyzed patient data and assisted in editing the discussion
section. JJX performed the surgical procedure on the patient. YCL,
JJM, ZRF, ZZ, LH, JPD, XHS, JQH, YCQ and JJX confirm the
authenticity of all the raw data. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent for publication was
obtained directly from the patient.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, AI tools were
used to improve the readability and language of the manuscript or
to generate images, and subsequently, the authors revised and
edited the content produced by the AI tools as necessary, taking
full responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Moore AJ, Parker RJ and Wiggins J:
Malignant mesothelioma. Orphanet J Rare Dis. 3:342008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Perrino M, De Vincenzo F, Cordua N, Borea
F, Aliprandi M, Santoro A and Zucali PA: Immunotherapy with immune
checkpoint inhibitors and predictive biomarkers in malignant
mesothelioma: Work still in progress. Front Immunol.
14:11215572023. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hung YP and Chirieac LR: Novel insights
and recent discoveries on the genetics and pathogenesis of
malignant mesothelioma. J Thorac Dis. 10:1314–1317. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pechriggl E, Blumer M, Tubbs RS, Olewnik
Ł, Konschake M, Fortélny R, Stofferin H, Honis HR, Quinones S,
Maranillo E and Sanudo J: Embryology of the abdominal wall and
associated Malformations-A review. Front Surg. 9:8918962022.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tsai YC, Chen HL, Lee TH, Chang HM, Wu KL,
Chuang CH, Chang YC, Tu YK, Hung JY, Yang CJ and Chong IW: Salvage
therapy for relapsed malignant pleural mesothelioma: A systematic
review and network Meta-analysis. Cancers (Basel). 14:1822021.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lettieri S, Bortolotto C, Agustoni F,
Lococo F, Lancia A, Comoli P, Corsico AG and Stella GM: The
evolving landscape of the molecular epidemiology of malignant
pleural mesothelioma. J Clin Med. 10:10342021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mezei G, Chang ET, Mowat FS and Moolgavkar
SH: Epidemiology of mesothelioma of the pericardium and tunica
vaginalis testis. Ann Epidemiol. 27:348–359.e11. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Marinaccio A, Binazzi A, Di Marzio D,
Scarselli A, Verardo M, Mirabelli D, Gennaro V, Mensi C, Merler E,
De Zotti R, et al: Incidence of extrapleural malignant mesothelioma
and asbestos exposure, from the Italian national register. Occup
Environ Med. 67:760–765. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chekol SS and Sun CC: Malignant
mesothelioma of the tunica vaginalis testis: Diagnostic studies and
differential diagnosis. Arch Pathol Lab Med. 136:113–117. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang N, Fu N, Peng S and Luo X: Malignant
mesothelioma of the tunica vaginalis testis: A case report and
literature review. Mol Clin Oncol. 7:1053–1056. 2017.PubMed/NCBI
|
|
11
|
Mazurek JM, Syamlal G, Wood JM, Hendricks
SA and Weston A: Malignant mesothelioma Mortality-united states,
1999–2015. MMWR Morb Mortal Wkly Rep. 66:214–218. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bisceglia M, Dor DB, Carosi I, Vairo M and
Pasquinelli G: Paratesticular mesothelioma. Report of a case with
comprehensive review of literature. Adv Anat Pathol. 17:53–70.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Grogg JB, Fronzaroli JN, Oliveira P, Bode
PK, Lorch A, Issa A, Beyer J, Eberli D, Sangar V, Hermanns T, et
al: Clinicopathological characteristics and outcomes in men with
mesothelioma of the tunica vaginalis testis: Analysis of published
case-series data. J Cancer Res Clin Oncol. 147:2671–2679. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yuan Z, Zheng N and Shao S: MRI findings
of well-differentiated mesothelioma of tunica vaginalis: One case
report. J Med Imaging. 33:1742–1743. 2023.
|
|
15
|
Chen Y, Pan J, Wang Z, Jin J and Jiang J:
Malignant mesothelioma of the tunica vaginalis testis with penile
invasion: A case report and literature review. National J Androl.
