Introduction
Hepatocellular carcinoma (HCC) is the third leading
cause of cancer-related mortalities worldwide, with 70–90% of cases
developing on a background of cirrhosis (1,2). The
integration of immune checkpoint inhibitors (ICIs) into the
treatment landscape has transformed therapeutic options for HCC,
offering renewed hope for patients who previously depended on
tyrosine kinase inhibitors (TKIs) such as sorafenib or lenvatinib
as first-line therapies (3,4). However, clinical responses to ICIs
remain variable, underscoring the need for predictive biomarkers
that can identify which patients are most likely to benefit from
treatment.
The skeletal muscle index at the third lumbar
vertebra (L3-SMI) is a widely validated, imaging-based metric for
diagnosing sarcopenia in both clinical and research settings. It is
calculated as the total cross-sectional area of skeletal muscle
(cm2) at the L3 level, divided by height squared
(m2) (5,6). L3-SMI is typically measured from
cross-sectional imaging, either using computed tomography (CT) or
magnetic resonance imaging obtained at the midpoint of the L3
vertebra. This region is selected because the lumbar spine provides
a stable anatomical landmark, and the skeletal muscle area at this
level correlates strongly with whole-body muscle mass (7). CT is particularly suitable for
clinical and research use due to wide availability, non-invasive
nature and standardized acquisition, which simplifies the
assessment of skeletal muscle mass. Currently, diagnostic cut-off
values for L3-SMI vary across regions, especially between Eastern
and Western populations, reflecting differences in ethnicity, liver
disease etiologies and measurement methodology. This variability
complicates both clinical practice and research, particularly in
the context of chronic liver disease (8). Despite the lack of uniform criteria,
the association between sarcopenia and adverse clinical outcomes in
patients with chronic liver disease has been consistently
demonstrated (5,9).
L3-SMI serves as a practical and cost-effective
surrogate marker for diagnosing sarcopenia and predicting outcomes
in patients with chronic liver disease (5,9,10). A
meta-analysis including 22 studies with 6,965 patients with
cirrhosis reported a pooled sarcopenia prevalence (defined by
L3-SMI) of 37.5%, and demonstrated that sarcopenia was associated
with increased mortality [adjusted hazard ratio (HR)=2.30], these
findings remained consistent across subgroups stratified by sex,
liver disease etiology and severity of hepatic dysfunction
(5). In patients with cirrhosis
awaiting liver transplantation (LT), lower L3-SMI has been
identified as a notable risk factor for waitlist mortality,
postoperative complications and post-LT mortality (11). Similarly, reduced L3-SMI is
associated with increased 1-year mortality after surgical resection
of HCC (12) and predicts shorter
median survival in patients with HCC (13). Previous studies also indicate that
lower L3-SMI predicts worse OS in patients with HCC treated with
trans-arterial chemoembolization or radiofrequency ablation
(14,15). While the relationship between low
skeletal muscle mass and prognosis in patients with HCC treated
with ICIs remains underexplored, evidence from locally advanced
esophageal cancer and biliary sepsis suggests plausible mechanistic
links and clinical relevance (16,17).
In the present single-center longitudinal
retrospective study, the prognostic value of multiple skeletal mass
indices SMIs-including SMI, SMI/weight, SMI/height and SMI/BMI were
evaluated for clinical outcomes in male patients with HCC receiving
ICI therapy.
Materials and methods
Patients
A retrospective analysis was conducted on 195 male
patients with HCC who were treated with ICIs. To eliminate the
potential confounding effect of sex, the present study was
restricted to male patients. All patients were admitted to Beijing
Youan Hospital (Beijing, China), between January 2018 and February
2022. Eligible participants were adults clinically diagnosed with
HCC according to the American Association for the Study of Liver
Diseases criteria (18), who were
male, aged ≥16 years and had undergone CT during hospitalization.
The median age of the enrolled patients was 59 years (range,
29–82). Exclusion criteria were as follows: i) Incomplete clinical
data; ii) confirmed or strongly suspected malignancy other than
HCC; iii) long-term bedridden status; iv) diagnosis of Acquired
Immune Deficiency Syndrome; and v) chronic renal and/or respiratory
insufficiency, severe heart disease or conditions associated with
intestinal nutrient malabsorption.
The study was performed in accordance with the
Declaration of Helsinki. All data were extracted from electronic
medical records. The protocol was approved by the Medical Ethics
Review Committee of Beijing Youan Hospital (approval no.
LL-2023-055K). Due to the retrospective nature of the study and
full protection of patient privacy, the requirement for informed
consent was waived.
Anthropometric measurements
Height and weight were measured during
hospitalization using standardized protocols. Body weight was
recorded in the morning using a digital scale, with patients
barefoot and wearing lightweight clothing. For patients with
cirrhosis and fluid retention, dry body weight was estimated by
subtracting a percentage of the current body weight to account for
ascites and edema: 5, 10 or 15% was deducted for mild, moderate or
severe ascites, respectively, with an additional 5% subtracted if
lower limb edema was present (19).
Height was measured with patients standing barefoot, heels
together, back straight and arms at their sides, as previously
described (20). BMI was then
calculated as the adjusted dry weight (in kg) divided by height
squared (in m2) (21).
