Introduction
Breast cancer remains the second most frequently
diagnosed malignancy and a leading cause of cancer-related
mortality among female patients worldwide. The average annual
age-standardized incidence rate in female patients is 131.8 per
100,000. Stage at diagnosis represents the most important
prognostic determinant, with 5-year relative survival rates >99%
in localized disease, decreasing to approximately 87% in regional
disease and 32% in metastatic disease (1). Among its molecular subtypes,
triple-negative breast cancer (TNBC) accounts for 10–20% of all
cases and is defined by the absence of estrogen receptor (ER),
progesterone receptor (PR) and HER2 expression or amplification
(2). TNBC is recognized as a
biologically aggressive subtype, typically composed of high-grade,
highly proliferative tumors and is associated with limited
therapeutic options and a markedly worse prognosis compared with
hormone receptor-positive or HER2-enriched breast cancer types,
exhibiting notably lower 5-year survival rates (3).
Calcium-activated nucleotidase 1 (CANT1) is a
calcium-dependent glycoprotein with nucleotidase activity that
shares cDNA sequence homology with adenosine triphosphatases and
diphosphatases. It is expressed in a range of human tissues,
particularly in cartilage and other connective tissue, as well as
epithelial organs such as the prostate, liver and kidney, and can
also be secreted extracellularly (4). Functionally, CANT1 is predominantly
localized to the endoplasmic reticulum and Golgi apparatus, where
it hydrolyzes nucleoside diphosphates (such as UDP and GDP),
thereby regulating nucleotide homeostasis and supporting proper
protein glycosylation (5). CANT1
serves a key role in cell proliferation, differentiation and
cartilage matrix organization and that loss-of-function mutations
in CANT1 are associated with congenital skeletal dysplasias
(5,6).
Emerging evidence has suggested that elevated CANT1
expression may influence tumor biology by modulating cell
proliferation, apoptosis and extracellular signaling through
alterations in nucleotide metabolism and glycoprotein processing
(7). However, the prognostic
importance of CANT1 is context-dependent. In numerous malignancies,
such as prostate cancer, higher CANT1 expression has been
associated with favorable outcomes, whereas in others, including
hepatocellular carcinoma, it has been associated with poor
prognosis (7,8). To the best of our knowledge, CANT1
expression in breast cancer has not been characterized and no prior
studies have specifically examined its role in TNBC. Given the lack
of hormone receptor and HER2 signaling in TNBC, investigation of
CANT1 as a potential component of alternative molecular pathways in
this subtype may provide novel insights into its biological and
prognostic relevance.
Therefore, the present study aimed to evaluate CANT1
expression in TNBC and investigate its association with
clinicopathologicalal features and overall survival (OS). By
clarifying the prognostic importance of CANT1 in this setting, the
aim was to identify a potential biomarker for risk stratification
in this clinically challenging breast cancer subtype. The present
study specifically focused on TNBC as this subtype lacks
established therapeutic targets and validated prognostic
biomarkers. In hormone receptor-positive or HER2-positive breast
cancer types, strong biological drivers such as ER signaling or
HER2 amplification may dominate tumor behavior and potentially
confound the prognostic impact of additional molecular markers,
such as Ki-67, TP53 alteration or PIK3CA mutation. Furthermore,
through restricting analysis to TNBC, the present study aimed to
evaluate the prognostic relevance of CANT1 in a biologically
homogeneous setting requiring novel biomarkers.
Materials and methods
Study design and patient cohort
Within the present retrospective, single-center
study, consecutive patients with TNBC who underwent definitive
breast surgery between May 2017 and December 2024 and were followed
at the Department of Medical Oncology of Ankara City Hospital
(Ankara, Turkey) were included. All immunohistochemistry (IHC)
assessments were performed on surgical resection specimens of
primary breast tumors and adjacent tissue (distance, >5 mm);
preoperative core needle biopsies and metastatic tissues were not
analyzed.
Eligible patients had histologically confirmed
non-metastatic TNBC, available surgical tumor tissue for
immunohistochemical analysis and complete clinicopathological and
follow-up data. Patients with metastatic disease at diagnosis,
prior malignancy, inadequate tissue samples or missing follow-up
data were excluded. An initial cohort of 60 surgically treated TNBC
cases was screened, with 1 case excluded due to inadequate tissue
quality for reliable IHC evaluation. The final cohort comprised 59
patients, all of whom were female, with a median age of 55 years
(range, 28–78 years). Clinicopathological parameters including age
at diagnosis, histologic type and grade, tumor size, nodal status,
lymphovascular invasion, resection margins and adjuvant treatments
were retrieved from institutional electronic medical records and
pathology reports.
A post hoc power calculation was performed based on
the observed hazard ratio (HR) for OS between the H3 (high CANT1)
and H1 (low CANT1) groups (HR=0.149; 95% CI, 0.031–0.715). Using
Schoenfeld's approximation for the log-rank test with two-sided
significance level α=0.05, the observed number of events between
these groups (n=11) provided an estimated statistical power of 88%,
demonstrating that the present study possessed adequate power to
detect the observed survival difference between high and low CANT1
expression groups.
Specimen handling and sectioning
Representative formalin-fixed, paraffin-embedded
blocks were selected by a breast pathologist to ensure adequate
tumor cellularity and morphological preservation. Surgical
specimens were fixed in 10% neutral buffered formalin at room
temperature for 24–48 h. Serial 3 µm sections were cut on a Leica
RM2125RT rotary microtome (Leica Biosystems), mounted on charged
glass slides and baked to enhance adhesion. Scoring was performed
on areas of invasive carcinoma, excluding in situ
components, stromal elements, necrotic tissue and artifacts. When
numerous tumor blocks were available, the block with the most
representative viable invasive tumor was prioritized for staining;
if necessary, additional blocks were evaluated and the predominant
pattern was used for scoring, with heterogeneity documented
qualitatively.
IHC for CANT1
IHC was performed using the BOND MAX Fully Automated
IHC and ISH Staining System (Leica Biosystems) according to the
manufacturer's protocol to minimize inter-assay variability.
Heat-induced epitope retrieval was performed using epitope
retrieval 2 solution (Tris-EDTA based buffer; alkaline pH) under
controlled temperature and time settings. Endogenous peroxidase
activity was quenched using the ready-to-use BOND Peroxide Block
reagent (Leica Biosystems GmbH) according to the manufacturer's
instructions on the BOND-MAX automated staining platform.
The primary antibody used was anti-CANT1 rabbit
polyclonal (cat. no. HPA019627; Prestige Antibodies®;
Merck KGaA). The antibody was applied at a 1:300 for 30 min at room
temperature. Signal detection employed the Leica HRP conjugate BOND
polymer refine detection system (cat. no. DS9800; Leica
Biosystems). Chromogenic development with 3,3′ diaminobenzidine was
followed by hematoxylin counterstaining for 5 min at room
temperature, graded alcohol dehydration, xylene clearing and
coverslipping.
