Introduction
Lung cancer is a leading cause of cancer-related
mortality worldwide and is a heterogeneous disease, requiring
personalized therapeutic approaches to improve patient outcomes. As
the predominant histological subtype of non-small cell lung cancer
(NSCLC), advanced lung adenocarcinoma presents a considerable
clinical challenge, with the 5-year survival rate of patients with
stage IIIB/IV disease remaining <20% (1). Consequently, advanced adenocarcinoma
has become a major focus of precision oncology, with treatment
decisions increasingly guided by genetic mutations, such as
epidermal growth factor receptor (EGFR) mutations, anaplastic
lymphoma kinase (ALK) gene rearrangements and/or programmed
death-ligand 1 (PD-L1) expression (1–3). Novel
therapeutic strategies involving tyrosine kinase inhibitors (TKIs)
and immune checkpoint inhibitors have been associated with improved
overall survival (OS) in randomized clinical trials, when compared
with conventional chemotherapy (2,4).
The present study integrates a decade of clinical
data (2010–2019), with machine learning methodologies to identify
key prognostic factors and establish predictive models. This has
several notable innovations: First, it combines traditional
statistical analyses with five machine learning models, including
decision trees, random forests and gradient boosting, to conduct
comparisons of survival prediction performance. Second, the study
includes 26 clinical characteristic variables encompassing
molecular biomarkers, treatment modalities and metastatic sites.
Third, it uses a concordance index (C-index) dynamic assessment
framework, to overcome the limitations associated with traditional
static performance indicators.
The present study examined the prognostic relevance
of specific genetic alterations, including EGFR mutations and PD-L1
expression, to OS. Mutant EGFR was a particular focus, with a
nuanced exploration of different EGFR mutation subtypes, including
exon 19 deletions and L858R mutations, to further characterize the
molecular underpinnings of lung adenocarcinoma.
Materials and methods
Study design and setting
Data of patients with lung cancer were collected
from Cheng-Hsin General Hospital (Taipei, Taiwan) between January
2010 and December 2019. The present retrospective study of the data
was approved by the Institutional Review Board of Cheng-Hsin
General Hospital [approval no. CHGH-IRB (922)111-01l; date of
approval, January 27, 2022], and the requirement for informed
consent was waived by the ethics committee. Clinical data were
collected from a total of 1,875 patients with lung cancer, of whom
1,190 were diagnosed with adenocarcinoma. Among these patients with
adenocarcinoma, 575 subjects (male/female, 291/284; mean age,
68.94±12.18 years) were initially diagnosed with advanced stage
[Lung American Joint Committee on Cancer (AJCC) 8th edition staging
system; stage IIIB or IV] disease (5). Due to not having undergone surgical
tumor excision, patients with advanced lung adenocarcinoma may vary
in prognosis according to their tumor genetic characteristics and
the therapeutic strategies chosen by clinicians. In addition to the
disease stage and histological subtype described, patients were
included if they had a confirmed diagnosis of lung adenocarcinoma
and available clinical data for analysis. No other specific
inclusion or exclusion criteria were applied.
Smoking status was categorized as never smoker,
current smoker, former smoker (quit <10 years ago) and long-term
former smoker (quit >10 years ago). Compound mutations were
defined as the presence of at least two gene mutations. Various
characteristics of the patients with advanced adenocarcinoma were
analyzed using multivariate regression analysis, including the
tumor-node-metastasis stage based on the Lung AJCC 8th edition
staging system, the location of the primary tumor, smoking status,
smoking quantity (pack-year) and biomarkers, including EGFR
mutation status and subtype, ALK status, PD-L1 expression and CEA
levels. EGFR mutation status and subtype were determined using
quantitative PCR or next-generation sequencing performed on tumor
tissue specimens as part of routine clinical care. ALK
rearrangement status was assessed by immunohistochemistry. PD-L1
expression was evaluated by immunohistochemistry using the
anti-PD-L1 monoclonal antibody clone 22C3 (PD-L1 IHC 22C3 pharmDx;
Agilent Technologies, Inc.) and reported as the tumor proportion
score. Serum CEA levels were measured using standard clinical
immunoassays. All biomarker data were obtained retrospectively from
patients' medical records and were not generated specifically for
the present study. The artificial intelligence-based algorithms
were applied to structured clinical data to evaluate OS and to
explore potential associations with treatment strategies.
Machine learning models
Five models were compared: i) A simple rule-based
model comprising a decision tree incorporating EGFR variant
patterns; ii) a random survival forest (RSF); iii) a Cox
proportional hazards model; iv) a support vector machine
(SVM)-based survival model; and v) a gradient-boosted survival
(GBS) analysis. Model performance was evaluated using the C-index,
where a C-index of <0.5 indicates random prediction, and a value
>0.7 indicates good concordance between the predicted and
observed survival times. For each model, the mean and standard
deviation of the C-index were calculated across repeated runs to
assess the average predictive accuracy and the robustness of the
model under repeated random data splits. In each run, the dataset
was randomly split into a training set (70%) and testing set (30%).
Model performance was also evaluated using the time-dependent area
under the curve (AUC) of the receiver operating characteristic
curve.
Simple rule-based model
A simple rule-based decision tree model was
implemented to establish a lower-bound performance benchmark. EGFR
status was encoded as a categorical variable in the dataset (0,
unknown; 1, wild-type; 2, mutant EGFR) according to the predefined
coding scheme. The model assigned a fixed risk score based solely
on the EGFR feature: Patients with EGFR=2 were assigned a predicted
risk score of 0.4 (lower risk), and all other patients were
assigned a risk score of 0.6 (higher risk). Because the C-index
depends only on relative risk ranking and is invariant to monotonic
transformations, the specific numeric values are arbitrary and
serve only to distinguish lower-risk (0.4) vs. higher-risk (0.6)
groups. Model performance was evaluated using the C-index to assess
whether this heuristic approach captured any prognostic signal in
the data.
RSF
RSFs extend the random forest framework to censored
survival data by growing an ensemble of survival trees using
bootstrap samples. Each tree recursively partitions the feature
space to maximize survival differences using log-rank statistics.
Ensemble predictions are obtained by aggregating survival
probabilities across all trees. In the present study, the
ExtraSurvivalTrees package (scikit-survival; version 0.26.0;
http://scikit-survival.readthedocs.io/en/stable/index.html)
in Python (version 3.9.7; http://www.anaconda.com/products/distribution) was
implemented with 100 trees (n_estimators=100). RSFs are
non-parametric models capable of capturing nonlinear relationships
and feature interactions without requiring the proportional hazards
assumption.
Cox proportional hazards model
The Cox proportional hazards model is a
semi-parametric regression approach that relates covariates to the
hazard function. The CoxnetSurvivalAnalysis package
(scikit-survival 0.26.0 in the Python library) was used, which
extends the classical Cox model by incorporating L1 and L2
regularization through an elastic net penalty. This regularization
enables variable selection and stabilizes coefficient estimates,
particularly in high-dimensional feature spaces.
