Introduction
External auditory canal cancer (EACC) is a rare and
aggressive malignancy that accounts for a small fraction (0.1–0.2%)
of head and neck cancers (1).
According to national cancer registry data, its annual incidence is
estimated to be around 0.1 per 100,000 population in Japan. The
management of EACC is characterized by anatomical complexity and a
lack of standardized treatment guidelines due to its rarity. The
proximity of the external auditory canal to critical structures,
such as the facial nerve, temporomandibular joint, and the brain,
complicates both surgical resection and high-dose radiation
delivery. These factors, combined with challenging diagnostic and
metastatic patterns, result in a survival rate that is
significantly lower than that of other head and neck cancers, such
as pharyngeal cancer. Several retrospective studies have indicated
that surgery or radiotherapy (RT) alone may be effective in
managing early-stage disease (2,3),
whereas a multimodal approach, including surgery, chemotherapy, and
RT, is often required for advanced-stage disease (4,5).
Traditionally, a combination of subtotal or total temporal bone
resection and postoperative RT is considered as the standard of
care for resectable tumors (6).
However, this approach is highly invasive and may considerably
affect the patient's quality of life. Definitive RT is a
potentially less invasive alternative to surgery, particularly for
locally advanced or unresectable cases, with the advantage of organ
function preservation (7). However,
identifying the patient's disease stage remains challenging; the
modified Pittsburgh system is the most commonly used (8). Disease stage correlates with survival
outcomes, although survival rates vary based on tumor stage. This
study addresses the urgent need for evidence-based strategies to
optimize RT for EACC.
Materials and methods
Study design and ethics
This retrospective study included patients with EACC
who underwent RT at Nagoya City University Hospital and five
regional hospitals (Ichinomiya Municipal Hospital, Nagoya Medical
Center, Japanese Red Cross Aichi Medical Center Nagoya Daini
Hospital, Okazaki City Hospital, and Kariya Toyota General
Hospital) between January 2000 and August 2023. This study was
approved by the institutional review board of Nagoya City
University Hospital (approval no. 60–23-0066). This study was
approved by the institutional review boards of five regional
hospitals: Ichinomiya Municipal Hospital (approval no. 1388),
Nagoya Medical Center (approval no. 2023-436), Japanese Red Cross
Aichi Medical Center Nagoya Daini Hospital (approval no. 5042),
Okazaki City Hospital (approval no. 2023-53), and Kariya Toyota
General Hospital (approval no. 961). The need for written informed
consent was waived owing to the retrospective study design, and its
content was disclosed in an opt-out form available on the website.
This study adhered to the guidelines of the Helsinki Declaration.
The eligibility criteria were: i) Pathologically diagnosed EACC,
ii) no history of head and neck region irradiation, and iii) no
distant organ metastasis. Patients who underwent palliative RT were
excluded.
Patient and treatment
characteristics
This study included 42 patients with EACC, including
23 men and 19 women with a median age of 65 years (range: 41–89
years) at the time of treatment. The Pittsburgh staging system was
used to assess the T-category. Lymph node (N category) and distant
(M category) metastases were classified based on the 8th edition of
the Union for International Cancer Control tumor, node, and
metastasis staging system for head and neck cancers. Patient
characteristics are summarized in Table
I. A tumor board comprising otorhinolaryngologists, diagnostic
radiologists, and radiation oncologists determined the treatment
approach.
 | Table I.Patient and treatment
characteristics. |
Table I.
Patient and treatment
characteristics.
| Characteristic | Value |
|---|
| Median age, years
(range) | 65 (41–89) |
| Sex, n (%) |
|
|
Male | 23 (55%) |
|
Female | 19 (45%) |
| Performance status,
n (%) |
|
| 0 | 13 (31%) |
| 1 | 21 (50%) |
| 2 | 7 (17%) |
| 3 | 1 (2%) |
| Histological
subtype, n (%) |
|
|
SCC | 39 (93%) |
|
ACC | 2 (5%) |
|
BCC | 1 (2%) |
| T
classification |
|
| T1 | 4 (10%) |
| T2 | 7 (17%) |
| T3 | 18 (42%) |
| T4 | 13 (31%) |
| N
classification |
|
| N0 | 39 (93%) |
| N1 | 3 (7%) |
| Stage, n |
|
| I | 4 (9%) |
| II | 7 (17%) |
|
III | 16 (38%) |
| IV | 15 (36%) |
| Treatment modality,
n (%) |
|
| Surgery
+ PORT | 10 (24%) |
|
Definitive RT | 32 (76%) |
| Surgery
technique |
|
| Lateral
temporal bone resection | 8 (80%) |
|
Subtotal temporal
resection | 2 (20%) |
| RT technique, n
(%) |
|
| Surgery
+ PORT, 3D-CRT/IMRT | 6 (60%)/4
(40%) |
|
Definitive RT,
3D-CRT/IMRT | 9 (28%)/23
(72%) |
| Median
RT dose, Gy (range) | 66 (47.5–70) |
| Chemotherapy, n
(%) |
|
|
Concurrent IA CDDP | 10 (23%) |
| Three
courses, 100/200 mg/m2 | 2/4 |
| Four
courses, 100/200 mg/m2 | 3/1 |
|
Concurrent IV CDDP | 7 (17%) |
| Three
triweekly courses, 80/100 mg/m2 | 3/4 |
| TPF | 7 (17%) |
|
Induction/concurrent | 5/2 |
|
Concurrent Cmab | 2 (5%) |
|
Concurrent S-1 | 2 (5%) |
|
None | 14 (33%) |
Surgery
Overall, 10 patients underwent surgery, including
lateral (n=8) and subtotal temporal bone resection (n=2). Among
these, five patients had clear margins, three had close margins,
and two had positive margins. In general, EACC surgery is more
invasive than RT. The decision to perform surgery was based on the
patient's overall condition and preferences, with surgical
candidates being required to have an ECOG performance status ≤2, a
maximum tumor diameter ≤3.5 cm, and no distant metastasis. Lateral
or subtotal temporal bone resection was decided based on curability
and the degree of invasiveness.