Each IHC run included negative controls prepared by
omitting the primary antibody and substituting using Rabbit
Negative Control Reagent (Leica Biosystems, Newcastle, United
Kingdom). Internal tissue controls were derived from adjacent
non-neoplastic ductal epithelium and benign breast parenchyma,
Stromal fibroblasts, lymphocytes and endothelial cells were
consistently non-reactive and served as internal negative
references.
Subcellular localization criteria
Based on the biochemical properties of CANT1, a
lumenal enzyme primarily localized to the Golgi apparatus and
endoplasmic reticulum, only cytoplasmic and perinuclear granular
staining in tumor cells was considered specific. Nuclear or
circumferential membranous staining, if present, was interpreted as
non-specific and excluded from scoring. Cases with extensive
necrosis, tissue folds, edge artifacts or inadequate antigen
preservation were recut and restained; persistently suboptimal
slides were excluded from analysis.
Evaluation and scoring of CANT1
immunoreactivity
When necessary, two board-certified pathologists,
blinded to clinical outcomes, independently evaluated all stained
slides using alight microscope (Olympus Corporation) for consensus.
Staining intensity in invasive tumor cells was graded on a 4-tier
scale: 0, no staining; 1+, weak cytoplasmic/perinuclear staining;
2+, moderate cytoplasmic staining; and 3+, strong coarse granular
cytoplasmic/perinuclear staing (7).
For each case, the percentage of invasive tumor cells demonstrating
1+, 2+ and 3+ intensity was estimated to the nearest 5%, with the
sum of all intensity categories reaching ~100% of invasive tumor
cells. An H-score was calculated using the following weighted
formula: H-score=(1 × percentage of weakly stained cells) + (2 ×
percentage of moderately stained cells) + (3 × percentage of
strongly stained cells).
To facilitate groupwise comparisons in subsequent
analyses, H-scores were also categorized a priori as: Low (H1),
1–100; moderate (H2), 101–200; and high (H3), 201–300. These
predefined H-score cut-off values were selected based on previously
published IHC literature employing similar semi-quantitative
H-score-based classification systems (7). Values of 0 were permitted by
definition but were not observed in the present cohort.
In cases with intratumoral heterogeneity, both
predominant fields and focal hotspots were examined; the final
score reflected the overall distribution across the tumor area
rather hotspot estimation alone. Discrepancies between observers
were resolved through consensus. Percentage maps, intensity grids
and representative microphotographs were archived in the digital
pathology system for documentation.
Quality assurance
All IHC runs were performed in uniform batches (≤20
slides per run) using identical retrieval and detection programs to
ensure technical consistency. Lot numbers, incubation times and
instrument logs were recorded. Slides were examined under
standardized illumination using an Olympus BX series light
microscope (Olympus Corporation). To minimize preanalytical
variability, blocks with prolonged fixation (>72 h), delayed
fixation or gross autolysis were avoided when possible; repeat
staining was performed from freshly cut sections when
necessary.
Statistical analysis
Statistical analyses were performed using SPSS
(version 22.0; IBM Corp.). Descriptive statistics were presented as
counts and percentages for categorical variables and as medians
with interquartile ranges for continuous variables. Continuous
variables were analyzed using the Mann-Whitney U test, while
categorical variables were assessed with Pearson's χ2
test or Fisher's exact test, as appropriate. Survival analyses were
conducted using the Kaplan-Meier method and the log-rank test with
survival curves generated using the R statistical software (version
4.3.2; Posit Software, PBC). Multivariable analysis was performed
using the Cox proportional hazards model. Adjusted HRs, 95% CIs and
P-values were reported, with P<0.05 being considered to indicate
a statistically significant difference.
With regard to missing data, Ki-67 values were
available for 56 of 59 patients (94.9%). The correlation between
CANT1 H-score and the Ki-67 proliferation index was evaluated using
Pearson's correlation coefficient, as both variables were treated
as continuous variables. Programmed death-ligand 1 (PD-L1) combined
positive scoring (CPS) was assessable in 28 patients (47.5%), with
24 patients (40.7%) scoring as negative (CPS 0) and 4 patients
(6.8%) demonstrating positive expression (CPS 1–25 or >25); 31
patients (52.5%) had unknown PD-L1 status due to unavailable tissue
or incomplete testing. BRCA mutational status was available in 24
patients (40.7%), with 35 patients (59.3%) having unknown results.
Missing data were handled using complete-case analysis; no
imputation methods were employed given the exploratory nature of
the present study and the low proportion of missingness for primary
prognostic variables.
Results
A total of 59 non-metastatic patients with TNBC
treated at The Department of Medical Oncology of Ankara City
Hospital were included in the present study. The median age was 55
(range, 28–78 years) and comorbidities were present in 62.7% of
patients. The median tumor size was 3.1 cm (range, 0.9–13.0 cm),
with a median Ki-67 index of 70%. According to primary tumor stage
(T), 23.7% of patients had T1, 49.2% had T2, 18.6% had T3 and 8.5%
had T4 disease. Pathologically, 27.1% of patients were lymph
node-negative (N0), 57.7% were N1 and 15.2% were N2. Based on
pathological TNM staging8.5% were stage I, 59.3% were stage II and
32.2% were stage III (9). BRCA
mutations were detected in 3.3% of patients, while the PD-L1 CPS
was 1–25 in 5.1% and >25 in 1.7% of the cohort. IHC analysis of
CANT1 was successfully performed in all 59 cases. Based on the
predefined H-score classification, 15 patients (25.4%) demonstrated
low expression (H1, 1–100), 29 patients (49.2%) showed moderate
expression (H2, 101–200) and 15 patients (25.4%) exhibited high
expression (H3, 201–300). No cases were completely negative for
CANT1 staining. The clinical and pathological characteristics of
the patients are summarized in Table
I.
 | Table I.Clinical and pathological
characteristics of patients in the present study. |
Table I.
