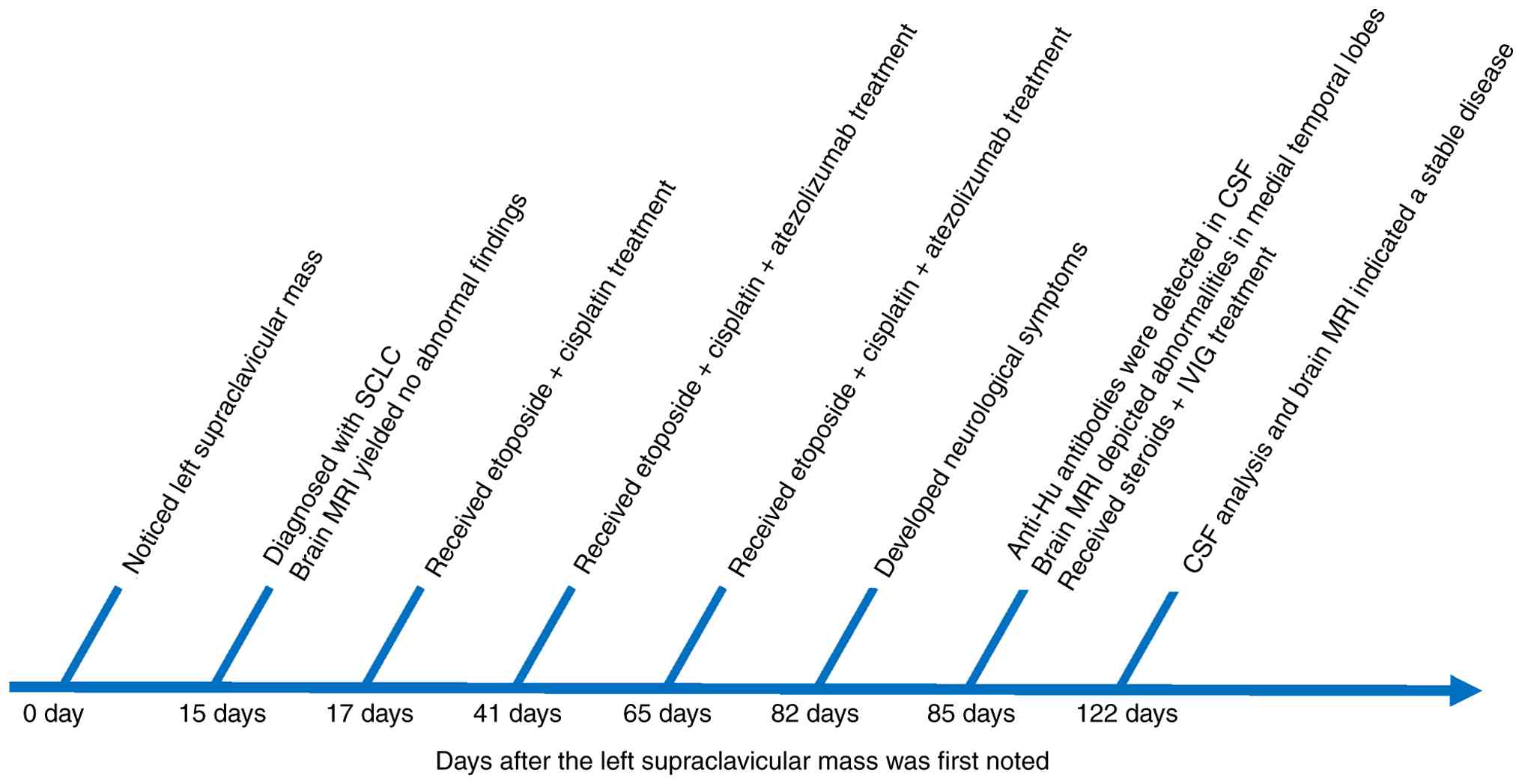
Introduction
Limbic encephalitis refers to an autoimmune process
localized to the limbic system. Paraneoplastic syndrome is the most
common etiology of limbic encephalitis, with small cell lung cancer
(SCLC) being the most frequently associated tumor. Cases have also
been reported in association with testicular cancer, breast cancer,
thymoma, Hodgkin lymphoma, and colon cancer. The clinical symptoms
of limbic encephalitis often precede the diagnosis of the
underlying malignancy by several months to years (1). However, with the emergence and
frequent use of immune checkpoint inhibitors (ICIs) in the
treatment of cancer, reports of immunotherapy-induced limbic
encephalitis began to emerge in the literature (2). In 2016, Williams et al
(3) first reported a case of limbic
encephalitis induced by combined nivolumab and ipilimumab therapy.
