Introduction
Prostate cancer is one of the most prevalent
malignancies in men, with an estimated 1.47 million new cases
worldwide in 2022, and its incidence continues to increase
annually, particularly in the aging population (1). Apalutamide, an androgen receptor
inhibitor, which is indicated for both non-metastatic
castration-resistant prostate cancer (nmCRPC) and metastatic
castration-sensitive prostate cancer (mCSPC) is widely used as a
standard therapeutic option (2–4).
While apalutamide demonstrates notable clinical
efficacy, including a marked reduction in prostate-specific antigen
(PSA) levels and prolonged time to metastasis, adverse events such
as cutaneous toxicities (including rashes and pruritus) have been
reported, potentially compromising the quality of life and
treatment adherence (3–5). Rashes associated with apalutamide have
been documented in 15.3–21.8% of patients, underscoring the
necessity of its management (3,4). In
addition, a notably high incidence of rashes (51.5%) has been
reported in Japanese patients, highlighting the need for vigilance
in this specific population (5).
Low body weight may be associated with increased systemic exposure
to apalutamide (6). Sasaki et
al (7) reported a markedly high
incidence of cutaneous adverse events in patients weighing <67
kg and with a BMI <24 kg/m2. Furthermore, Katsuta
et al (8) demonstrated a
markedly higher incidence of rashes in patients with a low body
weight. However, additional studies have reported no notable
association between body size and rash occurrence (5,9),
exhibiting how findings across these studies are inconsistent.
Therefore, a reduced initial dose of apalutamide may
be used in real-world clinical practice to mitigate the risk of
severe adverse events. Oishi et al (10) compared the safety and efficacy of
standard and reduced doses of apalutamide and found no notable
differences between the groups. However, this analysis was limited
to fixed-dose regimens at treatment initiation and did not evaluate
the dynamic strategies for dose adjustment. To the best of our
knowledge, to date, no studies have investigated the ‘reduced-dose
initiation with stepwise escalation’ approach, which may provide
clinically relevant insight into the safety and efficacy of
flexible dosing strategies tailored to individual patient
characteristics. Although the standard daily dose of apalutamide is
240 mg (1), the association between
dosage, adverse events and therapeutic efficacy remains
unclear.
Among surrogate measures used to assess treatment
efficacy, PSA kinetics, particularly PSA <0.2 ng/ml, are
regarded as indicators of a favorable treatment response.
Furthermore, the time required to reach this threshold reflects the
speed of response and has been suggested to correlate with
prognosis (11).
Subsequently, the present retrospective study
focused on the clinical impact of initiating apalutamide at a
reduced dose, followed by gradual escalation, to reduce the risk of
adverse events. Specifically, the effect of apalutamide dosage on
the incidence of cutaneous toxicities and PSA dynamics was
examined, with a particular emphasis on the time to achieve PSA
<0.2 ng/ml, as well as progression-free survival (PFS) between
the standard-dose and reduced-dose groups.
Materials and methods
Patients
A total of 46 patients who were newly prescribed
apalutamide at Ogaki Municipal Hospital (Ogaki, Japan) between May
2019 and September 2024, were included in the present study. All 46
patients were followed until treatment discontinuation or the
determined data cutoff date (March 2025), depending on whichever
occurred first. Prior to the analysis, predefined exclusion
criteria that were set to evaluate participants eligibility based
on treatment tolerability and outcomes included: i) Patients who
did not continue apalutamide beyond their first prescription; ii)
patients who transferred to another institution during the
treatment period, making adequate follow-up impossible; and iii)
patients deemed unsuitable due to severe comorbidity, poor
performance status or other conditions that precluded adequate
assessment of treatment outcomes.
Study methods
Patients were stratified into two groups according
to the initial prescribed dose of apalutamide, namely the
standard-dose (240 mg; n=26) and reduced-dose (120 mg; n=8 or 180
mg; n=12) groups. According to the Japanese package insert, the
standard approved dose of apalutamide is 240 mg once daily, with
stepwise dose reductions to 180 and 120 mg if necessary (2). The initial and subsequent dose
adjustments were determined at the discretion of the treating
physicians, based on the clinical characteristics of each patient,
including age, comorbidities, performance status and tolerance to
prior androgen receptor pathway inhibitors. No predefined
institutional criteria for dose selection, escalation or reduction
were established. In clinical practice, dose reductions from the
standard 240 mg/day to 180 or 120 mg/day were implemented stepwise
according to the emergence and severity of adverse events, graded
using the Common Terminology Criteria for Adverse Events (version
5.0) (12), together with physician
judgment.
Statistical analysis
Between the two groups, the relative dose intensity
(RDI), incidence and severity of adverse events (with a particular
focus on cutaneous toxicities), time to the first occurrence of
cutaneous toxicity after apalutamide initiation and treatment
discontinuation owing to cutaneous toxicity, were compared. As
efficacy endpoints, the time to achieve PSA <0.2 ng/ml and PFS
were evaluated. The RDI was calculated as the total number of
tablets prescribed from treatment initiation to the end of
treatment, divided by the planned standard dose (four tablets of
apalutamide at 60 mg per day) and further divided by the total
number of treatment days, including treatment interruptions.
Patient characteristics were compared using Mann-Whitney U tests
and the χ2 test or Fisher's exact test, as appropriate.
The incidence and severity of adverse events, management of the
first occurrence of cutaneous toxicity and reasons for treatment
discontinuation in the two groups were compared using Mann-Whitney
U tests and the χ2 test or Fisher's exact test, as
appropriate. Differences in the time to achieve PSA <0.2 ng/ml
and PFS between the two groups were analyzed using log-rank tests.
P<0.05 was considered to indicate a statistically significant
difference. EZR software (version 1.61; Division of Hematology,
Saitama Medical Center, Jichi Medical University) was used for all
analyses (13).