Clinical and pathological
characteristics of patients in the present study.
| Characteristic | No. of patients |
|---|
| Age, years | 55 (28–78) |
| Sex |
|
|
Female | 59 (100.0) |
| Comorbidity |
|
| Yes | 37 (62.7) |
| No | 22 (37.3) |
| Tumor size, cm | 3.1 (0.9–13.0) |
| Primary tumor |
|
| T1 | 14 (23.7) |
| T2 | 29 (49.2) |
| T3 | 11 (18.6) |
| T4 | 5 (8.5) |
| Pathological lymph
node |
|
| N0 | 16 (27.1) |
| N1 | 34 (57.7) |
| N2 | 9 (15.2) |
| TNM stage |
|
| IA | 5 (8.5) |
| IIA | 14 (23.7) |
| IIB | 21 (35.6) |
| IIIA | 13 (22.0) |
| IIIB | 4 (6.8) |
| IIIC | 2 (3.4) |
| Estrogen
receptor |
|
|
Negative | 59 (100.0) |
| Progesterone
receptor |
|
|
Negative | 59 (100.0) |
| HER2 |
|
|
Negative | 59 (100.0) |
| Ki-67, % | 70 (5–90) |
| BRCA mutation
status |
|
|
Wild-type | 22 (37.3) |
|
Mutant | 2 (3.3) |
|
Unknown | 35 (59.3) |
| PD-L1 CPS |
|
| 0 | 24 (40.7) |
| 1-25 | 3 (5.1) |
|
>25 | 1 (1.7) |
|
Unknown | 31 (52.5) |
| CANT1 H-score
category |
|
| H-score 1
(1–100) | 15 (25.4) |
| H-score 2
(101–200) | 29 (49.2) |
| H-score 3
(201–300) | 15 (25.4) |
Among the 59 patients, 40 (67.8%) received
neoadjuvant chemotherapy. The most common regimens were Adriamycin
and cyclophosphamide followed by paclitaxel in 17 patients (42.5%)
and Adriamycin and cyclophosphamide followed by carboplatin and
paclitaxel in another 17 patients (42.5%). In addition, 4 patients
(10%) received Adriamycin and cyclophosphamide alone, while 2
patients (5%) received immunotherapy-containing regimens
(Adriamycin, cyclophosphamide and pembrolizumab or carboplatin,
paclitaxel and pembrolizumab).
Regarding surgical procedures, 31 patients (52.5%)
underwent breast-conserving surgery with axillary lymph node
dissection, 16 (27.1%) underwent radical mastectomy with axillary
lymph node dissection and 12 (20.3%) had breast-conserving surgery
with sentinel lymph node biopsy. Among the 40 patients who received
neoadjuvant chemotherapy, 12 (30%) achieved a pathological complete
response, while 8 (20%) showed a partial pathological response
(data not shown). The remaining 20 patients (50%) exhibited limited
or minimal response to neoadjuvant treatment.
During follow-up, 26 patients (44.1%) developed
distant metastases. The most common metastatic sites were multiple
sites in 8 patients (30.8%), liver in 5 (19.2%), bone in 4 (15.4%),
lung in 3 (11.5%), brain or leptomeningeal involvement in 3
(11.5%), skin in 2 (7.7%) and adrenal gland in 1 patient (3.8%).
Among metastatic patients, the most common first-line treatments
were gemcitabine alone (19.2%), gemcitabine and cisplatin (19.2%),