As of January 2022, more than eight such cases have been reported
in the literature (4). Based on the
IMpower133 study, atezolizumab in combination with chemotherapy
(carboplatin plus etoposide) was approved by the United States Food
and Drug Administration as a first-line treatment for
extensive-stage SCLC (5). The
present study describes a case of anti-Hu limbic encephalitis
induced by atezolizumab in a patient with SCLC (Fig. 1).
Case report
A 63-year-old Chinese woman presented to an external
hospital with a self-detected left supraclavicular mass in May
2020. The patient had no history of smoking, autoimmune disease or
familial cancer, and denied any history of psychiatric or
neurological disorders. Neurological examination revealed no
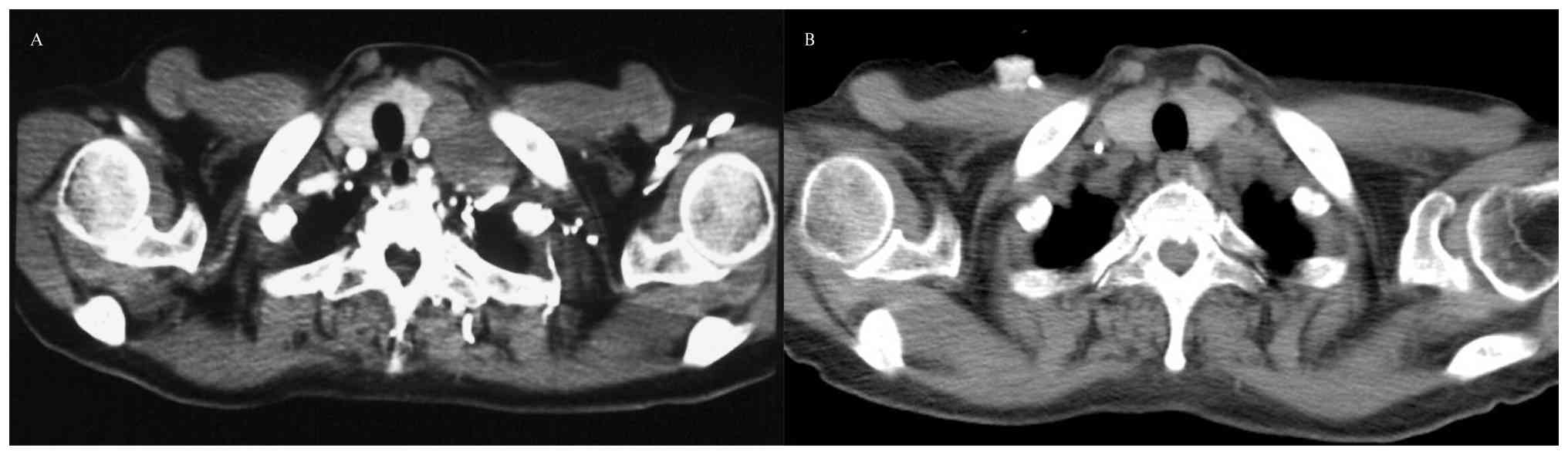
abnormalities. Computed tomography revealed a 6.9×5.0-cm mass at
the left hilum, with multiple enlarged lymph nodes in the
mediastinum and supraclavicular regions (Fig. 2A).
In June 2020, the patient was diagnosed with
extensive-stage SCLC based on the results of biopsy of the left
supraclavicular lymph node (hematoxylin and eosin staining and
immunohistochemical staining performed using standard procedures;
obtained from the pathology report; images are not available).