Results
Patient characteristics
No patients initiated on apalutamide during the
present study period fulfilled the predefined exclusion criteria,
hence no patients required exclusion. The patient characteristics
are summarized in Table I. The
median age of those in the standard-dose group was 73 (range,
48–87) years while that for those in the reduced-dose group was 78
(range, 58–90) years (P=0.156). The median body surface area (BSA)
was 1.73 m2 (range, 1.48–1.92) in the standard-dose
group and 1.60 m2 (range, 1.29–1.86) in the reduced-dose
group (P=0.049). The number of patients with mCSPC was 21/26 in the
standard-dose group and 13/20 in the reduced-dose group (P=0.385).
The median RDIs were 97.2 and 59.3% in the standard- and
reduced-dose groups, respectively (P<0.001). Dose escalation
during treatment was observed in 7 patients in the reduced-dose
group (P=0.001), whereas dose reductions occurred in 7 patients in
the standard-dose group and 6 patients in the reduced-dose group
(P>0.999). The median observation period was 336.0 days (range:
18.0–1,540.0) in the standard-dose group and 180.5 days (range,
14.0–1,405.0) in the reduced-dose group, with no significant
difference found between the groups (P=0.103; data not shown).
 | Table I.Patient characteristics of the
standard-dose group and reduced dose-group. |
Table I.
Patient characteristics of the
standard-dose group and reduced dose-group.
| Characteristic | Standard-dose group
(n=26) | Reduced-dose group
(n=20) | P-value |
|---|
| Age, years | 73 (48–87) | 78 (58–90) | 0.156 |
| Weight, kg | 63.7 (45.4–78.7) | 58.8 (37.9–73.0) | 0.064 |
| ≥67 | 10 | 5 |
|
|
<67 | 16 | 15 |
|
| BMI,
kg/m2 | 23.4 (17.6–27.5) | 22.4 (16.2–26.8) | 0.394 |
| ≥24 | 9 | 4 |
|
|
<24 | 17 | 16 |
|
| BSA,
m2 | 1.73 (1.48–1.92) | 1.60 (1.29–1.86) | 0.049 |
| Indication |
|
| 0.385 |
|
mCSPC | 21 | 13 |
|
|
nmCRPC | 5 | 7 |
|
| Initial PSA,
ng/ml |
|
|
|
|
mCSPC | 20.33
(0.41–968.13) | 12.36
(0.50–33.69) | 0.576 |
|
nmCRPC | 3.16 (0.44–4.88) | 11.55
(1.27–28.85) | 0.030 |
| RDI, % | 97.20
(23.53–100.00) | 59.30
(5.60–95.50) | <0.001 |
| Dose adjustment | 7 | 13 | 0.016 |
|
Escalation | 0 | 7 | 0.001 |
|
Reduction | 7 | 6 | >0.999 |
Incidence of adverse events
Results of the incidence and severity of adverse
events analysis are presented in Table
II. Adverse events occurred in 76.9% (20/26) and 65.0% (13/20)
of patients in the standard-dose and reduced-dose groups,
respectively (P=0.575). The incidence of cutaneous toxicities (all
grades) was 73.1% (19/26) in the standard-dose group and 40.0%
(8/20) in the reduced-dose group (P=0.036). No grade ≥3 cutaneous
toxicities were observed in the reduced-dose group.
 | Table II.Incidence and severity of adverse
events in the standard- and reduced dose-group. |
Table II.
Incidence and severity of adverse
events in the standard- and reduced dose-group.
| Adverse event
type | Standard-dose group
(n=26), n (%) | Reduced-dose group,
(n=20), n (%) | P-value |
|---|
| Any adverse
event |
|
|
|
| All
grade | 20 (76.9) | 13 (65.0) | 0.575 |
| Grade
≥3 | 5 (19.2) | 1 (5.0) | 0.212 |
| Cutaneous adverse
eventsa |
|
|
|
| All
grade | 19
(73.1)a | 8
(40.0)a | 0.036 |
| Grade
≥3 | 3 (11.5) | 0 (0.0) | 0.246 |
| Fatigue |
|
|
|
| All
grade | 2
(7.7)a | 2 (10.0) | >0.999 |
| Grade
≥3 | 0 (0.0) | 0 (0.0) | >0.999 |
| Pneumonia |
|
|
|
| All
grade | 2
(7.7)a | 0 (0.0) | 0.498 |
| Grade
≥3 | 2 (7.7) | 0 (0.0) | 0.498 |
| Somnolence |
|
|
|
| All
grade | 1
(3.8)a | 1 (5.0) | >0.999 |
| Grade
≥3 | 0 (0.0) | 0 (0.0) | >0.999 |
| Neutropenia |
|
|
|
| All
grade | 0 (0.0) | 1 (5.0) | 0.435 |
| Grade
≥3 | 0 (0.0) | 1 (5.0) | 0.435 |
| Edema of the
extremities |
|
|
|
| All
grade | 0 (0.0) | 1
(5.0)a | 0.435 |
| Grade
≥3 | 0 (0.0) | 0 (0.0) | >0.999 |
| Fever |
|
|
|
| All
grade | 0 (0.0) | 1 (5.0) | 0.435 |
| Grade
≥3 | 0 (0.0) | 0 (0.0) | >0.999 |
Time to the first occurrence of
cutaneous toxicity and management
Management at the first occurrence of cutaneous
toxicity is summarized in Table
III. The median time to the first cutaneous event was 63.0
(range, 14.0–142.0) days in the standard-dose group and 45.5
(range, 15.0–81.0) days in the reduced-dose group (P=0.193).
Furthermore, there were no significant differences between the two
groups in the management approaches at the first occurrence of
cutaneous toxicity (all P>0.05).
 | Table III.Management at first occurrence of
cutaneous toxicities in the standard- and reduced-dose group. |
Table III.
