Introduction
Tirabrutinib, an orally selective and irreversible
Bruton's tyrosine kinase (BTK) inhibitor, covalently binds to BTK
(1). Primary central nervous system
lymphomas (PCNSLs) are rare and aggressive extranodal lymphomas,
with a rising incidence particularly in the elderly population
(2). While the majority of lymphoma
cases are diffuse large B-cell lymphomas, PCNSL exhibits
significant clinicopathological and radiological diversity, and the
diagnosis remains challenging, often requiring invasive biopsies
(3). Furthermore, the non-germinal
center B-cell immunohistochemical subtype is more common in PCNSL
than the germinal center B-cell subtype, and is associated with
unfavorable prognostic factors, such as advanced age (>60
years), a high Ki-67 labeling index and a shorter overall survival
time (4). PCNSLs are commonly
treated with high-dose methotrexate (MTX)-based regimens or
induction therapy comprising whole-brain radiation therapy,
followed by high-dose systemic chemotherapy (5). Although highly responsive to
first-line chemotherapy and radiotherapy, ~50% of patients with
PCNSL experience relapse or refractory disease within 1 year
(6). Tirabrutinib, a
second-generation BTK inhibitor, was first approved in Japan for
the treatment of relapsed or refractory PCNSL. Tirabrutinib targets
key signaling pathways, such as the nuclear factor-κB pathway,
which are critical for tumor cell survival (7). Patients with PCNSL often present with
neurological deficits, and the clinical impact of dysphagia is
particularly important in this population. Tablets with a total
dimension >21 mm (length, width and thickness combined) have
been reported to affect adherence in patients with swallowing
disorders (8). The tirabrutinib
tablet has a combined dimension of 21.5 mm. Therefore,
administering tirabrutinib in patients with PCNSL can be difficult
in cases where oral administration is not feasible. If tirabrutinib
therapy is critical, administration of tirabrutinib as a suspension
may be necessary. However, the manufacturer does not provide
pharmacokinetic or safety data for tirabrutinib administered as a
suspension or in crushed form. Two previous reports have described
the administration of tirabrutinib suspension via a nasogastric
tube in patients with PCNSL (9,10), but
none have reported plasma concentrations. In the present case
report, the safety, efficacy and plasma concentration of
tirabrutinib administered as a suspension is evaluated in a patient
with an inability to swallow tablets. Additionally, the relevant
literature on the suspension administration of other tyrosine
kinase inhibitors (TKIs), similar to that described in the present
case, is reviewed.
Case report
A 68-year-old man first presented in February 2023
to Tokyo Metropolitan Bokutoh Hospital (Tokyo, Japan) with weakness
in the right leg and diplopia in the right eye. The diagnosis of
primary central nervous system lymphoma (PCNSL) was established
based on cerebrospinal fluid (CSF) cytology, immunocytochemical
analysis and flow cytometric immunophenotyping, in conjunction with
characteristic radiological findings. CSF cytology revealed
clusters of atypical large lymphoid cells with irregular nuclei and
prominent nucleoli. Immunocytochemical staining demonstrated strong
membranous CD20 expression in the majority of atypical cells, and
Ki-67 staining showed a high proliferative index. Flow cytometry
confirmed a dominant B-cell population expressing CD19 and CD20.