28:186–189. 2022.
|
|
16
|
Kazaz IO, Teoman AS and Mungan S:
Mesothelioma of the tunica vaginalis testis: A case report. Indian
J Pathol Microbiol. 63:475–477. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jones MA, Young RH and Scully RE:
Malignant mesothelioma of the tunica vaginalis. A clinicopathologic
analysis of 11 cases with review of the literature. Am J Surg
Pathol. 19:815–825. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Brimo F, Illei PB and Epstein JI:
Mesothelioma of the tunica vaginalis: A series of eight cases with
uncertain malignant potential. Mod Pathol. 23:1165–1172. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Huang K, Cao Y, Yao K, Zhou F, Liu Z and
Li X: Diagnosis and treatment of malignant mesothelioma of the
tunica vaginalis testis: A series of 7 cases. Zhonghua Wai Ke Za
Zhi. 61:812–817. 2023.(In Chinese). PubMed/NCBI
|
|
20
|
Plas E, Riedl CR and Pflüger H: Malignant
mesothelioma of the tunica vaginalis testis: Review of the
literature and assessment of prognostic parameters. Cancer.
83:2437–2446. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Drevinskaite M, Patasius A, Kevlicius L,
Mickys U and Smailyte G: Malignant mesothelioma of the tunica
vaginalis testis: A rare case and review of literature. BMC Cancer.
20:1622020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Park YJ, Kong HJ, Jang HC, Shin HS, Oh HK
and Park JS: Malignant mesothelioma of the spermatic cord. Korean J
Urol. 52:225–229. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Arda E, Arıkan MG, Cetin G, Kuyumcuoğlu U
and Usta U: Malignant mesothelioma of tunica vaginalis testis:
Macroscopic and microscopic features of a very rare malignancy.
Cureus. 9:e18602017.PubMed/NCBI
|
|
24
|
Yang C, Liang CZ, Zhang H, Ye YP, Zhang
XS, Hao ZY, Zhou J, Fan S, Tai S and Wang KX: Malignant
Mesothelioma of the Tunica Vaginalis: Report of a Case and Review
of the Literature. J Clin Urol. 25:621–625. 2010.(In Chinese).
|
|
25
|
de Sá Barrêto Callou Peixoto M, Bernardo
Soares MK, Libânio BB, Albuquerque KS and Bacchi CE: Malignant
mesothelioma of the tunica vaginalis testis: A rare cause of
hydrocele. Urol Case Rep. 43:1020482022.PubMed/NCBI
|
|
26
|
Chen DC, Yu FP and Gao Y: Malignant
mesothelioma of the tunica vaginalis: A case report. Clin J Med
Officers. 39:458–504. 2011.(In Chinese).
|
|
27
|
Du J, Wu T and Liang GB: Primary malignant
mesothelioma of the tunica vaginalis: A case report. Chin J Clin
Oncol. 51:427–428. 2024.
|
|
28
|
Huang CH, Li JY, Ding ZY, Liu S and Yu GX:
Malignant mesothelioma of the tunica vaginalis: A case report. Chin
J Diagnostic Pathol. 17:316–317. 2010.
|
|
29
|
Liu Q, Wang R, Ru N, Wu Y, Guo C, Chen L,
Liang J and Zhang F: Analysis of guide wire displacement in
robot-assisted spinal pedicle screw implantation. J Robot Surg.
