Introduction
Among the gynecological malignancies, ovarian cancer
has the highest mortality rate and seriously threatens the lives
and health of women. In 2022, there were an estimated 324,398 new
cases of ovarian cancer and 206,839 related deaths worldwide
(1). Currently, the first-line
treatments for advanced-stage epithelial ovarian cancer include
tumor reduction surgery, platinum-based chemotherapy and
maintenance therapy with a poly(ADP-ribose) polymerase inhibitor
(PARPi) (2,3). Neutropenia is the most common
hematological toxicity observed during chemotherapy. The National
Comprehensive Cancer Network guidelines recommend the use of
pegylated recombinant human granulocyte colony-stimulating factor
(PEG-rhG-CSF) for the prevention of chemotherapy-induced
neutropenia (4). rhG-CSF binds to
specific G-CSF receptors on the surface of granulocytes and
stimulates the differentiation and proliferation of neutrophil
progenitor cells. PEG-rhG-CSF is chemically modified with
polyethylene glycol to increase its molecular weight and reduce the
glomerular filtration rate, resulting in an extended half-life,
enhanced water solubility, reduced immunogenicity and a prolonged
action time within the body; some clinical studies have confirmed
its efficacy and safety in the context of chemotherapy for
malignant tumors (5–9). However, these studies only assessed
the short-term efficacy of PEG-rhG-CSF and failed to observe
long-term changes in neutrophils and leukocytes.
Studies have demonstrated that chemoradiotherapy can
induce the aging of hematopoietic stem cells (10–12).
The administration G-CSF following the repeated use of cytotoxic
drugs appears to damage bone marrow hematopoietic function
(13). This is related to the
direct or indirect induction of stem cell differentiation into more
lineage-committed hematopoietic cells by G-CSF, resulting in the
depletion of bone marrow reserves (14,15).
Patients with ovarian cancer may already have potential bone marrow
damage after undergoing multiple cycles of chemotherapy. However,
due to the administration of G-CSF, patients may still have normal
blood cell counts in the short term despite reduced hematopoietic
stem cell (HSC) reserves. Therefore, potential bone marrow damage
is difficult to detect.
Patients with advanced-stage epithelial ovarian
cancer usually undergo maintenance therapy with PARPi for 2–3 years
following first-line chemotherapy. During this process, there is
also a risk of developing neutropenia, with an incidence rate
ranging from 16 to 58.8% (16). The
mechanism is associated with the PARP1-mediated suppression of
myeloid progenitor cell growth and the impaired ability to repair
granulocyte DNA damage (17).
However, the question of whether the prophylactic use of
PEG-rhG-CSF following first-line chemotherapy for ovarian cancer
will aggravate chemotherapy-induced long-term bone marrow damage,
thus affecting patients with later PARPi maintenance treatment, has
not yet been fully addressed. Thus, this is worthy of further
investigation.
The present retrospective cohort study observed
long-term changes in neutrophils and leukocytes in patients with
ovarian cancer receiving prophylactic injections of PEG-rhG-CSF
following the completion of first-line chemotherapy during
maintenance therapy with PARPi. Furthermore, the present study
explored the independent factors influencing neutrophils and
leukocyte levels.
Patients and methods
Patients
The present study included 62 female patients with a
median age of 54 years (range, 45–68 years). The inclusion criteria
were as follows: i) Patients who were diagnosed with ovarian cancer
pathologically; ii) stage II–IV disease [International Federation
of Gynecology and Obstetrics (FIGO) staging system, 2014] (18); iii) patients who had received
prophylactic PEG-rhG-CSF injections during the interchemotherapy
interval; and iv) patients who had received PARPi maintenance
therapy. The exclusion criteria were as follows: i) Tumors
originating from other sites; ii) those for whom PARPi therapy
could not be continued for severe side-effects or other reasons;
and iii) cases where clinical data were missing as the patients
were lost to follow-up.
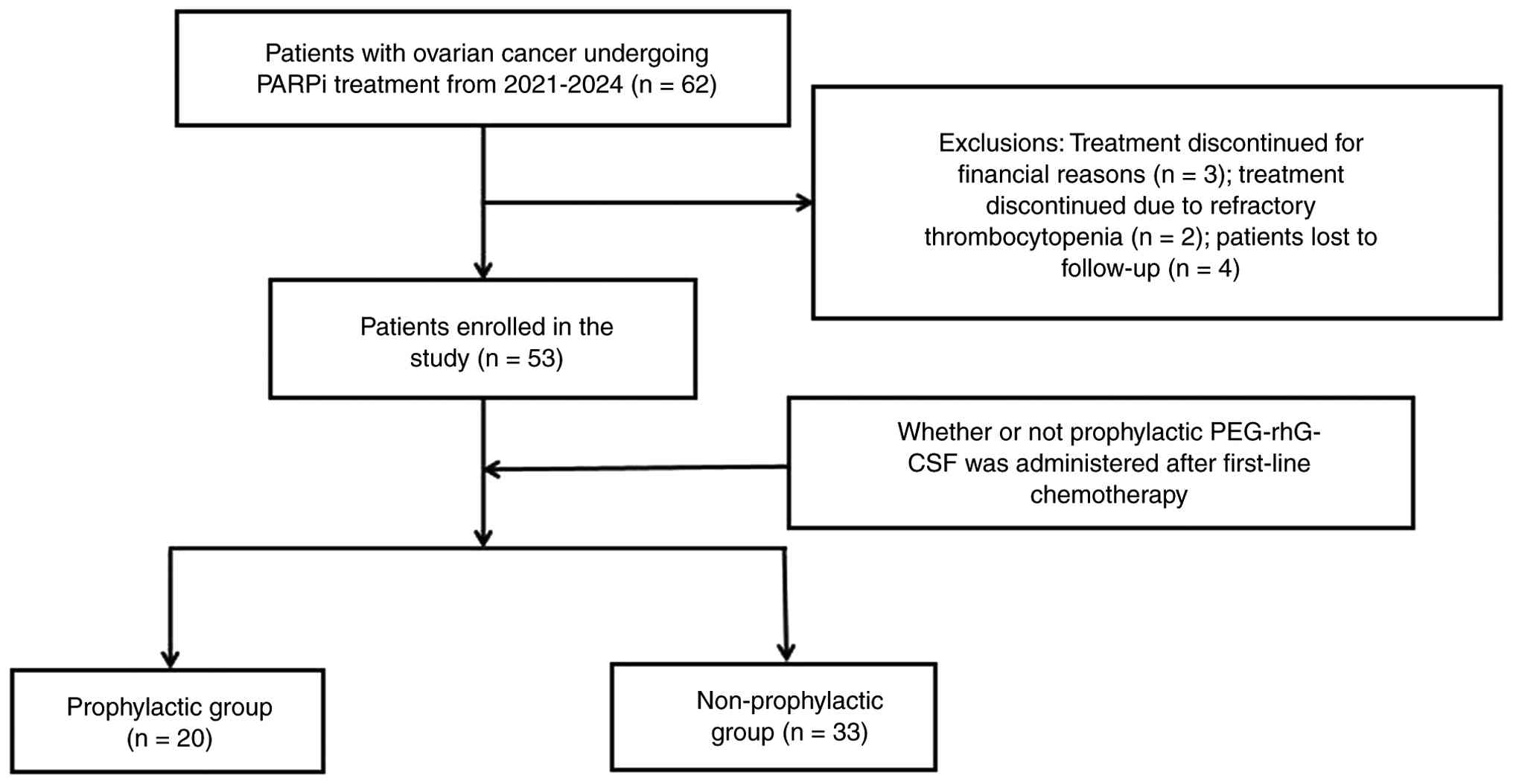
Study design and treatment
A total of 62 patients with primary ovarian cancer
who took niraparib/olaparib between March 2021 and January 2024
were included retrospectively. A total of 3 patients were excluded
for economic reasons, and 2 patients were excluded due to
refractory thrombocytopenia following treatment with PARPi. In
addition, 4 patients were lost to follow-up. Thus, a total of 53
patients were included in the study. The patients were divided into
the prophylactic group (n=20), receiving prophylactic PEG-rhG-CSF
injections at 24–48 h after the completion of first-line
chemotherapy, and the non-prophylactic group (n=33). The flow chart
of the study is presented in Fig.