Chemotherapy
Of the included patients, 28 underwent systemic
chemotherapy. The decision to administer chemotherapy and the
regimen were determined based on the patient's condition and
concurrent treatment strategy. Concurrent chemotherapy was
considered especially for patients in the T3 or advanced stages and
23 patients received concurrent chemotherapy. The decision was made
based on the patient's general condition, cardiac and renal
function, and preferences. The main regimen was cisplatin-based
chemotherapy. Detailed information on the administered regimens is
provided in Table I. Intra-arterial
chemotherapy was administered at facilities where it was feasible;
otherwise, intravenous or oral chemotherapy was administered. All
patients receiving the TPF regimen (docetaxel, cisplatin, and
5-fluorouracil) were in the definitive RT group. Among these, five
patients received TPF as induction chemotherapy, while two received
it as concurrent chemoradiotherapy.
Radiotherapy
Overall, 32 and 10 patients underwent definitive RT
and surgery followed by RT, respectively. All included patients
underwent RT planned using a computed tomography (CT) based
radiation treatment planning system that involved three-dimensional
conformal radiotherapy (3D-CRT) or intensity-modulated radiotherapy
(IMRT). In the definitive RT group, the primary tumor was
visualized using CT, magnetic resonance imaging (MRI), and/or
18F-fluorodeoxyglucose positron emission tomography CT
(PET-CT) and the gross tumor volume (GTV) was determined. The EACC
tumor encompassing the GTV and surrounding tissues was defined as
the clinical target volume (CTV). In the postoperative RT group,
the tumor bed was visualized on preoperative CT, MRI, and PET-CT
images and the tumor-infiltrated area was pathologically confirmed
and delineated as the CTV. In both groups, a 5-mm margin was added
to the CTV to define the planning target volume (PTV). In general,
the target volumes for patients without lymph node metastases
excluded the lymph node regions. The CTV in patients with lymph
node metastases included the metastatic lymph node levels and
adjacent lymph nodes. A 5-mm margin was added to the CTV of the
lymph node regions to define the PTV.
Assessment and statistical
analyses
Follow-up, including head and neck CT, was performed
at 2-month intervals after RT for 6 months and every 2–4 months
thereafter. The overall survival (OS), local control (LC), and
progression-free survival (PFS) rates were calculated from the time
of RT initiation using the Kaplan-Meier method. OS was defined as
the time from RT initiation to the last follow-up or all-cause
death. LC was defined as the time from RT initiation to local
relapse. PFS was defined as the time from RT initiation to
recurrence or death and was censored at the last date without
events. The log-rank test was used to compare Kaplan-Meier curves.
Univariate analyses were conducted using the Cox proportional
hazards model to identify factors associated with OS, LC, and PFS.
No multivariate analysis was conducted because of the limited
sample size. The difference in the patient characteristics of the
definitive and postoperative RT groups was investigated using the
Student's t-test or Mann-Whitney U test for continuous variables
and the Fisher's exact test for categorical variables. Toxicity was
assessed using the Common Terminology Criteria for Adverse Events
version 5.0. P<0.05 was considered to indicate a statistically
significant difference. All statistical analyses were conducted
using R version 4.4.0 (The R Foundation for Statistical Computing,
Vienna, Austria), an open-source software.