Assessment of L3-SMI
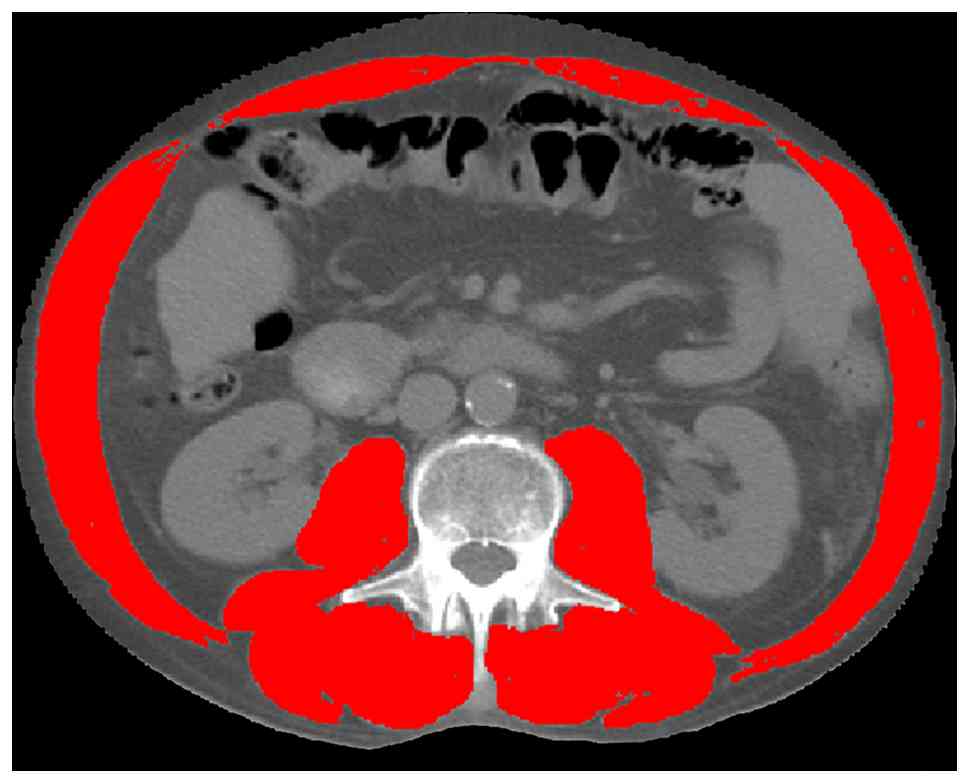
Abdominal non-contrast-enhanced CT scans were
acquired on a 64-slice LightSpeed VCT scanner (GE Healthcare) with
patients in the supine position. All scans used for skeletal muscle
analysis were obtained within a 2-week period prior to the
initiation of ICI therapy. For each scan, a single transverse image
at the L3 vertebral level was selected for analysis using
SliceOmatic software (version 5.0; TomoVision). Skeletal muscle
area (SMA; cm2) was measured at the L3 vertebral level
by segmenting muscle tissue within a Hounsfield unit window of −29
to +150, which included the psoas, erector spinae, quadratus
lumborum, transversus abdominis, external and internal obliques and
rectus abdominis muscles, as previously described (Fig. 1) (22). The cross-sectional SMA was
automatically computed and normalized to height squared to derive
the SMI (cm2/m2). All L3 SMA measurements
were independently reviewed by two radiologists. In cases of
disagreement, a third radiologist was consulted to reach
consensus.
Statistical analysis
Continuous variables were expressed as mean ±
standard deviation or median (interquartile range). Categorical
variables were expressed as numbers and percentages. Group
comparisons for continuous variables were performed using an
unpaired Student's t-test or Mann-Whitney-U test, as appropriate.
Categorical variables were compared using the χ2 test or
Fisher's exact probabilities.
Cox proportional hazards regression analysis with
forward LR selection (entry P<0.05; removal P<0.10) was used
to identify factors associated with 1-year OS. Candidate variables
included BMI, L3-SMI, L3-SMI/BMI, L3-SMI/weight (kg) and
L3-SMI/height (m). A risk-score plot was generated using the R
package ‘ggrisk’ (https://github.com/yikeshu0611/ggrisk). Optimal
cut-off values for each muscle-related index were determined using
the surv_cutpoint function from the R package ‘survminer’
(https://rpkgs.datanovia.com/survminer/index.html).
Using these optimal cutoffs, Kaplan-Meier survival curves were then
generated and visualized using the R packages ‘survival’
(https://github.com/therneau/survival)
and ‘survminer’. SHapley Additive exPlanations (SHAP) analysis,
implemented via the R package ‘shapviz’ (https://github.com/ModelOriented/shapviz), was used to
visualize and rank the importance of variables. Patients were
stratified according to the optimal cut-offs for survival
comparison. The predictive performance of each index was evaluated
by calculating the area under the receiver operating characteristic
(ROC) curve (AUC) using the R packages ‘reportROC’ (https://CRAN.R-project.org/package=reportROC). All
analyses were performed using IBM SPSS Statistics 22.0 (IBM Corp.)
or R software version 4.1.0 (R Foundation for Statistical
Computing). P<0.05 was considered to indicate a statistically
significant difference.
Results
Baseline clinical characteristics of
patients
Among the 195 male patients, the distribution of
first-line ICI regimens was as follows: 86 (44.10%) patients
received 200 mg of sintilimab every 3 weeks, 101 (51.79%) received
200 mg of camrelizumab every 3 weeks, 1 (0.51%) received 200 mg of
toripalimab every 3 weeks, 2 (1.03%) received 240 mg of
tremelimumab every 3 weeks and 5 (2.56%) received 1,200 mg of
atezolizumab every 3 weeks. All agents were administered as
first-line therapy.
The mean age of the patients was 58.13±9.32 years. A
total of 161 (82.56%) were HBV-infected, and 163 (83.59%) had
cirrhosis. The median follow-up duration was 14 months (range, 1–55
months). According to the Barcelona Clinic Liver Cancer (BCLC)
staging system, 53 patients (27.18%) were stage A, 84 (43.08%)
stage B, 31 (15.90%) stage C and 27 (13.85%) stage D.