1.
PEG-rhG-CSF medication regimen
Patients in the prophylactic group were administered
subcutaneously with 6 mg PEG-rhG-CSF at 24–48 h after the
completion of first-line chemotherapy. The non-prophylactic group
did not receive PEG-rhG-CSF following the completion of first-line
chemotherapy. According to the Chinese expert consensus, when the
neutrophil count of the patients was <1×109/l, 150 µg
rhG-CSF were injected until it reached ≥2×109/l
(19). The carboplatin plus
paclitaxel (TC) regimen is a chemotherapy regimen associated with a
moderate risk of developing febrile neutropenia (FN) (20). If the patient had ≥1 risk factor, or
if a FN or dose-limiting neutropenia event occurred in the previous
cycle of chemotherapy without the prophylactic use of G-CSF, and
the medication was agreed by the patient and their family,
PEG-rhG-CSF would be administered prophylactically at 24–48 h after
the completion of chemotherapy. The ensuing chemotherapy session
needed to be at least 14 days apart from the PEG-rhG-CSF
injection.
Chemotherapy medication regimen
Patients in both groups received platinum-based
first-line chemotherapy, with the majority completing 6–8 cycles.
The TC regimen was the most commonly used. Patients received
paclitaxel at a dose of 175 mg/m2 administered as a 3-h
infusion, followed by carboplatin administered as a 1-h infusion
with an area under the curve (AUC) of 5–6. The glomerular
filtration rate (GFR) was estimated using creatinine clearance. The
carboplatin dose was calculated with the Calvert formula:
Carboplatin dose in milligrams=AUC × (GFR + 25). Courses were
administered on a 21-day schedule. Prior to each medication,
patients were administered 8 mg ondansetron to prevent vomiting and
10 mg dexamethasone to prevent allergic symptoms. At the same time,
strict electrocardiographic monitoring was performed during the
medication.
PARPi medication regimen
If patients achieved a complete response or partial
response 1 month after the completion of first-line chemotherapy
with stable blood and organ function tests, PARPi treatment was
recommended for administration as soon as possible. The starting
dose of olaparib was 300 mg twice daily, and the starting dose of
niraparib was determined according to the weight and platelet level
of the patient. The starting dose for patients with a body weight
≥77 kg or a baseline platelet count ≥150×109/l was 300
mg once daily, while the starting dose for patients weighing <77
kg or with a baseline platelet count <150×109/l was
200 mg once daily. Following the initiation of PARPi treatment,
routine blood samples were monitored weekly in month 1, then every
month for year 1 and periodically thereafter. The drug dose was
adjusted according to the degree of adverse hematological reactions
observed, and the related symptoms were treated appropriately.
PARPi was permanently discontinued if hematological toxicity did
not resolve after a 28-day treatment interruption, and the patient
should be referred to a hematologist for additional evaluation and
treatment.
Outcomes
The primary study outcomes were the changes in
neutrophils and leukocytes in the two groups. The secondary study
outcomes were the changes in hemoglobin and platelet counts in the
two groups. Multivariate linear regression was used to analyze the
independent influencing factors of the primary outcome. The time
points observed were 1, 2, 3 and 6 months, and 1 year following the
administration of PARPi.
Statistical analysis
IBM SPSS Statistics 27.0 software (IBM Corp.) was
used for the data analysis. For continuous data, the data were
analyzed via the Mann-Whitney U test and are expressed as the
median (Q1, Q3). For classified data, the measured data were
analyzed using the χ2 test and Fisher's exact test. The
Mann-Whitney U test with Benjamini-Hochberg correction (false
discovery rate=0.1) was conducted to compare repeated measurement
indicators between the two groups. Statistical significance for
this analysis was defined as a corrected P-value below the adjusted
significance threshold, with the corrected P-values for each index
as follows: Leukocytes, P<0.08; neutrophils, P<0.06;
hemoglobin, P<0.04; and platelets, P<0.02. Multivariate
linear regression analysis was performed to identify independent
factors associated with the primary endpoint, for which two-sided
P<0.05 was considered to indicate a statistically significant
difference.
Results
In the present study, 62 patients with ovarian
cancer treated between March 2021 and January 2024 were included,
with a median age of 54.00 years (51.75–59.00 years) and a median
body mass index (BMI) of 22.83 kg/m2 (21.29–26.48
kg/m2). Among these, 58.49% of the patients had the
pathological type of high-grade serous carcinoma (HGSC), 92.45% had
stage IIIA-IVB disease and 88.68% were treated with niraparib
(Table I). As shown in Fig. 1, 2 patients could not continue PARPi
treatment due to refractory thrombocytopenia, the treatment of 3
patients was discontinued for economic reasons and 4 patients were
lost to follow-up; thus, 53 patients were ultimately included.