Results
Outcomes and prognostic factors
The median follow-up period of the 42 patients was
31.5 months (range: 6–120 months). Of the included patients, 14
died of the primary disease and 5 died of other causes. These five
deaths from other causes consisted of one patient in the
postoperative RT group (due to pneumonia) and four patients in the
definitive RT group (due to pneumonia, heart failure, and other
comorbidities). Recurrence was reported in 16 patients, including
14 with local recurrence, 5 with cervical lymph node recurrence,
and 5 with distant organ metastases. Four of the seven patients
(57%) who underwent the TPF regimen achieved local control without
recurrence. The median interval from local recurrence to death was
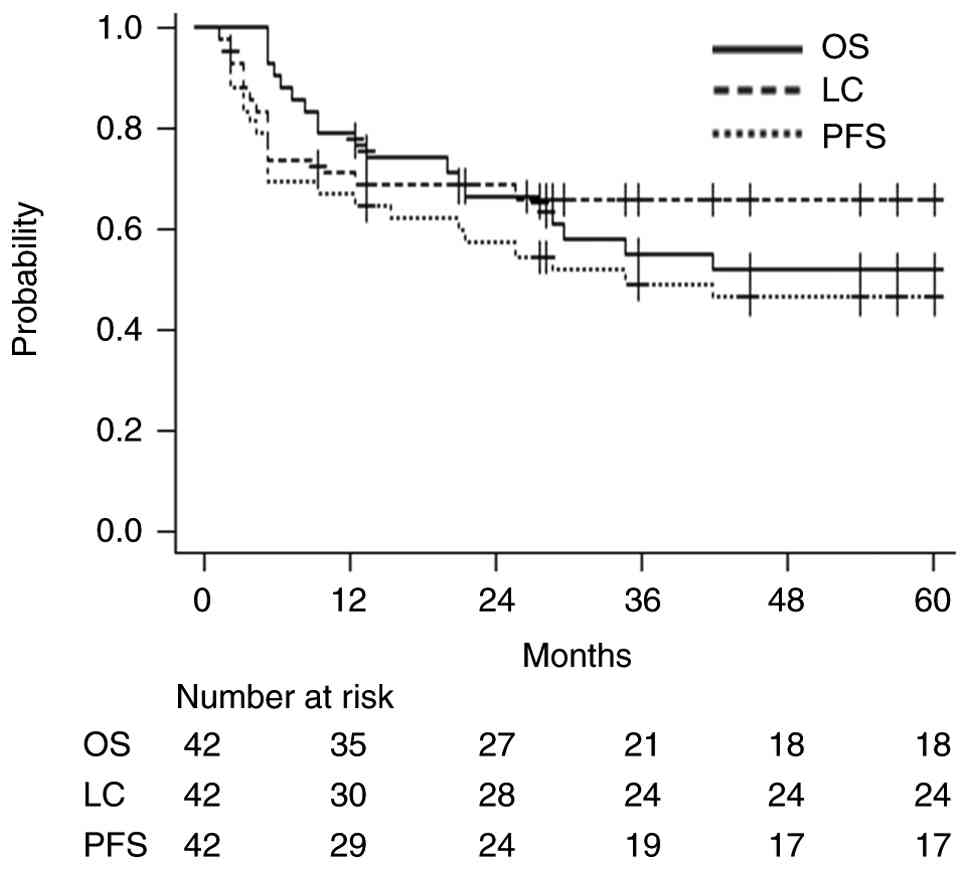
4 months (range: 1–68 months). In all patients, the 3-year OS, PFS,
and LC rates were 54.8% (95% confidence interval [CI]: 41.2–73.0%),
49.0% (95% CI: 37.7–69.1%), and 65.5% (95% CI: 54.1–83.7%),
respectively, and the median survival duration was 42 months.
Fig. 1 presents the OS, LC, and PFS
curves for all 42 patients. Table
II summarizes the prognostic factors on univariate analyses for
OS, LC, and PFS. Univariate analyses revealed that the treatment
method was a significant prognostic factor (surgery followed by RT
vs. definitive RT: OS: hazard ratio [HR]: 9.60, 95% CI: 1.27–72.5,
P=0.028; LC: HR: 12.22, 95% CI: 1.62–156.4, P=0.008; PFS: HR:
11.54, 95% CI: 1.54–86.5, P=0.017).
 | Table II.Univariate analyses of OS, LC and
PFS. |
Table II.
Univariate analyses of OS, LC and
PFS.
|
| OS | LC | PFS |
|---|
|
|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Age, years |
|
|
|
|
|
|
|
|
|
| <60
(n=15) | 1.00 | - | - | 1.00 | - | - | 1.00 | - | - |
| ≥60
(n=27) | 1.88 | 0.68–5.24 | 0.22 | 1.13 | 0.38–3.34 | 0.82 | 1.37 | 0.56–3.38 | 0.48 |
| Performance
status |
|
|
|
|
|
|
|
|
|
| 0 and 1
(n=34) | 1.00 | - | - | 1.00 | - | - | 1.00 | - | - |
| 2 and 3
(n=8) | 1.68 | 0.61–4.69 | 0.32 | 1.89 | 0.59–6.01 | 0.28 | 1.32 | 0.48–3.58 | 0.58 |
| T
classification |
|
|
|
|
|
|
|
|
|
| 1 and 2
(n=11) | 1.00 | - | - | 1.00 | - | - | 1.00 | - | - |
| 3 and 4
(n=31) | 1.04 | 0.39–2.74 | 0.94 | 6.48 | 0.84–49.5 | 0.072 | 1.34 | 0.52–3.43 | 0.54 |
| Treatment
methods |
|
|
|
|
|
|
|
|
|
| Surgery
+ PORT (n=10) | 1.00 | - | - | 1.00 | - | - | 1.00 | - | - |
|
Definitive RT (n=32) | 9.60 | 1.27–72.5 | 0.028 | 12.22 | 1.62–156.4 | 0.008 | 11.54 | 1.54–86.5 | 0.017 |
| RT dose, Gy |
|
|
|
|
|
|
|
|
|
| <60
(n=5) | 1.00 | - | - | 1.00 | - | - | 1.00 | - | - |
| ≥60
(n=37) | 1.51 | 0.35–6.55 | 0.58 | 2.22 | 0.29–17.02 | 0.44 | 1.84 | 0.43–7.90 | 0.41 |
| RT technique |
|
|
|
|
|
|
|
|
|
| 3D-CRT
(n=15) | 1.00 | - | - | 1.00 | - | - | 1.00 | - | - |
| IMRT
(n=27) | 1.20 | 0.47–3.06 | 0.69 | 1.64 | 0.51–5.28 | 0.40 | 1.49 | 0.60–3.67 | 0.38 |
| Chemotherapy |
|
|
|
|
|
|
|
|
|
| Yes
(n=28) | 1.00 | - | - | 1.00 | - | - | 1.00 | - | - |
| No
(n=14) | 1.11 | 0.44–2.84 | 0.81 | 0.72 | 0.22–2.30 | 0.58 | 0.97 | 0.41–2.33 | 0.95 |
Comparison of patient characteristics
and outcomes of the postoperative and definitive RT groups
Overall, 2 patients in T2 stage and 8 in T3/T4 stage
underwent surgery followed by RT. None of them had lymph node
metastases, whereas 5 (50%) patients underwent concurrent systemic
chemotherapy. In contrast, 9 patients in T1/T2 stage and 23 in
T3/T4 were included in the definitive RT group. Of these, 3 had
lymph node metastases and 29 patients did not. Moreover, 23 (72%)
patients underwent concurrent systemic chemotherapy in definitive
RT. The patient and treatment characteristics of the postoperative
and definitive RT groups are summarized in Table III. Compared with the definitive
RT group, the postoperative RT group had a significantly smaller
PTV and received a lower RT dose (median PTV: 71 cm3 vs.