Management at first occurrence of
cutaneous toxicities in the standard- and reduced-dose group.
| Item | Standard-dose
(n=19) | Reduced-dose group
(n=8) | P-value |
|---|
| Time to initial
onset of cutaneous adverse events, days | 63.0 (14–142) | 45.5 (15–81) | 0.193 |
| Prescription of
oral antihistamines | 17
(89.5)a | 5
(62.5)a | 0.136 |
| Single
agent | 14 (73.7) | 4 (50.0) | 0.375 |
| Two or
more agents | 3 (15.8) | 1 (12.5) | 1.000 |
| Treatment
interruption | 8
(42.1)a | 6
(75.0)a | 0.209 |
| Dose reduction with
continuation of therapy | 1 (5.3) | 0 (0.0) | >0.999 |
| Prescription of
topical agents |
|
|
|
|
Corticosteroids | 14 (73.7)
a | 6
(75.0)a | >0.999 |
|
Moisturizers | 8
(42.1)a | 5
(62.5)a | 0.420 |
|
Other | 2
(10.5)a | 0 (0.0) | >0.999 |
| Prescription of
oral agents |
|
|
|
|
Corticosteroids | 1
(5.3)a | 0 (0.0) | >0.999 |
|
Minocycline | 1
(5.3)a | 0 (0.0) | >0.999 |
Reasons for treatment
discontinuation
Reasons for treatment discontinuation are summarized
in Table IV. The discontinuation
rate owing to cutaneous toxicity was 19.2% (5/26) in the standard-
and 15.0% (3/20) in the reduced-dose group (P>0.999).
 | Table IV.Reasons for treatment discontinuation
in the standard-dose group and reduced-dose group. |
Table IV.
Reasons for treatment discontinuation
in the standard-dose group and reduced-dose group.
| Reason | Standard-dose group
(n=26) | Reduced-dose group
(n=20) | P-value |
|---|
| Adverse events
(%) | 7 (26.9) | 5 (25.0) | >0.999 |
|
Cutaneous adverse events | 5 (19.2) | 3 (15.0) | >0.999 |
|
Pneumonitis | 2 (7.7) | 0 (0.0) | 0.498 |
|
Somnolence | 0 (0.0) | 1 (5.0) | 0.435 |
|
Pyrexia | 0 (0.0) | 1 (5.0) | 0.435 |
| Disease
progression | 6 (23.1) | 6 (30.0) | 0.848 |
| Change in treatment
strategy | 3 (11.5) | 0 (0.0) | 0.246 |
| Observation without
active treatment | 2 (7.7) | 0 (0) | 0.498 |
| Surgery | 1 (3.8) | 0 (0.0) | >0.999 |
| Details
unknown | 0 (0.0) | 2 (10.0) | 0.184 |
| Ongoing | 10 (38.5) | 7 (35.0) | >0.999 |
Dose modifications
Details of the dose modifications are presented in
Table V. Among the 20 patients in
the reduced-dose group, 7 patients remained on the initial dose, 6
patients underwent further dose reductions and 7 patients
experienced dose escalation. The reasons for the additional dose
reduction included patient preference (n=1), rash (n=4) and
neutropenia (n=1). Among the 26 patients in the standard-dose
group, 19 were maintained on their initial dose, while 7 patients
required dose reduction, all due to a rash. The clinical course and
dose-escalation timeline in patients who commenced apalutamide at a
reduced dose are summarized in Table
VI. Notably, one patient (patient no. 1) was initiated at 120
mg/day, escalated to 180 mg/day on day 29 and 240 mg/day on day 57,
without the occurrence of cutaneous toxicity during this period.
However, cutaneous toxicity developed on day 92, leading to a dose
reduction to 120 mg/day. As cutaneous symptoms did not adequately
improve, treatment was discontinued on day 97. An additional
patient (patient no. 2) who was also initiated at 120 mg/day,
temporarily discontinued treatment on day 76 due to cutaneous
toxicity, but resumed therapy at 120 mg/day on day 148, after
symptoms improved. The dose was then escalated to 180 mg/day on day
442 and the patient continued treatment until disease progression.
No cutaneous toxicity was observed in the remaining 5 patients
either before or after dose escalation.
 | Table V.Dose modifications during
treatment. |
Table V.
Dose modifications during
treatment.
| Group | Dose modification
type | Dose pathway | No. of cases with
body weight <67 kg and BMI <24 kg/m2 | Reason for dose
reduction | No. of cases |
|---|
| Reduced-dose | Dose
escalation | 180-240 mg
(n=4) | 3 | - | - |
| (180 mg, n=12; |
| 120-180 mg
(n=2) | 1 | - | - |
| 120 mg, n=8) |
| 120-180-240 mg
(n=1) | 1 | - | - |
|
| Same dose | 180 mg (n=4) | 3 | - | - |
|
|
| 120 mg (n=3) | 2 | - | - |
|
| Dose reduction | 180-120 mg
(n=4) | 3 | Patient
preference | 1 |
|
|
|
|
| Skin rash |
|
|
|
|
|
| Grade 2 | 4 |
|
|
| 120-60 mg
(n=2) | 1 | Neutropenia |
|
|
|
|
|
| Grade 3 | 1 |
| Standard-dose | Same dose | 240 mg (n=19) | 10 | - | - |
| group (240 mg, | Dose reduction | 240-180 mg
(n=7) | 5 | Skin rash |
|
| n=26) |
|
|
| Grade 2 | 4 |
|
|
|
|
| Grade 3 | 3 |
 | Table VI.Clinical course and dose-escalation
timeline in patients who started apalutamide at a reduced dose. |
Table VI.