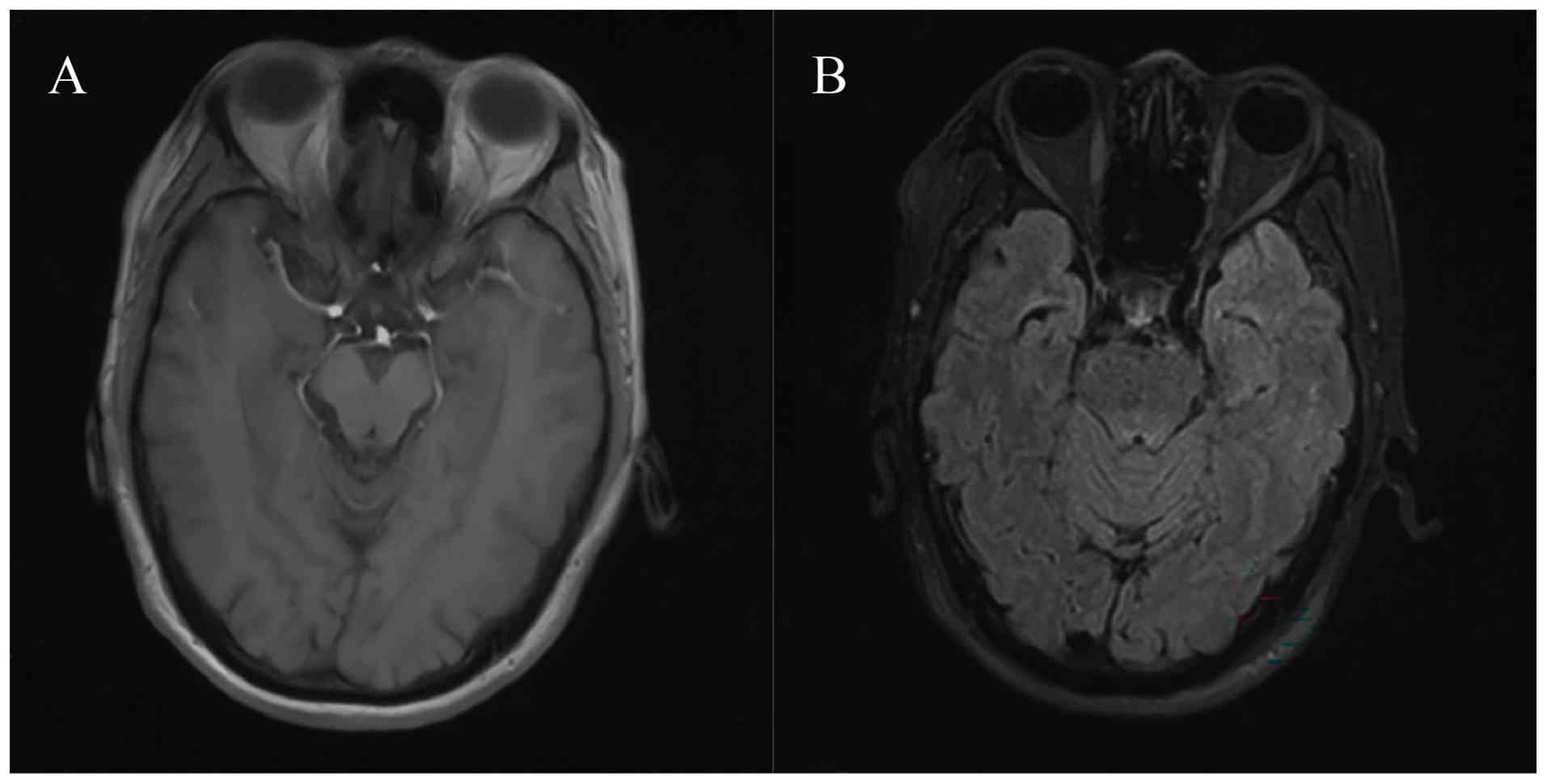
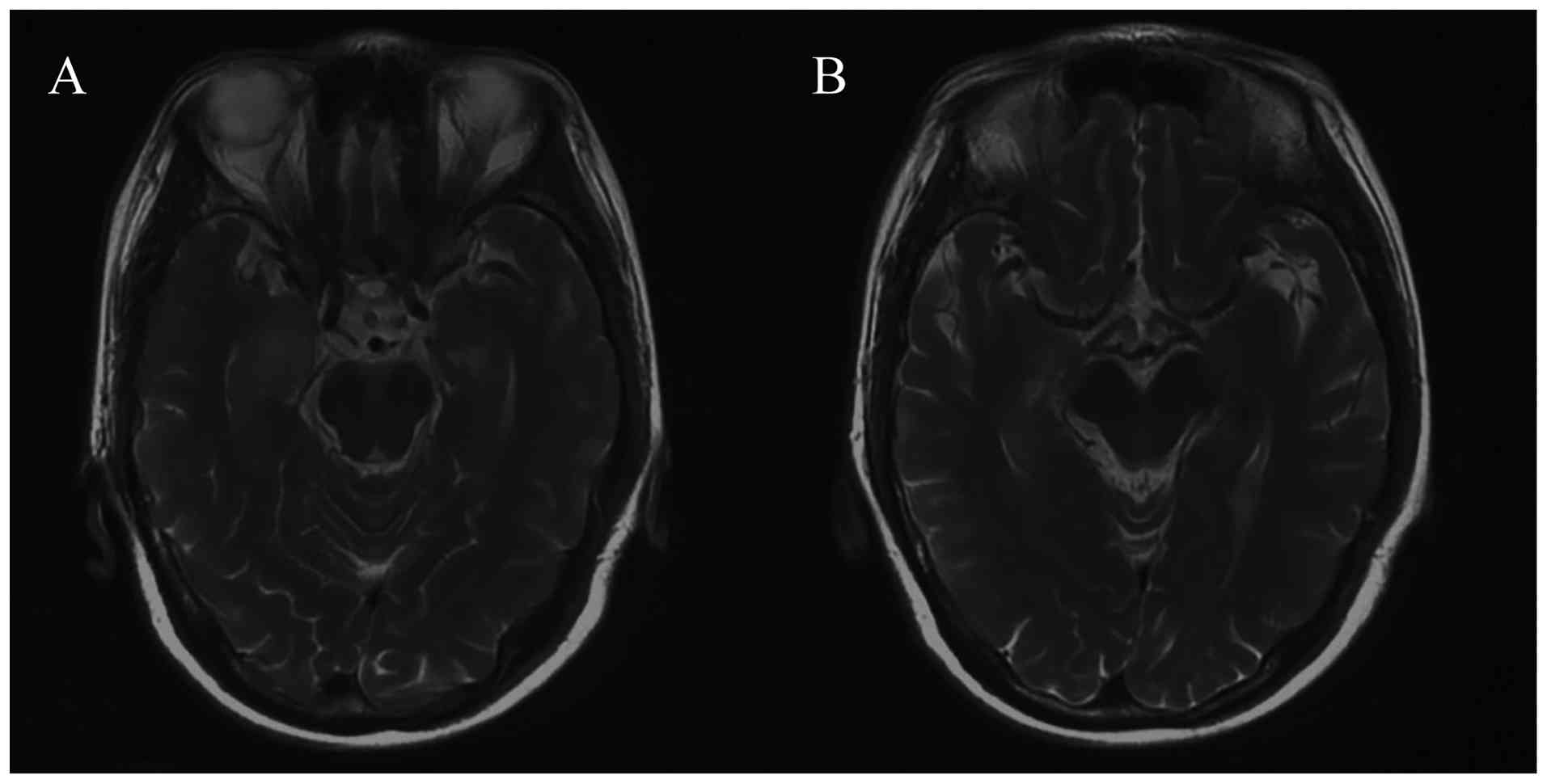
Magnetic resonance imaging (MRI) before medical treatment showed no
abnormal findings in the brain (Fig.