gemcitabine and carboplatin (15.4%) as well as capecitabine alone
(11.5%). Other first-line therapies included paclitaxel (7.7%),
cisplatin and capecitabine (7.7%), carboplatin and paclitaxel
(3.8%), radiotherapy (7.7%) and no systemic treatment (7.7%).
Second-line therapy was administered to 9 patients (15.3%), most
commonly vinorelbine (44.4%), followed by sacituzumab govitecan
(22.2%), capecitabine (11.1%), gemcitabine (11.1%) and paclitaxel
(11.1%). Third-line treatments were given to 6 patients (10.7%),
including capecitabine (33.3%), sacituzumab govitecan (33.3%),
gemcitabine and cisplatin (16.7%) as well as gemcitabine (16.7%).
Treatment modalities and clinical courses are summarized in
Table II.
 | Table II.Treatment modalities and clinical
course of the patients. |
Table II.
Treatment modalities and clinical
course of the patients.
| Variable | n (%) |
|---|
| Neoadjuvant
chemotherapy | 40 (67.8) |
| Neoadjuvant
chemotherapy regimen |
|
|
Adriamycin +
cyclophosphamide/paclitaxel | 17/40 (42.5) |
|
Adriamycin +
cyclophosphamide/carboplatin + paclitaxel | 17/40 (42.5) |
|
Adriamycin +
cyclophosphamide | 4/40 (10.0) |
|
Adriamycin + cyclophosphamide
+ pembrolizumab/carboplatin + paclitaxel + pembrolizumab | 2/40 (5.0) |
| Surgical
procedure |
|
|
Breast-conserving surgery +
axillary lymph node dissection | 31 (52.5) |
| Radical
mastectomy + axillary lymph node dissection | 16 (27.1) |
|
Breast-conserving surgery +
sentinel lymph node biopsy | 12 (20.3) |
| Adjuvant
chemotherapy | 47 (79.7) |
| Adjuvant
chemotherapy regimen |
|
|
Capecitabine | 26/47 (55.3) |
|
Adriamycin +
cyclophosphamide/paclitaxel | 14/47 (29.8) |
|
Adriamycin +
cyclophosphamide/carboplatin + paclitaxel | 2/47 (4.3) |
|
Pembrolizumab | 2/47 (4.3) |
|
Adriamycin +
cyclophosphamide | 1/47 (2.1) |
|
Cyclophosphamide +
methotrexate + 5-fluorouracil | 1/47 (2.1) |
|
Docetaxel +
cyclophosphamide | 1/47 (2.1) |
| Adjuvant
radiotherapy | 51 (86.4) |
| Metastasis after
adjuvant treatment | 26 (44.1) |
| Site of
metastasis |
|
|
Multiple sites | 8/26 (30.8) |
|
Liver | 5/26 (19.2) |
|
Bone | 4/26 (15.4) |
|
Lung | 3/26 (11.5) |
|
Brain/leptomeningeal | 3/26 (11.5) |
|
Skin | 2/26 (7.7) |
| Adrenal
gland | 1/26 (3.8) |
| First-line
treatments for metastatic disease | 26 (100.0) |
|
Gemcitabine | 5/26 (19.2) |
|
Gemcitabine + cisplatin | 5/26 (19.2) |
|
Gemcitabine + carboplatin | 4/26 (15.4) |
|
Capecitabine | 3/26 (11.5) |
|
Paclitaxel | 2/26 (7.7) |
|
Cisplatin + capecitabine | 2/26 (7.7) |
|
Carboplatin + paclitaxel | 1/26 (3.8) |
|
Radiotherapy | 2/26 (7.7) |
| No
treatment | 2/26 (7.7) |