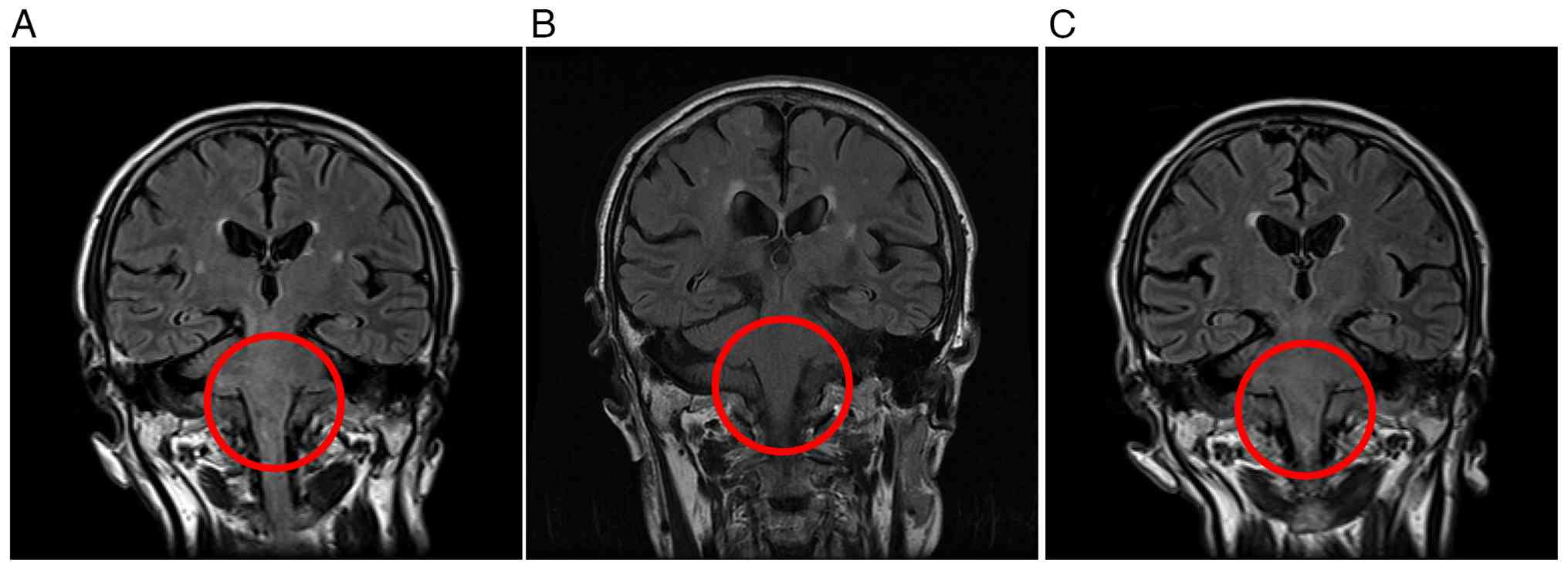
Given the typical MRI findings (Fig.
1A) and the clinical context, a diagnosis of PCNSL was made. A
stereotactic brain biopsy was not performed due to the deep
midbrain location of the lesion and the associated procedural risk.
After diagnosis, the patient received intrathecal chemotherapy
consisting of 40 mg/body prednisolone, 15 mg/body methotrexate and
40 mg/body cytarabine per administration. In parallel, the patient
underwent methotrexate-based systemic chemotherapy combined with
rituximab for four cycles. Rituximab was administered at 375
mg/m2 on day 1 of each 21-day cycle, and high-dose
methotrexate was administered at 3,500 mg/m2 on day 3.
Brain magnetic resonance imaging (Fig.
1B) demonstrated disappearance of the intracranial lesions, and
the patient was judged to have achieved a complete response (CR).
However, 3 months later, the patient relapsed and was treated again
with four cycles of intrathecal MTX, cytarabine and steroids using
the same dosing as previously applied. This was followed by
whole-brain radiotherapy delivered at a total dose of 36 Gy in 20
fractions, which resulted in another CR. At 1-month post-CR, the
patient presented with facial paralysis (inability to close the
right eye), dysphagia and gait instability. Brain magnetic
resonance imaging (MRI) confirmed a second relapse (Fig. 1C). Intrathecal MTX, cytarabine and
steroids were reintroduced at the same dosage as previously
applied, and tirabrutinib was initiated. Tirabrutinib was
administered at a dose of 480 mg once daily on an empty stomach,
which is the approved dose in Japan for the treatment of
relapsed/refractory PCNSL. Owing to severe dysphagia caused by
facial paralysis, a nasogastric tube was inserted and tirabrutinib
was administered as a suspension through this once daily. Following
a protocol similar to that reported by Yoshioka et al
(9), six tablets of tirabrutinib
(80 mg) were placed in a container, 20 ml of warm water at 55°C was
added and the mixture was left for 10 min to dissolve. After
confirming complete dissolution of the tirabrutinib tablets in the
container, the resulting suspension was drawn into a syringe and
administered via the nasogastric tube. To ensure complete
administration of the suspension and prevent tube obstruction, the
nasogastric tube was flushed with 20 ml of water after
administering the suspension. Upon admission in November 2023, the
patient received nutritional support via a nasogastric tube, and
the facial paralysis gradually improved. Notably, the patient's
dysphagia showed marked clinical improvement within 10 days of
tirabrutinib initiation, allowing the patient to transition to an
oral suspension (480 mg once daily) by day 11. By day 22, the
patient was able to swallow food and tirabrutinib tablets. The
patient was discharged on day 23, and oral tablets (480 mg once
daily) were continued thereafter. No adverse events were observed
during the suspension administration period.
Blood samples were collected on day 5 of
tirabrutinib treatment with the patient's consent. Plasma
tirabrutinib concentration (C2) was evaluated 2 h after
tirabrutinib administration. This was based on the assumption that
C2 would be close to maximum plasma concentration
(Cmax), given that the mean time to Cmax was
2.87 h (11). C2 samples
(50 µl plasma) were evaluated using high-performance liquid
chromatography (HPLC) coupled with ultraviolet (UV) spectroscopy.