3). Subsequently, 2 days later, the patient was administered
one 21-day cycle of chemotherapy with a combination of etoposide
[100 mg/m2 intravenously (iv), days 1–3] and cisplatin
(area under the curve 5 mg per ml/min iv, day 1). After three
cycles of treatment, computed tomography depicted partial
regression of the primary tumor and metastatic lymph nodes
(Fig. 2B). In August 2020 (2 weeks
after the second dose of atezolizumab), the patient developed
self-reported confusion and short-term memory loss. Further
physical examination revealed impairments in temporal and spatial
orientation as well as poor calculation ability.
Subsequently, she presented to Peking Union Medical
College Hospital and underwent evaluations. The results of
cerebrospinal fluid (CSF) analysis 3 days after the development of
these neurological problems included lymphocytic pleocytosis [white
blood cells, 16/µl (normal range, 0–8/µl); 90% lymphocytes (normal
range, 40–80%); 5% monocytes (normal range, 14–45%); and 5%
neutrophils (normal range, 0–6%)]; an elevated protein level [95
mg/dl (normal range, 15–45 mg/dl)]; a normal glucose level and IgG
index; and no oligoclonal bands or tumor cells were observed.
Polymerase chain reaction testing of CSF did not detect herpes
simplex virus, varicella-zoster virus, cytomegalovirus or human
herpesvirus-6 DNA. Anti-Hu antibodies were detected in the serum
and CSF using immunoblotting (Hu/Yo/Ri/Ma2/Ta/CV2/amphiphysin
antibodies; cat. no. DL 1111-1601-2G; EUROIMMUN; used in accordance
with the kit manufacturer's instructions). Neuronal cell surface
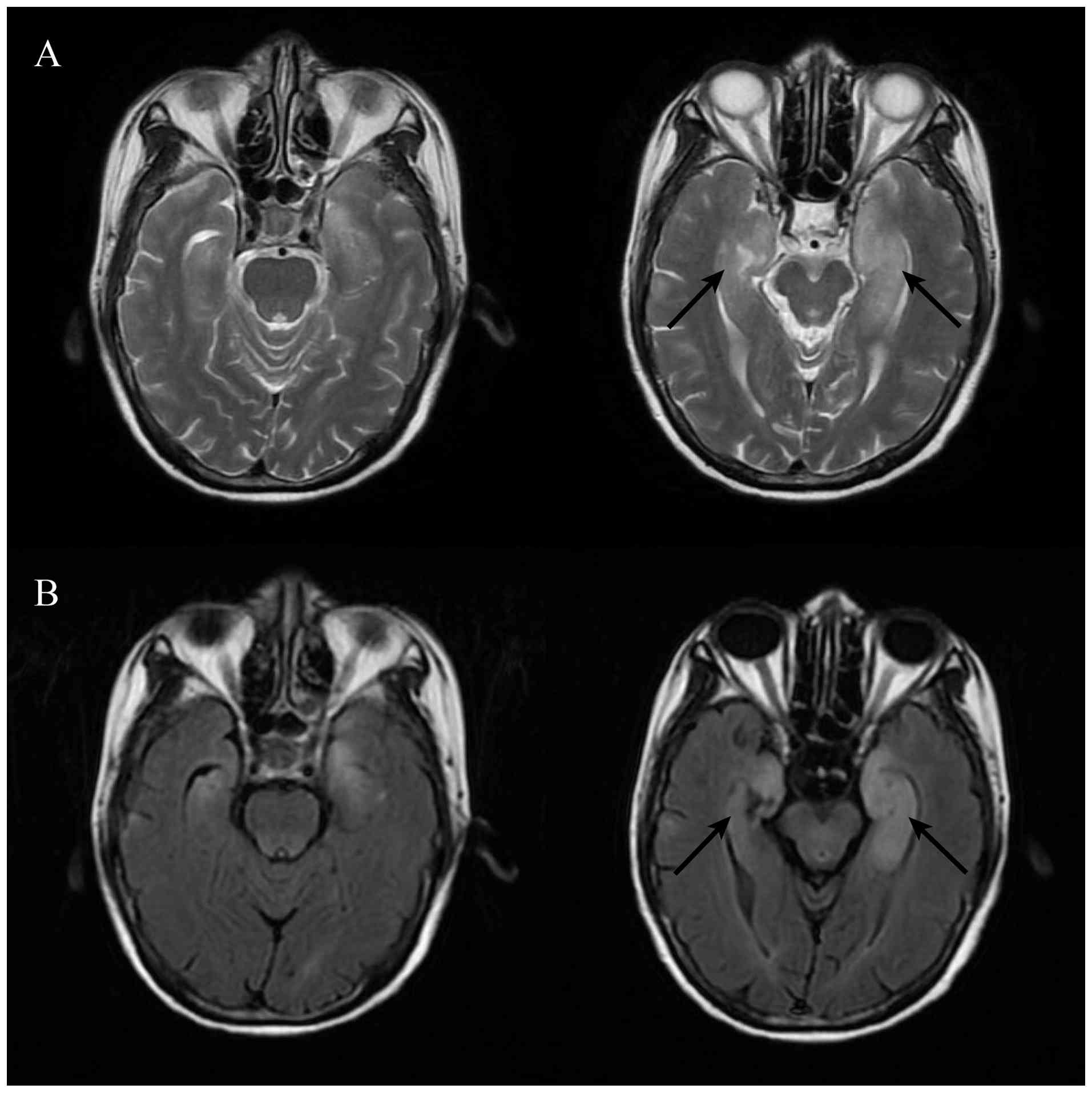
antibodies were all negative. Brain MRI depicted swelling of the
bilateral medial temporal lobes, with patchy prolonged T2 signal
and high fluid-attenuated inversion recovery (FLAIR) signals in
both hippocampi (Fig. 4).
A diagnosis of Hu-associated limbic encephalitis
induced by ICI was considered, and immunotherapy was therefore
withheld. High-dose steroids (methylprednisolone 1 g per day for 2
days, then 2 mg/kg per day for 5 days, then 1 mg/kg per day for 7
days, then tapered by 0.125 mg/kg per day every two weeks until
discontinuation) and intravenous immunoglobulin (400 mg/kg per day
for 5 days) were introduced. Subsequently, 1 month later, the