Among these, 20 patients received the prophylactic injection of
PEG-rhG-CSF following first-line chemotherapy (prophylactic group),
and 33 did not (non-prophylactic group).
 | Table I.Patient characteristics at baseline
before PARPi administration in the prophylactic (n=20) and
non-prophylactic (n=33) groups. |
Table I.
Patient characteristics at baseline
before PARPi administration in the prophylactic (n=20) and
non-prophylactic (n=33) groups.
|
Characteristics | All patients | Prophylactic
group | Non-prophylactic
group | P-value |
|---|
| Median age (Q1,
Q3), years | 54.0 (51.8,
59.0) | 54.5 (51.3,
58.8) | 54.0 (50.0,
59.5) | 0.934 |
| Median gravidity
(Q1, Q3) | 3.0 (2.0, 4.0) | 3.0 (2.0, 4.0) | 4.0 (3.0, 5.0) | 0.064 |
| Median parity (Q1,
Q3) | 2.0 (1.0, 2.0) | 2.0 (1.0, 2.0) | 2.0 (1.0, 2.0) | 0.732 |
| Median BMI (Q1,
Q3), kg/m2 | 22.8 (21.3,
26.5) | 21.9 (20.5,
25.4) | 23.7 (21.6,
27.1) | 0.061 |
| Classification of
diseases, n (%) |
|
|
| - |
|
EOC | 53 (100.0) | 20 (100.0) | 33 (100.0) |
|
| Pathological type,
n (%) |
|
|
| 0.121 |
|
HGSC | 31 (58.5) | 9 (45.0) | 22 (66.7) |
|
|
Others | 22 (41.5) | 11 (55.0) | 11 (33.3) |
|
| 2014 FIGO staging,
n (%) |
|
|
| 0.017 |
|
IIA-IIB | 4 (7.6) | 4 (20.0) | 0 (0.0) |
|
|
IIIA-IVB | 49 (92.5) | 16 (80.0) | 33 (100.0) |
|
| Type of PARPi, n
(%) |
|
|
| 0.269 |
|
Niraparib | 47 (88.7) | 16 (80.0) | 31 (93.9) |
|
|
Olaparib | 6 (11.3) | 4 (20.0) | 2 (6.1) |
|
| BRCA mutation, n
(%) |
|
|
| 0.081 |
|
BRCA1/2 | 14 (26.4) | 8 (40.0) | 6 (18.2) |
|
|
Others | 39 (73.6) | 12 (60.0) | 27 (81.8) |
|
| R0 status |
|
|
| 0.158 |
|
Yes | 20 (37.7) | 5 (25.0) | 15 (45.5) |
|
| No | 33 (62.3) | 15 (75.0) | 18 (54.5) |
|
| Received injections
of rhG-CSF, n (%) |
|
|
| 0.450 |
|
Yes | 23 (43.4) | 10 (50.0) | 13 (39.4) |
|
| No | 30 (56.6) | 10 (50.0) | 20 (60.6) |
|
| Number of
injections of PEG-rhG-CSF, n (%) |
|
|
| 0.093 |
| ≥5 | 26 (49.1) | 13 (65.0) | 13 (39.4) |
|
|
<5 | 27 (50.9) | 7 (35.0) | 20 (60.6) |
|
| Routine blood
(before PARPi) |
|
|
|
|
| Median
no. of leukocytes (Q1, Q3), ×109/l | 4.6 (3.9, 5.5) | 4.36 (3.8,
4.9) | 4.95 (3.9,
5.9) | 0.233 |
| Median
no. of neutrophils (Q1, Q3), ×109/l | 2.8 (2.2, 3.5) | 3.1 (2.2, 3.7) | 2.8 (2.0, 3.5) | 0.551 |
| Median
hemoglobin level (Q1, Q3), g/l | 114.0 (106.0,
122.5) | 111.5 (104.0,
123.8) | 115.0 (107.5,
121.5) | 0.274 |
| Median
no. of platelets (Q1, Q3), ×109/l | 159.0 (131.5,
204.5) | 148.5 (120.0,
192.5) | 162.0 (135.0,
209.5) | 0.263 |
The baseline characteristics of the two groups
before PARPi administration are summarized in Table I. The FIGO stages of the patients
were as follows: 4 patients in the prophylactic group had stage
IIA-IIB and 16 had stage IIIA-IVB, while all 33 patients in the
non-prophylactic group had stage IIIA-IVB. There were no
significant differences in gravidity, parity, age, BMI,
pathological classification, type of PARPi, BRCA mutation, the
complete removal of the macroscopic lesions (R0) status, the
receipt of injections of rhG-CSF or the number of injections of
PEG-rhG-CSF. Chemotherapy-related information is recorded in
Table SI. There were no
significant differences between the two groups in terms of the
chemotherapeutic regimen, the number of chemotherapies, cumulative
doses of paclitaxel and carboplatin, and the interval between the
end of chemotherapy and PARPi treatment. There were no significant
differences in leukocytes, neutrophils, hemoglobin and platelets
between the two groups prior to chemotherapy and prior to treatment
with PARPi (Tables I and SI).