87 cm3, P=0.021; median RT dose: 60 Gy vs. 66 Gy,
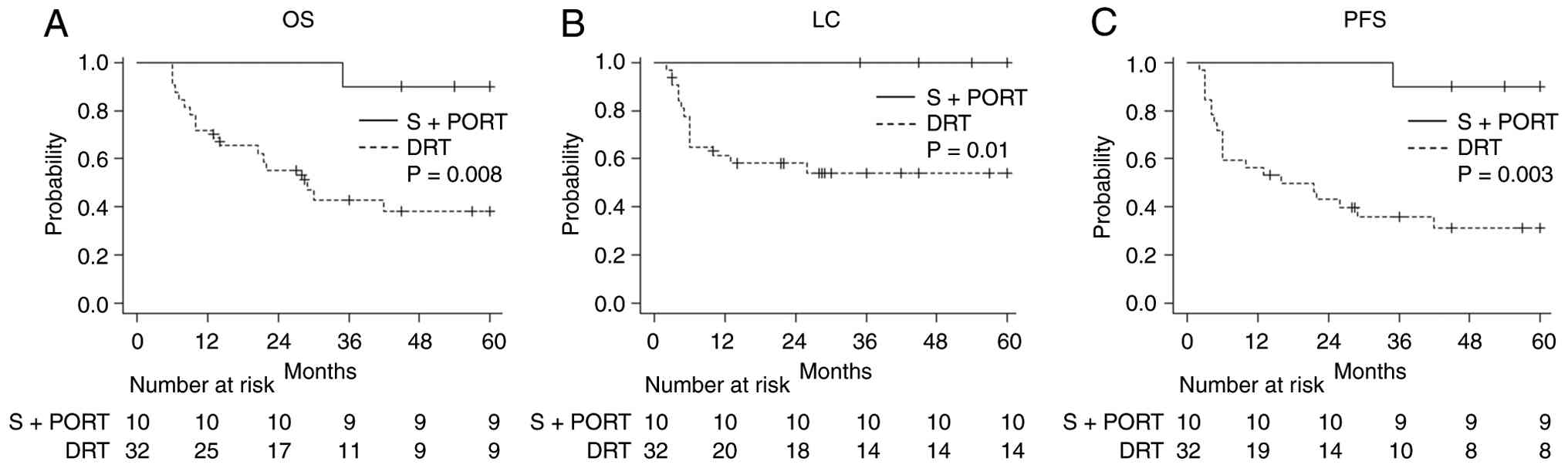
P=0.004). The 3-year OS, LC, and PFS rates in the postoperative and
definitive RT groups were 90% vs. 42.9%, 100% vs. 53.9%, and 90%
vs. 35.9%, respectively. P-values determined using the log-rank
test were 0.008, 0.01, and 0.003, respectively. Fig. 2 presents the OS, LC, and PFS curves
for both groups.
 | Table III.Patient characteristics of the
surgery combined with PORT group and definitive RT group. |
Table III.
Patient characteristics of the
surgery combined with PORT group and definitive RT group.
| Characteristic | Surgery + PORT
(n=10) | Definitive RT
(n=32) | P-value |
|---|
| Median age, years
(range) | 65 (41–77) | 65 (43–89) | 0.43 |
| Sex,
male/female | 6/4 | 17/15 | 1.0 |
| Performance status,
0/1/2/3 | 1/7/2/0 | 12/14/5/1 | 0.32 |
| T classification,
T1/T2/T3/T4 | 0/2/6/2 | 4/5/12/11 | 0.55 |
| N classification,
N0/N1 | 10/0 | 29/3 | 1.0 |
| Median PTV,
cm3 (range) | 71 (41.3–114) | 87 (14.4–350) | 0.021 |
| RT technique,
3D-CRT/IMRT | 6/4 | 9/23 | 0.69 |
| Median RT dose, Gy
(range) | 60 (47.5–70) | 66 (50–70) | 0.004 |
| Chemotherapy,
use/non-use | 5/5 | 23/9 | 0.25 |
Toxicities
After RT, 1 (2.4%) patient developed grade 3 brain
necrosis and 2 (4.7%) developed grade 3 and 4 brain abscesses. All
three T4 patients who developed severe toxicities were treated with
IMRT in the definitive RT group and received 70 Gy with concurrent
systemic chemotherapy. Toxicity occurred at 2, 12, and 17 months
after RT. Of the two patients with brain abscesses, one was
conservatively treated with antibiotics and the other underwent
brain surgery. The case of the patient who underwent surgery for
brain abscess has been reported in detail (9). Other toxicities ≥grade 3 did not
occur.