| Second-line
treatments | 9 (15.3) |
|
Vinorelbine | 4/9 (44.4) |
|
Sacituzumab govitecan | 2/9 (22.2) |
|
Capecitabine | 1/9 (11.1) |
|
Gemcitabine | 1/9 (11.1) |
|
Paclitaxel | 1/9 (11.1) |
| Third-line
treatments | 6 (10.7) |
|
Capecitabine | 2/6 (33.3) |
|
Sacituzumab govitecan | 2/6 (33.3) |
|
Gemcitabine + cisplatin | 1/6 (16.7) |
|
Gemcitabine | 1/6 (16.7) |
CANT1 cytoplasmic staining was independently
evaluated by two pathologists and graded as 0, 1+, 2+ or 3+ based
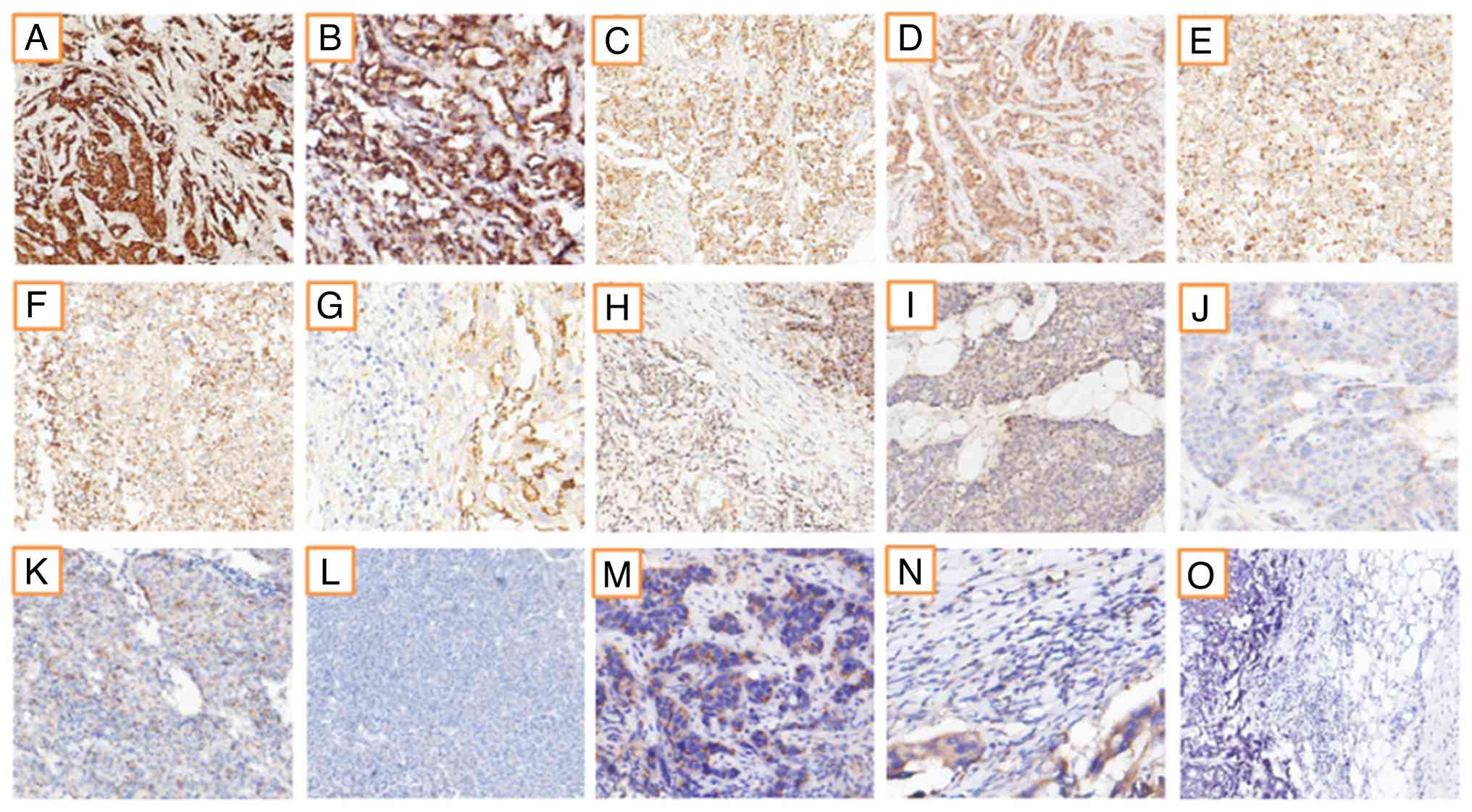
on staining intensity. Fig. 1
displays representative IHC staining patterns of CANT1 in the
present study cohort of 59 patients. CANT1 expression was
semi-quantitatively assessed using H-scores, which combine staining
intensity and the percentage of positively stained tumor cells. A
total of 9 patients demonstrated strong (3+) staining in 100% of
tumor cells, 15 showed moderate (2+) staining in 100% of tumor
cells and 6 exhibited weak (1+) staining in 100% of tumor cells
(Fig. 1). The remaining cases
displayed mixed staining patterns with variable intensity
distributions. Overall, 15 patients (25.4%) were classified as
H-score 3, 29 patients (49.2%) as H-score 2 and 15 patients (25.4%)
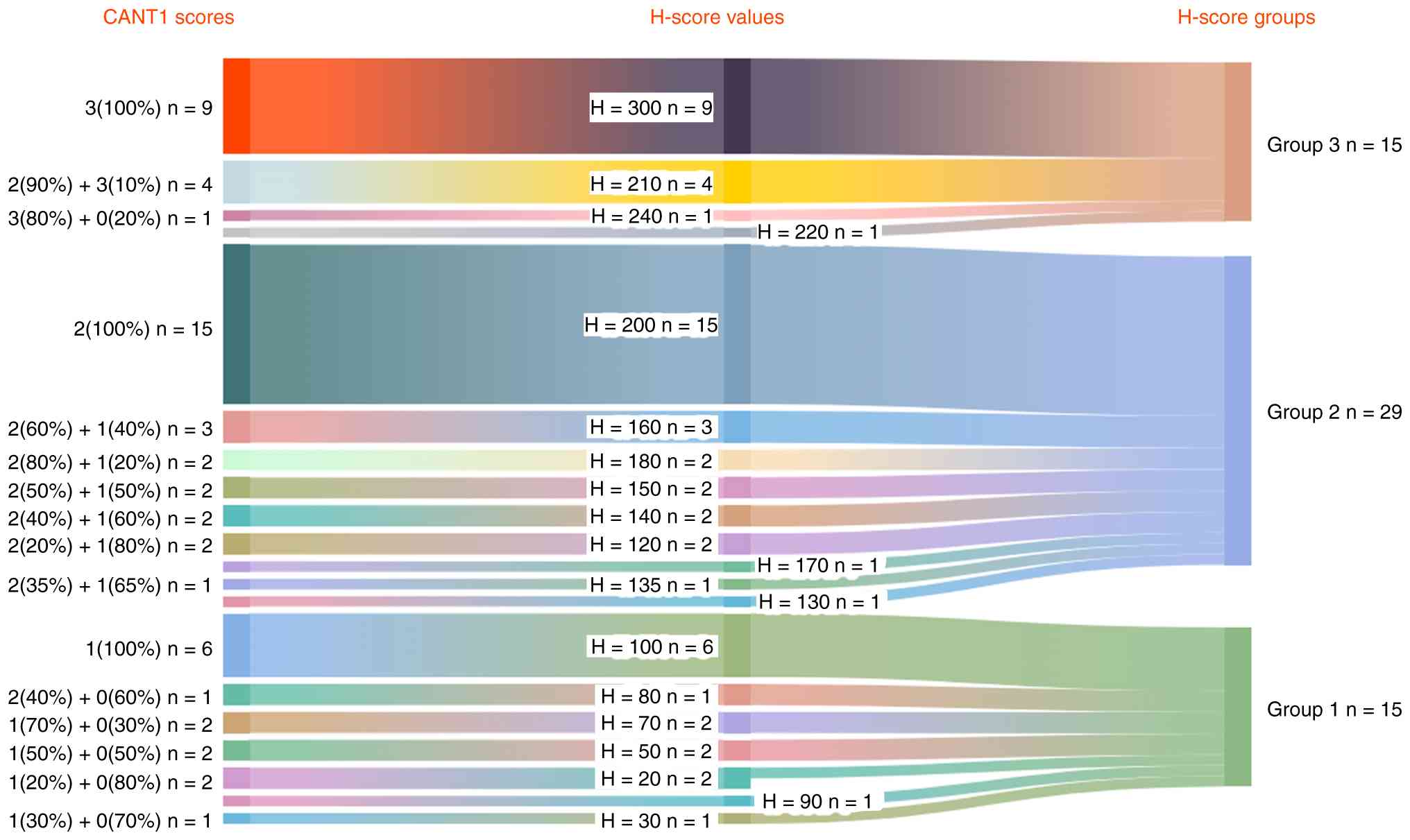
as H-score 1. Fig. 2 shows a Sankey
diagram summarizing CANT1 staining patterns and corresponding
H-score categories
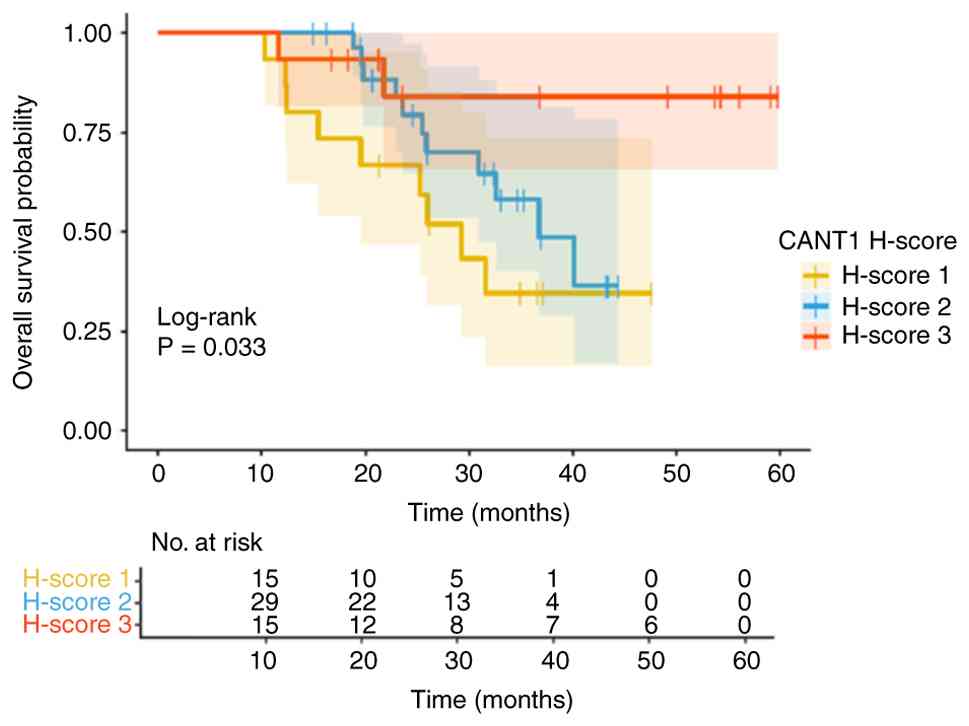
The median follow-up duration was 48.4 months and
the median OS (mOS) for the entire cohort was 40.2 months.
According to CANT1 expression levels, mOS was 29.2 months in the
H-score 1 group, 36.7 months in the H-score 2 group and not reached
in the H-score 3 group. The difference in OS among CANT1 H-score
groups was statistically significant (log-rank P=0.033).
Kaplan-Meier survival curves according to CANT1 H-score categories
are shown in Fig. 3. For patients
with H-score 1, the 1-, 2- and 3-year OS rates were 93.3, 66.7 and
34.6%, respectively. For the H-score 2 group, the corresponding
rates were 100, 79.3 and 58.1%, while for the H-score 3 group they
were 93.3, 84.0 and 84.0%, respectively. Patients with higher CANT1
expression (H-score 3) demonstrated more favorable long-term
survival compared with those with lower expression levels (log-rank
P=0.033). The 1-, 2- and 3-year OS rates according to CANT1 H-score
categories are summarized in Table
III.
 | Table III.Overall survival estimates at
selected time points according to CANT1 H-score categories. |
Table III.
Overall survival estimates at
selected time points according to CANT1 H-score categories.