Clinical course and dose-escalation
timeline in patients who started apalutamide at a reduced dose.
| Patient no. | Body weight,
kg | BMI,
kg/m2 | Day from
initiation | Event |
|---|
| 1 | 59.7 | 23.0 | 1 | Started apalutamide
at 120 mg/day |
|
|
|
| 29 | Increased to 180
mg/day |
|
|
|
| 57 | Increased to 240
mg/day |
|
|
|
| 92 | Dose reduced to 120
mg/day due to cutaneous toxicity |
|
|
|
| 97 | Treatment
discontinued due to persistent cutaneous toxicity |
| 2 | 73.0 | 24.7 | 1 | Started at 120
mg/day |
|
|
|
| 76 | Treatment
interrupted due to cutaneous toxicity |
|
|
|
| 148 | Restarted at 120
mg/day after cutaneous toxicity improvement |
|
|
|
| 442 | Increased to 180
mg/day |
|
|
|
| 469 | Discontinued due to
PD |
| 3 | 61.6 | 22.8 | 1 | Started at 180
mg/day |
|
|
|
| 127 | Increased to 240
mg/day |
|
|
|
| 694 | Discontinued due to
PD |
| 4 | 37.9 | 16.2 | 1 | Started at 120
mg/day |
|
|
|
| 127 | Increased to 180
mg/day |
|
|
|
| 183 | Discontinued due to
PD |
| 5 | 55.1 | 20.6 | 1 | Started at 180
mg/day |
|
|
|
| 57 | Increased to 240
mg/day (ongoing treatment) |
| 6 | 67.9 | 22.4 | 1 | Started at 180
mg/day |
|
|
|
| 36 | Increased to 240
mg/day (ongoing treatment) |
| 7 | 60.3 | 22.7 | 1 | Started at 180
mg/day |
|
|
|
| 43 | Increased to 240
mg/day (ongoing treatment) |
Time to achieve PSA <0.2 ng/ml and
PFS
Details of the time taken to achieve PSA <0.2
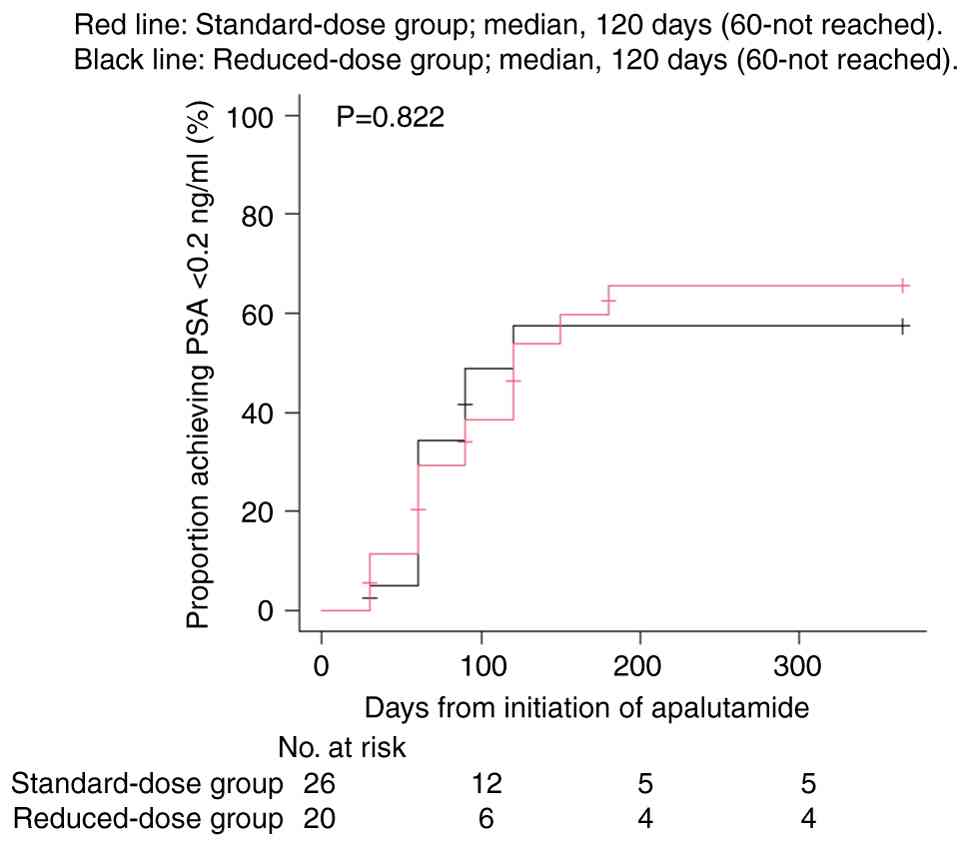
ng/ml are shown in Fig. 1. The
median time was 120.0 (95% CI, 60.0-not reached) days in both the
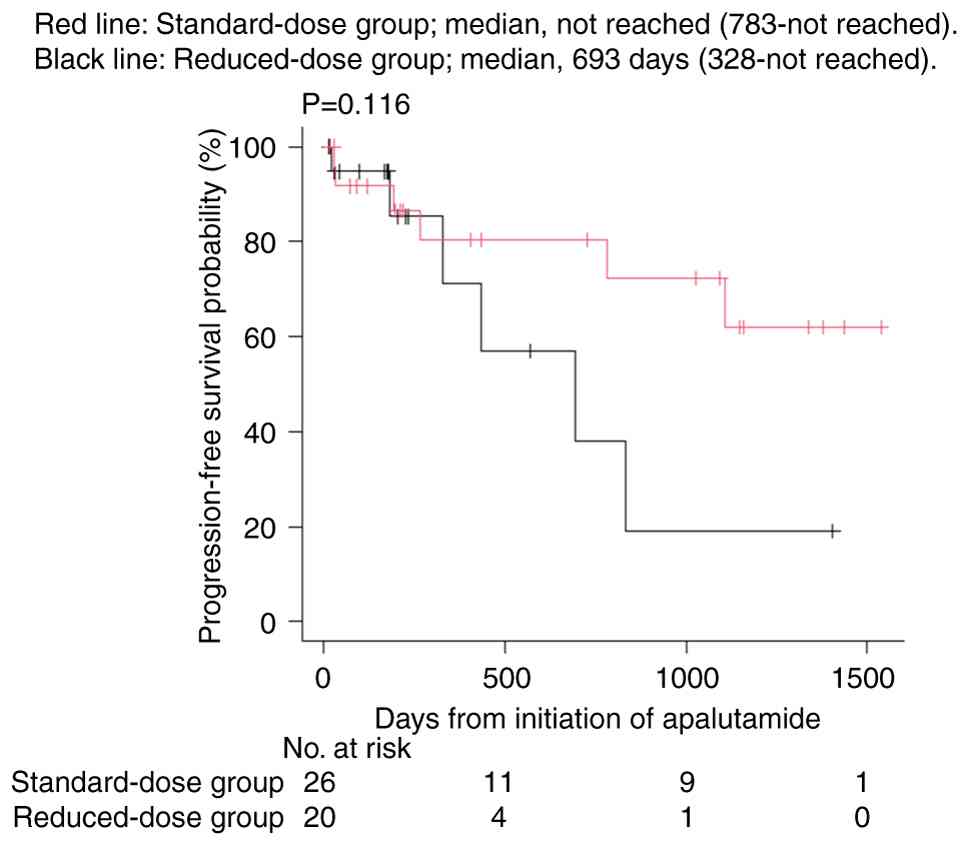
standard-dose and reduced-dose groups (P=0.822; Fig. 1). PFS is shown in Fig. 2. The median PFS was not reached in
the standard-dose group (95% CI, 783.0 days-not reached) and 693.0
(95% CI, 328.0-not reached) days in the reduced-dose group, with no
significant difference between the groups (P=0.116). Receiver
operating characteristic curve analysis of the RDI/BSA for
predicting cutaneous toxicity identified a cut-off value of 42.940,
with an area under the curve of 0.584, sensitivity of 0.481 and a
specificity of 0.737 (Fig.
S1).
Discussion
To the best of our knowledge, the present study is
the first retrospective report to reveal the effect of initiating
apalutamide at a reduced dose with subsequent stepwise escalation
on the incidence of cutaneous toxicities and PSA decline (a
surrogate marker of treatment efficacy). Overall, the incidence of
cutaneous adverse events was significantly lower in the
reduced-dose group (40.0%) compared with the standard-dose group
(73.1%) and no grade ≥3 events were observed in the reduced-dose
group. There was no significant difference in the rate of treatment
discontinuation due to rash between the two groups. Therefore,
initiating treatment at a reduced dose may help prevent severe
cutaneous toxicities, thereby minimizing treatment interruptions
and enabling patients to maintain continuous therapy without
compromising efficacy. By contrast, the median time to achieve PSA
<0.2 ng/ml, an indicator of therapeutic response, was 120.0 days
in both groups, with no significant difference between the groups.
Similarly, the median PFS was not reached in the standard-dose
group but was 693.0 days in the reduced-dose group, with no
significant difference between the two groups. Thus, initiating
treatment at a reduced dose with gradual escalation significantly