patient's neurological symptoms remained stable. In September 2020,
the white blood cell count in CSF was normal (2/µl), anti-Hu
antibodies continued to be detectable in the CSF, and MRI of the
brain indicated no significant change compared with the previous
MRI (Fig. 5). The patient died of
tumor progression in March 2021. During this period, her
neurological symptoms remained stable, and no additional magnetic
resonance imaging or cerebrospinal fluid examinations were
performed.
Discussion
Limbic encephalitis is a rapidly progressing
inflammatory disorder of the nervous system that primarily affects
the medial temporal lobes. The main clinical manifestations include
short-term memory impairment, behavioral changes, confusion and
seizures (6). According to the
diagnostic criteria proposed by Graus et al (6), the diagnosis of limbic encephalitis
requires the following: i) Subacute onset of symptoms suggestive of
limbic system involvement; ii) bilateral brain abnormalities on T2
or FLAIR sequence highly restricted to the medial temporal lobes;
iii) CSF pleocytosis or electroencephalography showing epileptiform
discharges involving the temporal lobes; and iv) reasonable
exclusion of alternative causes. Given the clinical presentation,
limbic encephalitis should be differentiated from infectious and
metabolic encephalopathies. However, metabolic encephalopathy
rarely involves the limbic system on MRI. In most cases of
infectious encephalitis, CSF shows a marked elevation in leukocyte
count, whereas in limbic encephalitis the increase is usually mild,
typically below 100/µl. Pathogen detection can provide additional
diagnostic evidence. Herpes simplex virus encephalitis represents
an exception: CSF leukocytes may show mild elevation, and
polymerase chain reaction results may be negative during the early
stage of the disease. Nevertheless, herpes simplex virus
encephalitis usually presents with unilateral temporal lobe
involvement rather than bilateral lesions, and the abnormalities
are often not confined to the limbic system; hemorrhagic changes
may also be observed. Stroke, epilepsy and glioma can also produce
MRI abnormalities localized to the temporal lobes; however, their
clinical features and disease courses differ distinctly from those
of limbic encephalitis (7).