The HPLC system included pumps (PU-4180), a UV detector (UV-4075)
and an autosampler (AS-4550) (Jasco Corporation). The mobile phase
comprised 0.5% potassium dihydrogen phosphate
(KH2PO4, pH 4.5) and acetonitrile (58:42,
v/v), pumped at a flow rate of 1.2 ml/m at 22°C. Tirabrutinib and
ibrutinib (internal standard) were detected at 215 nm using Capcell
Pak C18 MG II reversed-phase columns 250×4.6 mm (length × inner
diameter) (Osaka Soda Co., Ltd.). The accuracy, precision,
pretreatment recovery rate and sample stability of this
tirabrutinib detection method were validated according to the U.S.
Food and Drug Administration (12).
Hepatic function, which influences tirabrutinib
metabolism, was judged to be normal in the patient since blood
tests showed that hepatobiliary enzyme levels (aspartate
aminotransferase, alanine aminotransferase and total bilirubin)
were within the reference ranges. Concomitant previously prescribed
medications included 10 mg/day epinastine, 990 mg/day magnesium
oxide, 20 mg/day famotidine, 1 mg/day eszopiclone and 900 mg/day
acetaminophen, none of which interacted with tirabrutinib.
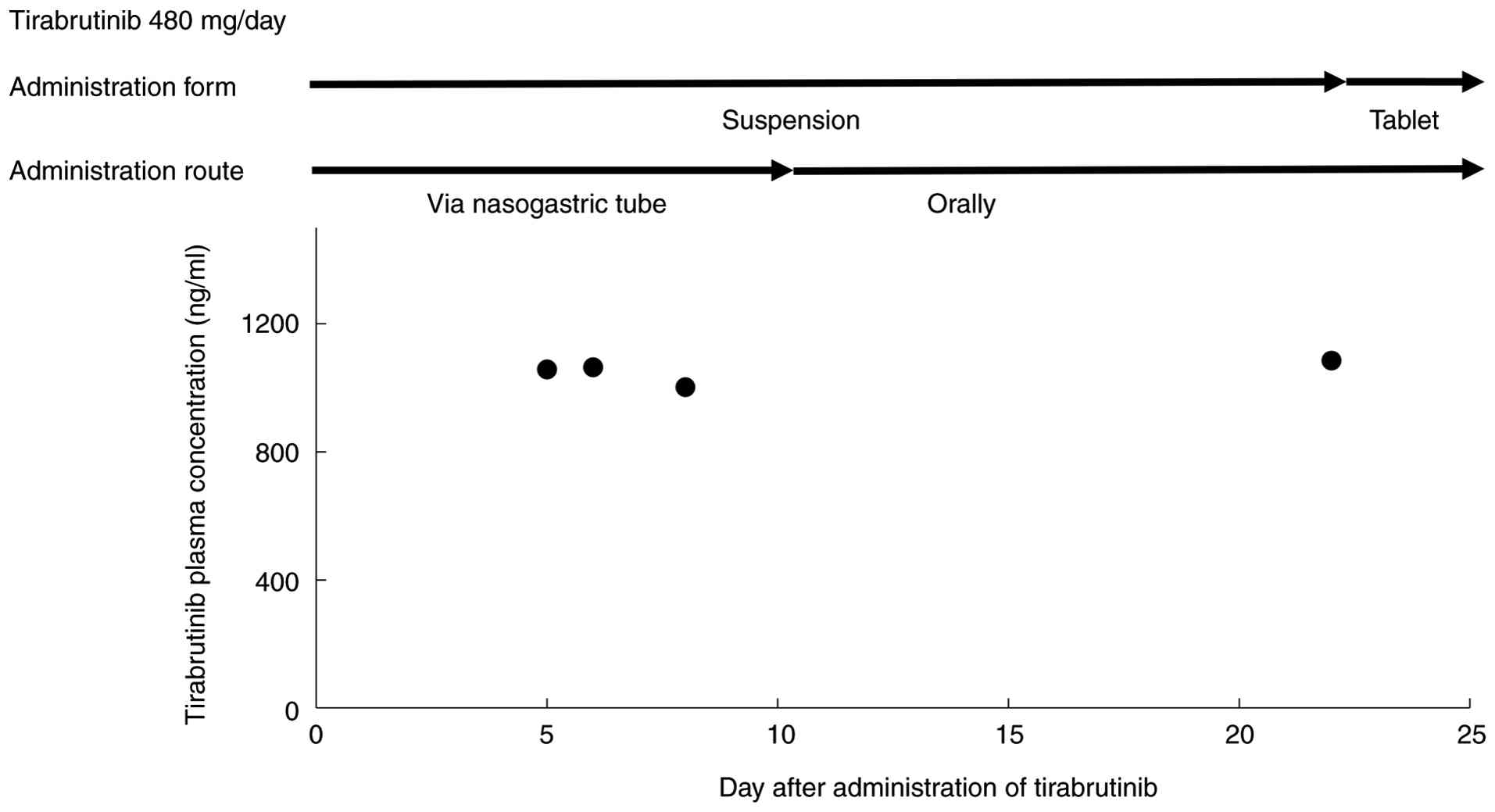
C2 values on days 5, 6, 8, 22 and 112 were 1,057, 1,064,
1,002, 1,085 and 1,042 ng/ml, respectively. Suspension was
administered via the nasogastric tube on days 5, 6 and 8, whereas
oral suspension was administered on day 22 (Fig. 2). Additionally, serum soluble
interleukin-2 receptor (siL-2R) levels (normal reference range,
157–474 U/ml) decreased from 795 U/ml at relapse to 607 U/ml on day
8 and to 532 U/ml on day 147 after the initiation of tirabrutinib.