SVM
SVMs formulates survival prediction as a ranking
problem, aiming to learn a function f(x) that preserves the
ordering of survival times. The FastKernelSurvivalSVM package
(implemented using the scikit-survival Python library; version
0.26.0) with a linear kernel was employed. This approach allows the
modeling of complex relationships while maintaining computational
efficiency.
GBS
Gradient boosting for survival data combines
multiple weak learners, typically decision trees, in a stage-wise
manner to optimize a loss function derived from the Cox partial
likelihood. The GradientBoostingSurvivalAnalysis package with 100
base learners was used
(sksurv.ensemble.GradientBoostingSurvivalAnalysis was implemented
using the scikit-survival in the Python library). This approach
sequentially refines the prediction by emphasizing samples with
larger prediction errors, resulting in a strong performance in
heterogeneous and high-dimensional survival datasets.
Statistical analysis
Categorical variables are presented as frequency and
percentage and continuous variables are presented as mean ±
standard deviation. Survival outcomes were analyzed using
time-to-event methods. Kaplan-Meier curves were generated to
visualize differences in survival patterns between and among
patient subgroups. The survival distributions of two or more
independent groups were compared using the log-rank test. However,
as the proportional hazards assumption of the log-rank test may be
violated when survival curves cross, the two-stage hazard rate
comparison (TSHRC) method was used when non-proportional hazards
were suspected, including situations with late-stage curve
crossover. In this method, a log-rank test was performed at stage I
to assess OS differences (α=0.05), and if the stage I test did not
indicate statistical significance, a stage II maximum-type test
sensitive to late or transient crossing hazards was then performed.
Finally, an overall two-stage P-value was calculated. A stabilizing
constant (ε=0.1) was applied to ensure numerical stability in
hazard estimation. These analyses were performed using the TSHRC
package in R (version 4.5.2; http://cran.r-project.org/web/packages/TSHRC).
The CoxPHSurvivalAnalysis model implemented in the
Python scikit-survival library was used to assess the effects of
clinical variables on OS and to generate risk predictions for
survival outcomes. Associations between survival and clinical
variables, including sex, age, smoking status, EGFR mutation
status, CEA level and tumor location, were assessed using
multivariable Cox proportional hazards regression to appropriately
account for censored time-to-event data. The strength and sources
of collinearity among variables in the multivariable Cox model were
assessed using the Belsley-Kuh-Welsch collinearity diagnostics.
Heteroscedasticity and deviation from normality were detected using
the White test and Shapiro-Wilk test, respectively. Patients with
missing data were excluded from the multivariable Cox analysis.
Statistical analyses were performed using EasyMedStat (version
3.17; www.easymedstat.com). P<0.05 was
considered to indicate a statistically significant result.
Results
Characteristics and OS of patients
with advanced adenocarcinoma of the lung
A total of 575 patients with advanced lung
adenocarcinoma were included in the present study. The median age
of the patients was 70 years. Patient characteristics are shown in
Table I. Bone metastasis was
detected in 38.4% of patients, liver metastasis in 17.9%, brain
metastasis in 36% and contralateral lung metastasis in 28.9%.
 | Table I.Demographic data of patients with
advanced lung adenocarcinoma. |
Table I.
Demographic data of patients with
advanced lung adenocarcinoma.
| Variables | N (%) | Mean ± SD | Median | Min-max | [95% CI] |
|---|
| Sex |
|
|
|
|
|
| Male | 291 (50.6) | - | - | - | - |
|
Female | 284 (49.4) | - | - | - | - |
| Age, years | 575 | 68.94±12.18 | 70.00 | 30.00–96.00 | [67.940 to
69.931] |
| Height, cm | 477 | 160.17±8.91 | 160.00 | 131.00–187.00 | [159.374 to
160.974] |
| Weight, kg | 480 | 59.39±12.42 | 59.00 | 27.00–113.00 | [58.275 to
60.496] |
| Smoking quantity,
pack-year | 441 | 12.60±24.26 |
| 0.00–160.00 | [10.338 to
14.866] |
| CEA, ng/ml | 565 | 481.16±2,735.94 | 25.70 | 1.00–42,412.00 | [255.558 to
706.758] |
| EGFR status |
|
|
|
|
|
|
Mutated | 229 (39.8) | - | - | - | - |
| Wild
type | 218 (37.9) | - | - | - | - |
|
Unknown | 118 (20.6) | - | - | - | - |
| ALK
positive | 10 (1.7) | - | - | - | - |
| Metastases |
|
|
|
|
|
|
Bone | 221 (38.4) | - | - | - | - |
|
Liver | 103 (17.9) | - | - | - | - |
|
Brain | 207 (36.0) | - | - | - | - |
|
Lung | 166 (28.9) | - | - | - | - |
| Overall survival,
months | 575 | 19.47±21.50 | 11.43 | 0.07–112.17 | [17.710 to
21.225] |
A total of 26 characteristic variables were included
in the analysis. A Cox proportional hazards model was fitted
separately for each variable, and its predictive power for OS was
recorded during training. The predictive power was determined using
this metric and presented in Table
II, with predictive power <0.5 indicating a poor predictive
performance. The five patient characteristics with the highest
predictive power were, in descending order, EGFR mutation status,
EGFR variant type, second-line treatment regimen, age and
first-line treatment regimen.
 | Table II.Predictive power of 26
characteristics for overall survival in patients with advanced lung
adenocarcinoma. |
Table II.
Predictive power of 26
characteristics for overall survival in patients with advanced lung
adenocarcinoma.
|
Characteristics | Predictive
power |
|---|
| EGFR mutation | 0.623 |
| EGFR variant
type | 0.607 |
| Second-line
treatment agent | 0.589 |
| Age | 0.580 |
| First-line
treatment agent | 0.573 |
| Third-line
treatment agent | 0.570 |
| Smoking
quantity | 0.564 |
| PD-L1 | 0.557 |
| Metastasis | 0.548 |
| ALK | 0.548 |
| CEA | 0.542 |
| Betel nut
chewing | 0.538 |
| Weight | 0.533 |
| Radiotherapy | 0.532 |
| Lymph node
involvement | 0.532 |
| Tumor size | 0.528 |
| Brain
metastasis | 0.526 |
| Bone
metastasis | 0.524 |
| Staging | 0.522 |
| Sex | 0.521 |
| Height | 0.515 |
| Liver
metastasis | 0.510 |
| Smoking status | 0.507 |
| Primary tumor
site | 0.507 |
| Lung
metastasis | 0.490 |
| Alcohol
consumption | 0.484 |
Multivariable analysis was performed using the
EasyMedStat online application to estimate and compare the effect
coefficients of different variables on the survival of patients
with advanced lung adenocarcinoma (Table III). Older age at diagnosis was
associated with poorer survival, suggesting that the cancer had a
higher malignancy and led to a poorer prognosis in older patients.