Regarding treatment history and combinations, 7
patients (3.58%) received anti-VEGF therapy, 45 (23.08%) received
TKIs (23 lenvatinib, 22 sorafenib), 40 (20.51%) underwent surgical
resection and 13 (6.67%) received interventional therapy. Overall,
127 patients (65.13%) had received three or more treatment
strategies during their course of care. Detailed demographic and
clinical characteristics of the participants are presented in
Table I.
 | Table I.Baseline characteristics of male
patients. |
Table I.
Baseline characteristics of male
patients.
| Parameters | Total patients
(n=195) | Alive (n=180) | Dead (n=15) | P-valuea |
|---|
| Age, years | 58.13±9.32 | 58.19±9.19 | 57.40±11.08 | 0.754 |
| BMI,
kg/m2 | 23.14 (21.10,
25.53) | 23.03 (20.93,
25.39) | 25.54 (23.15,
27.73) | 0.005 |
| ALT, U/l | 32.00 (20.00,
50.00) | 32.00 (20.00,
50.00) | 35.00 (1800,
44.00) | 0.781 |
| AST, U/l | 41.50 (28.00,
64.25) | 41.00 (28.00,
64.00) | 58.00 (29.00,
105.00) | 0.154 |
| AST/ALT ratio | 1.40 (1.13,
1.86) | 1.40 (1.12,
1.83) | 1.79 (1.18,
2.44) | 0.261 |
| TBIL, µmol/l | 22.30 (15.78,
28.63) | 21.90 (14.9,
28.60) | 28.10 (18.40,
37.90) | 0.080 |
| ALB, g/l | 36.85±5.03 | 36.90±4.89 | 36.24±6.70 | 0.714 |
| MELD score | 10.34 (9.60,
11.74) | 10.28 (9.51,
13.20) | 10.64 (10.06,
13.42) | 0.092 |
| Child-Pugh
score | 6.00 (5.00,
7.00) | 6.00 (5.00,
7.00) | 6.00 (5.00,
7.25) | 0.330 |
| HBV infection, n
(%) | 161 (82.56) | 150 (83.33) | 11 (73.33) | 0.531 |
| Cirrhosis Presence,
n (%) | 163 (83.59) | 150 (83.33) | 13 (86.67) | >0.999 |
| Decompensated
cirrhosis, n (%) | 66 (33.85) | 55 (30.56) | 8 (53.33) | 0.127 |
| Number of tumors
≥3, n (%) | 33 (16.92) | 29 (16.11) | 4 (26.67) | 0.491 |
| Maximum tumor
diameter, cm | 1.00 (1.00,