decreased the incidence of cutaneous toxicities without causing a
marked delay in the PSA response or a reduction in treatment
efficacy. However, the possibility of clinically relevant
differences being overlooked cannot be excluded due to the limited
sample size and number of events in the present study, yet the
present findings suggest that flexible dosing strategies may
mitigate adverse events without compromising efficacy, underscoring
the importance of individualized treatment approaches.
In current Japanese clinical practice guidelines for
prostate cancer, dose-adjustment strategies specific to apalutamide
are not described (14). The
approved apalutamide dosage is uniformly 240 mg once daily and
recommendations for dose reduction or re-escalation in response to
adverse events rely primarily on the Japanese package insert
(2). Stepwise dose reductions to
180 and 120 mg are permitted based on tolerability (2). However, no further guidance is
provided regarding individualized dose initiation based on patient
characteristics such as body size. Consequently, in the present
study, the dosing strategy used was guided by the package insert
and real-world clinical considerations rather than by
guideline-based recommendations.
In the reduced-dose group, the BSA at treatment
initiation was significantly smaller compared with that of the
standard-dose group. Previous studies have shown that patients with
low body weight or small BSA are at a higher risk of increased drug
exposure and cutaneous adverse events compared with those without
(7,8). Therefore, the prescribing physicians
may have selected reduced-dose at initiation for smaller-bodied
patients to prevent excessive drug exposure. Furthermore, the
incidence of cutaneous toxicity was significantly lower in the
reduced-dose group compared with that in the standard-dose group
and no severe cutaneous toxicities were observed. These findings
indicate that the lower incidence of cutaneous toxicity in the
reduced-dose group may have been attributable to the dose reduction
in patients with a small BSA. While studies have reported a
significant association between body size and the risk of cutaneous
toxicity (7,8), other investigations have found no such
relationship (5,9). A number of factors may account for
these inconsistencies. First, differences in patient
characteristics, including baseline disease severity, prior
systemic therapy and ethnic composition, may influence rash
susceptibility independent of body size. Second, variation in
dosing practices, including the timing and criteria for dose
escalation or reduction, could alter drug exposure and affect the
incidence of rash. Third, methodological differences, including
retrospective design, small sample sizes and inconsistent
definitions or grading of cutaneous events, may contribute to
divergent findings. Together, these factors may explain why
numerous studies fail to detect a clear association between body
size and rash risk. Accordingly, the present findings of a lower
incidence of rash with reduced-dose initiation in a cohort with
smaller body size should be interpreted in light of these
complexities and the limitations of real-world data. Among the
seven patients in the reduced-dose group who underwent dose
escalation, only 1 developed cutaneous toxicity after escalation.