Discussion
The results of this study suggest that surgery
followed by RT is associated with more favorable treatment outcomes
than definitive RT in patients with EACC. Table IV summarizes previous studies on RT
for EACC (10–20). Regarding the different outcomes for
the patients in the postoperative and definitive RT groups, the
treatment modality used may affect prognosis. Several factors may
contribute to this difference in outcomes. The outcomes of surgery
combined with postoperative RT vs. definitive RT are controversial,
with one report suggesting that surgery followed by RT is better,
(17) whereas another reported no
difference between the groups (21). However, it is crucial to acknowledge
the potential selection bias that is inherent in this retrospective
study design. In our study, although the definitive and
postoperative RT groups primarily included patients in T3/4 stages,
those selected for surgery likely possessed more favorable baseline
characteristics (e.g., more localized invasion or better general
condition). Therefore, the favorable outcomes observed in the group
that underwent surgery followed by RT must be interpreted with
caution as they likely reflect patient selection rather than
treatment modality efficacy itself.
 | Table IV.Studies of radiotherapy for external
auditory canal cancer. |
Table IV.
Studies of radiotherapy for external
auditory canal cancer.
| First author,
year | n | T stage | Treatment
modality | OS | LC or PFS | Toxicity (≥Grade
3) | (Refs.) |
|---|
| Fujiwara, 2015 | 13 | T3: 1; T4: 12 | DRT: 13 | 58.7% (2-year) | 53.8% (PFS,
2-year) | 15% | (10) |
| Koto, 2016 | 13 | T3: 4; T4: 9 | DRT: 13 (carbon
ion) | 40% (3-year) | 54% (LC,
3-year) | 15% | (11) |
| Choi, 2017 | 32 | T1-2: 12; T3-4:
20 | S + RT/DRT:
21/11 | 57% (5-year) | 52% (PFS,
5-year) | 0 | (12) |
| Matoba, 2018 | 25 | T1-2: 9; T3-4:
16 | S/DRT/C:
16/7/2 | 75.8% (2-year) | 58% (PFS,
2-year) | NA | (13) |
| Hayashi, 2019 | 31 | T1-2: 6; T3-4:
25 | DRT: 31 (carbon
ion) | 58.7% (3-year) | 55% (LC,
3-year) | 6.5% | (14) |
| Nagano, 2019 | 21 | T3: 8; T4: 13 | S+RT/DRT: 13/8 | 62% (2-year) | 71% (LC,
2-year) | 4.8% | (15) |
| Shiga, 2021 | 74 | T3: 8; T4: 66 | DRT: 74; TPF: 50;
CDDP: 24 | DRT: 54.6%
(5-year); TPF: 64.4% (5-year); CDDP: 36.7% (5-year) | DRT: NA; TPF: NA;
CDDP: NA | NA | (16) |
| Laskar, 2022 | 89 | T1-2: 16; T3-4:
73 | S + RT/DRT:
65/24 | 63.5% (5-year) | 66.2% (LC,
5-year) | NA | (17) |
| Jang, 2023 | 51 | T1-2: 19; T3-4:
32 | S or S + RT/DRT:
41/10 | 64% (5-year) | 71% (PFS,
5-year) | NA | (18) |
| Ooka, 2025 | 73 | T1-2: 37; T3-4:
36 | S + RT/DRT:
61/12 | 86.6% (3-year) | 81.9% (PFS,
3-year) | NA | (19) |
| Zhang, 2025 | 173 | T1-3: 46; T4:
127 | S + RT:173 | 80.6% (5-year) | 72.7% (PFS,
5-year) | 1.2% | (20) |
| Present study | 42 | T1-2: 11; T3-4:
31 | S + RT/DRT:
10/32 | 54.8% (3-year) | 65.5% (LC,
3-year) | 7.1% |
|
A study on completely resected early-stage EACC
reported that administering postoperative RT does not provide an
additional benefit, with no significant difference in the 5-year
disease-free survival of patients who underwent surgery followed by
RT and those who did not undergo RT (3). The superiority of surgery followed by
RT may be related to the reduction in tumor volume through surgical
resection and effective irradiation of microscopic disease, as well
as precise RT planning based on pathological information such as
margin status and invasion depth. Matoba et al reported that
surgery alone or surgery followed by RT tended to have better
treatment outcomes than definitive RT for patients with
advanced-stage disease (13). A
study including a larger number of patients reported that surgery
followed by RT is associated with improved outcomes compared with
definitive RT (17). A favorable
5-year OS rate of 80.6% was reported in the group that underwent
surgery combined with preoperative or postoperative RT (20). Positive surgical margins are
considered a risk factor for local recurrence and a poor prognostic
factor for patients who underwent surgery followed by RT (22). Although 50% of patients in the
postoperative RT group in our study, including advanced-stage
cases, had positive or close margins, their outcomes were good.
This study reports that no local recurrence was observed in the
postoperative RT group, even in patients with positive/close
margins (50% of the cohort), suggesting that postoperative RT
effectively eliminates microscopic residual disease. These findings
support reducing the PTV margin in postoperative RT to minimize
toxicity while maintaining efficacy.