Approximately 60% of patients with limbic
encephalitis have detectable specific antineuronal antibodies in
the CSF (1). Based on their
targets' locations, these antibodies can be categorized into those
against extracellular cell-surface or synaptic proteins (for
example, leucine-rich glioma-inactivated 1,
anti-contactin-associated protein-like 2,
α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid,
N-methyl-D-aspartate receptor and γ-aminobutyric acid type B
receptor) and those against intracellular proteins [for example,
Hu, membrane anchor 2 (Ma2), glutamic acid decarboxylase and
amphiphysin] (6). Previous studies
(8–10) have indicated that in cases
associated with cell-surface antibodies, neuronal injury is
primarily mediated by antibodies and complement activation. By
contrast, limbic encephalitis associated with intracellular
antibodies involves neuronal injury predominantly mediated by
CD8+ T cells, and the presence of these intracellular
antibodies likely reflects neuronal destruction rather than its
cause. Consequently, the symptoms in patients with intracellular
antibody-associated limbic encephalitis are often irreversible
(11). Paraneoplastic limbic
encephalitis is the most common subtype of limbic encephalitis. In
most patients, neurological symptoms precede the diagnosis of the
underlying tumor, and specific intracellular antibodies are
considered corresponding biomarkers of specific tumor types
(1). SCLC is the most common tumor
detected in paraneoplastic limbic encephalitis patients with immune
responses against Hu antigen (12).
The mechanisms that result in immune-related adverse
events are still being elucidated. Increased T-cell activity
against antigens that are present in healthy tissue and tumor
tissue is considered to be one, which is consistent with the
pathogenesis of paraneoplastic limbic encephalitis. In the current
case, the patient fulfilled the diagnostic criteria for limbic
encephalitis, and the presence of anti-Hu antibodies indicated that
the encephalitis was associated with a paraneoplastic syndrome
secondary to SCLC. The timing of the onset of neurological symptoms
strongly suggested the involvement of ICIs. In the present case, as
no other limbic encephalitis-related antibodies were detected in
CSF, ICIs may have accelerated autoimmune reactions targeting
paraneoplastic autoantigens, thereby exacerbating a pre-existing
subclinical paraneoplastic neurological syndrome (PNS). Vogrig
et al (13) reported two
cases of anti-Ma2 limbic encephalitis that occurred after ICI
treatment, in which anti-Ma2 antibodies were retrospectively
detected in the serum (blood) samples collected prior to treatment.
Therefore, detection of anti-Hu antibodies in CSF prior to
immunotherapy would further substantiate the above hypothesis.
However, in the current case, CSF samples were not collected before
the initiation of ICI treatment.
Clinicians should be alert to the risk of
ICI-induced PNSs in patients with tumors, such as SCLC, that are
traditionally associated with PNSs (14). Given the time-consuming nature of
antibody testing and low incidence of limbic encephalitis, such
screening is difficult to implement routinely before ICI treatment.
Future research is required to examine whether the use of ICIs in
patients with SCLC increases the risk of PNSs.
Acknowledgements
Not applicable.