5.50) | 1.00 (1.00,
5.50) | 1.00 (1.00,
6.50) | 0.305 |
| Tumor
differentiationb, n
(%) |
|
|
| 0.335 |
| Poor,
moderate | 17 (51.52) | 13 (76.47) | 4 (23.53) |
|
|
Well | 16 (48.48) | 15 (93.75) | 1 (6.25) |
|
| BCLC stage, n
(%) |
|
|
| 0.982 |
| A,
B | 137 (70.26) | 127 (92.70) | 10 (7.30) |
|
| C,
D | 58 (29.74) | 53 (91.38) | 5 (8.62) |
|
| Portal vein
invasion, n (%) | 27 (13.85) | 26 (14.44) | 1 (6.67) | 0.654 |
| Ki-67, % | 0.46±0.24 | 0.47±0.23 | 0.43±0.30 | 0.747 |
| AFP, ng/ml | 17.70 (4.46,
769.00) | 17.00 (5.16,
804.00) | 62.30 (2.43,
330.75) | 0.699 |
| SMI,
cm2/m2 | 46.46 (41.15,
51.46) | 46.89 (41.35,
52.17) | 41.15 (34.55,
49.47) | 0.015 |
| SMI/weight | 0.70 (0.63,
0.76) | 0.70 (0.65,
0.76) | 0.59 (0.51,
0.63) | <0.001 |
| SMI/height | 27.62 (24.31,
30.58) | 27.93 (24.71,
30.77) | 25.33 (20.53,
28.25) | 0.017 |
| SMI/BMI | 2.00 (1.83,
2.19) | 2.01 (1.85,
2.21) | 1.50 (1.47,
1.95) | <0.001 |
Relevance of multiple SMIs at baseline
for 1-year mortality in male patients with HCC treated with
ICIs
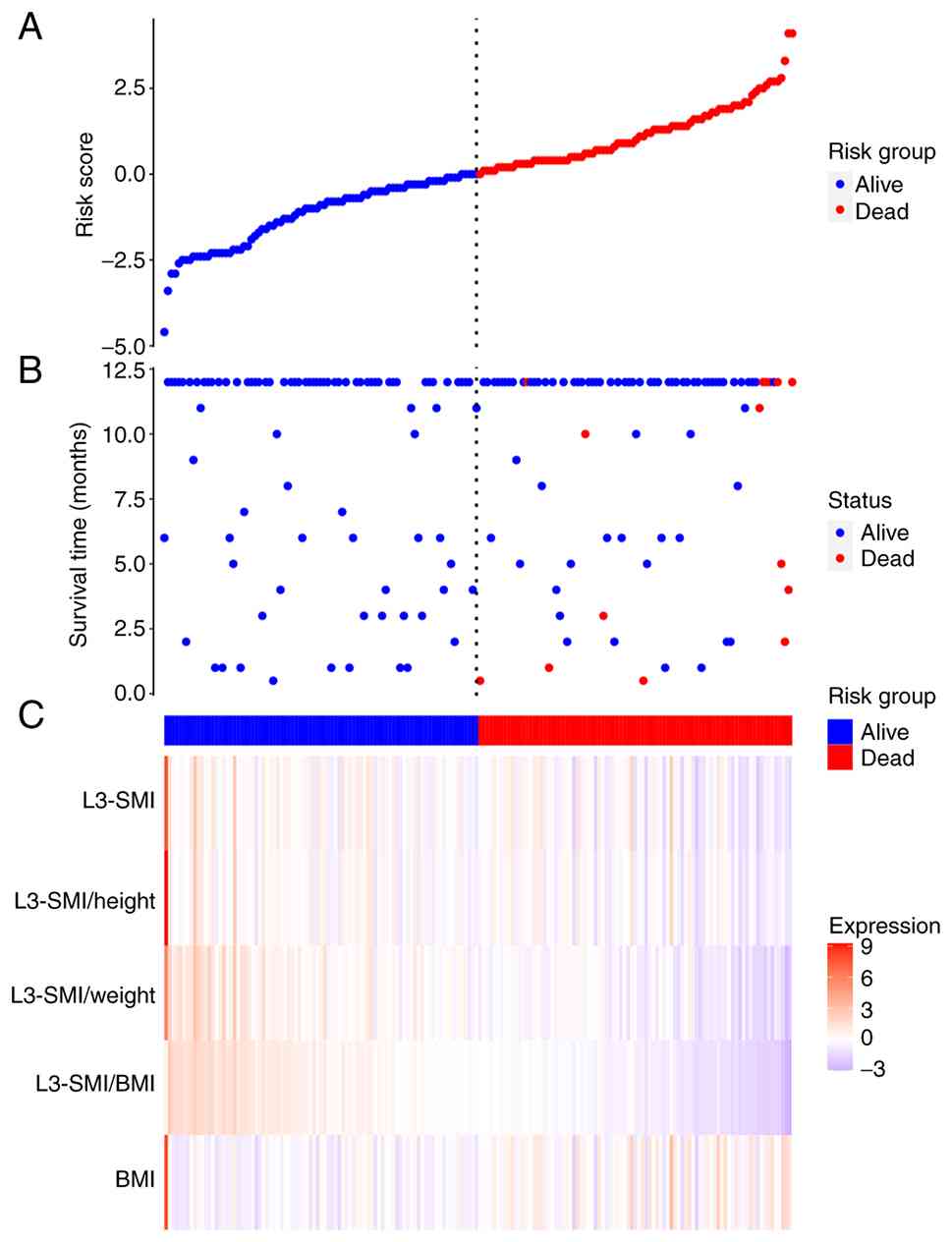
The distribution of risk score for each patient is
shown in Fig. 2A. A scatter plot of
survival time vs. risk score (Fig.
2B) demonstrated that a higher risk score was associated with
an increased number of mortality events and shorter survival times.
A heat map of the indices across high- and low-risk groups
(Fig. 2C) indicated that an
elevated risk of mortality was associated with a higher BMI and
lower levels of L3-SMI, L3-SMI/weight, L3-SMI/height and
L3-SMI/BMI.
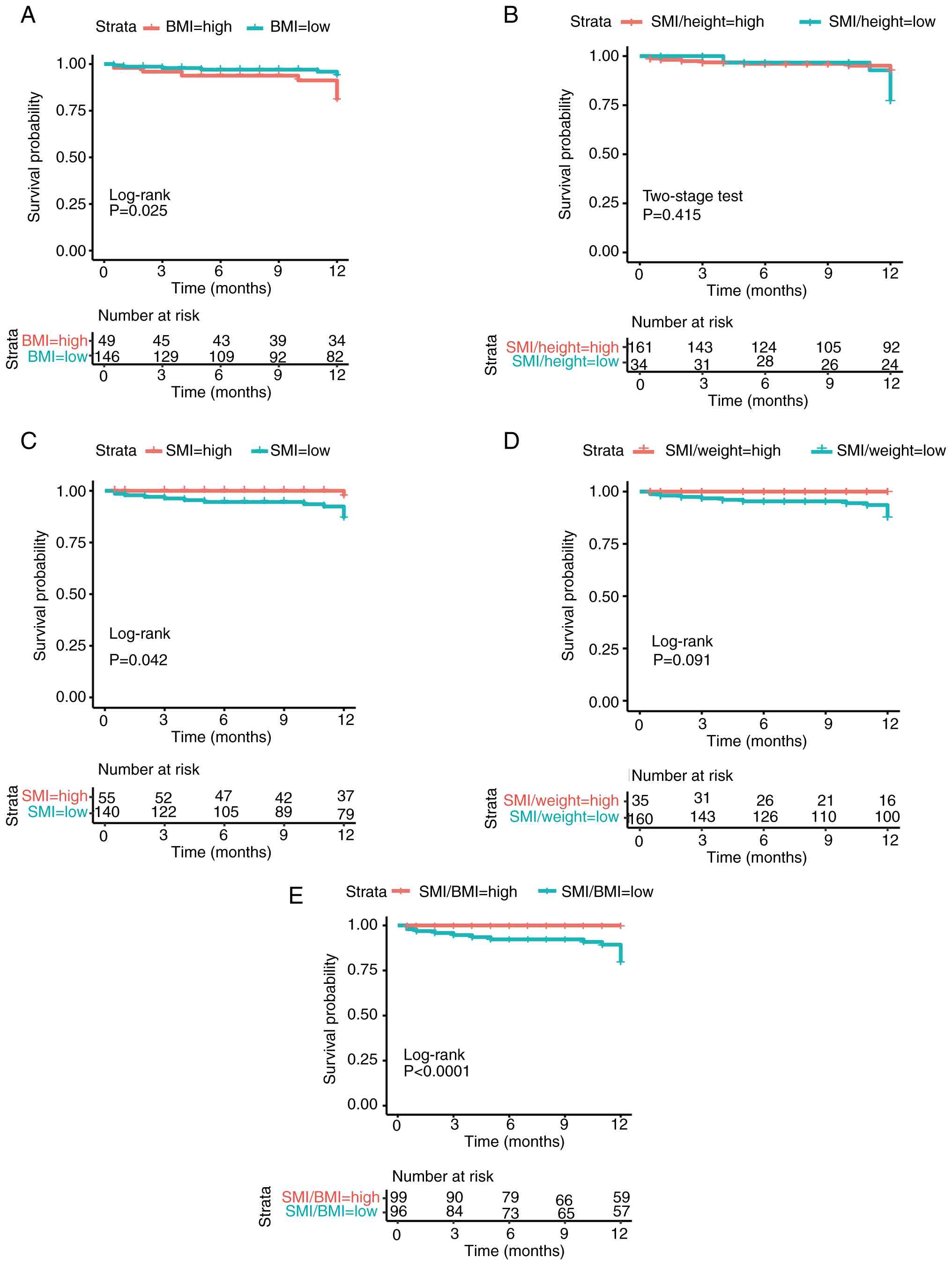
As shown in Fig. 3
and Table II, lower BMI and higher
values of L3-SMI and L3-SMI/BMI were significantly associated with
improved 1-year OS (all P<0.05; Fig.