Patients with left-sided lung cancer tended to have a worse
prognosis than those with right-sided tumors, although this finding
was not statistically significant. Smoking history was associated
with adverse survival outcomes, with the poorest OS observed among
those patients with former smoker status.
 | Table III.Multivariate regression analysis of
variables associated with survival in patients with advanced lung
adenocarcinoma. |
Table III.
Multivariate regression analysis of
variables associated with survival in patients with advanced lung
adenocarcinoma.
| Variables | Regression
coefficient, β [95% CI] | P-value |
|---|
| Intercept | 35.04 [24.1 to
45.97] | <0.0001 |
| Age, per 1-unit
increase | −0.252 [-0.396 to
−0.107] |
6.82×10−4 |
| Tumor location |
|
|
|
Right | Reference |
|
|
Left | −3.26 [-6.77 to
0.24] | 0.0678 |
| Smoking status |
|
|
|
Never | Reference |
|
|
Current | −3.62 [-9.24 to
1.99] | 0.205 |
|
Long-term former | 1.52 [-5.87 to
8.91] | 0.686 |
|
Former | −6.43 [-12.28 to
−0.58] | 0.0313 |
|
Unknown | −8.36 [-13.83 to
−2.9] | 0.00278 |
| EGFR |
|
|
| Wild
type | Reference |
|
|
Mutation | 9.23 [5.17 to
13.29] | <0.0001 |
|
Unknown | −0.961 [-5.59 to
3.67] | 0.683 |
| CEA, per 1-unit
increase | 0.0114
[-6.81×10−4 to 0.0236] | 0.0643 |
| Sex |
|
|
|
Female | Reference |
|
|
Male | 1.96 [-2.1 to
6.01] | 0.343 |
| Smoking quantity,
per 1-unit increase | −0.181 [-0.243 to
−0.119] | <0.0001 |
Effect of EGFR mutations on OS
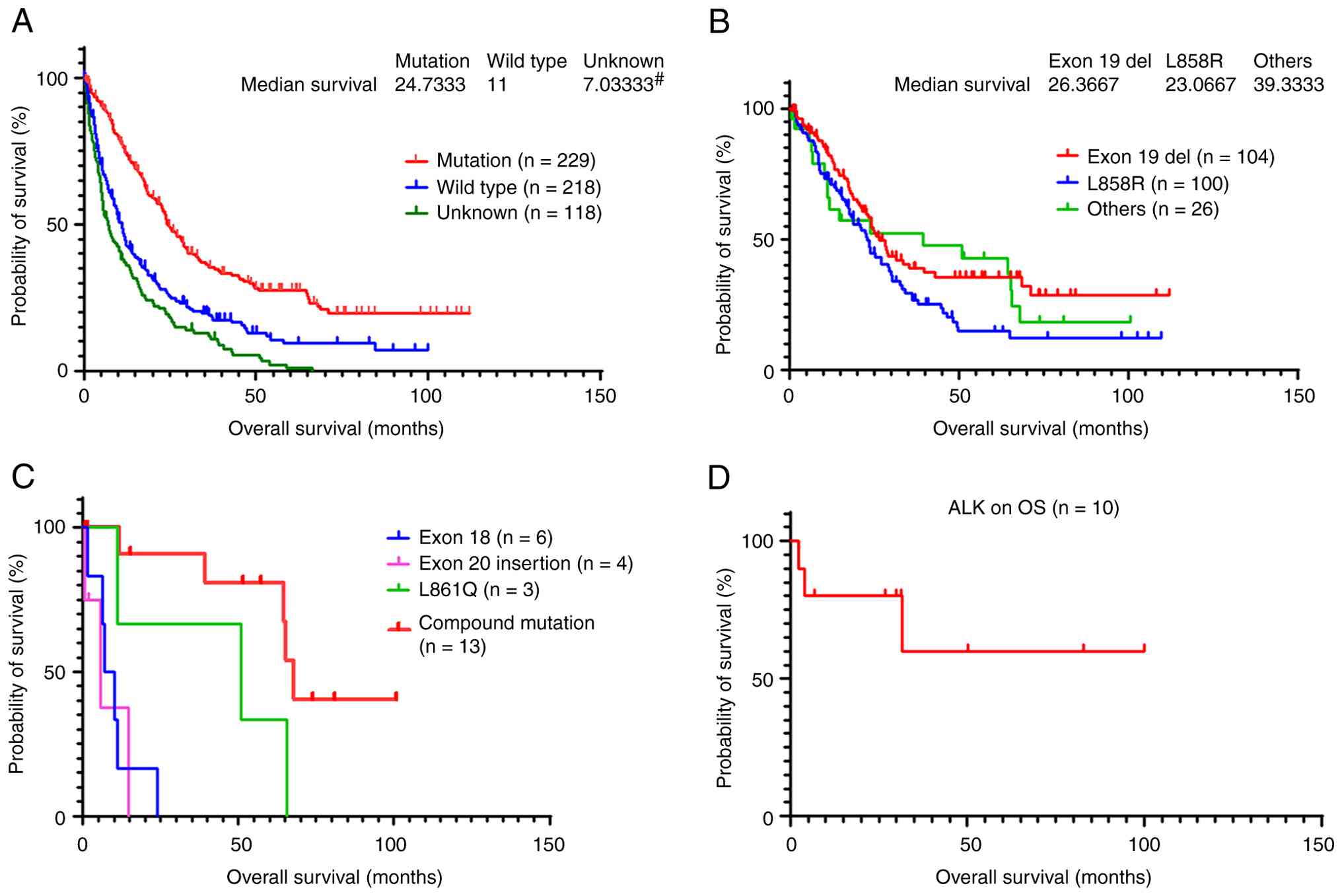
As shown in Fig. 1A,
patients with EGFR mutations had a longer OS than those without
EGFR mutations (n=229 vs. 218; median OS, 24.7 vs. 11 months,
respectively). Patients with an unknown EGFR mutation status had a
median OS of only 7.03 months (n=118). The lack of knowledge of
mutation status was primarily attributable to insufficient tumor
tissue being available for molecular testing, and the patients
refusing to undergo another biopsy.
In the analysis of the impact of EGFR mutation
subtypes on OS, no statistically significant difference in OS was
detected between the patients with the common EGFR mutation
subtypes exon 19 deletion and L858R (n=104 vs. 100; median OS, 26.4
vs. 23.1 months, respectively). Patients with other less common
EGFR variants had a longer median OS time (n=26; median OS, 39.3
months), as shown in Fig. 1B.