In the definitive RT group, the median time from
local recurrence to death was short at 4 months, emphasizing the
importance of LC. In advanced cases, hypoxic tumor regions may
reduce radiosensitivity and limit the efficacy of definitive RT to
cover the entire tumor (23).
Carbon ion RT can be administered to improve LC for EACC; however,
RT alone cannot achieve sufficient LC (11). This suggests that merely increasing
the intensity of RT is insufficient, indicating the importance of
chemotherapy. In this study, 23 patients in the definitive RT group
received concurrent chemotherapy but under different regimens. EACC
is a rare type of cancer; thus, no standard chemotherapy regimen
has been established, and platinum-based treatments similar to
those for head and neck squamous cell carcinoma are often currently
used. Recently, TPF therapy, which combines docetaxel, cisplatin,
and fluorouracil, has been used as a concurrent chemoradiotherapy
regimen, with reports suggesting its contribution to high response
rates and improved survival (24).
Further data validation in large cohorts is warranted to confirm
their efficacy and safety. Studies have reported achieving good
control with cetuximab as the most prevalent histological type of
head and neck cancer including EACC is the squamous cell carcinoma
(25,26). However, optimal drug selection,
dosage, and timing (concurrent, induction, adjuvant, etc.) remain
unclear. The findings of this study could not clarify the specific
role or optimal content of chemotherapy in definitive RT.
Furthermore, immunoradiotherapy is another promising treatment. The
synergistic mechanisms of programmed cell death-1 inhibitors and
RT, which can activate systemic anti-tumor immunity, were reviewed
(27). For rare malignancies like
EACC, where definitive RT alone often fails to achieve sufficient
local control in advanced stages, the addition of agents such as
pembrolizumab could theoretically improve outcomes. In the future,
multi-institutional collaborative research to identify the
molecular biological characteristics of EACC and conduct clinical
trials to establish more effective chemotherapy regimens with fewer
side effects is important to improve definitive RT outcomes.
The serious adverse event of brain abscess is
another concern. In this study, grade 3 and 4 brain abscess were
observed in two (4.7%) patients. Both were patients in the T4 stage
who had undergone definitive RT (70 Gy) and concurrent
chemotherapy. Brain abscess is a life-threatening, serious late
adverse event that significantly reduces the patient' quality of
life. The irradiation field often encompasses the brain parenchyma
and meninges as the tumor infiltrates the temporal bone and skull
base in the T4 stage. The incidence of central nervous system
infections is higher after RT in patients with nasopharyngeal
carcinoma with a compromised central nervous system barrier, a
condition frequently caused by high chronic otitis media prevalence
(28,29). Thus, the synergistic effect of
high-dose radiation and chemotherapy decreased the tolerance of
normal tissues and increased the risk of infection. However, not
all patients with skull base invasion develop brain abscesses after
the definitive RT. Specific causative factors for early-onset brain
abscess after definitive RT for EACC remained unclear in these
cases. IMRT is useful for improving dose conformity and reducing
the risk of toxicity (30–32). Recently, there's been an increase in
head and neck cancer patients receiving particle therapy (33). Regarding future directions, carbon
ion RT is an emerging modality for EACC. While the integration of
carbon ion RT into postoperative RT strategies could theoretically
enhance local control, this approach warrants extreme caution.
Given the high relative biological effectiveness of carbon ions,
the potential for severe late toxicities such as brain necrosis or
cranial nerve injury, must be carefully weighed, especially when
irradiating the surgical bed where anatomical barriers may be
compromised. Therefore, careful patient selection and precise dose
constraints are mandatory before carbon ion RT combined with
postoperative RT can be considered a viable clinical option.
Moreover, combining high-dose irradiation and chemotherapy in
advanced cases carries the risk of serious complications such as
brain abscess. Therefore, treatment indications, dose, and
chemotherapy intensity must be carefully determined. Furthermore,
close post-treatment follow-up for early detection and appropriate
intervention is crucial.
This study has several limitations. First, the
retrospective study design is subject to potential selection bias,
which can be particularly pronounced in rare cancers. Second, the
limited number of patients precluded a multivariate analysis,
emphasizing the need for a larger sample size for more detailed
investigation, especially since the postoperative RT group included
a very small number of patients. Third, the extended recruitment
period, spanning 23 years, resulted in chronological changes in
treatment methodologies. Fourth, the concurrent chemotherapy
regimens were heterogeneous. While these agents differ in their
mechanisms of action and treatment intensity, the small sample size
precluded a detailed analysis of the efficacy of each specific
regimen. In conclusion, surgery followed by RT was associated with
more favorable outcomes than definitive RT in patients with EACC.
We suggest a stratified treatment approach. For T1-T2 stage,
definitive RT remains a viable option, offering the benefit of
organ preservation. However, for T3-T4 stage, surgery followed by
postoperative RT may be considered as a primary strategy to
mitigate the high risk of local failure associated with definitive
RT. A multidisciplinary approach is crucial for optimizing
treatment outcomes, especially in patients in advanced stages.
Hence, future prospective studies are warranted to establish the
optimal treatment method.
Acknowledgements
Not applicable.
Funding
This work was supported by JSPS KAKENHI (grant no.
24K18769).
Availability of data and materials
The data generated in the present study are not
publicly available due to restrictions such as their containing
information that could compromise the privacy of research
participants, but may be requested from the corresponding
author.