3A, C and E). Using the forward linear regression method,
variables with a univariate P-value <0.05 were entered into a
multivariate Cox proportional hazards model, and only a higher
L3-SMI/BMI was retained as an independent predictor of improved
1-year OS (HR=0.02; 95% CI: 0.001–0.940; P=0.047). This association
persisted after further adjustment for the potential confounding
effect of age (age-adjusted HR=0.02; 95% CI: 0.001–0.934;
P=0.046).
 | Table II.Univariate and multivariate analyses
of indicators for 1-year mortality. |
Table II.
Univariate and multivariate analyses
of indicators for 1-year mortality.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
| Parameters | Cut-off value | χ2 | P-value | HR (95% CI) | P-value |
|---|
| BMI,
kg/m2 | 25.39 | 5.051 | 0.025 | - | 0.077 |
| SMI/height | 22.91 | - | 0.415a |
|
|
| SMI,
cm2/m2 | 50.59 | 4.120 | 0.042 | - | 0.511 |
| SMI/weight | 0.78 | 2.857 | 0.091 |
|
|
| SMI/BMI | 2.00 | 15.827 | <0.0001 | 0.02
(0.001–0.940) | 0.047 |
Kaplan-Meier analysis showed accumulated mortality
rates of 4, 5 and 15% at 6, 9 and 12 months, respectively. Among
the 189 patients with follow-up data, the objective response rate
was 23.91% (45/189), the disease control rate was 36.51% (69/189)
and the median progression-free survival was 7 months (95% CI:
5.56–8.44). These favorable outcomes are consistent with the
predominance of early- to intermediate-stage disease (70.26% BCLC
A/B) and preserved liver function in the cohort. Notably, patients
with an L3-SMI/BMI ≥2.00 had a significantly lower 1-year
cumulative mortality compared with those with an L3-SMI/BMI
<2.00 (0 vs. 20%; log rank χ2=15.827; P<0.001;
Fig. 3E). Corresponding survival
curves of other indices are presented in Fig. 3A-D.
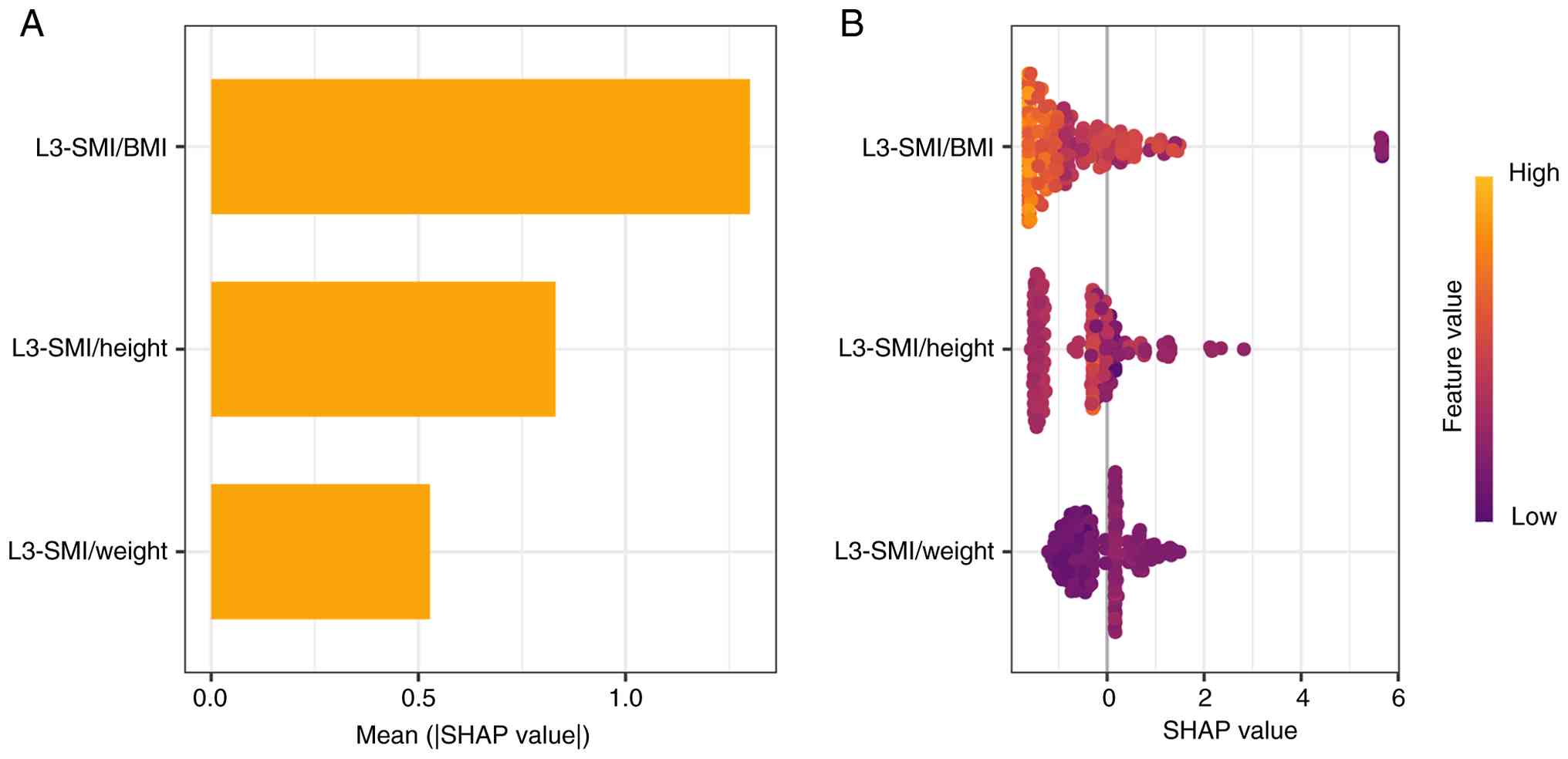
To visually interpret the contributions of each
variable, SHAP values were calculated to quantify the effect of the
derived ratios on 1-year mortality predictions. Fig. 4A ranks the variables by average
absolute SHAP values, illustrating their relative importance.