| CANT1 H-score
group | Time, months | No. at risk | No. of events | Survival, % | 95% CI
(lower-upper) |
|---|
| H-score 1 | 12 | 14 | 1 | 93.3 | 81.5–100.0 |
|
| 24 | 9 | 4 | 66.7 | 46.6–95.3 |
|
| 26 | 3 | 4 | 34.6 | 16.2–73.6 |
| H-score 2 | 12 | 29 | 0 | 100.0 | 100.0–100.0 |
|
| 24 | 18 | 5 | 79.3 | 64.7–97.3 |
|
| 36 | 6 | 4 | 58.1 | 40.0–84.4 |
| H-score 3 | 12 | 14 | 1 | 93.3 | 81.5–100.0 |
|
| 24 | 8 | 1 | 84.0 | 65.6–100.0 |
|
| 36 | 8 | 0 | 84.0 | 65.6–100.0 |
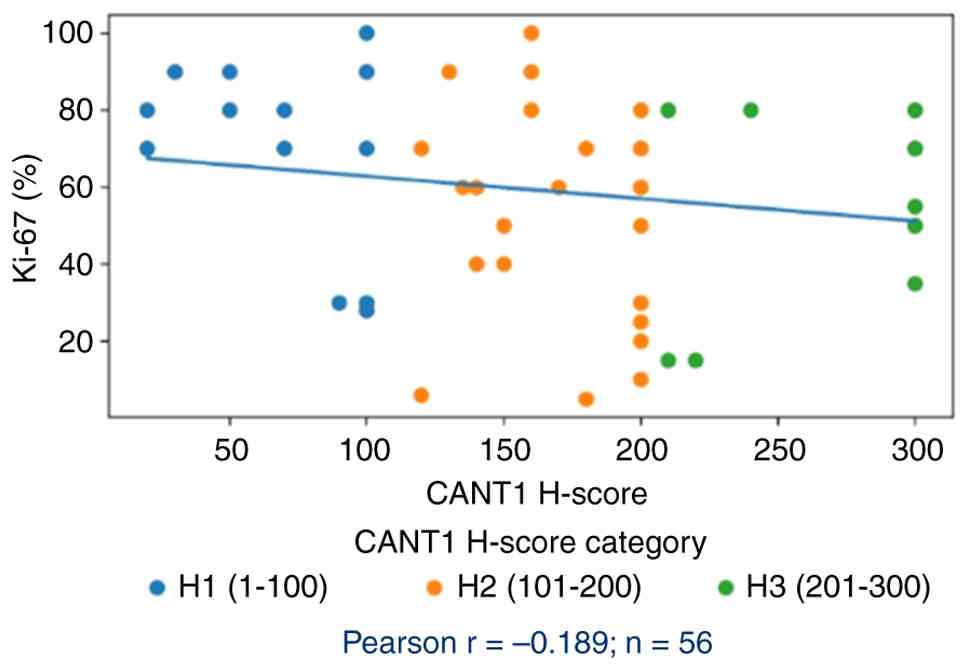
To further characterize the biological importance of
CANT1 expression, its correlation with established prognostic
biomarkers and clinicopathological parameters was examined. A weak
negative correlation was observed between CANT1 H-score and Ki-67
proliferation index (Pearson r=−0.189; n=56). Notably, patients in
the H-score 3 group (high CANT1 expression) demonstrated a mean
Ki-67 index of 52.3%, which was markedly lower compared with the
H-score 1 group (68.3%), suggesting an inverse relationship between
CANT1 expression and tumor cell proliferative activity (Fig. 4). This finding was consistent with
the improved OS observed in the high CANT1 expression group and may
indicate that CANT1 contributes to suppression of proliferative
signaling in TNBC. Analysis of staging parameters revealed no
significant association between CANT1 H-score categories and T
stage (mean T stage, H1=2.27, H2=2.28 and H3=1.87) or N stage (mean
N stage, H1=1.07, H2=0.90 and H3=0.87; data not shown). These
results suggest that CANT1 expression is independent of
conventional anatomic staging and may represent a distinct
biological pathway affecting prognosis in TNBC. With regard to
PD-L1 CPS, insufficient data points precluded meaningful
statistical analysis of its correlation with CANT1 expression;
future studies with more complete PD-L1 assessment in larger
cohorts are warranted to elucidate potential interactions between
CANT1 and immune checkpoint molecule expression.
The prognostic impact of clinicopathological factors
on OS was evaluated using univariate and multivariate Cox
regression analyses. For multivariable analysis, only variables
that demonstrated statistical significance in univariate analysis
(P<0.05) were entered into the Cox proportional hazards model;
the proportional hazards assumption was assessed using Schoenfeld
residuals and was satisfied for all variables. Age was dichotomized
at the median value of 55 years. In univariate analysis, patients
aged ≥55 years demonstrated significantly longer OS compared with
those aged <55 years (P=0.023). This association remained
independently significant in multivariate analysis (P=0.024;
HR=0.354; 95% CI, 0.144–0.875), indicating that a younger age
(<55 years) was independently associated with worse prognosis.
Other variables, including comorbidity, TNM stage, surgical
approach and administration of neoadjuvant or adjuvant
chemotherapy, were not significantly associated with OS in
univariate analysis and thus were not included in the multivariate
model. CANT1 H-score was significantly associated with OS in
univariate analysis (P=0.033) and remained independently
significant in the multivariate model (P=0.048). Pairwise
comparisons revealed that the survival difference was primarily
driven by the contrast between the H-score 3 and H-score 1 groups,
whereby patients with high CANT1 expression (H3) exhibited
significantly longer survival (HR=0.149; 95% CI, 0.031–0.715;
P=0.017). The difference between the H-score 3 and H-score 2 groups
was not statistically significant (HR=0.539; 95% CI, 0.220–1.300;
P=0.172). These results are summarized in Table IV.
 | Table IV.Univariate and multivariate Cox
regression analysis of prognostic factors for overall survival. |
Table IV.
Univariate and multivariate Cox
regression analysis of prognostic factors for overall survival.
| Variable | mOS, months | Univariate
P-value | Multivariate HR
(95% CI) | Multivariate
P-value |
|---|
| Age |
| 0.023 | HR=0.354
(0.144–0.875) | 0.024 |
|
<55 | 30.9 |
|
|
|
|
≥55 | NR |
|
|
|
| Comorbidity |
| 0.253 |
|
|
|
Yes | 31.5 |
|
|
|
| No | 42.5 |
|
|
|
| Stage |
| 0.059 |
|
|
|
I–II | NR |
|
|
|
|
III | 32.6 |
|
|
|
| Neoadjuvant
chemotherapy |
| 0.971 |
|
|
|
Yes | NR |
|
|
|
| No | 40.1 |
|
|
|
| Adjuvant
chemotherapy |
| 0.089 |
|
|
|
Yes | NR |
|
|
|
| No | 30.9 |
|
|
|
| Surgical
procedure |
| 0.870 |
|
|
| Radical
mastectomy | 36.8 |
|
|
|
|
Breast-conserving surgery | 42.4 |
|
|
|
| CANT1 H-score |
| 0.033 |
| 0.048a |
| H-score
1 | 29.2 |
| H-score 3 vs. 1,
HR=0.149 (0.031–0.715); P=0.017 |
|
| H-score
2 | 36.7 |
| H-score 3 vs. 2,
HR=0.539 (0.220–1.300); P=0.172 |
|
| H-score
3 | NR |
|
|
|
Discussion
TNBC is one of the most aggressive molecular
subtypes of breast cancer, with limited targeted treatment options
and unsatisfactory long-term outcomes despite advances in systemic
therapy. In this context, reliable prognostic biomarkers that go
beyond conventional anatomical staging remain an unmet clinical
need. In the present cohort of 59 non-metastatic patients with
TNBC, CANT1 expression, assessed by H-score, 25.4% of patients were
classified as H-score 1, 49.2% as H-score 2 and 25.4% as H-score 3
and higher CANT1 expression (H-score 3) was associated with
significantly longer OS compared with lower expression groups. In
multivariable analysis, high CANT1 expression remained
independently associated with improved OS, while traditional
clinicopathological variables such as stage and treatment
modalities were not retained as independent predictors. . Taken
together, these results suggest that CANT1 expression may reflect
tumor biological characteristics not indicated by conventional TNM
staging and could provide additional prognostic information for
risk stratification in TNBC.