Oral tirabrutinib tablets were administered on day 112 during the
outpatient visit. Follow-up visits were conducted approximately
every 2 months, and brain magnetic resonance imaging was performed
approximately every 6 months. At the 1-year follow-up in November
2024, the patient remained clinically stable, with no evidence of
intracranial or intraorbital lymphoma recurrence on brain MRI.
Tirabrutinib treatment has been continuously maintained, and
complete remission has been sustained as of December 2025.
Literature review
Using PubMed (https://pubmed.ncbi.nlm.nih.gov/), 17 studies
describing 19 patients who were administered TKI suspension were
identified (9,10,13–27).
These cases are summarized in Table
I. The duration of suspension administration ranged from 2 days
to 14 months. As in the present case, tirabrutinib was the most
frequently used TKI (4 patients), followed by alectinib (3
patients). Lung cancer was the most common malignancy (10
patients). Pharmacokinetics study and blood levels of TKIs were not
evaluated in any of the 17 articles.
 | Table I.Cases of administration of tyrosine
kinase inhibitor suspension. |
Table I.
Cases of administration of tyrosine
kinase inhibitor suspension.
| First author,
year | Tyrosine kinase
inhibitor | Malignancy | Age, years | Sex | Administration route
and daily dosage (duration) | (Refs.) |
|---|
| Yoshioka et
al, 2021 | Tirabrutinib | PCNSL | 76 | Female | Nasogastric tube, 480
mg (14 days) | (9) |
| Yoshioka et
al, 2022 | Tirabrutinib | PCNSL | 77/77/70 |
Female/female/female | Nasogastric tube, 480
mg (data not shown) | (10) |
| Okahashi et
al, 2020 | Ibrutinib | Mantle cell
lymphoma | 70 | Male | Nasogastric tube, 560
mg (90 days) | (13) |
| Alsuliman et
al, 2018 | Ibrutinib | Mantle cell
lymphoma | 69 | Male | Nasogastric tube,
560 mg (2 days) | (14) |
| Molinaro et
al, 2019 | Lenvatinib | Thyroid cancer | 62 | Female | Nasogastric tube,
20 mg (1 month); 10 mg (1 month) | (15) |
| Kawano et
al, 2020 | Lenvatinib | Thyroid cancer | 54 | Female | Nasogastric tube,
10 mg (10 days) | (16) |
| Tani et al,
2022 | Osimertinib | Lung cancer | 78 | Female | Nasogastric tube,
80 mg (20 days) | (17) |
| Takeda et
al, 2017 | Osimertinib | Lung cancer | 73 | Female | Nasogastric tube,
80 mg (20 days) | (18) |
| Suzumura et
al, 2014 | Gefitinib | Lung cancer | 72 | Female | Nasogastric tube,
250 mg (9 months) | (19) |
| Bejarano Varas
et al, 2019 | Alectinib | Lung cancer | 66 | Female | Nasogastric tube
and percutaneous endoscopic gastrostomy tube, 1,200 mg (30
days) | (20) |
| Kanai et al,
2017 | Alectinib | Lung cancer | 76 | Female | Nasogastric tube,
600 mg (14 months) | (21) |
| Thomas et
al, 2021 | Alectinib | Lung cancer | 90 | Male | Nasogastric tube
and percutaneous endoscopic gastrostomy tube, 1,200 mg (6
months) | (22) |
| Bosch-Barrera et
al, 2014 | Erlotinib | Lung cancer | 60 | Female | Nasogastric tube,
150 mg (5 days) | (23) |
| Sasaki et
al, 2021 | Lorlatinib | Lung cancer | 71 | Male | Nasogastric tube,
100 mg (4 weeks) | (24) |
| Wang et al,
2023 | Lorlatinib | Lung cancer | 49 | Male | Nasogastric tube,
100 mg (18 days) | (25) |
| Jang et al,
2024 | Trametinib,
dabrafenib | Lung cancer | 44 | Female | Nasogastric tube,
300 mg (33 days); Nasogastric tube, 2 mg (33 days) | (26) |
| Muta et al,
2010 | Imatinib | Chronic myeloid
leukemia | 64 | Female | Nasogastric tube,
400 mg (3 days); 600 mg (5 days) | (27) |
Discussion
Treatment options for refractory PCNSL are limited,
and tirabrutinib remains a crucial therapeutic choice. For patients
with dysphagia, alternative administration routes such as
nasogastric or gastrostomy tubes may be essential. To the best of
our knowledge, this is the first report to evaluate plasma
tirabrutinib concentrations following suspension administration via
a nasogastric tube or oral administration in a patient with
PCNSL.