Fig. 1C shows the
effect of four uncommon EGFR mutation subtypes on OS, including
exon 18 gene mutation, exon 20 insertion, L861Q and compound
mutations (n=6 vs. 4 vs. 3 vs. 13; median OS, 8.6, 5.8, 50.9 and
67.9 months, respectively). Among these subgroups, patients with
exon 20 insertions exhibited the shortest survival.
Effect of ALK positivity on OS
A total of 255 patient samples were tested for ALK
rearrangements, of which 10 were positive, accounting for 3.92% of
all tested patients. However, the median OS was not reached, as
shown in Fig. 1D.
Effect of therapeutic strategies on
OS
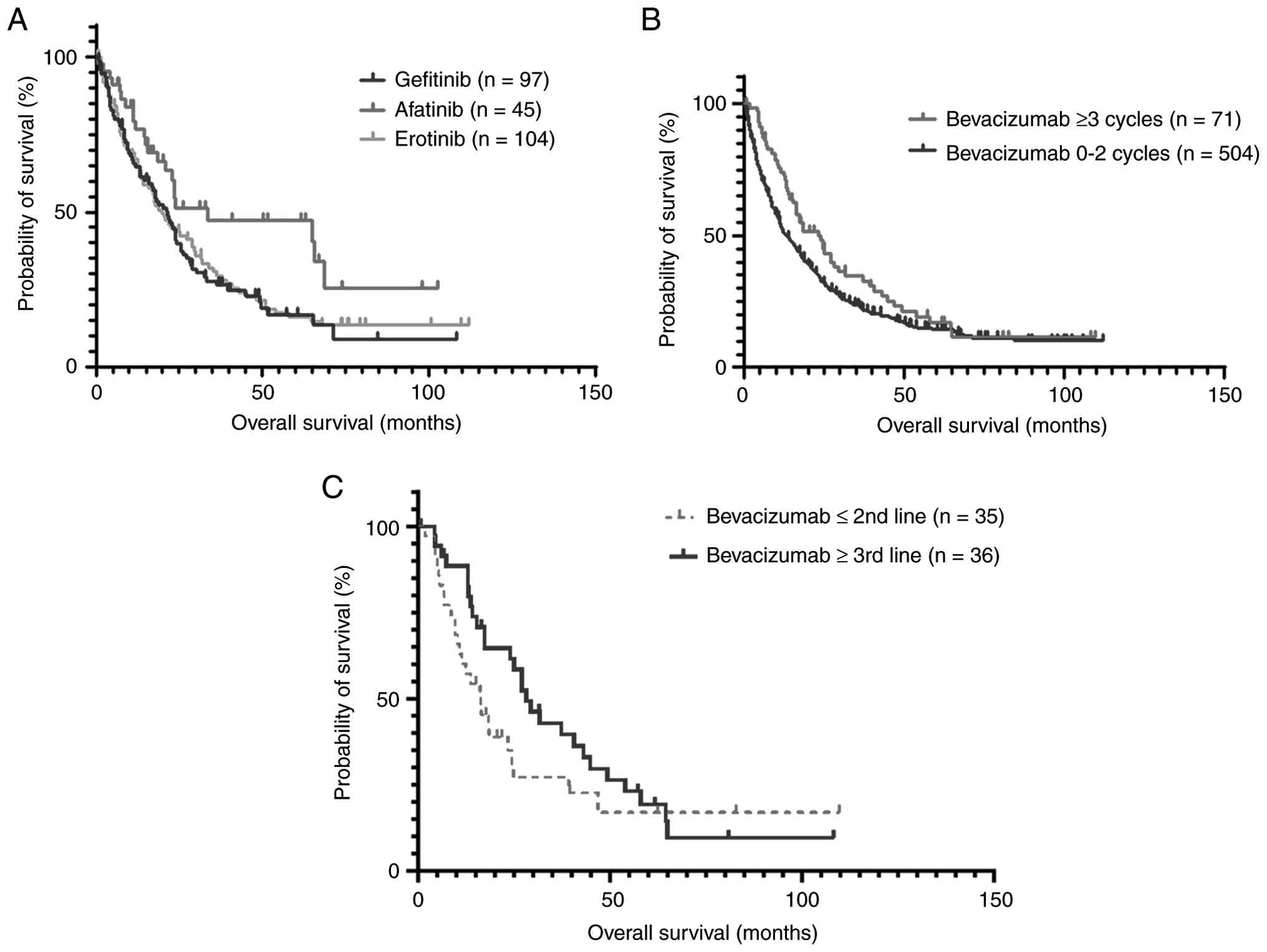
Fig. 2A shows the
impact of different first-line targeted therapies on the OS of
patients with EGFR mutations. First-line treatment with the
second-generation EGFR TKI afatinib was associated with a longer OS
than that obtained with the first-generation EGFR TKIs gefitinib
and erlotinib (n=45 vs. 97 and 104; median OS, 33.4 vs. 19.9 and
21.3 months, respectively; P=0.0358).
In the treatment of clinically advanced lung
adenocarcinoma, angiogenesis inhibitors have been shown to improve
progression-free survival (PFS) (6). In the present study, the OS of
patients who received three or more courses of bevacizumab was
longer than that of patients who had not been treated with
bevacizumab or had received fewer than three cycles (n=71 vs. 504;
median OS, 23.9 vs. 13.4 months; respectively; hazard ratio, 0.74;
CI, 0.57 to 0.96), as shown in Fig.
2B. The TSHRC analysis revealed stage-I P=0.039 and final
two-stage P=0.0253, confirming that the survival advantage was
statistically significant after adjustment for potential hazard
crossing.
Regarding the effect of angiogenesis inhibitors, the
present data suggested that the use of bevacizumab in late
treatment lines, defined as the third line or later, was associated
with a longer OS than use in earlier lines; (second line or earlier
vs. third line or later; n=35 vs. 36; median OS, 16.4 vs. 28.1
months; hazard ratio, 1.7; 95% CI, 1.0 to 2.9), as shown in
Fig. 2C. However, this apparent
advantage was not statistically significant when hazard crossing
was accounted for, as reflected by stage I log-rank P=0.138, stage
II P=0.056 and final two-stage P=0.080.