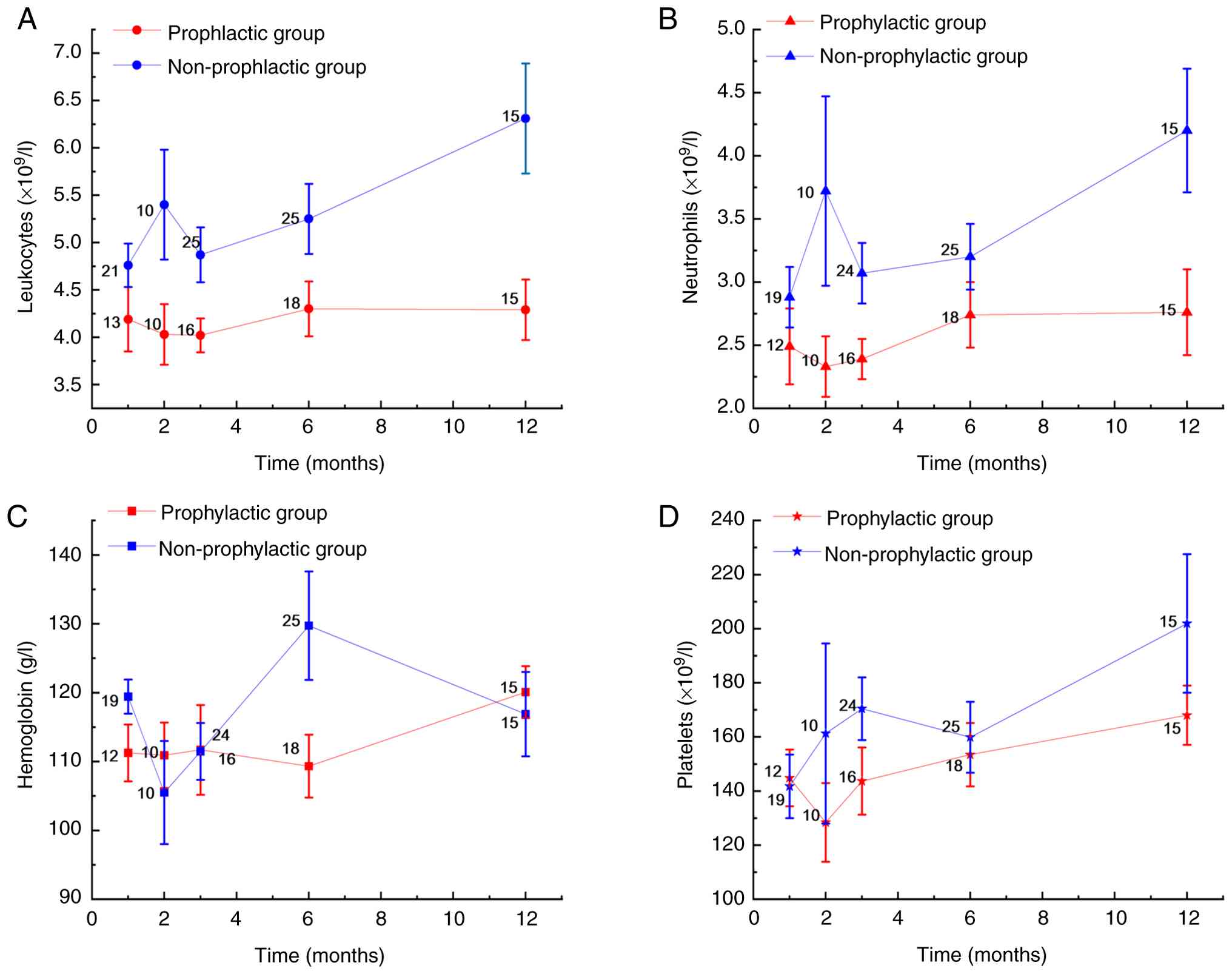
The changes in neutrophils, leukocytes, hemoglobin
and platelets following treatment with PARPi in both groups are
presented in Table II. Compared
with the non-prophylactic group, in the prophylactic group, the
leukocyte counts decreased significantly at month 2, month 6 and
year 1 (based on corrected P<0.08), the neutrophil counts
decreased significantly at month 3 and year 1 (based on corrected
P<0.06) and the hemoglobin level decreased significantly at
month 6 (based on corrected P<0.04). There were no statistically
significant differences in platelet counts between the two groups
at any time point. The trajectory of the routine blood test results
over time is illustrated in Fig. 2.
To investigate the significant difference between the two groups in
leukocyte and neutrophil counts at the 1-year, multiple linear
regression models were constructed separately with these counts as
dependent variables. Multivariate linear regression analysis
revealed that at 1 year following PARPi treatment, whereas parity
was positively associated with leukocytes and neutrophils
(P<0.05) (Tables III and
IV). Leukocytes in patients with
HGSC decreased by 1.964×109/l, and the prophylactic
injection of PEG-rhG-CSF following first-line chemotherapy reduced
the leukocyte level by 2.370×109/l (P<0.05) (Table III). The number of neutrophils in
patients with HGSC decreased by 1.816×109/l, and the
prophylactic injection of PEG-rhG-CSF reduced the number of
neutrophils by 2.181×109/l (P<0.05) (Table IV). In general, parity,
pathological type and the prophylactic injection following the
completion of first-line chemotherapy were significantly associated
with lower levels of leukocytes and neutrophils at 1 year following
the administration of PARPi (P<0.05) (Tables III and IV).
 | Table II.Changes in leukocytes, neutrophils,
hemoglobin and platelets after poly(ADP-ribose) polymerase
inhibitor administration in the prophylactic (n=20) and
non-prophylactic group (n=33) groups. |
Table II.
Changes in leukocytes, neutrophils,
hemoglobin and platelets after poly(ADP-ribose) polymerase
inhibitor administration in the prophylactic (n=20) and
non-prophylactic group (n=33) groups.
| A, First month |
|---|
|
|---|
| Routine blood | Prophylactic
group | Non-prophylactic
group | P-value (raw) | P(corr)
(FDR=0.1) |
|---|
| Median no. of
leukocytes (Q1, Q3), ×109/l | 3.9 (3.1, 5.0) | 3.6 (3.4, 3.7) | 0.246 | 0.10 |
| Median no. of
neutrophils (Q1, Q3), ×109/l | 2.5 (0.9, 2.9) | 2.1 (1.5, 2.6) | 0.484 | 0.10 |
| Median hemoglobin
level (Q1, Q3), g/l | 108.5 (93.5,
118.8) | 109.5 (105.8,
116.3) | 0.367 | 0.04 |
| Median no. of
platelets (Q1, Q3), ×109/l | 136.5 (102.0,
174.3) | 99.5 (87.3,
168.0) | 0.646 | 0.04 |
|
| B, Second
month |
|
| Routine
blood | Prophylactic
group | Non-prophylactic
group | P-value
(raw) | P(corr)
(FDR=0.1) |
|
| Median no. of
leukocytes (Q1, Q3), ×109/l | 3.7 (3.3, 4.4) | 4.9 (3.9,
10.7) | 0.027 | 0.04 |
| Median no. of
neutrophils (Q1, Q3), ×109/l | 1.8 (1.6, 2.5) | 2.8 (2.4, 8.2) | 0.063 | 0.06 |
| Median hemoglobin
level (Q1, Q3), g/l | 104.5 (91.3,
121.8) | 112.5 (79.0,
113.8) | 0.684 | 0.08 |
| Median no. of
platelets (Q1, Q3), ×109/l | 102.0 (75.8,
130.5) | 114.0 (71.3,
355.5) | 0.853 | 0.08 |
|
| C, Third
month |
|
| Routine
blood | Prophylactic
group | Non-prophylactic
group | P-value
(raw) | P(corr)
(FDR=0.1) |
|
| Median no. of
leukocytes (Q1, Q3), ×109/l | 4.1 (2.9, 4.6) | 4.1 (3.0, 5.6) | 0.119 | 0.08 |
| Median no. of
neutrophils (Q1, Q3), ×109/l | 1.8 (1.3, 2.6) | 2.5 (1.7, 3.6) | 0.048 | 0.04 |
| Median hemoglobin
level (Q1, Q3), g/l | 99.5 (77.5,
132.0) | 107.5 (94.5,
119.8) | 0.594 | 0.06 |
| Median no. of
platelets (Q1, Q3), ×109/l | 129.0 (84.3,
144.0) | 141.0 (80.5,
272.0) | 0.075 | 0.02 |
|
| D, Sixth
month |
|
| Routine
blood | Prophylactic
group | Non-prophylactic
group | P-value
(raw) | P(corr)
(FDR=0.1) |
|
| Median no. of