The C2 values obtained in the present
case during nasogastric and oral administration of tirabrutinib
suspension (from days 5 to 22) were comparable to the
Cmax (1,220 ng/ml) reported in a previous study
(28) involving a dose of 480
mg/day under fasting conditions. Additionally, the C2
value on day 112 was consistent with that reported in the previous
study (28). However, it must be
noted that the reliance on C2 (a single-point
measurement) provides only a limited snapshot of the drug's
bioavailability. Without area under the curve (AUC) data,
bioequivalence cannot be definitively confirmed. Future
pharmacokinetic evaluations should consider the 24-h AUC rather
than C2 alone. In the present case, dysphagia markedly
improved in the patient within 10 days of initiating tirabrutinib
suspension. This clinical improvement is consistent with previous
reports (9,10), which showed that tirabrutinib
suspension can induce a partial response within 2–4 weeks. In the
present case, this clinical recovery was accompanied by a rapid
decrease in serum sIL-2R levels. This reduction in the tumor marker
is consistent with effective drug absorption and an early
antineoplastic effect, which likely contributed to the resolution
of neurological symptoms. While the clinical improvement with
regard to dysphagia may not be solely attributed to tirabrutinib,
as the effects of intrathecal MTX, cytarabine and steroids must be
considered, the administration of suspension was nonetheless the
essential facilitator for treatment initiation. No adverse events
were observed during either nasogastric or oral administration of
the suspension. Although, to the best of our knowledge, only 5
cases of PCNSL treated with tirabrutinib suspension have been
published to date (9,10), including the present case, the
administration of tirabrutinib suspension appears to be a viable
treatment option for patients with relapsed or refractory PCNSL who
are unable to swallow tablets. However, it cannot be definitively
concluded that the tirabrutinib suspension and tablet formulations
are bioequivalent based on this single-point measurement. This
suspension administration method has off-label use, but it was
performed with informed consent to save the patient's life.
Therefore, suspension administration requires careful handling.
Future prospective studies with serial blood sampling are warranted
to evaluate the full pharmacokinetic profile and long-term efficacy
of tirabrutinib suspension in this patient population.
An aging population likely results in an increased
number of elderly patients with dysphagia and poor adherence to TKI
therapy. Feeding tube placement has not been associated with
improved survival or post-discharge outcomes in older hospitalized
adults with dementia (29).