Funding
The present case report was funded by the National High Level
Hospital Clinical Research Fund (grant no. 2022-PUMCH-A-131).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
ZL was involved in the conceptualization of the
study and performed data curation and performed writing - original
draft and visualization. HW was involved in the conceptualization
of the study, performed the writing - review & editing and
provided supervision. ZL and HW confirm the authenticity of all the
raw data. Both authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of any potentially identifiable images
or data included in this article.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
CSF
|
cerebrospinal fluid
|
|
ICI
|
immune checkpoint inhibitor
|
|
MRI
|
magnetic resonance imaging
|
|
SCLC
|
small cell lung cancer
|
References
|
1
|
Gultekin SH, Rosenfeld MR, Voltz R, Eichen
J, Posner JB and Dalmau J: Paraneoplastic limbic encephalitis:
Neurological symptoms, immunological findings and tumour
association in 50 patients. Brain. 123:1481–1494. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fan S, Ren H, Zhao L, Yin J, Feng G, Wang
J and Guan H: Neurological immune-related adverse events associated
with immune checkpoint inhibitors: A review of the literature. Asia
Pac J Clin Oncol. 16:291–298. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Williams TJ, Benavides DR, Patrice KA,
Dalmau JO, de Ávila AL, Le DT, Lipson EJ, Probasco JC and Mowry EM:
Association of autoimmune encephalitis with combined immune
checkpoint inhibitor treatment for metastatic cancer. JAMA Neurol.
73:928–933. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nakashima K, Demura Y, Kurokawa K, Takeda
T, Jikuya N, Oi M, Tada T, Akai M and Ishizuka T: Immune checkpoint
inhibitor-induced limbic encephalitis during treatment with
atezolizumab in a patient with small-cell lung cancer: A case
report and review of the literature. Case Reports Immunol.
2022:92909222022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Horn L, Mansfield AS, Szczęsna A, Havel L,
Krzakowski M, Hochmair MJ, Huemer F, Losonczy G, Johnson ML, Nishio
M, et al: First-line atezolizumab plus chemotherapy in
extensive-stage small-cell lung cancer. N Engl J Med.
397:2220–2229. 2018. View Article : Google Scholar
|
|
6
|
Graus F, Titulaer MJ, Balu R, Benseler S,
Bien CG, Cellucci T, Cortese I, Dale RC, Gelfand JM, Geschwind M,
et al: A clinical approach to diagnosis of autoimmune encephalitis.
Lancet Neurol. 15:391–404. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Budhram A, Leung A, Nicolle MW and Burneo
JG: Diagnosing autoimmune limbic encephalitis. CMAJ. 191:E529–E534.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bien CG, Vincent A, Barnett MH, Becker AJ,
Blümcke I, Graus F, Jellinger KA, Reuss DE, Ribalta T, Schlegel J,
et al: Immunopathology of autoantibody-associated encephalitides:
Clues for pathogenesis. Brain. 135:1622–1638. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dalmau J, Geis C and Graus F:
Autoantibodies to synaptic receptors and neuronal cell surface
proteins in autoimmune diseases of the central nervous system.
Physiol Rev. 97:839–887. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sechi E, Markovic SN, McKeon A, Dubey D,
Liewluck T, Lennon VA, Lopez-Chiriboga AS, Klein CJ, Mauermann M,
Pittock SJ, et al: Neurologic autoimmunity and immune checkpoint
inhibitors: Autoantibody profiles and outcomes. Neurology.
95:e2442–e2452. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kyritsis AP, Markoula S, Alexiou G,
Asimakopoulos A, Jabbour P, Fotopoulos A and Sioka C: Diagnosis and
treatment of limbic encephalitis in the cancer patient. Future
Oncol. 16:1647–1655. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Graus F, Keime-Guibert F, Reñe R, Benyahia
B, Ribalta T, Ascaso C, Escaramis G and Delattre JY:
Anti-Hu-associated paraneoplastic encephalomyelitis: Analysis of
200 patients. Brain. 124:1138–1148. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Vogrig A, Fouret M, Joubert B, Picard G,
Rogemond V, Pinto AL, Muñiz-Castrillo S, Roger M, Raimbourg J,
Dayen C, et al: Increased frequency of anti-Ma2 encephalitis
associated with immune checkpoint inhibitors. Neurol Neuroimmunol
Neuroinflamm. 6:e6042019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gozzard P, Woodhall M, Chapman C, Nibber
A, Waters P, Vincent A, Lang B and Maddison P: Paraneoplastic
neurologic disorders in small cell lung carcinoma: A prospective
study. Neurology. 85:235–239. 2015. View Article : Google Scholar : PubMed/NCBI
|