In this patient, the dose was increased within the first month of
treatment, a rash developed on day 92 and therapy was subsequently
discontinued. In the remaining 6 patients, no new adverse events
were observed following dose escalation, suggesting that an initial
dose reduction followed by gradual escalation after confirming
tolerability may represent a safe and effective treatment strategy.
These findings highlight the importance of avoiding early dose
escalation and closely monitoring adverse events during the first
2–3 months of treatment.
In the standard-dose group, >50% of the patients
weighed <67 kg or had a BMI <24 kg/m2, with these
populations having been previously reported to be at an increased
risk of cutaneous toxicity (7). It
is conceivable that the reduced initial dose in these patients may
have prevented the occurrence of rash. The overall incidence of
cutaneous toxicity in the present study of 58.7% (27/46) was
slightly higher compared with the 51.5% reported in an integrated
analysis of Japanese clinical trials (5). Similarly, in other Asian populations,
the incidence of cutaneous adverse events was reported to be 32.0%
in Chinese patients and 36.4% in Korean patients (15,16).
This discrepancy between the present study and previous reports may
be explained by the inclusion of patients with small BSA in the
standard-dose group, who did not receive a dose reduction at
initiation. The incidence and severity of cutaneous toxicities were
significantly higher in the standard-dose group compared with the
reduced-dose group and although some patients developed severe
events, there was no significant difference in treatment
discontinuation rates between the groups. This finding suggests
that early recognition of rash and appropriate management,
including temporary interruption, dose reduction and supportive
care, enabled patients to continue therapy despite experiencing
adverse events. Conversely, initiating apalutamide at a reduced
dose may further reduce the risk of severe cutaneous toxicities and
minimize the need for treatment interruption or additional
interventions, representing a more practical approach in real-world
clinical practice. Therefore, although treatment continuation was
achievable in both groups, initiating therapy at a reduced dose may
offer advantages in improving tolerability and ensuring treatment
safety.
The median time to the first occurrence of cutaneous
toxicity was 63.0 days in the standard-dose group and 45.5 days in
the reduced-dose group, which is consistent with previous reports
of a median onset of 66.0 days in Japanese patients (5). Comparable findings have been reported
in other Asian populations, with a median onset of 57.5 days in
Chinese and 65.5 days in Korean patients (15,16).
In the TITAN trial, the median time to the first rash was 80.5 days
in the overall population, compared with that of the Japanese
subpopulation at 60.0 days, suggesting that patients with smaller
body size may develop rashes earlier (4). Although the difference was not
significant in the present study, the slightly earlier onset of
cutaneous toxicity observed in the reduced-dose group may reflect
the higher proportion of patients with smaller BSA in this group,
consistent with the findings of the TITAN trial. This observation
emphasizes the importance of early patient education and prompt
management of adverse events, particularly during the initial phase
of treatment in Japanese patients with smaller body size. The
management of rashes at the first occurrence did not differ between
the two groups with the majority of patients receiving oral
antihistamines and topical corticosteroids. This finding is
consistent with a previous report indicating that 71.4% of Japanese
patients who developed apalutamide-related rash were treated with
antihistamines (5).
Although the median RDI was significantly lower in
the reduced-dose group than in the standard-dose group (59.3 vs.
97.2%), the time to achieve PSA <0.2 ng/ml and PFS did not
differ significantly between the groups. This suggests a plateau
effect of apalutamide, whereby the therapeutic efficacy may not
increase proportionally beyond a certain drug exposure threshold
(17). Interindividual variability
in the PSA response, influenced by body size, drug metabolism and
tumor biology, may also account for the observed findings (6,7).
Considering that PSA is the only surrogate marker of treatment
efficacy, short-term PSA kinetics may not fully capture overall
therapeutic benefits. In the present study, no significant
difference in PFS was observed between the standard-dose and
reduced-dose groups. This finding is consistent with a previous
report on patients with mCSPC, which demonstrated no notable
difference in castration-resistant prostate cancer-free survival
between standard- and reduced-dose groups (10). However, both groups in the present
study included patients with nmCRPC. Conversely, another previous
study suggested that initiating apalutamide at a reduced dose may
reduce the time to castration-resistant prostate cancer compared
with that of standard-dose initiation (18). A number of factors may explain the
differing results between the previous study and the present
analysis. First, the prior study only included patients with mCSPC,
whereas the present cohort comprised patients with both mCSPC and
nmCRPC, creating different baseline risk profiles. Second, the
reduced-dose group in the previous study may have included patients
who were clinically frail or those with adverse prognostic
features, introducing confounding by indication. Third, variations
in dose-escalation strategies, namely fixed reduced-dose initiation
in the previous study vs. the flexible stepwise-escalation approach
used in the present study, may have influenced the overall drug
exposure and therapeutic outcomes. Given these considerations and
the limited sample size of the present study, findings should be
interpreted with caution. Prospective studies are further needed to
determine whether reduced-dose initiation can reliably maintain
oncologic outcomes across diverse patient populations.
Collectively, the initial reduced dose may mitigate adverse events
without compromising efficacy, however patient-specific
characteristics must be considered when tailoring dosing regimens.
Sasaki et al (7) reported
that patients weighing <67 kg and with a BMI <24
kg/m2 had a markedly higher incidence of rash,
supporting the clinical relevance of body size-based dose
adjustments. However, these criteria remain provisional and further
research is required to establish evidence-based dosing algorithms
that integrate multiple patient factors.