Alectinib suspension has been reported to be safe and effective in
patients with anaplastic lymphoma kinase-positive non-small cell
lung cancer and poor performance status after failure of 14-month
crizotinib therapy with continuous treatment via a feeding tube
(21). In several case reports,
alternative administration of TKI suspensions to patients with
transient dysphagia associated with cancer progression and
inability to be treated orally improved the dysphagia and other
tumor-related symptoms (such as dyspnea and impaired
consciousness), allowing the initiation of oral therapy
(9,10,13-17,20,23-26). The patient in the present report also
showed improvement in swallowing function after 10 days of
administration of tirabrutinib suspension. However, the
administration of TKIs in suspension form is not officially
approved or recommended by pharmaceutical companies, making it an
off-label use in clinical practice. Consequently, in routine
clinical practice, TKI suspensions may have to be administered via
nasogastric tubes. Pharmacokinetic studies on the administration of
TKI suspensions have been conducted for gefitinib (30), alectinib (31), dacomitinib (32), dasatinib (33) and pazopanib (34). For gefitinib and alectinib
administered to healthy volunteers, the AUC and Cmax
were generally similar for suspensions, tablets and capsules
(30,31). For dacomitinib, Cmax and
AUC were significantly decreased when administered via percutaneous
feeding gastrostomy tubes compared with oral administration of
tablets, in patients with locally advanced head and neck squamous
cell carcinoma (32). Compared with
oral tablet administration, greater mean drug exposure, decreased
half-life and higher Cmax were observed in patients
receiving dasatinib via percutaneous endoscopic gastrostomy tubes
(33). In a phase I study
evaluating pharmacokinetics, the administration of a single dose of
pazopanib suspension increased AUC and Cmax and
decreased time to Cmax in patients with advanced cancer,
indicating an increased rate and extent of oral absorption compared
with administration of the whole tablet (34). Pharmacokinetic changes in suspension
vary by TKI; for instance, dacomitinib shows decreased exposure via
tubes, while pazopanib shows increased absorption (32,34).
Such variability necessitates caution when extrapolating results.
Therefore, further studies are warranted to evaluate the
pharmacokinetics, safety and clinical outcomes of tirabrutinib and
other TKIs in suspension form across a broader patient population.
Furthermore, as tirabrutinib and other TKIs are antineoplastic
agents, healthcare staff must follow hazardous drug handling
guidelines, such as using closed systems or personal protective
equipment during preparation of suspensions to prevent occupational
exposure.
In conclusion, the present case demonstrates the
potential feasibility and efficacy of tirabrutinib suspension for
patients with PCNSL and severe dysphagia. While C2
levels were comparable across different administration routes, the
lack of comprehensive AUC data and the small sample size remain
significant limitations. In the relevant literature, 19 cases of
administration of TKI suspension have been reported; however,
pharmacokinetic studies were not evaluated in these cases.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
TY contributed to the conception and design of the
study, analyzed the data and drafted the manuscript. TY and YG were
responsible for measuring plasma tirabrutinib concentrations and
analyzing the pharmacokinetic data. MK performed the clinical
diagnosis and examinations, interpreted the radiological findings,
and was responsible for collecting and analyzing the patient's
clinical data. HO contributed to the literature review, collected
comparative data and analyzed the patient's clinical data. TY and
YG confirm the authenticity of all the raw data. All authors have
read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient for publication.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
PCNSL
|
primary central nervous system
lymphoma
|
|
BTK
|
Bruton's tyrosine kinase
|
|
MTX
|
methotrexate
|
|
TKI
|
tyrosine kinase inhibitor
|
|
CR
|
complete response
|
|
C2
|
plasma concentration of tirabrutinib
at 2 h after administration of tirabrutinib
|
|
Cmax
|
maximum plasma concentration
|
|
HPLC
|
high-performance liquid
chromatography
|
|
UV
|
ultraviolet
|
|
AUC
|
area under the curve
|
|
MRI
|
magnetic resonance imaging
|
References
|
1
|
Liclican A, Serafini L, Xing W,
Czerwieniec G, Steiner B, Wang T, Brendza KM, Lutz JD, Keegan KS,
Ray AS, et al: Biochemical characterization of tirabrutinib and
other irreversible inhibitors of Bruton's tyrosine kinase reveals
differences in on- and off-target inhibition. Biochim Biophys Acta
Gen Subj. 1864:1295312020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Korfel A, Schlegel U, Johnson DR, Kaufmann
TJ, Giannini C and Hirose T: Case-based review: Primary central
nervous system lymphoma. Neurooncol Pract. 4:46–59. 2017.PubMed/NCBI
|
|
3
|
Fiorentino V, Pizzimenti C, Pierconti F,
Lentini M, Ieni A, Caffo M, Angileri F, Tuccari G, Fadda G, Martini