Fig. 3B displays how each variable
influences the prediction. Notably, a higher L3-SMI/BMI value was
associated with a lower predicted probability of 1-year mortality,
underscoring its protective role in survival outcomes. This
association remained strong after additional adjustment for age as
a potential confounder (Fig.
S1).
Predicting performance of SMIs for
1-year mortality
The predictive performance of each SMI was assessed
via ROC analysis. The AUC for L3-SMI/BMI in predicting 1-year
mortality was 0.86 (95% CI: 0.76–0.96), which was notably higher
than the AUCs for BMI, L3-SMI, L3-SMI/weight and L3-SMI/height, as
shown in Table III.
 | Table III.AUC analysis of indicators for 1-year
mortality. |
Table III.
AUC analysis of indicators for 1-year
mortality.
| Indicators | AUC (95% CI) | Cut-off value | Se | Sp | PPV | NPV |
|---|
| BMI,
kg/m2 | 0.72
(0.59–0.85) | 22.94 | 1.00 | 0.49 | 0.93 | 0.15 |
| SMI,
cm2/m2 | 0.69
(0.55–0.83) | 41.74 | 0.73 | 0.60 | 0.96 | 0.16 |
| SMI/weight | 0.83
(0.71–0.95) | 0.64 | 0.79 | 0.87 | 0.98 | 0.28 |
| SMI/height | 0.69
(0.56–0.82) | 29.06 | 0.39 | 0.93 | 0.99 | 0.11 |
| SMI/BMI | 0.86
(0.76–0.96) | 1.51 | 1.00 | 0.60 | 0.96 | 1.00 |
Discussion
In the present study, the prognostic value of four
L3 SMIs were assessed, including L3-SMI, L3-SMI/weight,
L3-SMI/height and L3-SMI/BMI in male patients with HCC treated with
ICIs. L3-SMI/BMI was demonstrated to be independently and
positively associated with improved 1-year OS (as higher values
predicted improved survival), outperforming the other indices.
Furthermore, L3-SMI/BMI demonstrated the highest area under the ROC
curve for predicting 1-year mortality, underscoring its high
predictive performance as a prognostic marker in this
population.
Compared with survivors, deceased patients exhibited
significantly lower levels of L3-SMI, L3-SMI/weight, L3-SMI/height
and L3-SMI/BMI. These results support the protective role of
skeletal muscle mass, which was assessed using L3-SMI, and are
consistent with prior studies reporting lower L3-SMI in deceased
vs. surviving patients with HCC (12,23).
Additionally, earlier research has indicated that when appendicular
lean mass (ALM) is adjusted by height alone, individuals with
normal or low body weight may be misclassified as having lower
muscle mass compared with obese individuals (24). By contrast, weight-adjusted methods
reduce the likelihood of overestimating muscle mass in obesity
(25). A previous study suggested
that adjusting ALM for both height and weight better identifies
metabolic impairment than height-adjusted ALM alone (26). Similarly, in this cohort, L3-SMI
adjusted for weight or height remained positively associated with
1-year OS, supporting the notion that these indices partially
reflect muscle mass in patients with HCC. Thirdly, deceased
patients had higher BMI, a finding aligned with a prior report by
Yang et al (27).
Conversely, another study by Akce et al (6) found that higher BMI was associated
with longer median OS following anti-programmed cell death protein
1 therapy. This discrepancy may stem from heterogeneity in the
study population, including differences in underlying etiology,
treatment settings or demographic characteristics.
In the present analysis, L3-SMI/BMI demonstrated
superior predictive performance for 1-year mortality compared with
L3-SMI, L3-SMI/weight and L3-SMI/height. This advantage may be
explained by the fact that adjusting L3-SMI for BMI mitigates the
overestimation of muscle mass in individuals with high body weight,
thereby offering a more accurate identification of low muscle mass
in patients with HCC. Previous work has also shown that SMI/BMI is
the most commonly used metric for defining low muscle mass and is
considered an optimal predictor of metabolic syndrome, further
supporting its clinical relevance (28,29).
In addition, myokines secreted by skeletal muscle are considered to
modulate inflammatory processes positively, which may in turn
influence HCC prognosis (30,31).
Nevertheless, the precise molecular mechanisms involved warrant
further investigation. Furthermore, integrating this index into
clinical workflows could prompt a comprehensive nutritional
assessment and personalized interventions, such as targeted
protein-calorie supplementation and supervised exercise therapy. By
identifying high-risk patients at the start of treatment, this
biomarker may facilitate early, multimodal interventions designed
to modify a key prognostic factor-skeletal muscle mass. Future
prospective studies should investigate whether this risk-stratified
pathway, triggered by a low L3-SMI/BMI, leads to improvements in
muscle mass, enhanced tolerance to ICI therapy and ultimately,
improved survival outcomes in patients with HCC.