At the molecular level, CANT1 encodes a
calcium-dependent lumenal nucleotidase that localizes predominantly
to the endoplasmic reticulum and Golgi apparatus, whereby it
regulates nucleotide metabolism and contributes to protein
glycosylation and vesicular trafficking (4,5). By
hydrolyzing nucleoside diphosphates such as UDP and guanosine
diphosphate (GDP) into their corresponding monophosphates, CANT1
prevents the accumulation of inhibitory nucleoside diphosphates
within the Golgi lumen, thereby facilitating nucleotide recycling
and sustaining glycosylation reactions required for proper
secretory pathway function. The clinical relevance of this pathway
is illustrated by loss-of-function mutations in CANT1, which cause
autosomal recessive Desbuquois dysplasia, a skeletal disorder
characterized by growth restriction, limb shortening and severe
abnormality of cartilage and extracellular matrix organization
(10). These observations
underscore the importance of CANT1 in tissue growth and matrix
homeostasis, providing a plausible mechanistic basis for its
involvement in tumor biology.
Beyond its intracellular role, CANT1 may also be
secreted into the extracellular matrix, where its nucleotidase
activity hydrolyzes extracellular nucleoside diphosphates such as
UDP and GDP into monophosphates, thereby decreasing local
diphosphate concentrations and potentially attenuating P2
receptor–mediated purinergic signaling (11). Through this mechanism, CANT1 may
influence cell-cell communication, stromal remodeling and immune
cell recruitment within the tumor microenvironment. Such
context-dependent effects provide a biologically plausible
explanation for why CANT1 expression might be associated with
either favorable or unfavorable prognosis depending on tumor type
and microenvironmental context.
Evidence from prostate cancer supports this
context-dependent behavior. Gerhardt et al (7) systematically evaluated CANT1
expression in prostate cancer using large tissue microarrays and
demonstrated marked upregulation of CANT1 in primary prostate
carcinoma compared with benign tissue, with predominantly
Golgi-type and cytoplasmic staining patterns. Notably,
castration-resistant tumors showed decreased CANT1 expression and
loss of CANT1 was associated with clinically more aggressive
disease, suggesting that higher CANT1 expression may be associated
with a more differentiated, less aggressive phenotype in prostate
cancer (7). This pattern was
concordant with the present findings in TNBC, whereby higher CANT1
expression was associated with improved OS, supporting the
hypothesis that CANT1 may act as a favorable prognostic marker in
at least a subset of solid tumors.
Within the present cohort, no completely
CANT1-negative tumors were observed. Even in the lowest expression
group, invasive carcinoma cells consistently exhibited at least
weak cytoplasmic staining (1+) in ~20% of cells, resulting in
minimum H-scores of 20. This near-consistent expression suggested
that CANT1 may be constitutively expressed in TNBC or may represent
a key feature of the TNBC phenotype. Whether this reflects a
specific metabolic requirement of TNBC cells for secretory pathway
homeostasis or a broader mammary tissue-specific expression
pattern, could not be determined from the present data, but it
underscored that prognostic information was carried not by the
presence or absence of CANT1, but by the level of its
expression.
By contrast, numerous studies have associated high
CANT1 expression with adverse outcomes in other tumor types. Liu
et al (8) reported that
CANT1 was markedly upregulated in hepatocellular carcinoma compared
with adjacent non-tumorous liver tissue and that high CANT1 levels
were associated with an advanced stage, higher grade and shorter
OS. Functional analyses in this study indicated that CANT1
upregulation may promote cell cycle progression, DNA replication
and oncogenic signaling pathways in hepatocellular carcinoma
(8). Similarly, pan-cancer
bioinformatics and IHC work by Yang et al (12) showed that CANT1 was upregulated at
both mRNA and protein levels across a number of malignancies and
that elevated expression was associated with advanced T and N
stages as well as worse OS and disease-specific survival in lung
adenocarcinoma, kidney renal papillary cell carcinoma and
lower-grade glioma, amongst others.
Yao et al (13) further demonstrated that high CANT1
expression was an independent predictor of poor OS in lung
adenocarcinoma, both in The Cancer Genome Atlas and Gene Expression
Omnibus datasets, with elevated CANT1 levels remaining notably
associated with adverse prognosis after multivariable adjustment.
These data stand in clear contrast with the present findings in
TNBC, whereby high CANT1 expression was independently associated
with improved OS. Collectively, the available evidence indicates
that the prognostic impact of CANT1 is strongly tissue- and
context-dependent: It may act as an oncogenic driver in HCC and
lung adenocarcinoma, whereas in prostate cancer and TNBC, higher
expression appears to be associated with more favorable clinical
behavior.
The mechanisms underlying the protective association
of high CANT1 expression in TNBC are speculative but a number of
hypotheses may be considered. In the present cohort, higher CANT1
expression was accompanied by a trend toward lower proliferative
activity, with the H-score 3 group exhibiting a markedly lower mean
Ki-67 index compared with the H-score 1 group, consistent with the
observed survival advantage. This inverse relationship suggested
that CANT1 may be associated with a less proliferative, more
differentiated phenotype in TNBC, potentially through its role in
glycoprotein processing and maintenance of secretory pathway
integrity. Enhanced CANT1-mediated glycosylation may support more
orderly expression and trafficking of adhesion molecules and
receptors, thereby constraining invasive behavior and metastatic
potential. In addition, secreted CANT1 could modulate purinergic
signaling within the tumor microenvironment, possibly reducing
immunosuppressive nucleotide signaling and indirectly facilitating
antitumor immune responses. Notably, the present study did not
observe meaningful associations between CANT1 expression and T or N
stage, suggesting that its prognostic effect is not simply a
surrogate for tumor burden, but may reflect a distinct biological
axis.
The present study exhibits certain limitations that
should be acknowledged when interpreting the results. Its
retrospective, single-center design carries the inherent risk of
selection bias and incomplete capture of clinical and molecular
covariates, despite systematic case identification. CANT1
expression was assessed using a semi-quantitative H-score method
based on manual pathologist evaluation; although scoring was
performed in a blinded fashion with consensus review, some degree
of inter-observer variability cannot be excluded. The overall
sample size was modest (n=59), which limits the precision of effect
estimates and the number of covariates that can be included in
multivariable models. Nevertheless, a post hoc power calculation
indicated 88% power to detect the observed difference between the
highest and lowest H-score groups, suggesting that the main signal
is unlikely to be a chance finding. Finally, the present study was
designed primarily as a clinicopathological and prognostic analysis
and did not include mechanistic experiments or external validation
cohorts. In addition, missing data were present for certain
clinical variables, including PD-L1 expression, BRCA status and
Ki-67, which should be considered when interpreting the results,
although the main analyses remained robust.
Furthermore, the inclusion of patients who did and
did not receive neoadjuvant chemotherapy represents a source of
clinical heterogeneity that may have influenced survival outcomes.