Authors' contributions
YI, TT, NT and AH conceptualized the study. MK, AM,
CS, SO, KU, DO, MN, TM, DK and SI conducted the investigation and
data acquisition. AT, NK, MO and ST performed the formal analysis.
YI wrote the original draft. TM, DK, SI and AH performed review and
editing. YI and TT confirm the authenticity of all the raw data.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
This study was performed after approval by the
institutional review board of Nagoya City University Hospital
(approval no. 60-23-0066). This study was approved by the
institutional review boards of five regional hospitals: Ichinomiya
Municipal Hospital (approval no. 1388), Nagoya Medical Center
(approval no. 2023-436), Japanese Red Cross Aichi Medical Center
Nagoya Daini Hospital (approval no. 5042), Okazaki City Hospital
(approval no. 2023-53), and Kariya Toyota General Hospital
(approval no. 961). Written informed consent was waived due to the
retrospective nature of this study, its content was disclosed in an
opt-out form available on the website. This study has been
conducted in compliance with the guidelines of the Helsinki
Declaration.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
CI
|
confidence interval
|
|
CT
|
computed tomography
|
|
CTV
|
clinical target volume
|
|
EACC
|
external auditory canal cancer
|
|
GTV
|
gross tumor volume
|
|
HR
|
hazard ratio
|
|
IMRT
|
intensity-modulated radiotherapy
|
|
LC
|
local control
|
|
OS
|
overall survival
|
|
PET
|
positron emission tomography
|
|
PFS
|
progression-free survival
|
|
PTV
|
planning target volume
|
References
|
1
|
Gandhi AK, Roy S, Biswas A, Raza MW,
Saxena T, Bhasker S, Sharma A, Thakar A and Mohanti BK: Treatment
of squamous cell carcinoma of external auditory canal: A tertiary
cancer centre experience. Auris Nasus Larynx. 43:45–49. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shinomiya H, Uehara N, Teshima M, Kakigi
A, Otsuki N and Nibu KI: Clinical management for T1 and T2 external
auditory canal cancer. Auris Nasus Larynx. 9:785–789. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nabuurs CH, Kievit W, Leemans CRR, Smit
CFGM, van den Brekel MWM, Pauw RJ, van der Laan BFAM, Jansen JC,
Lacko M, Braunius WW, et al: Postoperative radiotherapy for pT1-
and pT2-classified squamous cell carcinoma of the external auditory
canal. Cancers (Basel). 16:40262024. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shinomiya H, Fujita T and Nibu KI: Current
treatment strategies for external auditory canal cancer. Jpn J Clin
Oncol. 55:699–706. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhou P, de Brito R, Cui Y, Lloyd S, Kunst
H, Kutz JW, Mani N, Moon IS, Mostafa BE, Nabuurs C, et al: The
international expert consensus on management of external auditory
canal carcinoma. Eur Arch Otorhinolaryngol. 282:1677–1691. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
McCracken M, Pai K, Cabrera CI, Johnson
BR, Tamaki A, Gidley PW and Manzoor NF: Temporal bone resection for
squamous cell carcinoma of the lateral skull base: Systematic
review and meta-analysis. Otolaryngol Head Neck Surg. 168:154–164.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Takenaka Y, Cho H, Nakahara S, Yamamoto Y,
Yasui T and Inohara H: Chemoradiation therapy for squamous cell
carcinoma of the external auditory canal: A meta-analysis. Head
Neck. 37:1073–1080. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Moody SA, Hirsch BE and Myers EN: Squamous
cell carcinoma of the external auditory canal: an evaluation of a
staging system. Am J Otol. 21:582–588. 2000.PubMed/NCBI
|
|
9
|
Iwaki S, Kawakita D, Matoba T, Minohara K
and Iwasaki S: Brain abscess following definitive radiotherapy in
patients with external auditory canal carcinoma: Report of two
cases. Cureus. 17:e815922025.PubMed/NCBI
|
|
10
|
Fujiwara M, Yamamoto S, Doi H, Takada Y,
Odawara S, Niwa Y, Ishikura R, Kamikonya N, Terada T, Uwa N, et al:
Arterial chemoradiotherapy for carcinomas of the external auditory
canal and middle ear. Laryngoscope. 125:685–689. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Koto M, Hasegawa A, Takagi R, Sasahara G,
Ikawa H, Mizoe JE, Jingu K, Tsujii H, Kamada T and Okamoto Y;
Organizing Committee for the Working Group for Head and Neck
Cancer, . Carbon ion radiotherapy for locally advanced squamous
cell carcinoma of the external auditory canal and middle ear. Head
Neck. 38:512–516. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Choi J, Kim SH, Koh YW, Choi EC, Lee CG
and Keum KC: Tumor stage-related role of radiotherapy in patients
with an external auditory canal and middle ear carcinoma. Cancer
Res Treat. 49:178–184. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Matoba T, Hanai N, Suzuki H, Nishikawa D,
Tachibana E, Okada T, Murakami S and Hasegawa Y: Treatment and
outcomes of carcinoma of the external and middle ear: The validity
of en bloc resection for advanced tumor. Neurol Med Chir (Tokyo).
58:32–38. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hayashi K, Koto M, Demizu Y, Saitoh JI,
Suefuji H, Okimoto T, Ohno T, Shioyama Y, Takagi R, Ikawa H, et al:
A retrospective multicenter study of carbon-ion radiotherapy for
external auditory canal and middle ear carcinomas. Cancer Med.