18:1382024. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Matsuki R, Ishii S, Suzuki T and Sugiyama
H: A case of simultaneous diagnosis of tunica vaginalis testis and
pleural mesothelioma. Respirol Case Rep. 10:e09372022. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bertolotto M, Boulay-Coletta I, Butini R,
Dudea SM, Grenier N, Oltmanns G, Ramchandani P, Stein MW, Valentino
M and Derchi LE: Imaging of mesothelioma of tunica vaginalis
testis. Eur Radiol. 26:631–638. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Dacic S: Pleural mesothelioma
classification-update and challenges. Mod Pathol. 35 (Suppl
1):S51–S56. 2022. View Article : Google Scholar
|
|
33
|
Hocking AJ, Thomas EM, Prabhakaran S,
Jolley A, Woods SL, Soeberg MJ and Klebe S: Molecular
characterization of testicular mesothelioma and the role of
asbestos as a causative factor. Arch Pathol Lab Med. 147:1446–1450.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Janes WCI, Al-Asaaed S and Johnston PH:
Malignant mesothelioma of the testes with retroperitoneal
recurrence and resection in an 80-Year-Old male and review of the
literature. Case Rep Oncol. 16:698–704. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Amin KM, Litzky LA, Smythe WR, Mooney AM,
Morris JM, Mews DJ, Pass HI, Kari C, Rodeck U, Rauscher FJ III, et
al: Wilms' tumor 1 susceptibility (WT1) gene products are
selectively expressed in malignant mesothelioma. Am J Pathol.
146:344–356. 1995.PubMed/NCBI
|
|
36
|
Chu AY, Litzky LA, Pasha TL, Acs G and
Zhang PJ: Utility of D2-40, a novel mesothelial marker, in the
diagnosis of malignant mesothelioma. Mod Pathol. 18:105–110. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hinterberger M, Reineke T, Storz M, Weder
W, Vogt P and Moch H: D2-40 and calretinin-a tissue microarray
analysis of 341 malignant mesotheliomas with emphasis on
sarcomatoid differentiation. Mod Pathol. 20:248–255. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang GY and Liu YM: Paradidymal malignant
mesothelioma: A case report and literature review. China Modern
Doctor. 60:167–197. 2022.
|
|
39
|
Yang LH, Yu JH, Xu HT, Lin XY, Liu Y, Miao
Y, Wang L, Fan CF, Jiang GY, Ding SL, et al: Mesothelioma of the
tunica vaginalis testis with prominent adenomatoid features: A case
report. Int J Clin Exp Pathol. 7:7082–7087. 2014.PubMed/NCBI
|
|
40
|
Yuan J, Wu JR, Song HJ, Yu B, Lu ZF, Jiang
SJ and Zhou XJ: Malignant mesothelioma of the tunica vaginalis
testis: a clinicopathological observation of 3 cases and review of
the literatures. J Clin Exp Pathol. 29:40–44. 2013.(In
Chinese).
|
|
41
|
Arslan A, Ozcakir-Tomruk C, Deniz E and
Akin O: A case report of metastasis of malignant mesothelioma to
the retromolar trigone. World J Surg Oncol. 14:1882016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Koschel SG and Wong LM: Radical inguinal
orchidectomy: The gold standard for initial management of
testicular cancer. Transl Androl Urol. 9:3094–3102. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Gupta NP and Kumar R: Malignant gonadal
mesothelioma. Curr Treat Options Oncol. 3:363–367. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zucali PA, De Vincenzo F, Perrino M,
Digiacomo N, Cordua N, D'Antonio F, Borea F, Fazio R, Pirozzi A and
Santoro A: Advances in drug treatments for mesothelioma. Expert
Opin Pharmacother. 23:929–946. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zalcman G, Mazieres J, Margery J,
Greillier L, Audigier-Valette C, Moro-Sibilot D, Molinier O, Corre
R, Monnet I, Gounant V, et al: Bevacizumab for newly diagnosed
pleural mesothelioma in the Mesothelioma Avastin Cisplatin
Pemetrexed Study (MAPS): A randomised, controlled, open-label,
phase 3 trial. Lancet. 387:1405–1414. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Hassan R, Kindler HL, Jahan T, Bazhenova
L, Reck M, Thomas A, Pastan I, Parno J, O'Shannessy DJ, Fatato P,