leukocytes (Q1, Q3), ×109/l | 3.9 (3.7, 4.3) | 4.7 (4.1, 6.5) | 0.053 | 0.06 |
| Median no. of
neutrophils (Q1, Q3), ×109/l | 2.1 (1.6, 2.6) | 3.0 (2.7, 4.0) | 0.115 | 0.08 |
| Median hemoglobin
level (Q1, Q3), g/l | 117.5 (109.8,
114.0) | 123.0 (124.5,
265.5) | 0.005 | 0.02 |
| Median no. of
platelets (Q1, Q3), ×109/l | 131.0 (100.3,
174.8) | 107.0 (96.5,
141.5) | 0.951 | 0.10 |
|
| E, First
year |
|
| Routine
blood | Prophylactic
group | Non-prophylactic
group | P-value
(raw) | P(corr)
(FDR=0.1) |
|
| Median no. of
leukocytes (Q1, Q3), ×109/l | 3.4 (3.1, 3.9) | 7.6 (4.3, 9.5) | 0.001 | 0.02 |
| Median no. of
neutrophils (Q1, Q3), ×109/l | 1.8 (1.5, 2.5) | 5.2 (2.9, 6.8) | 0.021 | 0.02 |
| Median hemoglobin
level (Q1, Q3), g/l | 122.0 (107.0,
127.3) | 105.0 (80.3,
140.3) | 0.967 | 0.10 |
| Median no. of
platelets (Q1, Q3), ×109/l | 146.5 (115.3,
185.5) | 177.5 (125.0,
401.8) | 0.683 | 0.06 |
 | Table III.Association between clinical
characteristics and leukocytes 1 year after PARPi
administration. |
Table III.
Association between clinical
characteristics and leukocytes 1 year after PARPi
administration.
| Variables | B | 95% CI | P-value | VIF |
|---|
| Age | −0.006 | (−0.109,
0.097) | 0.907 | 1.873 |
| BMI | −0.075 | (−0.286,
0.137) | 0.460 | 2.336 |
| Gravidity | −0.563 | (−1.334,
0.208) | 0.140 | 3.092 |
| Parity | 1.239 | (0.236, 2.242) | 0.019 | 3.944 |
| Pathology type | 1.964 | (0.509, 3.419) | 0.012 | 1.691 |
| 2014 FIGO
stage | −0.309 | (−2.472,
1.853) | 0.763 | 1.734 |
| Received
neoadjuvant chemotherapy | −0.208 | (−1.807,
1.391) | 0.785 | 1.970 |
| R0 status | −0.459 | (−2.065,
1.148) | 0.550 | 1.740 |
| Chemotherapy
medication regimen | 0.710 | (−1.166,
2.586) | 0.431 | 2.623 |
| Received
bevacizumab | 0.122 | (−1.866,
2.110) | 0.897 | 1.762 |
| Number of
chemotherapy cycles | −0.024 | (−0.758,
0.711) | 0.946 | 1.915 |
| Received injections
of rhG-CSF | 0.153 | (−1.850,
2.156) | 0.872 | 3.091 |
| Number of
PEG-rhG-CSF injections | −0.608 | (−2.366,
1.150) | 0.471 | 2.204 |
| Injection of
PEG-rhG-CSF after the last chemotherapy | 2.370 | (0.566, 4.173) | 0.014 | 2.610 |
| Type of PARPi | 1.419 | (−1.239,
4.076) | 0.271 | 2.040 |
 | Table IV.Association between clinical
characteristics and neutrophils after PARPi administration. |
Table IV.
Association between clinical
characteristics and neutrophils after PARPi administration.
| Variables | B | 95% CI | P-value | VIF |
|---|
| Age | −0.006 | (−0.095,
0.083) | 0.882 | 1.873 |
| BMI | −0.050 | (−0.233,
0.133) | 0.567 | 2.336 |
| Gravidity | −0.618 | (−1.284,
0.049) | 0.067 | 3.902 |
| Parity | 1.104 | (0.237, 1.971) | 0.016 | 3.944 |
| Pathology type | 1.816 | (0.558, 3.073) | 0.008 | 1.691 |
| 2014 FIGO
stage | −0.445 | (−2.314,
1.423) | 0.617 | 1.734 |
| Received
neoadjuvant chemotherapy | 0.244 | (−1.139,
1.626) | 0.711 | 1.970 |
| R0 status | −0.813 | (−2.202,
0.576) | 0.230 | 1.740 |
| Chemotherapy
medication regimen | 0.560 | (−1.061,
2.182) | 0.471 | 2.623 |
| Whether received
bevacizumab | 0.008 | (−1.711,
1.726) | 0.992 | 1.762 |
| Number of
chemotherapy cycles | −0.168 | (−0.802,
0.467) | 0.580 | 1.915 |
| Received injections
of rhG-CSF | −0.398 | (−2.219,
1.334) | 0.630 | 3.091 |
| Number of
PEG-rhG-CSF injections | −0.343 | (−1.863,
1.176) | 0.636 | 2.204 |
| Injection of
PEG-rhG-CSF after the last chemotherapy | 2.181 | (0.622, 3.740) | 0.010 | 2.610 |
| Type of PARPi | 2.002 | (−0.295,
4.299) | 0.083 | 2.040 |
Discussion
The present retrospective study investigated the
long-term effects of the prophylactic use of PEG-rhG-CSF on routine
blood parameters during PARPi maintenance therapy in patients with
ovarian cancer. It was found that the low levels of neutrophils,
leukocytes and hemoglobin in patients with ovarian cancer receiving
PARPi maintenance therapy may be associated with the prophylactic
injection of PEG-rhG-CSF following first-line chemotherapy. The
mechanism may be associated with impaired bone marrow
hematopoiesis.