With regard to the time taken to achieve PSA <0.2
ng/ml and PFS, the limited sample size and number of events in the
present study may have reduced the statistical power to detect
modest differences. Therefore, although no significant differences
were observed, the possibility of a type II error cannot be
excluded. However, the similarity in the PSA response patterns
between the groups suggests that reduced-dose initiation may
preserve short-term antitumor efficacy, which is clinically
important.
Akagi et al (19) reported a stronger association of
rash risk with RDI adjusted for BSA compared with RDI adjusted for
body weight. While this analysis focused on toxicity, the effects
of RDI or RDI/BSA on the PSA response remain unexplored. To the
best of our knowledge, to date, no studies have directly examined
the association between RDI and PSA decline; however, previous
reports have suggested that dose adjustments aimed at reducing
cutaneous toxicity do not markedly affect PFS (10,17)
and the present findings were consistent with these observations.
Exploratory analysis identified an RDI/BSA cut-off value of 42.940
(area under the curve=0.5838: 95% CI, 0.415–0.752; specificity,
0.737; sensitivity, 0.481) for predicting cutaneous toxicity.
RDI/BSA demonstrated limited discriminative ability for predicting
cutaneous toxicity, suggesting this index alone may be insufficient
as a robust clinical predictor. As the present exploratory analysis
included a small, single-center cohort comprising exclusively
Japanese patients, the generalizability of the estimated cut-off
value is limited. Differences in body size, pharmacokinetics and
treatment practices across ethnic groups may influence apalutamide
exposure and toxicity. Therefore, the optimal RDI/BSA threshold may
vary among populations. Notably, the cut-off value identified in
the present study was lower than that previously reported (RDI/BSA
≥56) (19), which may reflect
variations in patient characteristics, study design or sample size.
Validation in larger, multiethnic cohorts is required to further
determine exposure thresholds that minimize cutaneous toxicity
while maintaining treatment efficacy.
The present single-center retrospective
observational study has a number of limitations. First, despite
existing predefined exclusion criteria, no patients were excluded
on account of these, resulting in an unselected, real-world cohort.
While this enhances the practical relevance of the present study,
the lack of patient exclusions also raises the possibility that
unmeasured comorbidities, pre-existing dermatologic conditions or
concomitant medications may have contributed to toxicity or
treatment outcome variabilities. Second, inherent biases associated
with the retrospective design, including information and selection
biases, could not be excluded and reliance on medical records
introduces the possibility of incomplete data collection. Third,
findings were based on a limited patient population and differences
in baseline characteristics such as BSA and initial PSA levels
between the groups may have influenced the observed outcomes.
Although additional analyses adjusting for these variables were
conducted using propensity score matching (data not shown), these
analyses showed no notable differences in the incidence of
cutaneous toxicity or progression-free survival between the
standard- and reduced-dose groups. However, the limited sample size
warrants cautious interpretation, as residual confounding cannot be
completely excluded. Consequently, baseline differences,
particularly those related to factors such as body size or tumor
burden, may have influenced toxicity or treatment response
independent of the dosing strategy. These limitations should be
considered when interpreting the association between the
dose-initiation approach and clinical outcomes. Fourth, the lack of
significant differences in some endpoints (such as time to achieve
PSA <0.2 ng/ml) may reflect insufficient statistical power
rather than true equivalence. Furthermore, as this was a
single-center study conducted exclusively in Japanese patients,
caution should be exercised when extrapolating the findings to
other ethnic populations with different pharmacogenetic backgrounds
or body compositions. In addition, published evidence on
apalutamide-related cutaneous toxicity in non-Asian populations is
limited, making it difficult to determine whether the
exposure-toxicity patterns observed in East Asian cohorts apply to
other racial or ethnic groups. The absence of comparable data from
Western or African populations further constrains the global
generalizability of the present findings. Future multicenter
prospective studies, the development of individualized dosing
strategies based on patient body size and the evaluation of
long-term outcomes such as overall survival and PFS are
warranted.
In conclusion, the findings of the present study
suggest that initiating apalutamide at a reduced dose with stepwise
escalation may decrease the risk of cutaneous toxicities in
Japanese patients with a small BSA, while potentially mitigating
adverse events, without compromising short-term efficacy.
Furthermore, the present study highlights the importance of careful
monitoring and dose adjustment during the first 2–3 months of
treatment. Establishing individualized dosing protocols that
incorporate body size and adverse event risk is an important step
toward optimizing apalutamide therapy.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study are not
publicly available due to restrictions imposed by patient consent
and institutional regulations but may be requested from the
corresponding author.
Authors' contributions
TM and MK designed the study. TM, MG, SY, MN, HM and
RM collected the data. TM analyzed and interpreted the data and
wrote the manuscript. MG, SY, MN, HM and RM interpreted data. MK
and EU interpreted data and revised the manuscript. All authors
have read and approved the final manuscript. TM and MK confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
The present study was conducted in accordance with
the Ethical Guidelines for Medical and Health Research Involving
Human Subjects in Japan and was approved by the Ethics Committee of
Ogaki Municipal Hospital (approval no. 20251225-7h). The present
study did not involve the collection of any new samples or
information but was conducted solely using existing data, therefore
written or verbal consent from the participants was not obtained.