et al: Phase II clinical trial of amatuximab, a chimeric
antimesothelin antibody with pemetrexed and cisplatin in advanced
unresectable pleural mesothelioma. Clin Cancer Res. 20:5927–5936.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Fennell DA, Ewings S, Ottensmeier C,
Califano R, Hanna GG, Hill K, Danson S, Steele N, Nye M, Johnson L,
et al: Nivolumab versus placebo in patients with relapsed malignant
mesothelioma (CONFIRM): A multicentre, double-blind, randomised,
phase 3 trial. Lancet Oncol. 22:1530–1540. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Baas P, Scherpereel A, Nowak AK, Fujimoto
N, Peters S, Tsao AS, Mansfield AS, Popat S, Jahan T, Antonia S, et
al: First-line nivolumab plus ipilimumab in unresectable malignant
pleural mesothelioma (CheckMate 743): A multicentre, randomised,
open-label, phase 3 trial. Lancet. 397:375–386. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Mishra K, Siddiquee S and Mislang AR: A
rare presentation of malignant mesothelioma of the tunica vaginalis
managed with immunotherapy and review of the literature. Clin Case
Rep. 11:e76102023. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Naing A, Meric-Bernstam F, Stephen B, Karp
DD, Hajjar J, Rodon Ahnert J, Piha-Paul SA, Colen RR, Jimenez C,
Raghav KP, et al: Phase 2 study of pembrolizumab in patients with
advanced rare cancers. J Immunother Cancer. 8:e0003472020.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
D'Angelo SP, Russell J, Lebbé C,
Chmielowski B, Gambichler T, Grob JJ, Kiecker F, Rabinowits G,
Terheyden P, Zwiener I, et al: Efficacy and safety of First-line
avelumab treatment in patients with Stage IV metastatic merkel cell
carcinoma: A preplanned interim analysis of a clinical trial. JAMA
Oncol. 4:e1800772018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mehnert JM, Panda A, Zhong H, Hirshfield
K, Damare S, Lane K, Sokol L, Stein MN, Rodriguez-Rodriquez L,
Kaufman HL, et al: Immune activation and response to pembrolizumab
in POLE-mutant endometrial cancer. J Clin Invest. 126:2334–2340.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Marabelle A, Fakih M, Lopez J, Shah M,
Shapira-Frommer R, Nakagawa K, Chung HC, Kindler HL, Lopez-Martin
JA, Miller WH Jr, et al: Association of tumour mutational burden
with outcomes in patients with advanced solid tumours treated with
pembrolizumab: Prospective biomarker analysis of the multicohort,
open-label, phase 2 KEYNOTE-158 study. Lancet Oncol. 21:1353–1365.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Wu G, Fang Y, Bi D, Yang W and Sun Y: Case
report: Immunotherapy in rare high TMB pancreatic acinar carcinoma.
Front Oncol. 14:13572332024. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chen Y, Du X, Gao Y, Wu H, Zhao H and Su
Y: Methylthioadenosine phosphorylase and breast cancer 1
Protein-associated protein 1 as biomarkers for the peritoneal
mesothelioma. Cancer Control. 30:107327482312208052023. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Vrugt B, Kirschner MB, Meerang M, Oehl K,
Wagner U, Soltermann A, Moch H, Opitz I and Wild PJ: Deletions of
CDKN2A and MTAP detected by Copy-number variation array are
associated with loss of p16 and MTAP protein in pleural
mesothelioma. Cancers (Basel). 15:49782023. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Chung CT, Santos Gda C, Hwang DM,
Ludkovski O, Pintilie M, Squire JA and Tsao MS: FISH assay
development for the detection of p16/CDKN2A deletion in malignant
pleural mesothelioma. J Clin Pathol. 63:630–634. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Chapel DB, Hornick JL, Barlow J, Bueno R
and Sholl LM: Clinical and molecular validation of BAP1, MTAP, P53,
and Merlin immunohistochemistry in diagnosis of pleural
mesothelioma. Mod Pathol. 35:1383–1397. 2022. View Article : Google Scholar : PubMed/NCBI
|