As hematopoietic cells renew, long-term
myelosuppression can be defined as myelosuppression persisting 3
months after the completion of chemotherapy, primarily
characterized by neutrophil, platelet or hemoglobin levels below
the normal ranges (21,22). The present study found that
injecting PEG-rhG-CSF prophylactically following first-line
chemotherapy was an independent influencing factor affecting
neutrophils and leukocytes at 1 year following PARPi treatment. In
other words, prophylactically injecting PEG-rhG-CSF following the
completion of first-line chemotherapy may be associated with low
levels of neutrophils and leukocytes, increasing the risk of
long-term myelosuppression. Although PEG-rhG-CSF is widely used in
clinical practice to prevent chemotherapy-induced myelosuppression,
little is known about the combined effects of chemotherapy and
PEG-rhG-CSF on the primitive stem cells responsible for long-term
hematopoietic support. van Os et al (13) reported that G-CSF administered after
multiple doses of cytotoxic agents appeared to impair long-term
hematopoiesis and marrow stem cell reserves. Other studies have
demonstrated that G-CSF combined with a cytotoxic agent can lead to
HSC damage and the loss of the bone marrow reserves, and that this
effect is more evident in animals with a damaged HSC compartment.
The mechanism involved is associated with G-CSF directly or
indirectly inducing stem cells to differentiate into more
hematopoietic-oriented cells, leading to loss of the bone marrow
reserve (14,15). These studies provide a theoretical
basis for the possible bone marrow injury caused by G-CSF after
multiple doses of chemotherapy. Platinum drugs and paclitaxel are
common chemotherapeutics used to treat ovarian cancer, and they
exert cytotoxic effects by inhibiting DNA replication and cell
mitosis, respectively (23–25). Patients with ovarian cancer who are
injected with PEG-rhG-CSF following the repeated use of cytotoxic
drugs may experience the loss of the bone marrow stem cell reserve,
with lower levels of neutrophils and leukocytes. Neutrophils are
the most abundant type of granulocyte in the human body, accounting
for 40–70% of white blood cells; they are a key component of the
innate immune system and are mainly responsible for resisting
extracellular pathogens and acute inflammatory reactions. Patients
with neutropenia are susceptible to pathogen attacks and are at an
increased risk of acquiring infections (26).
In addition, studies have reported that G-CSF
stimulates neutrophil elevation, while causing the suppression of
bone marrow erythropoiesis (27,28),
which may be related with the results observed within the present
study at 6 months after PARPi treatment. This may be linked to the
induction of the over-mobilization of bone marrow cells by G-CSF,
leading to the disruption of the bone marrow microenvironment,
which in turn leads to bone marrow dysfunction (29). Moreover, for patients requiring
maintenance treatment, PARPi itself can also cause myelosuppression
(16,30), and the overall incidence of anemia
is 21.0–70.8%. In the event that the bone marrow stem cell reserve
is already impaired prior to PARPi treatment, patients may be at a
high risk of myelosuppression during PARPi maintenance therapy.
Therefore, for patients with ovarian cancer requiring PARPi
maintenance therapy, the use of prophylactic PEG-rhG-CSF following
the completion of first-line chemotherapy needs to be
reconsidered.
HGSC is the most common subtype of ovarian cancer,
with a high degree of malignancy and a poor prognosis (31,32).
In the present study, patients with HGSC had lower levels of
neutrophils and leukocytes than those with other pathological types
during subsequent PARPi maintenance therapy for ovarian cancer.
Huang et al (22) reported
that among patients with long-term myelosuppression and ovarian
cancer, the proportion of patients with pathological grade 2–3 and
stage IV disease was greater. The findings of the present study are
consistent with these results. Notably, the present study also
found that the parity of the patients was positively associated
with neutrophil and leukocyte counts during PARPi treatment. The
mechanism involved may be associated with the suppression of
retrotransposons in HSCs during pregnancy, which activate cyclic
GMP-AMP synthase and stimulator of interferon gene signaling and
trigger the interferon response, thus promoting the activation of
HSCs and increasing the production of neutrophils (33).
To reduce the incidence of severe myelosuppression
and FN in patients during chemotherapy, PEG-rhG-CSF is increasingly
being used for malignant tumors (34–36).
However, only a limited number of studies have mentioned its effect
on long-term myelosuppression in patients. For patients with
advanced-stage epithelial ovarian cancer, PARPi can significantly
prolong progression-free survival time (37–39).
Therefore, the indications for the use of PEG-rhG-CSF following the
completion of first-line chemotherapy should be strictly controlled
to avoid affecting the subsequent maintenance therapy in patients
with ovarian cancer. It is also critical to develop novel treatment
methods and ensure the safety of patients administered medication
during maintenance treatment.
The present study strictly followed clinical
medication specifications. PEG-rhG-CSF was injected 24–48 h after
the completion of chemotherapy, and the evaluation of whether to
implement PARPi treatment began at least 14 days after the
injection of PEG-rhG-CSF. This avoided the short intervals between
chemotherapy, PEG-rhG-CSF injection and targeted therapy, which
induce potential bone marrow suppression. However, the present
study also has some limitations. First, the opportunistic,
single-center, retrospective study design resulted in the cohort
being established based on the available clinical data of patients
treated over a specific 3-year period, rather than via prospective
enrollment. The use of multivariate linear regression adds value;
however, the relatively small sample, resulting from the low
incidence of ovarian cancer and strict inclusion criteria requiring
concomitant PEG-rhG-CSF and PARP inhibitor therapy, may limit
statistical power and model stability. Second, although a detailed
analysis of potential outcome-related factors (such as baseline
bone marrow reserve, number of chemotherapy cycles, chemotherapy
regimen, and chemotherapy dosage) was conducted, the complexity of
clinical decision-making and the potential biases it may introduce
may be unavoidable. For example, during PARPi therapy, patients may
face treatment interruptions and dose adjustment. Third, the
present retrospective study relied on previous medical records, and
thus, data on some indicators may be lacking. Nonetheless, this
pragmatic design was critical for investigating this specific
clinical situation and offers a foundational real-world evaluation
that can guide future, larger prospective studies. Additionally,
the study planned to analyze the impact of neutropenia and FN on
long-term myelosuppression. However, since patients with ovarian
cancer have some risk factors for the occurrence of FN, most
patients in the study received primary prevention according to
expert consensus, resulting in an extremely low incidence of
neutropenia and FN. Analyzing their impact on long-term
myelosuppression was somewhat challenging. Therefore, further
prospective studies or even multicenter studies are required in the
future to explore the potential impact of the prophylactic
injection of PEG-rhG-CSF during maintenance treatment in patients
with ovarian cancer in order to expand the current findings and
ensure the accuracy of the results. Further basic research is
warranted to verify the findings presented, to determine whether
patients with ovarian cancer already experience HSC aging following
multiple rounds of chemotherapy and to determine whether the
prophylactic injection of PEG-rhG-CSF following multiple rounds of
chemotherapy can aggravate bone marrow damage.