Several limitations of the present study should be
acknowledged. First, this was a retrospective, single-center
analysis with a relatively small sample size and short follow-up
period and an exclusively male cohort, which may limit the
generalizability of the findings to broader HCC populations.
Second, as the cohort was predominantly composed of patients with
BCLC stage A/B disease and preserved performance status, the
limited number of mortality events during follow-up may have
resulted in an underestimation of cumulative mortality. These
factors potentially limit the generalizability of the findings to
patients with more advanced HCC or compromised liver function.
Third, the analysis was confined to radiologically derived indices
of skeletal muscle quantity (L3-SMI and its ratios) and did not
include direct measures of muscle quality-such as CT radiodensity,
detailed body composition from dedicated software or physical
function tests. Future studies should integrate both quantitative
and qualitative assessments to clarify their distinct prognostic
contributions. Future large-scale, multicenter prospective studies
are needed to externally validate the conclusions and to further
clarify the prognostic role of L3-SMI/BMI in patients with HCC
receiving ICIs.
In conclusion, L3-SMI/BMI is a strong independent
predictor of 1-year OS in male patients with HCC treated with ICIs.
This finding enhances our understanding of the clinical utility of
L3-SMI/BMI in stratifying outcomes during HCC immunotherapy and
highlights its potential as a valuable prognostic biomarker.
Prospective studies with larger cohorts are warranted to establish
optimal cut-off values for L3-SMI/BMI and to evaluate its ability
to predict long-term prognosis in this population.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
This research is funded by Scientific Research Project of
Beijing Youan Hospital (grant no. 2022BJYAYY-YN2022-08).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YW contributed to the visualization, methodology,
formal analysis, drafting, and editing of the manuscript. SL, HuW
and XH contributed to data curation (organising and managing
collected datases, validating daata, documenting metadata,
preparing data in appropriate formats for analysis) and
acquisition, review and editing of the manuscript. QM contributed
to study design, interpretation of data, project administration,
resources, review and editing of the manuscript. JL contributed to
conceptualization, funding acquisition, study design,
interpretation of data for the work, supervision and critical
revision of the manuscript. YW and JL confirm the authenticity of
all the raw data. All authors read and approved the final version
of the manuscript.
Ethics approval and consent to
participate
All patient's data were retrieved from electronic
medical records. The study was approved by the Medical Ethics
Review Committee of Beijing Youan Hospital (approval no.
LL-2023-055K). All patients provided written informed consent
authorizing access to their medical records for research purposes,
and additional, study-specific informed consent was waived.
Patient consent for publication
The authors confirm that written informed consent
for publication of their case (including any accompanying images
and/or data) has been obtained from the patient.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
L3-SMI
|
skeletal muscle index at the lumbar 3
vertebra
|
|
BMI
|
body mass index
|
|
CT
|
computed tomography
|
|
HCC
|
hepatocellular carcinoma
|
|
ROC
|
receiver operating characteristic
|
References
|
1
|
Jiang Y, Tie C, Wang Y, Bian D, Liu M,
Wang T, Ren Y, Liu S, Bai L, Chen Y, et al: Upregulation of serum
sphingosine (d18:1)-1-P potentially contributes to distinguish HCC
Including AFP-Negative HCC from cirrhosis. Front Oncol.
10:17592020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Xiao CL, Zhong ZP, Lu C, Guo BJ, Chen JJ,
Zhao T, Yin ZF and Li B: Physical exercise suppresses
hepatocellular carcinoma progression by alleviating hypoxia and
attenuating cancer stemness through the Akt/GSK-3beta/beta-catenin
pathway. J Integr Med. 21:184–193. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Donne R and Lujambio A: The liver cancer
immune microenvironment: Therapeutic implications for
hepatocellular carcinoma. Hepatology. 77:1773–1796. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhou X, Xing Z, Dong R, Zhang X, Liang X,
Lu Z and Yang G: Cell function experiments and bioinformatics
analysis jointly revealed the antineoplastic effect of lumican on
hepatocellular carcinoma. Phenomics. 5:252–269. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tantai X, Liu Y, Yeo YH, Praktiknjo M,
Mauro E, Hamaguchi Y, Engelmann C, Zhang P, Jeong JY, van Vugt JLA,
et al: Effect of sarcopenia on survival in patients with cirrhosis:
A meta-analysis. J Hepatol. 76:588–599. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Akce M, Liu Y, Zakka K, Martini DJ, Draper
A, Alese OB, Shaib WL, Wu C, Wedd JP, Sellers MT, et al: Impact of
sarcopenia, BMI, and inflammatory biomarkers on survival in
advanced hepatocellular carcinoma treated with Anti-PD-1 antibody.
Am J Clin Oncol. 44:74–81. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kong M, Lin N, Wang L, Geng N, Xu M, Li S,
Song W, Zhou Y, Piao Y, Han Z, et al: Age-specific reference values
for low psoas muscle index at the L3 vertebra level in healthy
populations: A multicenter study. Front Nutr. 9:10338312022.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang Y, Wei L, Chang C, Duan F, Quan M
and Yang S: Sarcopenia defined with L3-SMI is an independent
predictor of survival in male patients with ARLD in mainland China.
Front Nutr. 10:12384332023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li T, Xu M, Kong M, Song W, Duan Z and
Chen Y: Use of skeletal muscle index as a predictor of short-term
mortality in patients with acute-on-chronic liver failure. Sci Rep.