M and Larocca LM: Unusual localization and clinical presentation of
primary central nervous system extranodal marginal zone B-cell
lymphoma: A case report. Oncol Lett. 26:4082023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rizzuto I, Pizzimenti C, Fiorentino V,
Scarcella SC, Martini M, Granata F, Germanò A, Ieni A and Tuccari
G: Morphological, immunophenotypic and neuroradiological
characteristics of primitive B-large cell diffuse lymphoma of the
central nervous system: A retrospective cohort analysis. Oncol
Lett. 30:4332025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Horbinski C, Nabors LB, Portnow J,
Baehring J, Bhatia A, Bloch O, Brem S, Butowski N, Cannon DM, Chao
S, et al: NCCN guidelines® insights: Central nervous
system cancers, version 2.2022. J Natl Compr Canc Netw. 21:12–20.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schaff L, Nayak L and Grommes C: Bruton's
tyrosine kinase (BTK) inhibitors for the treatment of primary
central nervous system lymphoma (PCNSL): Current progress and
latest advances. Leuk Lymphoma. 65:882–894. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ferreri AJM, Calimeri T, Cwynarski K,
Dietrich J, Grommes C, Hoang-Xuan K, Hu LS, Illerhaus G, Nayak L,
Ponzoni M and Batchelor TT: Primary central nervous system
lymphoma. Nat Rev Dis Primers. 9:292023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kabeya K, Satoh H, Hori S, Miura Y and
Sawada Y: Threshold size of medical tablets and capsules: Based on
information collected by Japanese medical wholesaler. Patient
Prefer Adherence. 14:1251–1258. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yoshioka H, Okuda T, Nakao T, Fujita M and
Takahashi JC: Experience with nasogastric tube administration of
tirabrutinib in the treatment of an elderly patient with primary
central nervous system lymphoma. Int Cancer Conf J. 10:290–293.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yoshioka H, Okuda T, Nakao T, Fujita M and
Takahashi JC: Experience with tirabrutinib in the treatment of
primary central nervous system lymphoma that is difficult to treat
with standard treatment. Anticancer Res. 42:4173–4178. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Narita Y, Nagane M, Mishima K, Terui Y,
Arakawa Y, Yonezawa H, Asai K, Fukuhara N, Sugiyama K, Shinojima N,
et al: Phase I/II study of tirabrutinib, a second-generation
Bruton's tyrosine kinase inhibitor, in relapsed/refractory primary
central nervous system lymphoma. Neuro Oncol. 23:122–133. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
National Library of Medicine, . Guidance
for Industry: Bioanalytical Method Validation. Center for Drug
Evaluation and Research; Rockville, MD: 2018, Available:.
http://resource.nlm.nih.gov/101734209
|
|
13
|
Okahashi N, Uchihara M and Hoshino E:
Mantle cell lymphoma with central nervous system relapse
successfully treated with nasogastric-tube administration of
ibrutinib. Rinsho Ketsueki. 61:1508–1510. 2020.(In Japanese).
PubMed/NCBI
|
|
14
|
Alsuliman T, Belghoul M and Choufi B:
Ibrutinib treatment through nasogastric tube in a comatose patient
with central nervous system localization of mantle cell lymphoma.
Case Rep Hematol. 2018:57616272018.PubMed/NCBI
|
|
15
|
Molinaro E, Viola D, Viola N, Falcetta P,
Orsolini F, Torregrossa L, Vagli P, Ribechini A, Materazzi G, Vitti
P and Elisei R: Lenvatinib administered via nasogastric tube in
poorly differentiated thyroid cancer. Case Rep Endocrinol.
2019:68312372019.PubMed/NCBI
|
|
16
|
Kawano F, Yonekawa T, Yamaguchi H, Shibata
N, Tashiro K, Ikenoue M, Munakata S, Higuchi K, Tanaka H, Sato Y,
et al: Nasogastric administration of lenvatinib solution in a
mechanically ventilated patient with rapidly growing anaplastic
thyroid cancer. Endocrinol Diabetes Metab Case Rep. 2020:20–0064.
2020.PubMed/NCBI
|
|
17
|
Tani N, Takatsuka S, Kataoka N, Kunimatsu
Y, Tsutsumi R, Sato I, Tanimura M, Nakano T, Tanimura K, Kato D and
Takeda T: Nasogastric administration of osimertinib suspension for
an epidermal growth factor receptor-mutated lung cancer causing an
esophageal stricture: Case report. Ann Palliat Med. 11:1542–1545.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Takeda T, Itano H, Takeuchi M, Nishimi Y,
Saitoh M and Takeda S: Osimertinib administration via nasogastric
tube in an EGFR-T790M-positive patient with leptomeningeal
metastases. Respirol Case Rep. 5:e002412017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Suzumura T, Yonesaka K, Tsukuda H and
Fukuoka M: Successful gefitinib treatment administration via
gastrostomy tube in a patient with non-small cell lung cancer with
dysphagia. BMJ Case Rep. 2014:bcr20132027052014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bejarano Varas MT, Gould S and Charlot M:
Response to alectinib oil-based suspension in anaplastic lymphoma
kinase-positive non-small cell lung cancer in a patient unable to
swallow: A case report. J Oncol Pharm Pract. 25:1722–1725. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kanai O, Kim YH, Nakatani K, Fujita K and
Mio T: Nasogastric tube-administered alectinib achieved long-term
survival in a crizotinib-refractory nonsmall cell lung cancer
patient with a poor performance status. Clin Case Rep. 5:927–930.