In conclusion, myelosuppression may lead to the
interruption of PARPi treatment, thereby affecting the treatment
efficacy in patients with ovarian cancer. The present study found
that low levels of neutrophils, leukocytes and hemoglobin in
patients with ovarian cancer undergoing PARPi maintenance therapy
may be associated with the prophylactic injection of PEG-rhG-CSF
following first-line chemotherapy. Therefore, in order to improve
the safety of PARPi therapy and reduce the risk of neutropenia and
subsequent infection in patients with ovarian cancer, the
prophylactic use of PEG-rhG-CSF following first-line chemotherapy
needs to be carefully considered. Future initiatives should also
concentrate on creating cutting-edge methods for managing and
preventing neutropenia. Furthermore, in order to confirm these
results and remove the inherent constraints of confounding by
indication in retrospective investigations such as the present
study, prospective, randomized controlled trials are necessary.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
This research was partially supported by the Beijing Life Oasis
Public Welfare Service Center (grant no. cphcf-2022-104), the
Sichuan Provincial Medical Science and Technology Innovation
Research Association Project (grant no. YCH-KY-YCZD2024-091) and
the Doctoral Initiation Fund Project (Natural Science) (grant no.
CBY21-QD23).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YZ and MY contributed to the design of the study and
revised the manuscript. WZ contributed to the analysis of data and
drafted the manuscript. NT, YW and TF contributed to the
acquisition, analysis and interpretation of data. WZ and YZ confirm
the authenticity of all the raw data. All authors have read and
approved the manuscript.
Ethics approval and consent to
participate
The present retrospective cohort study was approved
by the Ethics Committee of North Sichuan Medical College (Nanchong,
China; approval no. 2025ER698-1). The committee granted a waiver of
informed consent to participate as the study posed no more than
minimal risk to participants and as the waiver did not adversely
affect their rights or welfare.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Caruso G, Weroha SJ and Cliby W: Ovarian
cancer: A review. JAMA. 334:1278–1291. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
O'Malley DM, Krivak TC, Kabil N, Munley J
and Moore KN: PARP inhibitors in ovarian cancer: A review. Target
Oncol. 18:471–503. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liu J, Berchuck A, Backes FJ, Cohen J,
Grisham R, Leath CA, Martin L, Matei D, Miller DS, Robertson S, et
al: NCCN Guidelines® Insights: Ovarian cancer/fallopian
tube cancer/primary peritoneal cancer, version 3.2024. J Natl Compr
Canc Netw. 22:512–519. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Becker PS, Griffiths EA, Alwan LM,
Bachiashvili K, Brown A, Cool R, Curtin P, Dinner S, Gojo I, Hicks
A, et al: NCCN guidelines insights: Hematopoietic growth factors,
version 1.2020. J Natl Compr Canc Netw. 18:12–22. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen H, Ma Y, Wang D, Wu H, Zhang M, Xu Y
and Cheng S: The efficacy and safety of the prophylactic
application of PEG-rhG-CSF in radiotherapy with weekly concurrent
chemotherapy for cervical cancer. BMC Cancer. 24:14052024.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Petru E, Singer CF, Polterauer S, Galid A,
Schauer C, Klocker J, Seifert M, Reinthaller A, Benedicic C,
Hubalek M, et al: Prophylactic long-acting granulocyte-colony
stimulating factors (G-CSF) in gynecologic malignancies: An
oncologic expert statement. Wien Med Wochenschr. 165:387–394. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Huang J, Zhu J, Jiang L, Xu J, Lin X,
Chang J, Zhang X, Lu S, Sun F, Wang J, et al: Efficacy, safety, and
cost-effectiveness of pegylated PEG-rhg-CSF in pediatric patients
receiving high-intensity chemotherapy: results from a phase II
study. Front Pharmacol. 15:14193692024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
You J, Yuan Y, Gu X, Wang W and Li X:
Pegylated recombinant human granulocyte colony-stimulating factor
for primary prophylaxis of neutropenia in patients with cervical
cancer receiving concurrent chemoradiotherapy: a prospective study.
BMC Cancer. 24:8332024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jiang Y, Zhang J, Zhong J, Liao H, Zhang
J, Liu Y, Liang Y and Li H: Efficacy and safety of PEG-rhG-CSF
versus rhG-CSF in preventing chemotherapy-induced-neutropenia in
early-stage breast cancer patients. BMC Cancer. 23:7022023.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Meng A, Wang Y, Van Zant G and Zhou D:
Ionizing radiation and busulfan induce premature senescence in
murine bone marrow hematopoietic cells. Cancer Res. 63:5414–5419.
2003.PubMed/NCBI
|
|
11
|
Wang Y, Schulte BA and Zhou D:
Hematopoietic stem cell senescence and long-term bone marrow
injury. Cell Cycle. 5:35–38. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Obeng EA and Gruber TA: The development of
therapy related myeloid neoplasms in childhood cancer survivors.
Trends Cancer. 8:790–791. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
van Os R, Robinson S, Sheridan T, Mislow
JM, Dawes D and Mauch PM: Granulocyte colony-stimulating factor
enhances bone marrow stem cell damage caused by repeated
administration of cytotoxic agents. Blood. 92:1950–1956. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
van Os R, Robinson S, Sheridan T and Mauch
PM: Granulocyte-colony stimulating factor impedes recovery from
damage caused by cytotoxic agents through increased differentiation
at the expense of self-renewal. Stem Cells. 18:120–127. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li C, Lu L, Zhang J, Huang S, Xing Y, Zhao
M, Zhou D, Li D and Meng A: Granulocyte colony-stimulating factor
exacerbates hematopoietic stem cell injury after irradiation. Cell
Biosci. 5:652015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hopkins TA, Ainsworth WB, Ellis PA,
Donawho CK, DiGiammarino EL, Panchal SC, Abraham VC, Algire MA, Shi
Y, Olson AM, et al: PARP1 Trapping by PARP inhibitors drives
cytotoxicity in both cancer cells and healthy bone marrow. Mol
Cancer Res. 17:409–419. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ponath V, Heylmann D, Haak T, Woods K,
Becker H and Kaina B: Compromised DNA repair and signalling in
human granulocytes. J Innate Immun. 11:74–85. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Prat J; FIGO Committee on Gynecologic
Oncology, : Staging classification for cancer of the ovary,
fallopian tube, and peritoneum. Int J Gynaecol Obstet. 124:1–5.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ba Y, Shi Y, Jiang W, Feng J, Cheng Y,
Xiao L, Zhang Q, Qiu W, Xu B, Xu R, et al: Current management of
chemotherapy-induced neutropenia in adults: key points and new
challenges: Committee of Neoplastic Supportive-Care (CONS), China
anti-cancer association committee of clinical chemotherapy, China
anti-cancer association. Cancer Biol Med. 17:896–909. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
The Society of Chemotherapy and Chinese
Anti-Cancer Association, Committee of Neoplastic Supportive-Care
(CONS), China Anti-Cancer Association, . Consensus on the clinical
diagnosis, treatment, and prevention of chemotherapy- induced
neutropenia in China (2019 edition). Chin J Clin Oncol. 46:876–822.