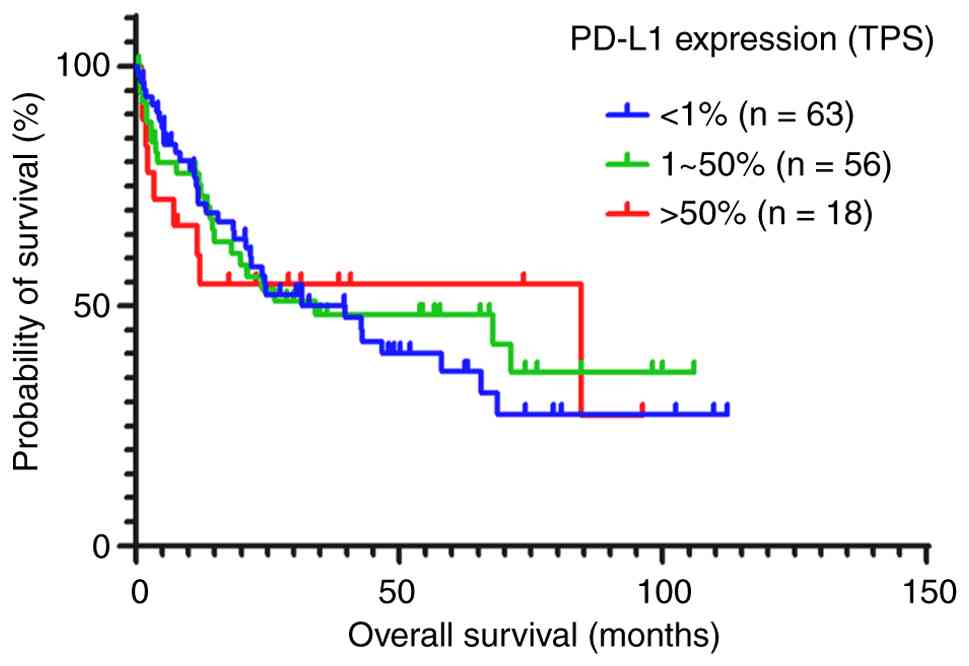
PD-L1 expression in lung adenocarcinoma tissue was
assessed to evaluate the potential benefit of immunotherapy. The
association between PD-L1 expression and OS was analyzed (Fig. 3). Immunohistochemistry using a 22C3
PD-L1 monoclonal antibody was performed in 137 patients. Of these
patients, 63 had <1% PD-L1, 56 had 1–50% PD-L1 and 18 had
>50% PD-L1 staining. The median OS times of these groups were
39.7, 34.1 and 84.5 months respectively; however, the differences
among the groups were not statistically significant.
Machine learning models for survival
analysis
In survival prediction, patients with a higher
estimated risk are expected to have shorter survival times. Model
performance was evaluated using the C-index, which predicts the
proportion of concordant patient pairs. Table IV summarizes the average C-index
values for the different machine learning models. Among the
evaluated models, the SVM achieved the highest C-index (~0.7018).
Evaluation time points were defined at 3-month intervals from 1 to
60 months, providing a uniform temporal grid for performance
assessment throughout the follow-up period. For each subject in the
test set, the RSF, GBS and Cox proportional hazard models were
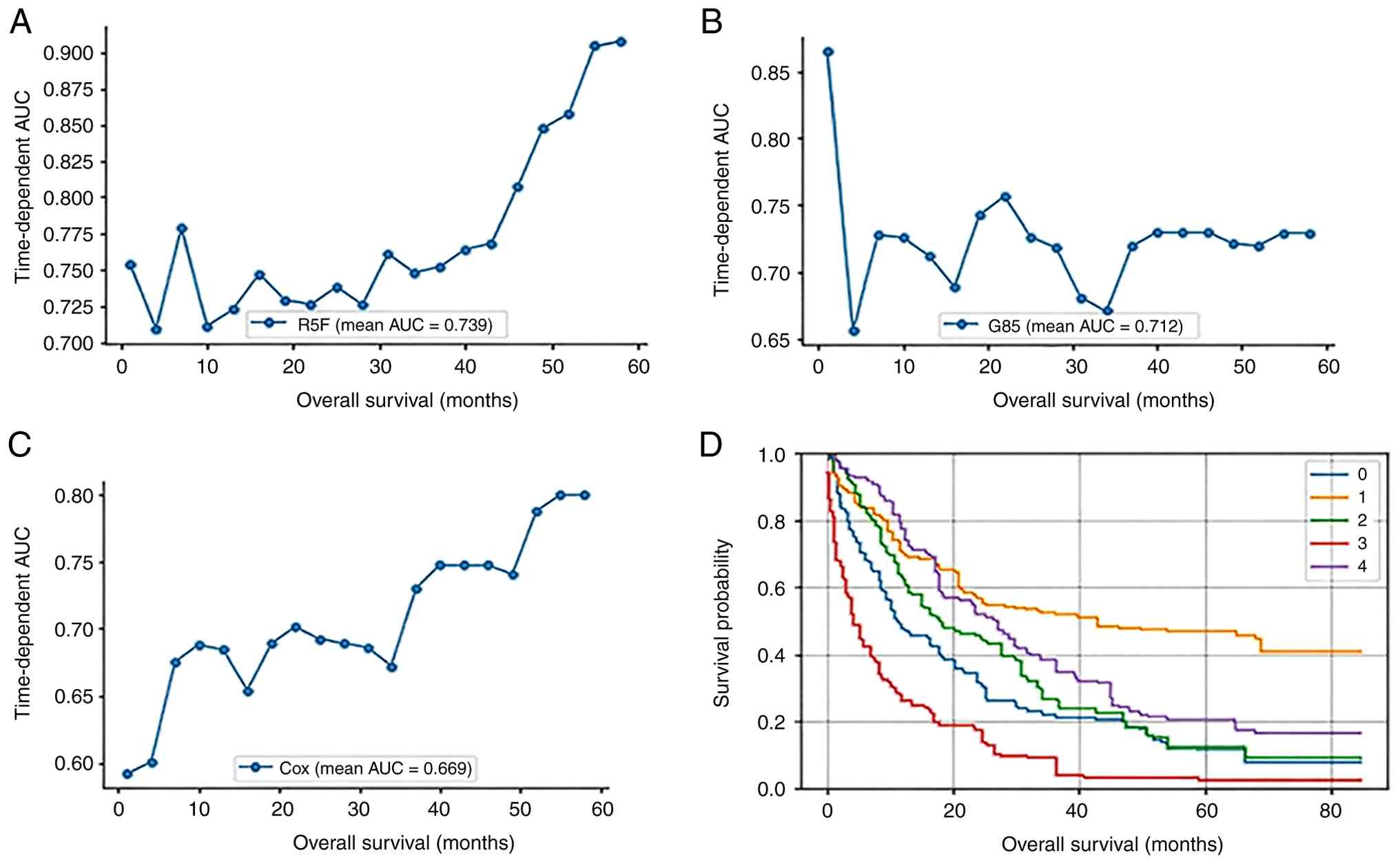
trained separately to estimate individual cumulative hazard
functions (Fig. 4A-C). The GBS
model maintained a relatively stable AUC ranging from 0.65 to 0.75.
By contrast, the AUC values of the RSF and Cox models gradually
increased over 40 months, and reached their highest values at ~60
months. To further illustrate the individualized survival
predictions generated by the RSF model, survival functions were
estimated for a subset of test patients. Specifically, the first
five subjects from the test set were selected, and their
Kaplan-Meier survival curves are shown in Fig. 4D. The figure demonstrates distinct
survival patterns among these subjects, with clear differences in
survival probability over the entire follow-up period, highlighting
heterogeneity in individual survival outcomes.
 | Table IV.C-index of different machine learning
models in survival analysis. |
Table IV.