2017. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Thomas QD, Pautas M, Guilhaume MN, Fiteni
F, Ge T and Girard N: Enteral administration of alectinib for
ALK-positive non-small cell lung cancer in an elderly patient: A
case report. Medicine (Baltimore). 100:e276112021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bosch-Barrera J, Sais E, Lorencio C, Porta
R, Izquierdo A, Menéndez JA, Brunet J, Sirvent JM and Rosell R:
Successful empirical erlotinib treatment of a mechanically
ventilated patient newly diagnosed with metastatic lung
adenocarcinoma. Lung Cancer. 86:102–104. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sasaki K, Yokota Y, Isojima T, Fujii M,
Hasui K, Chen Y, Saito K, Takahata T, Kindaichi S and Sato A:
Enteral lorlatinib after alectinib as a treatment option in
anaplastic lymphoma kinase-positive non-small cell lung cancer with
triple problems: Carcinomatous meningitis, poor performance status,
and dysphagia-a case report. Respirol Case Rep. 9:e007962021.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang H, Wu Z, Shi G, Zhou J and Xiao Z:
Enteral lorlatinib after immune hyperprogression as a treatment
option for anaplastic lymphoma kinase-positive non-small cell lung
cancer: A case report. Oncol Lett. 26:5262023. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Jang C, Lau SC and Velcheti V: To crush or
not to crush: Administering dabrafenib and trametinib through a
nasogastric tube in a critically ill patient with nonsmall cell
lung cancer-a case report and review of literature of targeted
therapies given through enteral feeding tubes. Clin Lung Cancer.
25:e124–e128. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Muta T, Sawada Y, Moriyama Y, Seike Y,
Tokuyama T, Ueda Y and Fujisaki T: Chronic myeloid leukemia
complicated with cerebellar hemorrhage and acute hydrocephalus
successfully treated with imatinib and intensive supportive care.
Rinsho Ketsueki. 51:1769–1774. 2010.(In Japanese). PubMed/NCBI
|
|
28
|
Munakata W, Ando K, Hatake K, Fukuhara N,
Kinoshita T, Fukuhara S, Shirasugi Y, Yokoyama M, Ichikawa S,
Ohmachi K, et al: Phase I study of tirabrutinib (ONO-4059/GS-4059)
in patients with relapsed or refractory B-cell malignancies in
Japan. Cancer Sci. 110:1686–1694. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hartford AM, Li W, Qureshi D, Talarico R,
Fung SG, Bush SH, Casey G, Isenberg SR, Webber C and Tanuseputro P:
Use of feeding tubes among hospitalized older adults with dementia.
JAMA Netw Open. 8:e24607802025. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Cantarini MV, McFarquhar T, Smith RP,
Bailey C and Marshall AL: Relative bioavailability and safety
profile of gefitinib administered as a tablet or as a dispersion
preparation via drink or nasogastric tube: Results of a randomized,
open-label, three-period crossover study in healthy volunteers.
Clin Ther. 26:1630–1636. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu SN, Agarwal P, Heinig K, Datye A,
Sturm-Pellanda C, Crugnola A, Arca M and Miles D: Relative
bioavailability and food effect study of an oral suspension of
alectinib in healthy volunteers using venipuncture and capillary
microsampling. Clin Transl Sci. 16:1085–1096. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chiu JW, Chan K, Chen EX, Siu LL and Abdul
Razak AR: Pharmacokinetic assessment of dacomitinib (pan-HER
tyrosine kinase inhibitor) in patients with locally advanced head
and neck squamous cell carcinoma (LA SCCHN) following
administration through a gastrostomy feeding tube (GT). Invest New
Drugs. 33:895–900. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Brooks HD, Glisson BS, Bekele BN, Johnson
FM, Ginsberg LE, El-Naggar A, Culotta KS, Takebe N, Wright J, Tran
HT and Papadimitrakopoulou VA: Phase 2 study of dasatinib in the
treatment of head and neck squamous cell carcinoma. Cancer.
117:2112–2119. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Heath EI, Forman K, Malburg L, Gainer S,
Suttle AB, Adams L, Ball H and LoRusso P: A phase I pharmacokinetic
and safety evaluation of oral pazopanib dosing administered as
crushed tablet or oral suspension in patients with advanced solid
tumors. Invest New Drugs. 30:1566–1574. 2012. View Article : Google Scholar : PubMed/NCBI
|