11:125932021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yao J, Zhou X, Yuan L, Niu LY, Zhang A,
Shi H, Duan Z and Xu J: Prognostic value of the third lumbar
skeletal muscle mass index in patients with liver cirrhosis and
ascites. Clin Nutr. 39:1908–1913. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhou D, Zhang D, Zeng C, Zhang L, Gao X
and Wang X: Impact of sarcopenia on the survival of patients
undergoing liver transplantation for decompensated liver cirrhosis.
J Cachexia Sarcopenia Muscle. 14:2602–2612. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Salman MA, Omar HSE, Mikhail HMS, Tourky
M, El-Ghobary M, Elkassar H, Omar MG, Matter M, Elbasiouny AM,
Farag AM, et al: Sarcopenia increases 1-year mortality after
surgical resection of hepatocellular carcinoma. ANZ J Surg.
90:781–785. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen J, Huang X, Wei Q, Liu S, Song W and
Liu M: The relationship between systemic therapies and low skeletal
muscle mass in patients with intermediate and advanced
hepatocellular carcinoma. Front Immunol. 16:15578392025. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Salman A, Salman M, Moustafa A, Shaaban
HE, El-Mikkawy A, Labib S, Youssef A, Omar MG, Matter M and
Elkassar H: Impact of sarcopenia on Two-Year mortality in patients
with HCV-associated hepatocellular carcinoma after radiofrequency
ablation. J Hepatocell Carcinoma. 8:313–320. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang S, Zhang X, Chen Q, Jin ZC, Lu J and
Guo J: A novel Neutrophil-to-Lymphocyte ratio and sarcopenia based
TACE-Predict model of hepatocellular carcinoma patients. J
Hepatocell Carcinoma. 10:659–671. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Harada T, Tsuji T, Ueno J, Koishihara Y,
Konishi N, Hijikata N, Ishikawa A, Kotani D, Kojima T, Fujiwara H
and Fujita T: Prognostic impact of the loss of skeletal muscle mass
during neoadjuvant chemotherapy on older patients with esophageal
cancer. Ann Surg Oncol. 29:8131–8139. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chun SY, Cho YS and Kim HB: Association
between reduced muscle mass and poor prognosis of biliary sepsis.
Sci Rep. 14:18572024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Heimbach JK, Kulik LM, Finn RS, Sirlin CB,
Abecassis MM, Roberts LR, Zhu AX, Murad MH and Marrero JA: AASLD
guidelines for the treatment of hepatocellular carcinoma.
Hepatology. 67:358–380. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
European Association for the Study of the
Liver, . EASL clinical practice guidelines on nutrition in chronic
liver disease. J Hepatol. 70:172–193. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Boulhosa R, Lourenco RP, Cortes DM,
Oliveira LPM, Lyra AC and de Jesus RP: Comparison between criteria
for diagnosing malnutrition in patients with advanced chronic liver
disease: GLIM group proposal versus different nutritional screening
tools. J Hum Nutr Diet. 33:862–868. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li Q, Zhang X, Tang M, Song M, Zhang Q,
Zhang K, Ruan G, Zhang X, Ge Y, Yang M, et al: Different muscle
mass indices of the Global Leadership Initiative on Malnutrition in
diagnosing malnutrition and predicting survival of patients with
gastric cancer. Nutrition. 89:1112862021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
van Vugt JL, Levolger S, Gharbharan A,
Koek M, Niessen WJ, Burger JW, Willemsen SP, de Bruin RW and
IJzermans JN: A comparative study of software programmes for
cross-sectional skeletal muscle and adipose tissue measurements on
abdominal computed tomography scans of rectal cancer patients. J
Cachexia Sarcopenia Muscle. 8:285–297. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Portal D, Melamed G, Segal G and Itelman
E: Sarcopenia as manifested by L3SMI is associated with increased
long-term mortality amongst internal medicine patients-A
prospective cohort study. J Clin Med. 11:35002022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Newman AB, Kupelian V, Visser M, Simonsick
E, Goodpaster B, Nevitt M, Kritchevsky SB, Tylavsky FA, Rubin SM
and Harris TB; Health ABC Study Investigators, : Sarcopenia:
Alternative definitions and associations with lower extremity
function. J Am Geriatr Soc. 51:1602–1609. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kim NH, Park Y, Kim NH and Kim SG:
Weight-adjusted waist index reflects fat and muscle mass in the
opposite direction in older adults. Age Ageing. 50:780–786. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Buchmann N, Nikolov J, Spira D, Demuth I,
Steinhagen-Thiessen E, Eckardt R and Norman K: Identifying
sarcopenia in metabolic syndrome: Data from the berlin aging Study
II. J Gerontol A Biol Sci Med Sci. 71:265–272. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang L, Wang J, Yao L, Chen C, Pan J, Peng
L and Chen F: Body mass index and serum alpha-fetoprotein level
associated with PD1 expression and prognosis in patients with
hepatocellular carcinoma. Heliyon. 9:e144602023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Carvalho CJ, Longo GZ, Kakehasi AM,
Pereira PF, Segheto KJ, Juvanhol LL and Ribeiro AQ: Association
between skeletal mass indices and metabolic syndrome in brazilian
adults. J Clin Densitom. 24:118–128. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tinggaard AB, Skou MK, Jessen N, Norrelund
H and Wiggers H: ALM/BMI: A clinically superior index for
identifying skeletal muscle dysfunction in patients with heart
failure. J Am Heart Assoc. 13:e0335712024. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Dao T, Green AE, Kim YA, Bae SJ, Ha KT,
Gariani K, Lee MR, Menzies KJ and Ryu D: Sarcopenia and muscle
aging: A brief overview. Endocrinol Metab (Seoul). 35:716–732.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dumond Bourie A, Potier JB, Pinget M and
Bouzakri K: Myokines: Crosstalk and consequences on liver
physiopathology. Nutrients. 15:17292023. View Article : Google Scholar : PubMed/NCBI
|