8:51–57. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nagano T, Yoshimura RI, Kojima M, Nakagawa
K and Toda K: Outcomes of radiotherapy in advanced external
auditory canal cancer. J Radiat Res. 60:380–386. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shiga K, Nibu KI, Fujimoto Y, Asakage T,
Homma A, Mitani H, Ogawa T, Okami K, Murono S, Hirano S, et al:
Multi-institutional survey of squamous cell carcinoma of the
external auditory canal in Japan. Laryngoscope. 131:E870–E874.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Laskar SG, Sinha S, Pai P, Nair D,
Budrukkar A, Swain M, Kumar A, Moiyadi A, Shetty P, Ray V, et al:
Definitive and adjuvant radiation therapy for external auditory
canal and temporal bone squamous cell carcinomas: Long term
outcomes. Radiother Oncol. 170:151–158. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jang IJH, Thong JF, Teo CEH and Sommat K:
Analysis of prognostic factors for external auditory canal
carcinoma: A 22-year experience. Laryngoscope. 133:2203–2210. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ooka T, Ariizumi Y, Asakage T and Tsutsumi
T: Treatment outcomes of 73 cases of external auditory canal
squamous cell carcinoma: A single-center six-year analysis in
Japan. Auris Nasus Larynx. 52:158–166. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang S, Yan L, Li R, Zhao Y, Wang X,
Zhang Y and Zhu Y: Development and validation of a nomogram to
predict overall survival in patients with External auditory canal
cancer. Radiother Oncol. 203:1106912025. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Katano A, Takenaka R, Yamashita H, Ando M,
Yoshida M, Saito Y, Asakage T, Abe O and Nakagawa K: A
retrospective analysis of radiotherapy in the treatment of external
auditory canal carcinoma. Mol Clin Oncol. 14:452021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Morris LG, Mehra S, Shah JP, Bilsky MH,
Selesnick SH and Kraus DH: Predictors of survival and recurrence
after temporal bone resection for cancer. Head Neck. 34:1231–1239.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Takaoka T, Shibamoto Y, Matsuo M, Sugie C,
Murai T, Ogawa Y, Miyakawa A, Manabe Y, Kondo T, Nakajima K, et al:
Biological effects of hydrogen peroxide administered intratumorally
with or without irradiation in murine tumors. Cancer Sci.
108:1787–1792. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yamada A, Shinomiya H, Uehara N, Iritani
K, Tatehara S, Furukawa T, Teshima M, Miyawaki D, Fujita T, Kakigi
A, et al: Oncological outcomes of concurrent chemoradiotherapy with
docetaxel, cisplatin, and 5-fluorouracil for locally advanced
squamous cell carcinoma of the external auditory canal: A
single-center study. Head Neck. 45:2498–2504. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ebisumoto K, Okami K, Hamada M, Maki D,
Sakai A, Saito K, Shimizu F, Kaneda S and Iida M: Cetuximab with
radiotherapy as an alternative treatment for advanced squamous cell
carcinoma of the temporal bone. Auris Nasus Larynx. 45:637–639.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Imai C, Saeki H, Yamamoto K, Ichikawa A,
Arai M, Tawada A, Suzuki T, Takiguchi Y, Hanazawa T and Ishii I:
Radiotherapy plus cetuximab for locally advanced squamous cell head
and neck cancer in patients with cisplatin-ineligible renal
dysfunction: A retrospective study. Oncol Lett. 23:1522022.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wei C, Lan X, Qiu M, Cui R, Fu Q, Shinge
SAU, Muluh TA and Jiang O: Expanding the role of combined
immunochemotherapy and immunoradiotherapy in the management of head
and neck cancer (Review). Oncol Lett. 26:3722023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fang PH, Lin WC, Tsai NW, Chang WN, Huang
CR, Chang HW, Huang TL, Lin HC, Lin YJ, Cheng BC, et al: Bacterial
brain abscess in patients with nasopharyngeal carcinoma following
radiotherapy: Microbiology, clinical features and therapeutic
outcomes. BMC Infect Dis. 12:2042012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chuang JM, Lin WC, Fang FM, Huang YJ, Ho
JT and Lu CH: Bacterial brain abscess formation in post-irradiated
patients: What is the possible pathogenesis? Clin Neurol Neurosurg.
136:132–138. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tomita N, Kodaira T, Tachibana H, Nakamura
T, Nakahara R, Inokuchi H, Mizoguchi N and Takada A: A comparison
of radiation treatment plans using IMRT with helical tomotherapy
and 3D conformal radiotherapy for nasal natural killer/T-cell
lymphoma. Br J Radiol. 82:756–763. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Torii A, Tomita N, Takaoka T, Kondo T,
Yamamoto S, Sugie C, Nagai A, Miyakawa A, Kuno M, Uchiyama K, et
al: Salvage radiotherapy for locoregional recurrence of esophageal
cancer after surgery. Jpn J Clin Oncol. 55:59–66. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xue T, Zhou J, Wang B, Xiao L and Sun Y:
Dosimetric comparison and prognostic analysis of helical
tomotherapy vs. intensity-modulated radiation therapy in locally
advanced cervical cancer. Oncol Lett. 30:3962025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nomura M: Definitive treatment for head
and neck mucosal melanoma. Jpn J Clin Oncol. 53:1112–1118. 2023.
View Article : Google Scholar : PubMed/NCBI
|