2019.(In Chinese).
|
|
21
|
Chinese Society of Clinical Oncology
(CSCO) Expert Committee on Integrative Medicine, . Expert consensus
on the integrated traditional Chinese and Western medicine
treatment of bone marrow suppression caused by antitumor drugs.
Chinese Clinical Oncology. 26:1020–1027. 2021.(In Chinese).
|
|
22
|
Huang C, Wang T, Wu Z, Liang H, Wu Y and
Guo H: Preliminary Analysis on the Prognostic Factors of Long-term
Myelosuppression in Patients With Ovarian Epithelial Cancer After
Initial Treatment. Chin J Min Inv Surg. 23:241–246. 2023.(In
Chinese).
|
|
23
|
Wood GE and Ledermann JA: Adjuvant and
post-surgical treatment in high-grade epithelial ovarian cancer.
Best Pract Res Clin Obstet Gynaecol. 78:64–73. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Todd RC and Lippard SJ: Inhibition of
transcription by platinum antitumor compounds. Metallomics.
1:280–291. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schiff PB and Horwitz SB: Taxol stabilizes
microtubules in mouse fibroblast cells. Proc Natl Acad Sci USA.
77:1561–1565. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang F, Xia Y, Su J, Quan F, Zhou H, Li
Q, Feng Q, Lin C, Wang D and Jiang Z: Neutrophil diversity and
function in health and disease. Signal Transduct Target Ther.
9:3432024. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jing W, Guo X, Qin F, Li Y, Wang G, Bi Y,
Jin X, Han L, Dong X and Zhao Y: G-CSF shifts erythropoiesis from
bone marrow into spleen in the setting of systemic inflammation.
Life Sci Alliance. 4:e2020007372020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu M, Jin X, He X, Pan L, Zhang X and
Zhao Y: Macrophages support splenic erythropoiesis in 4T1
tumor-bearing mice. PLoS One. 10:e01219212015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cook KM, Sifri ZC, Baranski GM, Mohr AM
and Livingston DH: The role of plasma granulocyte colony
stimulating factor and bone marrow dysfunction after severe trauma.
J Am Coll Surg. 216:57–64. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Farrés J, Llacuna L, Martin-Caballero J,
Martínez C, Lozano JJ, Ampurdanés C, López-Contreras AJ, Florensa
L, Navarro J, Ottina E, et al: PARP-2 sustains erythropoiesis in
mice by limiting replicative stress in erythroid progenitors. Cell
Death Differ. 22:1144–1157. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
De Leo A, Santini D, Ceccarelli C,
Santandrea G, Palicelli A, Acquaviva G, Chiarucci F, Rosini F,
Ravegnini G, Pession A, et al: What is new on ovarian carcinoma:
Integrated morphologic and molecular analysis following the new
2020 world health organization classification of female genital
tumors. Diagnostics (Basel). 11:6972021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Armstrong DK, Alvarez RD, Bakkum-Gamez JN,
Barroilhet L, Behbakht K, Berchuck A, Chen LM, Cristea M, DeRosa M,
Eisenhauer EL, et al: Ovarian cancer, version 2.2020, NCCN clinical
practice guidelines in oncology. J Natl Compr Canc Netw.
19:191–226. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Phan J, Chen B, Zhao Z, Allies G,
Iannaccone A, Paul A, Cansiz F, Spina A, Leven AS, Gellhaus A, et
al: Retrotransposons are co-opted to activate hematopoietic stem
cells and erythropoiesis. Science. 386:eado68362024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sun L, Tian Y, Zhang S, Huang L, Ma J and
Han C: Impact of prophylactic Use of PEG-rhG-CSF on First-Line
immunochemotherapy in advanced NSCLC: A cohort study. JTO Clin Res
Rep. 6:1007802024.PubMed/NCBI
|
|
35
|
Yang L, Yu L, Du X, Cui Y and Du G: Study
of PEG-rhG-CSF for the prevention of neutropenia in concurrent
chemoradiotherapy for nasopharyngeal carcinoma. PLoS One.
20:e03150012025. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tominaga Y and Furukawa K: Clinical
experience with preventing chemotherapy-induced neutropenia in
breast cancer patients with different timings of pegylated
granulocyte colony-stimulating factor (PEG-G-CSF) injection: A case
series. Cureus. 17:e912582025.PubMed/NCBI
|
|
37
|
Banerjee S, Moore KN, Colombo N, Scambia
G, Kim BG, Oaknin A, Friedlander M, Lisyanskaya A, Floquet A, Leary
A, et al: Maintenance olaparib for patients with newly diagnosed
advanced ovarian cancer and a BRCA mutation (SOLO1/GOG 3004):
5-year follow-up of a randomised, double-blind, placebo-controlled,
phase 3 trial. Lancet Oncol. 22:1721–1731. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ray-Coquard I, Leary A, Pignata S, Cropet
C, González-Martín A, Marth C, Nagao S, Vergote I, Colombo N,
Mäenpää J, et al: Olaparib plus bevacizumab first-line maintenance
in ovarian cancer: final overall survival results from the
PAOLA-1/ENGOT-ov25 trial. Ann Oncol. 34:681–692. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
González-Martín A, Pothuri B, Vergote I,
DePont Christensen R, Graybill W, Mirza MR, McCormick C, Lorusso D,
Hoskins P, Freyer G, et al: Niraparib in patients with newly
diagnosed advanced ovarian cancer. N Engl J Med. 381:2391–2402.
2019. View Article : Google Scholar : PubMed/NCBI
|