C-index of different machine learning
models in survival analysis.
| Machine learning
models | Mean C-index | Standard
deviation |
|---|
| EFGR variant
patterns | 0.614 | 0.007 |
| Cox proportional
hazards | 0.695 | 0.021 |
| Support vector
machine | 0.702 | 0.023 |
| Random survival
forest | 0.689 | 0.020 |
| Gradient boosting
survival | 0.683 | 0.022 |
Discussion
The comprehensive retrospective analysis of 575
patients in the present study provides a nuanced understanding of
the complex treatment landscape and prognostic factors associated
with advanced lung adenocarcinoma. Numerous previous studies have
used PFS as a surrogate endpoint because results for this can be
obtained relatively quickly; however, there is inevitably a
temporal bias in PFS due to its intermittent assessment (7). By contrast, the present study used OS
as the evaluation outcome, as the time of death is definitive and
accurately recorded, thereby providing a more accurate reflection
of clinical outcomes (8).
One notable aspect of the present study is the
diversity of metastatic sites included within the patient cohort.
The identified prognostic factors, including smoking status, EGFR
mutation status and age, contribute to a deeper understanding of
patient outcomes. The recognition that certain demographic and
molecular characteristics are poor prognostic indicators
underscores the importance of personalized and targeted
interventions. For example, the presence of bone metastases was
identified as a factor associated with poor outcomes, and possibly
poor clinical condition, indicating that heightened clinical
attention and tailored therapeutic approaches are warranted for
this subgroup.
According to previous studies, approximately
one-third of patients with stage IV lung adenocarcinoma have bone
metastasis, and nearly one-quarter have lung metastasis, with liver
metastasis being associated with the poorest prognosis, followed by
bone metastasis (9,10). In the present study, the OS of
patients with advanced lung adenocarcinoma was comparable between
men and women; although women exhibited a slightly worse prognosis,
the difference between sexes was not statistically significant. The
most common metastatic locations in the present cohort were the
bones, brain, lungs and liver. The prognostic impact was indicated
to be in the order of brain, bone, liver and lung. The discrepancy
between these findings and those of previous studies may be
attributable to differences in the incidence of metastasis at each
location, which may affect the predictive ability for survival
outcomes.
It has previously been reported that smoking volume
is associated with reduced OS in patients with advanced lung
adenocarcinoma (11). Consistent
with this, the present study showed that the patients who had quit
smoking within the last 10 years and current smokers had a worse OS
compared with that of patients who had never smoked. Whether
smoking contributes to the development of drug resistance requires
further investigation. In addition, left-sided primary lung cancer
showed a non-significant trend toward being associated with poorer
OS, possibly due to its proximity to critical structures such as
the heart, aorta and esophagus.
Among all the patient characteristics evaluated,
EGFR mutation status was found to have the highest predictive power
for OS. A molecular epidemiological analysis of lung adenocarcinoma
conducted in Taiwan in 2016, based on 1 year of cumulative EGFR
gene testing, revealed that 56.1% of patients were aged ≤45 years
and 60.6% harbored EGFR mutations (12). By contrast, the present study
focused primarily on patients with advanced lung adenocarcinoma, of
which ~40% had confirmed EGFR gene mutations, while 20.6% had
unknown or untested EGFR status. The therapeutic efficacy of
targeted agents for EGFR-mutated lung adenocarcinoma is known to
vary according to mutation subtype, with common subtypes including
exon 19 deletion and L858R; first-line targeted therapy has been
shown to result in a longer PFS in cases with exon 19 deletions
(13). In the present study, the
population proportions and OS outcomes of these two mutation
subtypes were comparable, with no statistically significant
difference observed. The cohort also included a small group of
patients with unknown EGFR mutation status. Patients with unknown
EGFR mutation status were analyzed as a separate category and had
the shortest median OS, which likely reflected their poorer
clinical condition and inability to undergo repeat molecular
testing or aggressive treatment at diagnosis.
Among rare EGFR mutation subtypes, exon 20
insertions were observed in a small number of cases in the present
study; they were found to be associated with a short OS (median,
5.8 months) and are reportedly associated with a poor response to
targeted drugs and chemotherapy (14). The second most common rare mutation
was exon 18 mutation G719X, which was also associated with a short
survival period (median, 8.6 months), consistent with previous
reports (15). Notably, most cases
within this category involved compound mutations, 13 in total,
including L858/T790M, L858R/L861Q and exon19 del/T790M. These EGFR
compound mutations were revealed to be associated with a relatively
long OS time, consistent with the findings of a previous study
(16). Patients with the third most
common rare mutation, the simple exon 21-point mutation L861Q, had
a median OS of 50.8 months, and exhibited a favorable response to
the second-generation TKI afatinib.
With respect to ALK-positive cases, only 10 of the
255 patients tested were found to harbor ALK rearrangements,
accounting for 3.92% of the study cohort. Due to the small number
of cases, definitive conclusions cannot be drawn; however, treated
ALK-positive patients exhibited a long OS, which is consistent with
general clinical observations and previously published studies
(17,18).
Comparative analysis of first-line TKIs revealed a
clear disparity in OS, with the OS of afatinib being longer than
that of erlotinib and gefitinib. This finding provides insight into
the relative efficacy of first-line TKIs, and may be used to inform
treatment decisions and guide clinicians toward the most effective
therapeutic options for patients with advanced adenocarcinoma.
Angiogenesis inhibitors such as bevacizumab have
been widely used to treat ovarian cancer, and their incorporation
into combination regimens is an important strategy for optimizing
therapeutic outcomes. In EGFR-mutated metastatic NSCLC, the
combination of bevacizumab and erlotinib has been shown to
significantly improve PFS and overall response rates, but is also
associated with high toxicity (6).
Despite this efficacy, no improvement in OS has been reported for
this combination (6,19,20).
Bevacizumab is also used as an adjuvant treatment for lung
adenocarcinoma, often in combination with other targeted therapies
or chemotherapy. However, evidence regarding whether the inclusion
of bevacizumab can prolong OS when used as a first-line therapy is
inconsistent (21,22).
The present study showed that, regardless of EGFR
mutation status, patients with lung adenocarcinoma who received
more than three doses of bevacizumab had a significantly prolonged
OS time. Further analysis suggested that the use of bevacizumab in
the third or later treatment line did not significantly prolong OS
compared with earlier use after accounting for potential hazard
crossing. Therefore, the findings do not provide definitive
evidence that adding anti-angiogenic therapy in later lines confers
a greater survival benefit than is achieved when it is used in
earlier lines. Notably, these findings may be influenced by
selection bias, as patients who are able to receive third-line
therapy may inherently have improved prognoses and longer OS. As a
result, the analysis may not allow detection of a true
treatment-line-dependent effect of bevacizumab.