While exploratory analyses did not reveal a consistent association
between CANT1 expression and pathological response, this should be
interpreted cautiously due to the limited sample size. Future work
should validate these findings in larger, prospective, multicenter
series with standardized IHC protocols, automated image analysis
and comprehensive molecular profiling and to investigate the
functional role of CANT1 in TNBC through both in vitro and
in vivo models, as well as analyses in independent
transcriptomic datasets. In addition, future studies should aim to
specifically address the prognostic and biological importance of
CANT1 expression in de novo metastatic TNBC cohorts. Given
the distinct tumor biology, treatment intent and survival dynamics
characterizing de novo metastatic disease, evaluating CANT1
in this setting may provide complementary insights and help clarify
whether its prognostic impact differs between curative-intent
early-stage and metastatic TNBC.
In conclusion, the present study provided primary
evidence that CANT1 expression carries prognostic information in
TNBC. In a cohort of 59 non-metastatic patients with TNBC treated
with curative surgery, higher CANT1 expression, quantified by
H-score, was independently associated with improved OS, whereas
conventional clinicopathological factors did not retain
significance in multivariable analysis. These findings, which
contrast with reports of adverse prognostic effects of CANT1 in
hepatocellular carcinoma and lung adenocarcinoma, support a
context-dependent role for CANT1 in human cancer biology and
suggest that it may act as a favorable prognostic marker in TNBC.
Although the retrospective design and limited sample size warrant
cautious interpretation, CANT1 emerges as a promising candidate
biomarker for risk stratification in this high-risk breast cancer
subtype. Validation in larger, independent cohorts and mechanistic
studies clarifying how CANT1 modulates TNBC behavior are required
before routine clinical implementation can be considered.
Acknowledgements
Not applicable.
Funding
The present study was supported by the GILEAD Türkiye ‘Hayat
Bulan Fikirler’ Project (GILEAD Türkiye ‘Ideas That Give Life’
Initiative), which provided financial support for the procurement
of the CANT1 IHC kit.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
DB and GU conceptualized the present study. DB, GU
and BC were responsible for the methodology. DB, EH and SNÖC
screened the hospital information system and patient medical
records to identify patients with triple-negative breast cancer and
extract the relevant clinical data. AC and DÖT conducted
pathological examination and interpretation. SK performed tissue
processing and CANT1 IHC staining. DB, ÖB and EA conducted formal
analysis and statistical analysis. DB wrote the original manuscript
draft. MANŞ, SY interpreted data and revised the manuscript . GU
and BC provided supervision. BC contributed to the
conceptualization and design of the study, and was involved in the
analysis and interpretation of the data. DB and GU confirm the
authenticity of all the raw data. All authors read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
The present study protocol was reviewed and approved
by The Ankara City Hospital Clinical Research Ethics Committee
(approval no. TABED 1–25-1032). Due to the retrospective design and
the use of archival formalin-fixed paraffin-embedded specimens, the
requirement for written informed consent was waived in accordance
with national regulations and institutional policy. All procedures
were performed in accordance with the principles of The Declaration
of Helsinki and applicable local data protection regulations.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, artificial
intelligence tools were used to improve the readability and
language of the manuscript or to generate images, and subsequently,
the authors revised and edited the content produced by the
artificial intelligence tools as necessary, taking full
responsibility for the ultimate content of the present
manuscript.
Glossary
Abbreviations
Abbreviations:
|
CANT1
|
calcium-activated nucleotidase 1
|
|
TNBC
|
triple-negative breast cancer
|
|
ER
|
estrogen receptor
|
|
PR
|
progesterone receptor
|
|
IHC
|
immunohistochemistry
|
|
OS
|
overall survival
|
References
|
1
|
Giaquinto AN, Sung H, Newman LA, Freedman
RA, Smith RA, Star J, Jemal A and Siegel RL: Breast cancer
statistics 2024. CA Cancer J Clin. 74:477–495. 2024.PubMed/NCBI
|
|
2
|
Garrido-Castro AC, Lin NU and Polyak K:
Insights into molecular classifications of Triple-negative breast
cancer: Improving patient selection for treatment. Cancer Discov.
9:176–198. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zagami P and Carey LA: Triple negative
breast cancer: Pitfalls and progress. NPJ Breast Cancer. 8:952022.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Smith TM, Hicks-Berger CA, Kim S and
Kirley TL: Cloning, expression, and characterization of a soluble
calcium-activated nucleotidase, a human enzyme belonging to a new
family of extracellular nucleotidases. Arch Biochem Biophys.
406:105–115. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kodama K, Takahashi H, Oiji N, Nakano K,
Okamura T, Niimi K, Takahashi E, Guo L, Ikegawa S and Furuichi T:
CANT1 deficiency in a mouse model of Desbuquois dysplasia impairs
glycosaminoglycan synthesis and chondrocyte differentiation in
growth plate cartilage. FEBS Open Bio. 10:1096–1103. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Daşar T, Ürel Demir G, İmren G, Utine GE,
Yilmaz G and Şimşek Kiper P: From desbuquois dysplasia to multiple
epiphyseal dysplasia: The clinical impact of a CANT1 variant across
five unrelated families. Am J Med Genet A. 197:e639502025.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gerhardt J, Steinbrech C, Büchi O, Behnke
S, Bohnert A, Fritzsche F, Liewen H, Stenner F, Wild P, Hermanns T,
et al: The androgen-regulated Calcium-Activated Nucleotidase 1
(CANT1) is commonly overexpressed in prostate cancer and is
tumor-biologically relevant in vitro. Am J Pathol. 178:1847–1860.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liu T, Li ZZ, Sun L, Yang K, Chen JM, Han
XY, Qi LM, Zhou XG and Wang P: Upregulated CANT1 is correlated with
poor prognosis in hepatocellular carcinoma. BMC Cancer.
23:10072023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Teichgraeber DC, Guirguis MS and Whitman
GJ: Breast cancer staging: Updates in the AJCC cancer staging
manual, 8th edition, and current challenges for radiologists, from
the AJR special series on cancer staging. AJR Am J Roentgenol.
217:278–290. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Balasubramanian K, Li B, Krakow D, Nevarez
L, Ho PJ, Ainsworth JA, Nickerson DA, Bamshad MJ, Immken L, Lachman
RS and Cohn DH: MED resulting from recessively inherited mutations
in the gene encoding calcium-activated nucleotidase CANT1. Am J Med
Genet A. 173:2415–2421. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu X, Yang Z, Luo X, Luo J, Fu W, Fang Z,
Xia D, Li L and Xu J: Calcium-activated nucleotidase 1 silencing
inhibits proliferation, migration, and invasion in human clear cell
renal cell carcinoma. J Cell Physiol. 234:22635–22647. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yang W, Liu Z and Liu T: Pan-cancer
analysis predicts CANT1 as a potential prognostic, immunologic
biomarker. Cell Signal. 117:1111072024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yao Q, Yu Y, Wang Z, Zhang M, Ma J, Wu Y,
Zheng Q and Li J: CANT1 serves as a potential prognostic factor for
lung adenocarcinoma and promotes cell proliferation and invasion in
vitro. BMC Cancer. 22:1172022. View Article : Google Scholar : PubMed/NCBI
|