Instead, information regarding the present study was disclosed to
the participants (through postings within the hospital or on the
hospital website), thereby ensuring that the participants had the
opportunity to opt out of the present study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
BSA
|
body surface area
|
|
PSA
|
prostate-specific antigen
|
|
RDI
|
relative dose intensity
|
|
PFS
|
progression-free survival
|
|
nmCRPC
|
non-metastatic castration-resistant
prostate cancer
|
|
mCSPC
|
metastatic castration-sensitive
prostate cancer
|
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:17–48. 2024.
|
|
2
|
Janssen Pharmaceutical K.K., . Erleada
(apalutamide) package insert, 2024. https://www.pmda.go.jp/PmdaSearch/iyakuDetail/ResultDataSetPDF/800155_4291059F1029_1_10November
18–2025(In Japanese).
|
|
3
|
Smith MR, Saad F, Chowdhury S, Oudard S,
Hadaschik BA, Graff JN, Olmos D, Mainwaring PN, Lee JY, Uemura H,
et al: Apalutamide treatment and metastasis-free survival in
prostate cancer. N Engl J Med. 378:1408–1418. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chi KN, Agarwal N, Bjartell A, Chung BH,
de Santana Gomes AJ, Given R, Soto ÁJ, Merseburger AS, Özgüroğlu M,
Uemura H, et al: Apalutamide for metastatic, castration-sensitive
prostate cancer. N Engl J Med. 381:13–24. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Uemura H, Koroki Y, Iwaki Y, Imanaka K,
Kambara T, Lopez-Gitlitz A, Smith A and Uemura H: Skin rash
following administration of apalutamide in Japanese patients with
advanced prostate cancer: An integrated analysis of the phase 3
SPARTAN and TITAN studies and a phase 1 open-label study. BMC Urol.
20:1392020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Uemura H, Satoh T, Tsumura H, Arai G,
Imanaka K, Shibayama K, Fujii K, Rooney B, Lopez-Gitlitz A, Espina
B, et al: Efficacy and safety of apalutamide in Japanese patients
with nonmetastatic castration-resistant prostate cancer: A subgroup
analysis of a randomized, double-blind, placebo-controlled, phase-3
study. Prostate Int. 8:190–197. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sasaki D, Hatakeyama S, Tanaka T, Okamoto
T, Yoneyama T and Ohyama C: Impact of body size on skin-related
adverse events in advanced prostate cancer treated with
apalutamide: A multicenter retrospective study. Int J Urol.
29:772–773. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Katsuta M, Kimura T, Tashiro K, Murakami
M, Hata K, Yanagisawa T, Onuma H, Yamamoto T, Sugaya S, Watanabe Y,
et al: Low body weight as a risk factor for apalutamide-related
cutaneous adverse events. Anticancer Res. 42:2023–2028. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tohi Y, Kato T, Fukuhara H, Kobayashi K,
Ohira S, Ikeda K, Daizumoto K, Katayama S, Shimizu R, Nishimura K,
et al: Real-world analysis of apalutamide-associated skin adverse
events in Japanese patients with advanced prostate cancer: A
multi-institutional study in the Chu-shikoku Japan Urological
Consortium. Int J Clin Oncol. 27:1348–1355. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Oishi T, Hatakeyama S, Tabata R, Fujimori
D, Kawashima Y, Tanaka R, Ishii N, Miura H, Tanaka T, Okamoto T, et
al: Effects of apalutamide dose reduction on skin-related adverse
events in patients with advanced prostate cancer: A multicenter
retrospective study. Prostate. 83:198–203. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Saad F, Small EJ, Feng FY, Graff JN, Olmos
D, Hadaschik BA, Oudard S, Londhe A, Bhaumik A, Lopez-Gitlitz A, et
al: Deep prostate-specific antigen response following addition of
apalutamide to ongoing androgen deprivation therapy and long-term
clinical benefit in SPARTAN. Eur Urol. 81:184–192. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
National Cancer Institute, . Common
Terminology Criteria for Adverse Events (CTCAE) Version 5.0. U.S.
Department of Health and Human Services; 2017, https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5×7.pdfNovember
18–2025
|
|
13
|
Kanda Y: Investigation of the freely
available easy-to-use software ‘EZR’ for medical statistics. Bone
Marrow Transplant. 48:452–458. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Japanese Urological Association: Clinical
Practice Guidelines for Prostate Cancer 2023. Kanehara & Co.,
Ltd. (Tokyo). 2023.
|
|
15
|
Yang Z, Shao Y, Huang H, Liu Y, Wang Z and
Wang Y: Real-world analysis of apalutamide-associated skin rash in
Chinese patients with prostate cancer. World J Urol. 42:1712024.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kim WT, Han HH, Yun SJ, Yu SH, Kang TW, Ha
YS, Lee JN, Kwon TG, Kim BH, Seo WI, et al: Skin rash in metastatic
hormone sensitive prostate cancer patients treated with
apalutamide: A retrospective multicenter study in Korea. Prostate
Int. 13:15–21. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
T'jollyn H, Ackaert O, Chien C,
Lopez-Gitlitz A, McCarthy S, Ruixo CP, Karsh L, Chi K, Chowdhury S,
Ruixo JP and Agarwal N: Efficacy and safety exposure-response
relationships of apalutamide in patients with metastatic
castration-sensitive prostate cancer: Results from the phase 3
TITAN study. Cancer Chemother Pharmacol. 89:629–641. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tohi Y, Kato T, Kobayashi K, Daizumoto K,
Fukuhara H, Ohira S, Katayama S, Shimizu R, Takamoto A, Nishimura
K, et al: Real-world prostate-specific antigen response and
progression to castration-resistant prostate cancer among men with
metastatic castration-sensitive prostate cancer treated with
apalutamide: A multi-institutional study in the Chu-shikoku Japan
Urological Consortium. Jpn J Clin Oncol. 54:167–174. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Akagi N, Obayashi R, Yamamoto A, Nagoshi
A, Fujiwara T, Igarashi A, Hattori Y, Shibasaki N, Kawakita M and
Yamasaki T: Skin rash induced by apalutamide correlated with age
and relative dose intensity adjusted by body surface area in
Japanese patients with prostate cancer. Jpn J Clin Oncol.
55:963–969. 2025. View Article : Google Scholar : PubMed/NCBI
|