Immunotherapy is an additional therapeutic option
for the treatment of lung adenocarcinoma. Since immunotherapy was
first used for melanoma, its use has gradually expanded to the
treatment of various types of cancer. The expression of PD-L1 in
tumor tissue is considered to inhibit the cytotoxic activity of
immune cells against cancer cells and is commonly used to predict
the response to immunotherapy (23). Increased PD-L1 expression on
tumor-infiltrating cells has been associated with more aggressive
tumor behavior (24). In studies of
NSCLC, patients with high PD-L1 expression in the absence of EGFR
mutation exhibited poorer OS than those with lower PD-L1 expression
levels (25,26). In the present analysis, 137 patients
underwent PD-L1 testing using the 22C3 monoclonal antibody, of whom
63 (46%) had PD-L1 <1%, 56 (41%) had PD-L1 between 1 and 50%,
and only 18 (13%) had PD-L1 >50%. Among these groups, the PD-L1
>50% group appeared to have the longest median OS, although no
statistically significant difference among the groups was observed.
Variability in treatment approaches among the groups may have
influenced the observed outcomes. A previous study suggested that
maintenance permetrexed chemotherapy was associated with improved
progression-free survival and OS in patients with lung
adenocarcinoma exhibiting high PD-L1 expression levels compared
with those with low PD-L1 expression (27). Among the 18 patients with PD-L1
>50% in the present study, five harbored EGFR mutations (three
had exon 19 deletions and two had the L858R mutation), while of the
remaining 13 patients without EGFR mutations, only three received
immunotherapy and the others were treated with permetrexed
chemotherapy. Therefore, we hypothesize that the use of permetrexed
chemotherapy to treat patients with PD-L1 >50% may explain the
prolongation of OS.
The survival analysis of patients with lung cancer
is complicated by the presence of numerous patient characteristics
and clinical variables. To address this, machine learning models
were used to process these high-dimensional variables and predict
the risk of patients. A previous study has used machine learning
models to investigate patients with early-stage (stage I to III)
NSCLC, importing 127 features (28). By contrast, the present study
focused on patients with advanced lung adenocarcinoma, included 28
features, and used the C-index to estimate the consistency between
the predicted results and observed outcomes. A random forest-based
prediction model was used to calculate the C-index at different
time points. The C-index gradually increased after the observation
period exceeded 3 years,. This may indicate that the patients
underwent marked changes in clinical condition during the first 3
years of treatment, resulting in greater discrepancies between
predicted and actual outcomes and consequently lower C-index values
during this period. These findings suggest that clinicians should
closely monitor changes in patient condition during the first 3
years of treatment. Different prediction models were selected
according to the C-index at different time points to improve the
prediction accuracy for patient survival.
The present study had several limitations. In
Taiwan, osimertinib is not broadly used and was reimbursed only for
second-line treatment in patients with the T790M mutation following
failure of treatment with gefitinib, erlotinib, afatinib or
dacomitinib from 2020 onwards. Consequently, OS outcomes associated
with first-line osimertinib were not available in the database used
in the current study. Although osimertinib has been shown to extend
PFS as a first-line treatment for Taiwanese patients with advanced
EGFR-mutated NSCLC, it has not exhibited a statistically
significant OS advantage over second-generation EGFR-TKIs (29). In addition, immunotherapy and
next-generation sequencing-based genetic testing have not been
widely used or integrated into routine clinical practice for lung
cancer.
In the era of machine learning, the present analysis
was extended to predictive modeling by evaluating concordance
statistics for OS based on diverse features within the patient
cohort. This approach not only refines our understanding of
prognostic factors but also contributes to the development of
predictive tools that may support more accurate clinical
decision-making. In summary, the present study aimed to elucidate
the complex interrelationships among therapeutic modalities,
prognostic markers and patient outcomes in advanced lung
adenocarcinoma. By synthesizing evidence from a large-scale
retrospective analysis, the findings of the study advance current
knowledge of optimal treatment strategies and may pave the way for
personalized interventions in this challenging clinical
landscape.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
WTC designed the methodology, analyzed the data and
wrote the original draft. SWC and SY participated in data analysis
and revised the manuscript. IC was involved in conception and
design of the analysis plan, critically revised the manuscript for
important intellectual content, and provided substantive scientific
input during manuscript amendments. SY participated in the coding
and design of the methodology. WTC and SWC confirm the authenticity
of all the raw data. All authors read and approved the final
version of the manuscript.
Ethics approval and consent to
participate
This retrospective study was approved by the
Institutional Review Board of Cheng-Hsin General Hospital [approval
no. CHGH-IRB (922)111-01]. The requirement for informed consent was
waived.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jeon DS, Kim HC, Kim SH, Kim TJ, Kim HK,
Moon MH, Beck KS, Suh YG, Song C, Ahn JS, et al: Five-year overall
survival and prognostic factors in patients with lung cancer:
Results from the Korean association of lung cancer registry
(KALC-R) 2015. Cancer Res Treat. 55:103–111. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Osmani L, Askin F, Gabrielson E and Li QK:
Current WHO guidelines and the critical role of immunohistochemical
markers in the subclassification of non-small cell lung carcinoma
(NSCLC): Moving from targeted therapy to immunotherapy. Semin
Cancer Biol. 52:103–109. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Greenhalgh J, Boland A, Bates V, Vecchio
F, Dundar Y, Chaplin M and Green JA: First-line treatment of
advanced epidermal growth factor receptor (EGFR) mutation positive
non-squamous non-small cell lung cancer. Cochrane Database Syst
Rev. 3:CD0103832021.PubMed/NCBI
|
|
4
|
Duma N, Santana-Davila R and Molina JR:
Non-small cell lung cancer: Epidemiology, screening, diagnosis, and
treatment. Mayo Clin Proc. 94:1623–1640. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goldstraw P, Chansky K, Crowley J,
Rami-Porta R, Asamura H, Eberhardt WE, Nicholson AG, Groome P,
Mitchell A, Bolejack V, et al: The IASLC lung cancer staging
project: Proposals for revision of the TNM stage groupings in the
forthcoming (Eighth) Edition of the TNM classification for lung
cancer. J Thorac Oncol. 11:39–51. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Motta-Guerrero R, Leon Garrido-Lecca A,
Failoc-Rojas VE, Calle-Villavicencio A, Villacorta-Carranza R,
Huerta-Collado Y, Torres-Mera A, Valladares-Garrido MJ,
Rivera-Francia V, Carracedo C and Raez L: Effectiveness and safety
of the bevacizumab and erlotinib combination versus erlotinib alone
in EGFR mutant metastatic non-small-cell lung cancer: Systematic
review and meta-analysis. Front Oncol. 13:13353732024. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zeng L, Cook RJ, Wen L and Boruvka A: Bias
in progression-free survival analysis due to intermittent
assessment of progression. Stat Med. 34:3181–3193. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Imai H, Kaira K and Minato K: Clinical
significance of post-progression survival in lung cancer. Thorac
Cancer. 8:379–386. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li J, Zhu H, Sun L, Xu W and Wang X:
Prognostic value of site-specific metastases in lung cancer: A
population based study. J Cancer. 10:3079–3086. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li Y, Wong M, Zhan L, Corke L, Brown MC,
Cheng S, Khan K, Balatnaram K, Chowdhury M, Sabouhanian A, et al:
Single organ metastatic sites in non-small cell lung cancer:
Patient characteristics, treatment patterns and outcomes from a
large retrospective Canadian cohort. Lung Cancer. 192:1078232024.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tseng CH, Chiang CJ, Tseng JS, Yang TY,
Hsu KH, Chen KC, Wang CL, Chen CY, Yen SH, Tsai CM, et al: EGFR
mutation, smoking, and gender in advanced lung adenocarcinoma.
Oncotarget. 8:98384–98393. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hsu CH, Tseng CH, Chiang CJ, Hsu KH, Tseng
JS, Chen KC, Wang CL, Chen CY, Yen SH, Chiu CH, et al:
Characteristics of young lung cancer: Analysis of Taiwan's
nationwide lung cancer registry focusing on epidermal growth factor
receptor mutation and smoking status. Oncotarget. 7:46628–46635.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kim TH, Choi JH, Ahn MS, Lee HW, Kang SY,
Choi YW, Koh YW and Sheen SS: Differential efficacy of tyrosine
kinase inhibitors according to the types of EGFR mutations and
agents in non-small cell lung cancer: A real-world study. BMC
Cancer. 24:702024. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hou J, Li H, Ma S, He Z, Yang S, Hao L,
Zhou H, Zhang Z, Han J, Wang L and Wang Q: EGFR exon 20 insertion
mutations in advanced non-small-cell lung cancer: Current status
and perspectives. Biomark Res. 10:212022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Beau-Faller M, Prim N, Ruppert AM,
Nanni-Metellus I, Lacave R, Lacroix L, Escande F, Lizard S, Pretet
JL, Rouquette I, et al: Rare EGFR exon 18 and exon 20 mutations in
non-small-cell lung cancer on 10 117 patients: A multicentre
observational study by the French ERMETIC-IFCT network. Ann Oncol.
25:126–131. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rossi S, Damiano P, Toschi L, Finocchiaro
G, Giordano L, Marinello A, Bria E, D'Argento E and Santoro A:
Uncommon single and compound EGFR mutations: Clinical outcomes of a
heterogeneous subgroup of NSCLC. Curr Probl Cancer. 46:1007872022.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lin CW, Huang KY, Lin CH, Hou MH and Lin
SH: Diverse clinical outcomes for the EGFR-mutated and
ALK-rearranged advanced non-squamous non-small cell lung cancer.
Oncol Lett. 29:1252025. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shimamura SS, Shukuya T, Asao T, Hayakawa
D, Kurokawa K, Xu S, Miura K, Mitsuishi Y, Tajima K, Shibayama R,
et al: Survival past five years with advanced, EGFR-mutated or
ALK-rearranged non-small cell lung cancer-is there a ‘tail plateau’
in the survival curve of these patients? BMC Cancer. 22:3232022.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Colomban O, Tod M, Peron J, Perren TJ,
Leary A, Cook AD, Sajous C, Freyer G and You B: Bevacizumab for
newly diagnosed ovarian cancers: Best candidates among high-risk
disease patients (ICON-7). JNCI Cancer Spectr. 4:pkaa0262020.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tewari KS, Burger RA, Enserro D, Norquist
BM, Swisher EM, Brady MF, Bookman MA, Fleming GF, Huang H, Homesley
HD, et al: Final overall survival of a Randomized trial of
bevacizumab for primary treatment of ovarian cancer. J Clin Oncol.
37:2317–2328. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Reck M, Von Pawel J, Zatloukal P, Ramlau
R, Gorbounova V, Hirsh V, Leighl N, Mezger J, Archer V, Moore N, et
al: Overall survival with cisplatin-gemcitabine and bevacizumab or
placebo as first-line therapy for nonsquamous non-small-cell lung
cancer: Results from a randomised phase III trial (AVAiL). Ann
Oncol. 21:1804–1809. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sato H, Nagashima H, Akiyama M, Ito T,
Hashimoto T, Saikawa H, Utsumi Y and Maemondo M: Analysis of
bevacizumab treatments and metastatic sites of lung cancer. Cancer
Treat Res Commun. 26:1002902021.PubMed/NCBI
|
|
23
|
Chen G, Huang AC, Zhang W, Zhang G, Wu M,
Xu W, Yu Z, Yang J, Wang B, Sun H, et al: Exosomal PD-L1
contributes to immunosuppression and is associated with anti-PD-1
response. Nature. 560:382–386. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ren M, Dai B, Kong YY, Lv JJ and Cai X:
PD-L1 expression in tumour-infiltrating lymphocytes is a poor
prognostic factor for primary acral melanoma patients.
Histopathology. 73:386–396. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhou ZJ, Zhan P and Song Y: PD-L1
over-expression and survival in patients with non-small cell lung
cancer: A meta-analysis. Transl Lung Cancer Res. 4:203–208. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tang Y, Fang W, Zhang Y, Hong S, Kang S,
Yan Y, Chen N, Zhan J, He X, Qin T, et al: The association between
PD-L1 and EGFR status and the prognostic value of PD-L1 in advanced
non-small cell lung cancer patients treated with EGFR-TKIs.
Oncotarget. 6:14209–14219. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Qin Y, Jiang L, Yu M, Li Y, Zhou X, Wang
Y, Gong Y, Peng F, Zhu J, Liu Y, et al: PD-L1 expression is a
promising predictor of survival in patients with advanced lung
adenocarcinoma undergoing pemetrexed maintenance therapy. Sci Rep.
10:161502020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kinoshita F, Takenaka T, Yamashita T,
Matsumoto K, Oku Y, Ono Y, Wakasu S, Haratake N, Tagawa T,
Nakashima N and Mori M: Development of artificial intelligence
prognostic model for surgically resected non-small cell lung
cancer. Sci Rep. 13:156832023. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen HY, Chen CH, Liao WC, Lin YC, Chen
HJ, Hsia TC, Cheng WC and Tu CY: Optimal first-line treatment for
EGFR-mutated NSCLC: A comparative analysis of osimertinib and
second-generation EGFR-TKIs. BMC Pulm Med. 24:5172024. View Article : Google Scholar : PubMed/NCBI
|